Introduction
Navigating the landscape of research ethics can be daunting, particularly in Belize, where the Institutional Review Board (IRB) plays a critical role in safeguarding human subjects. Researchers face the dual challenge of adhering to rigorous ethical standards while ensuring their studies are scientifically valid and impactful. This guide serves as a comprehensive roadmap to achieving IRB approval in Belize, detailing essential requirements and common pitfalls to avoid.
How can researchers streamline their applications and enhance their chances of success amidst these complexities? By understanding the nuances of the IRB process, researchers can better position themselves for approval and contribute meaningfully to the field of clinical research.
Understand the Role of Institutional Review Boards (IRBs)
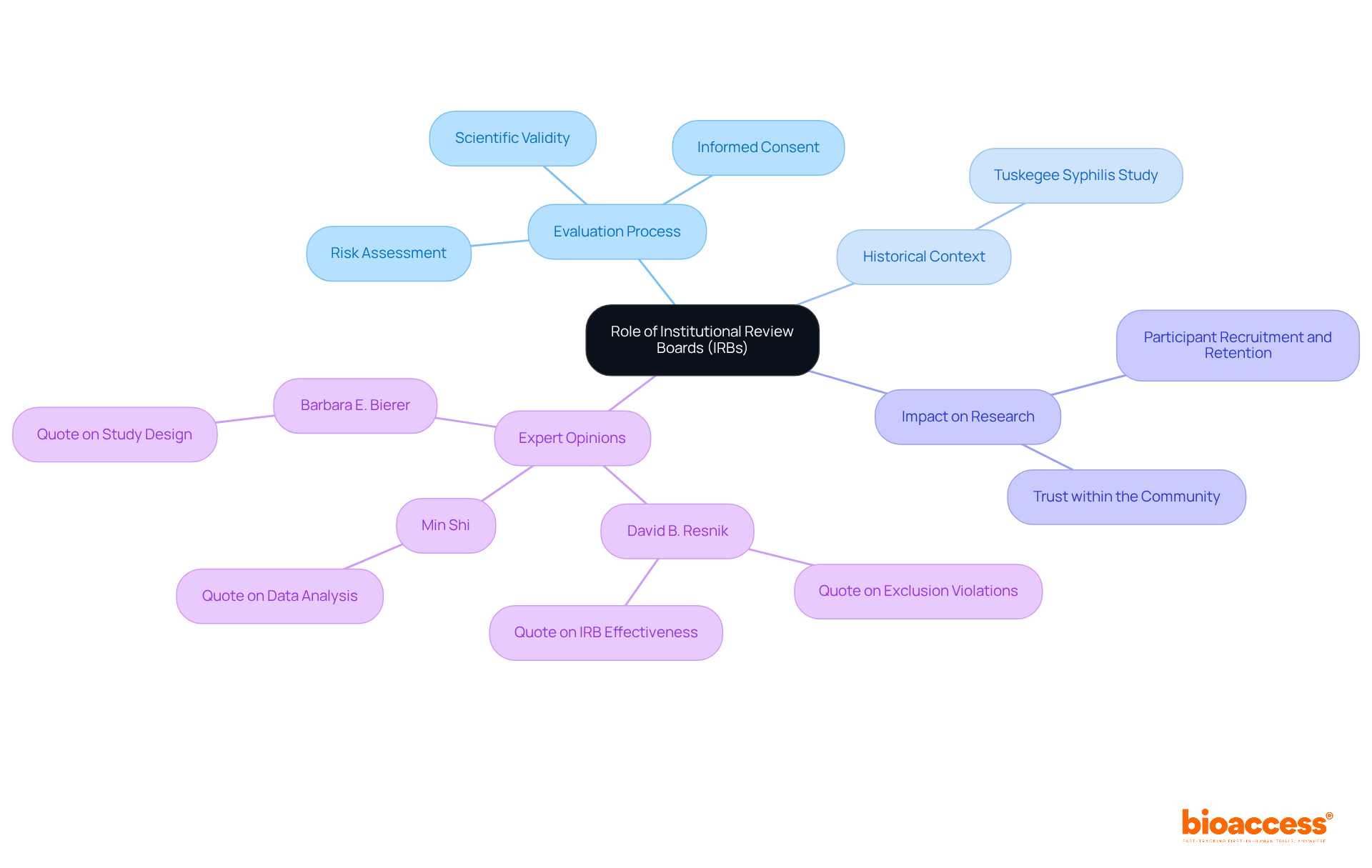
Institutional Review Boards (IRBs) are essential committees that evaluate and approve studies involving human subjects, with a primary focus on safeguarding the rights, welfare, and privacy of participants. In Belize, irb approval belize plays a pivotal role in ensuring that research adheres to ethical standards and regulatory requirements. This is particularly significant given the historical context of ethics in research, influenced by past violations like the Tuskegee Syphilis Study, which highlighted the urgent need for robust protective measures.
