Introduction
Chile has become a pivotal player in clinical research, especially in medical devices, due to its efficient regulatory environment and cost advantages. Companies aiming to conduct trials in this vibrant market stand to gain not only from reduced expenses but also from a diverse patient population that enhances research robustness. Navigating local regulations can be daunting for sponsors, making the selection of a CRO critical to success.
What key factors should sponsors consider to partner with a CRO that aligns with their goals and enhances their success in this competitive environment?
Understand the Benefits of Conducting Trials in Chile
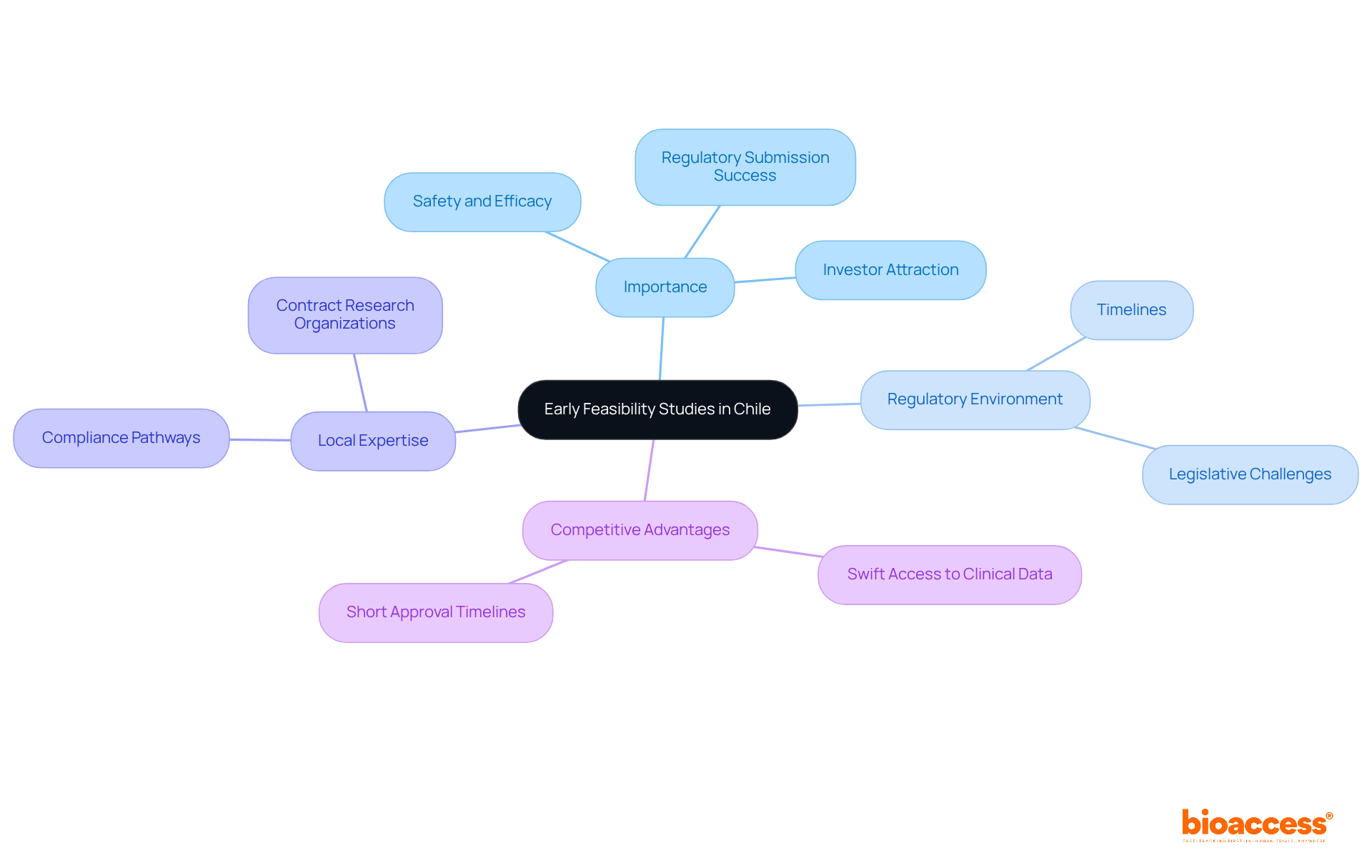
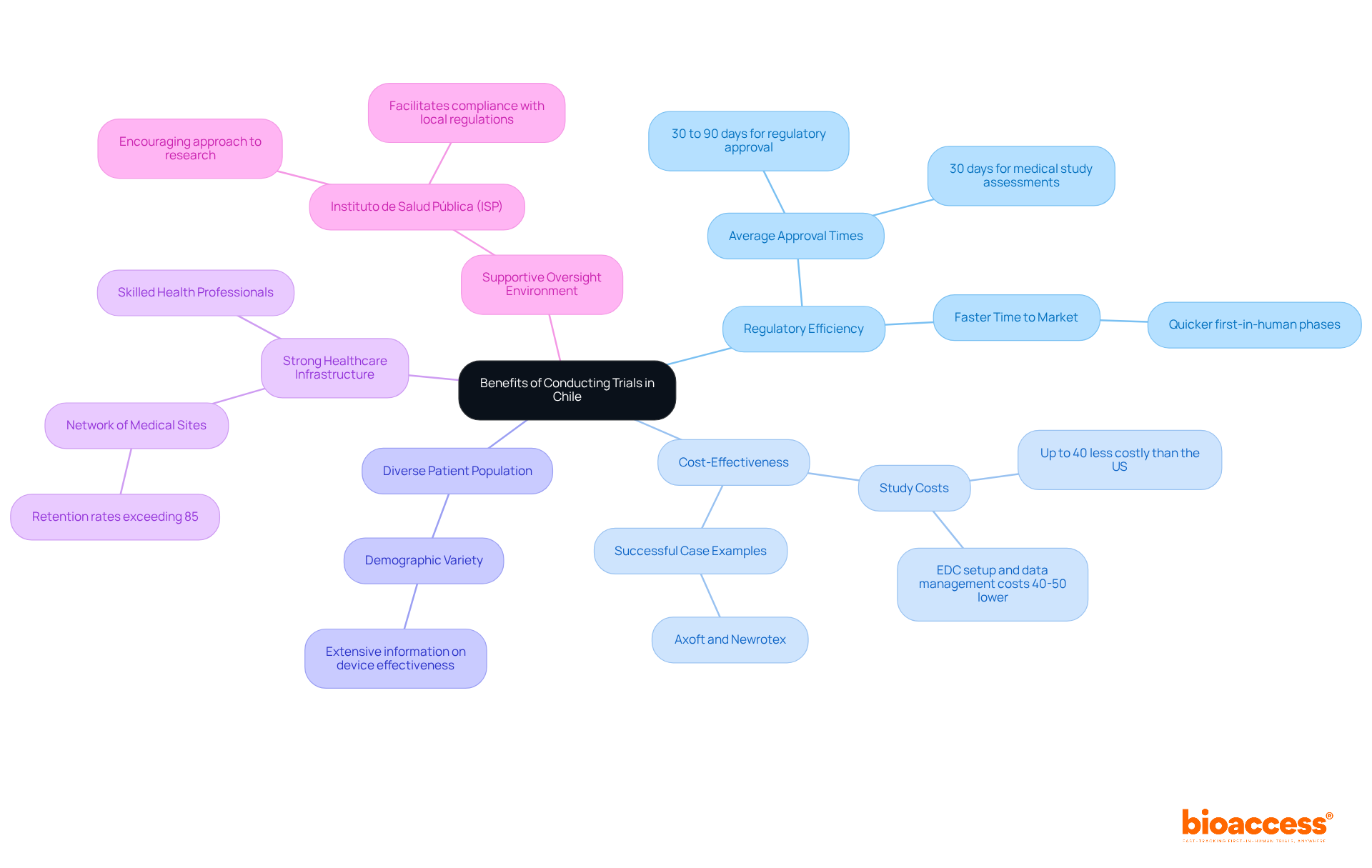
Chile is recognized as a prime location for a clinical research organization specializing in medical device clinical trials, particularly beneficial for MedTech and Biopharma companies, due to its unique advantages.
- Regulatory Efficiency: The regulatory landscape in Chile is notably streamlined, with average approval times ranging from 30 to 90 days. The assessment period for medical studies typically takes around 30 days, significantly speeding up the time to market for new medical devices and therapies. This efficiency allows companies, particularly clinical research organization Chile medical device firms, to bring their innovations to market faster, a vital element for those using LATAM pathways to achieve first-in-human phases quicker than ever.