The process for irb approval belize typically involves a comprehensive evaluation of research proposals, emphasizing critical aspects such as risk assessment, informed consent, and the scientific validity of study designs. This process is vital for researchers, as it not only helps them comply with guidelines but also enhances the credibility of their studies.
Statistics reveal that IRB panels in Belize convene regularly to review applications, providing timely feedback and support for researchers. For instance, studies indicate that the irb approval belize timelines can be as efficient as those in other regions, with panels meeting monthly to assess new proposals. Successful irb approval belize in the region showcases the effectiveness of these boards in fostering ethical inquiry practices. A recent study that navigated the IRB process successfully reported improved participant recruitment and retention, alongside enhanced trust within the community.
Experts underscore the importance of IRBs in maintaining ethical standards in research. As David B. Resnik, a bioethicist at NIEHS/NIH, noted, “Unwarranted exclusion of individuals with uncertain or impaired decision-making capacity from involvement in studies violates principles of justice and fairness and adversely impacts the health and welfare of these populations.” This statement emphasizes the delicate balance IRBs must maintain between facilitating research and protecting human subjects.
Understanding the role of irb approval belize is crucial for investigators aiming to conduct ethically sound studies that respect participant rights and contribute positively to the field of inquiry.
Identify Requirements for IRB Approval in Belize
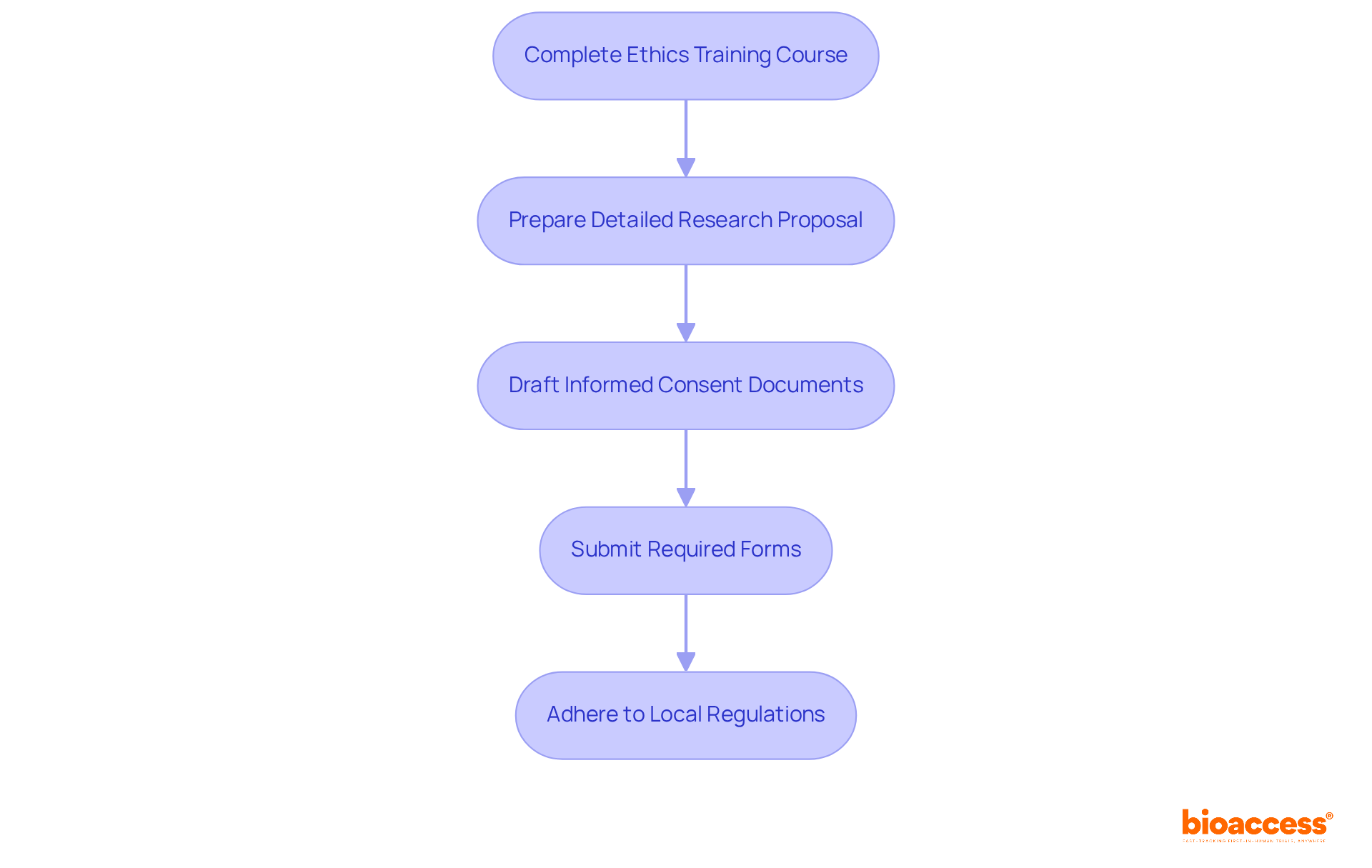
To obtain IRB approval in Belize, researchers must fulfill several key requirements:
- Complete an Ethics Training Course: Participation in a recognized ethics training program, such as the Collaborative Institutional Training Initiative (CITI) program, is essential. CITI training plays a vital role in promoting integrity and principled practices at UB, equipping scholars with the necessary knowledge in study design and moral conduct, ensuring alignment with international standards.
- Prepare a Detailed Research Proposal: The proposal should clearly outline the study’s objectives, methodology, participant recruitment strategies, and data management plans. Successful proposals often demonstrate a thorough understanding of ethical considerations and participant safety.
- Informed Consent Documents: Researchers must draft clear and comprehensive informed consent forms that detail the study’s purpose, procedures, risks, and benefits. These documents are crucial for guaranteeing that participants are fully informed before consenting to take part in the study.
- Submit Required Forms: The IRB application form, along with all supporting documents, including the study proposal and consent forms, must be submitted for IRB approval evaluation. A written approval notification, including an IRB number, will be sent via email once the study is approved. Note that submissions to the IRB will undergo administrative screening for completeness and eligibility, followed by a formal review that may take approximately thirty working days.
- Adherence to Local Regulations: It is crucial that the study conforms to Belizean laws and moral guidelines, especially those set by the Ministry of Health and Wellness. Effective March 1, all public health studies involving human participants must be submitted through the new MOHW IRB process, which emphasizes ethical oversight and participant protection.