- Cost-Effectiveness: Conducting studies in Chile can be up to 40% less costly than in the United States. This cost efficiency is crucial for startups aiming to optimize their funding and resources while ensuring high-quality research outcomes. Costs associated with EDC setup, data cleaning, query management, and biostatistical analysis are also 40-50% lower in LATAM. Successful cases like Axoft and Newrotex demonstrate how leveraging bioaccess®’s services can lead to rapid advancements.
- Diverse Patient Population: The demographic variety in Chile provides a valuable resource for a clinical research organization Chile medical device studies, which is crucial for collecting extensive information on the effectiveness and safety of medical devices across different population groups. This diversity enhances the robustness of research findings, making them more applicable to broader markets.
- Strong Healthcare Infrastructure: The nation features a solid network of medical sites and skilled health professionals, guaranteeing high-quality data collection and patient care throughout the research process. Notably, Chile has retention rates exceeding 85%, surpassing global averages, which highlights the effectiveness of its research infrastructure in supporting trials. This infrastructure is essential for companies like Sparta Biomedical, a clinical research organization Chile medical device, which have successfully navigated the trial landscape with the help of bioaccess®.
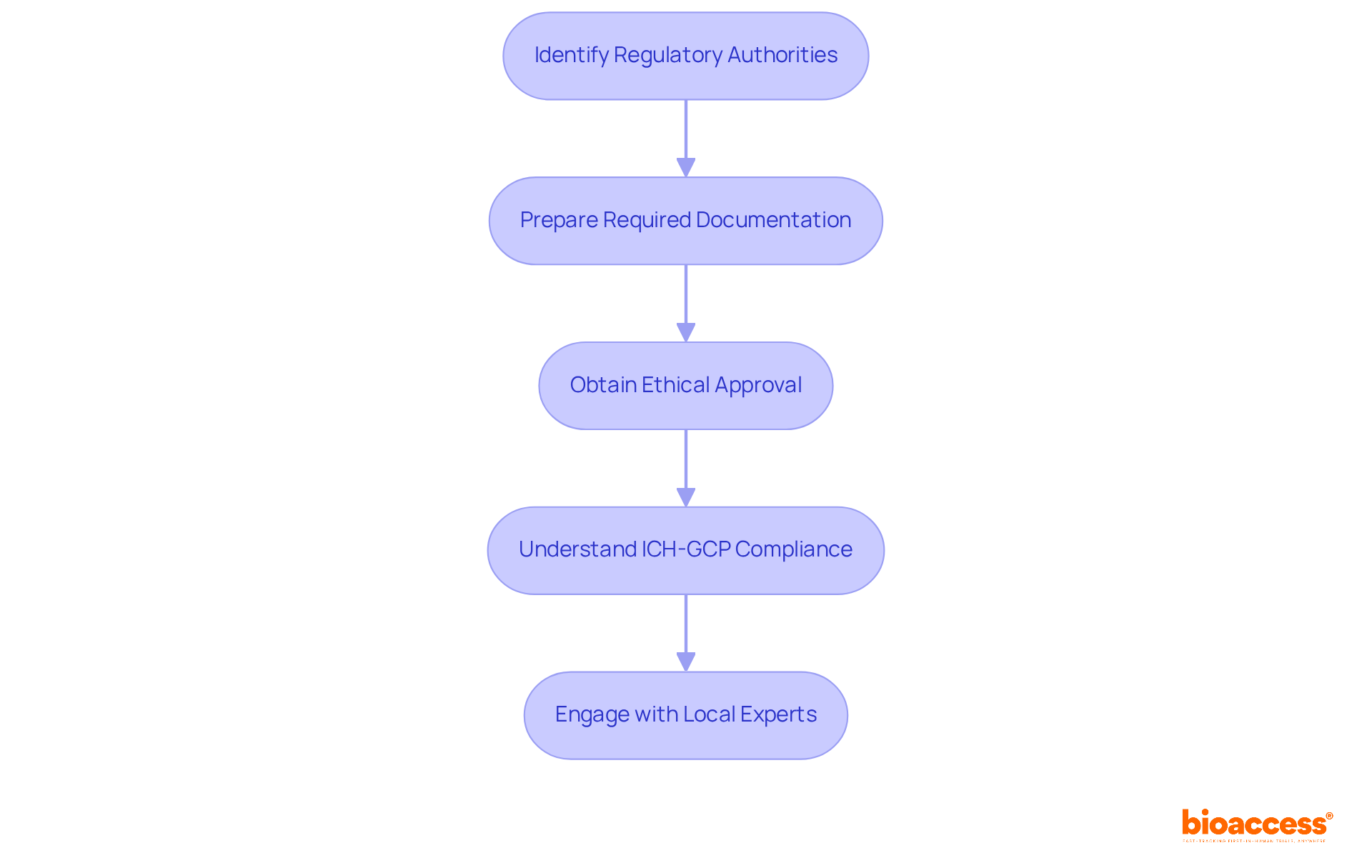
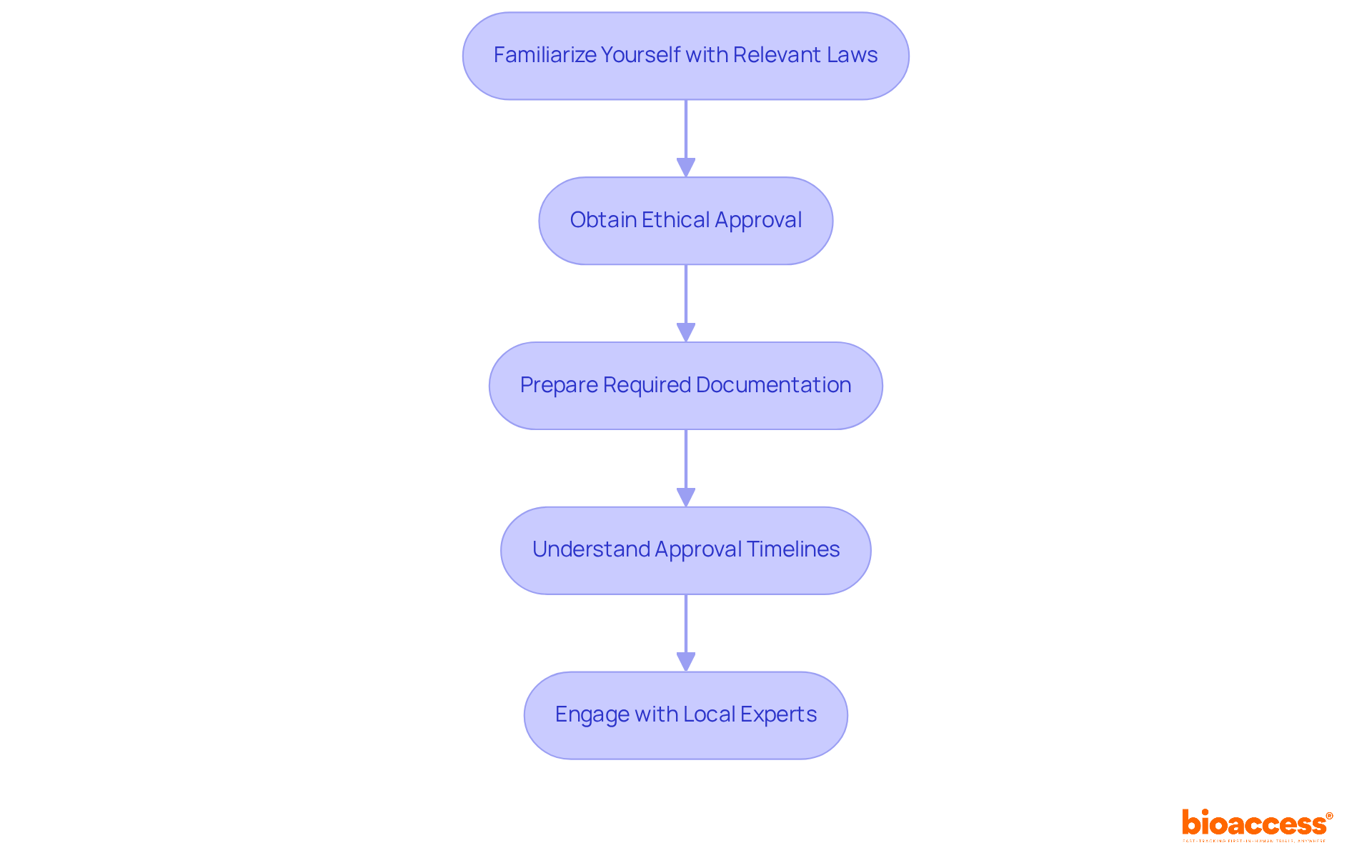
- Supportive Oversight Environment: The Instituto de Salud Pública (ISP) acts as the primary oversight authority, recognized for its encouraging approach to research involving human subjects. This facilitates a conducive environment for innovation and development, enabling sponsors to navigate the approval process effectively. Comprehensive documentation preparation is essential for navigating this process, ensuring compliance with local regulations, including ICH-GCP standards.