Submit Your IRB Application: A Step-by-Step Process
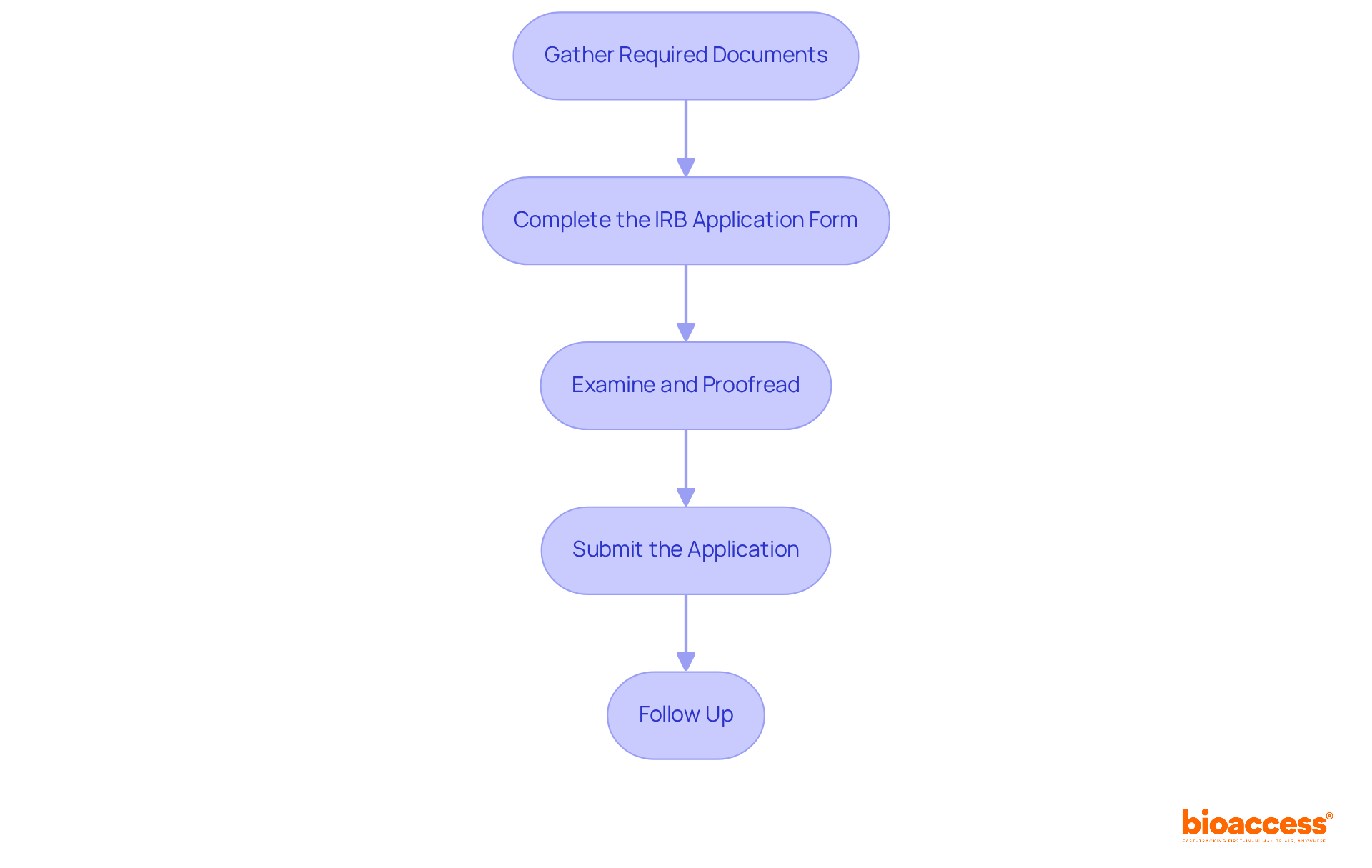
To successfully submit your IRB application in Belize, follow these essential steps:
- Gather Required Documents: Compile crucial documents, including your research proposal, informed consent forms, and certificates of ethics training. Ensure that all materials are current and relevant to your study.
- Complete the IRB Application Form: Fill out the IRB application form accurately, making sure every section is thoroughly completed. Incomplete forms often lead to delays, accounting for 80% of significant holdups in IRB assessments due to slow responses from investigators regarding requested modifications.
- Examine and Proofread: Before submission, meticulously assess your application for clarity and completeness. Organize all documents logically and check for typographical or formatting errors, as these can lead to unnecessary complications.
- Submit the Application: Submit your application either electronically or in person to the designated IRB office, along with any applicable fees. Keep a copy of your submission for your records.
- Follow Up: After submission, promptly check in with the IRB to confirm receipt of your application and inquire about the anticipated evaluation timeline. As highlighted by specialist Daniel E Hall, ‘The most crucial action investigators can take to accelerate the evaluation is to respond swiftly (within a week).’ Be prepared to respond quickly to any requests for additional information or clarifications, as delays in responses can significantly extend approval times. Remember, the median time for full board evaluation is 131 days, so setting realistic expectations is crucial.
By following these steps, you can enhance your chances of a smooth and efficient IRB approval Belize process.
Troubleshoot Common Challenges in the IRB Approval Process
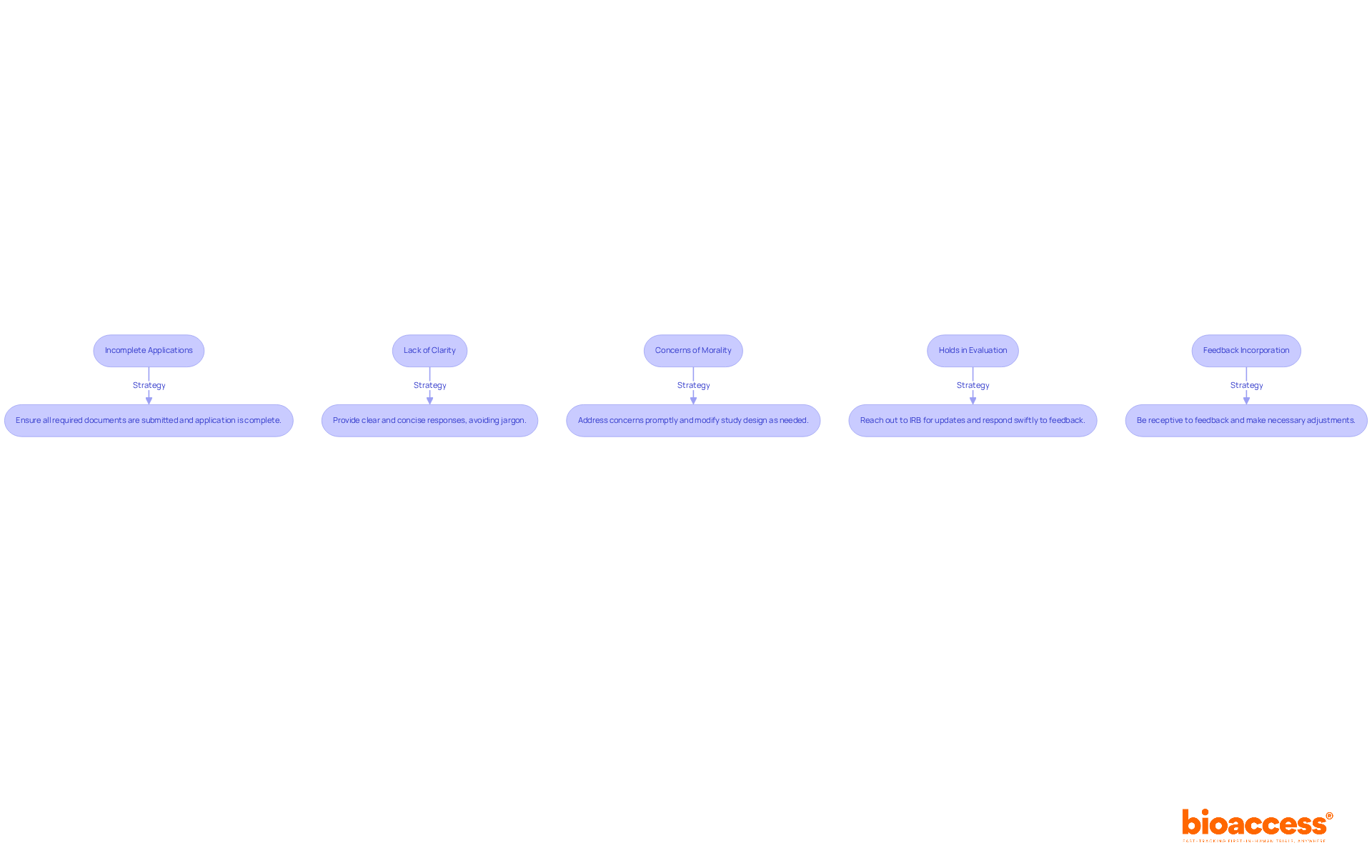
Researchers frequently encounter various challenges during the irb approval belize process. Here are effective strategies to troubleshoot these issues:
- Incomplete Applications: Ensure that all required documents are submitted and that the application form is fully completed. Missing signatures or documents can lead to significant delays, as over 80% of severe delays in IRB evaluation stem from slow responses to requested changes. As noted by Daniel E. Hall, “The most important thing investigators can do to expedite the review is to respond quickly (within a week).”
- Lack of Clarity: When reviewers request clarifications, provide clear and concise responses. Avoid jargon and ensure that your explanations are easily understandable. Clear communication can drastically reduce the time spent in back-and-forth exchanges.
- Concerns of Morality: Address any concerns raised by the IRB promptly. Be prepared to modify your study design or consent forms to meet ethical standards. A thorough risk/benefit analysis that aligns across all documents can enhance the submission’s quality and minimize delays in obtaining irb approval belize.
- Holds in Evaluation: If the evaluation process is taking longer than expected, reach out to the IRB for updates. Understanding their timeline can help you manage your project schedule effectively. It is crucial to respond swiftly to any pre-review feedback to avoid unnecessary delays.
- Feedback Incorporation: Be receptive to feedback from the IRB and willing to make necessary adjustments to your proposal. This collaborative approach can facilitate a smoother approval process. As observed by clinical researchers, prompt responses to IRB requests can significantly accelerate the evaluation. For instance, a case study on “Incomplete Submission – Missing or Inadequate Supporting Documents” illustrates how proactive communication can resolve submission issues and expedite the review process.
Conclusion
Achieving IRB approval in Belize stands as a pivotal milestone for researchers intent on conducting ethical studies involving human subjects. Grasping the function of Institutional Review Boards (IRBs) and the specific criteria for approval is crucial in navigating this intricate process. By prioritizing participant welfare and adhering to ethical standards, researchers can ensure their work not only advances the field but also benefits society as a whole.
This guide has delineated the essential steps to secure IRB approval, encompassing everything from completing ethics training and crafting detailed research proposals to submitting applications and tackling common challenges. Key insights highlight the necessity of clear communication, meticulous documentation, and responsiveness to feedback-elements that significantly bolster the chances of a seamless approval process.
Given the vital role that IRBs play in protecting research participants, it is imperative for researchers to approach the IRB approval process with diligence and a deep respect for ethical standards. By adhering to the outlined steps and proactively addressing potential hurdles, researchers can streamline their approval journey while enhancing the integrity and credibility of their research endeavors. Embracing these practices not only ensures compliance with local regulations but also cultivates trust and collaboration within the research community.
Frequently Asked Questions
What is the primary role of Institutional Review Boards (IRBs)?
The primary role of IRBs is to evaluate and approve studies involving human subjects, focusing on safeguarding the rights, welfare, and privacy of participants.
Why is IRB approval important in Belize?
IRB approval in Belize is crucial for ensuring that research adheres to ethical standards and regulatory requirements, particularly in light of historical violations in research ethics.
What does the IRB approval process typically involve?
The IRB approval process involves a comprehensive evaluation of research proposals, emphasizing risk assessment, informed consent, and the scientific validity of study designs.
How does IRB approval benefit researchers?
IRB approval helps researchers comply with guidelines and enhances the credibility of their studies.
How often do IRB panels in Belize meet to review applications?
IRB panels in Belize typically meet monthly to assess new proposals, providing timely feedback and support for researchers.
What impact does successful IRB approval have on research studies?
Successful IRB approval can lead to improved participant recruitment and retention, as well as enhanced trust within the community.
What do experts say about the importance of IRBs?
Experts emphasize that IRBs are essential for maintaining ethical standards in research and protecting participants, particularly vulnerable populations.
Why is understanding the role of IRBs important for investigators?
Understanding the role of IRBs is crucial for investigators to conduct ethically sound studies that respect participant rights and contribute positively to the field of inquiry.
List of Sources
- Understand the Role of Institutional Review Boards (IRBs)
- An Analysis of Institutional Review Board Policies for Enrollment of Adults with Impaired or Uncertain Decision-Making Capacity – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC12227281)
- Institutional Review Boards: Purpose and Challenges – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC4631034)
- The IRB and me: What is the Institutional Review Board? – @theU (https://attheu.utah.edu/facultystaff/irb-1)
- Institutional Review Boards Must Address the Ethics of Inclusion in Clinical Research | American Association for the Advancement of Science (AAAS) (https://aaas.org/news/institutional-review-boards-must-address-ethics-inclusion-clinical-research)
- Overview of IRB Purpose and Practices (https://statisticssolutions.com/overview-of-irb-purpose-and-practices)
- Identify Requirements for IRB Approval in Belize
- Health Ministry Tightens Public Health Research Oversight with IRB Transition (https://lovefm.com/health-ministry-tightens-public-health-research-oversight-with-irb-transition)
- CITI Program Training | University of Belize Research (https://ub.edu.bz/research-citi-program-training)
- Institutional Review Board (IRB) | University of Belize Research (https://ub.edu.bz/research-about-the-irb)
- Submit Your IRB Application: A Step-by-Step Process
- Time Required for Institutional Review Board Review at One Veterans Affairs Medical Center – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC4527305)
- Institutional Review Board (IRB) | University of Belize Research (https://ub.edu.bz/research-about-the-irb)
- Timing Your IRB Application for Success – Solutions IRB (https://solutionsirb.com/timing-your-irb-application-for-success)
- Troubleshoot Common Challenges in the IRB Approval Process
- Time Required for IRB Review (https://jamanetwork.com/journals/jamasurgery/fullarticle/2020866)
- 70 Research Quotes to Inspire Your Work – Qualtrics (https://qualtrics.com/articles/strategy-research/research-quotes)
- IRB: Common Application Problems | Office of Research (https://sjsu.edu/research/research-compliance/irb/irb-help/common-problems.php)
- Top Reasons for IRB Submission Delays | Office of the Vice President for Research | University of Connecticut (https://ovpr.uchc.edu/top-reasons-for-irb-submission-delays)

Leave a Reply