In conclusion, Chile’s administrative efficiency, cost benefits, varied patient demographics, and robust infrastructure make it a compelling location for executing early-stage studies, especially for firms concentrating on first-in-human research. By leveraging Chile’s strengths, companies can not only enhance their research outcomes but also position themselves for success in a competitive landscape.
Evaluate Key Criteria for Selecting a CRO
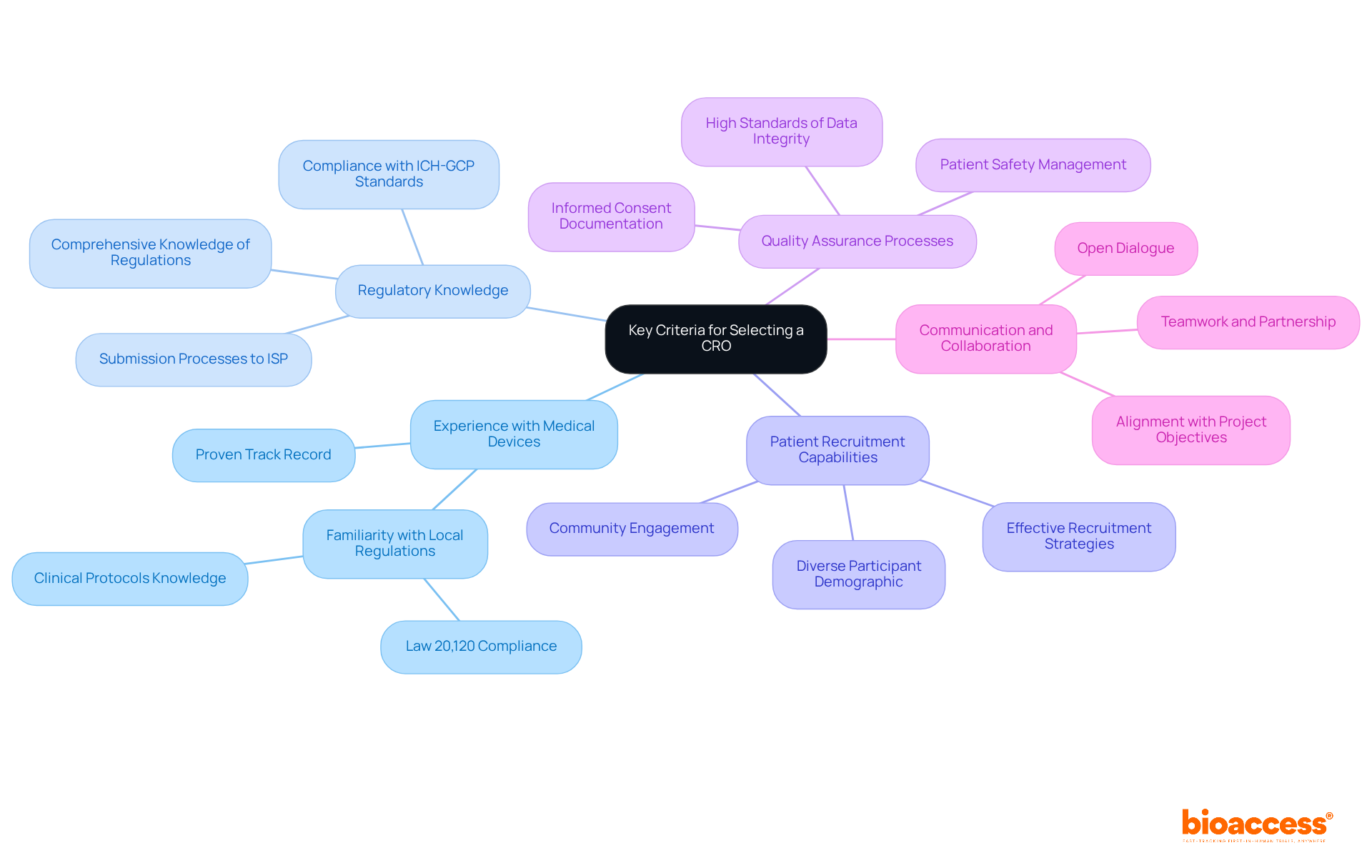
Selecting the right clinical research organization in Chile for medical device trials is crucial for success, yet many sponsors overlook key criteria that can make or break their studies in the clinical research organization Chile medical device sector. Here are several key criteria to prioritize:
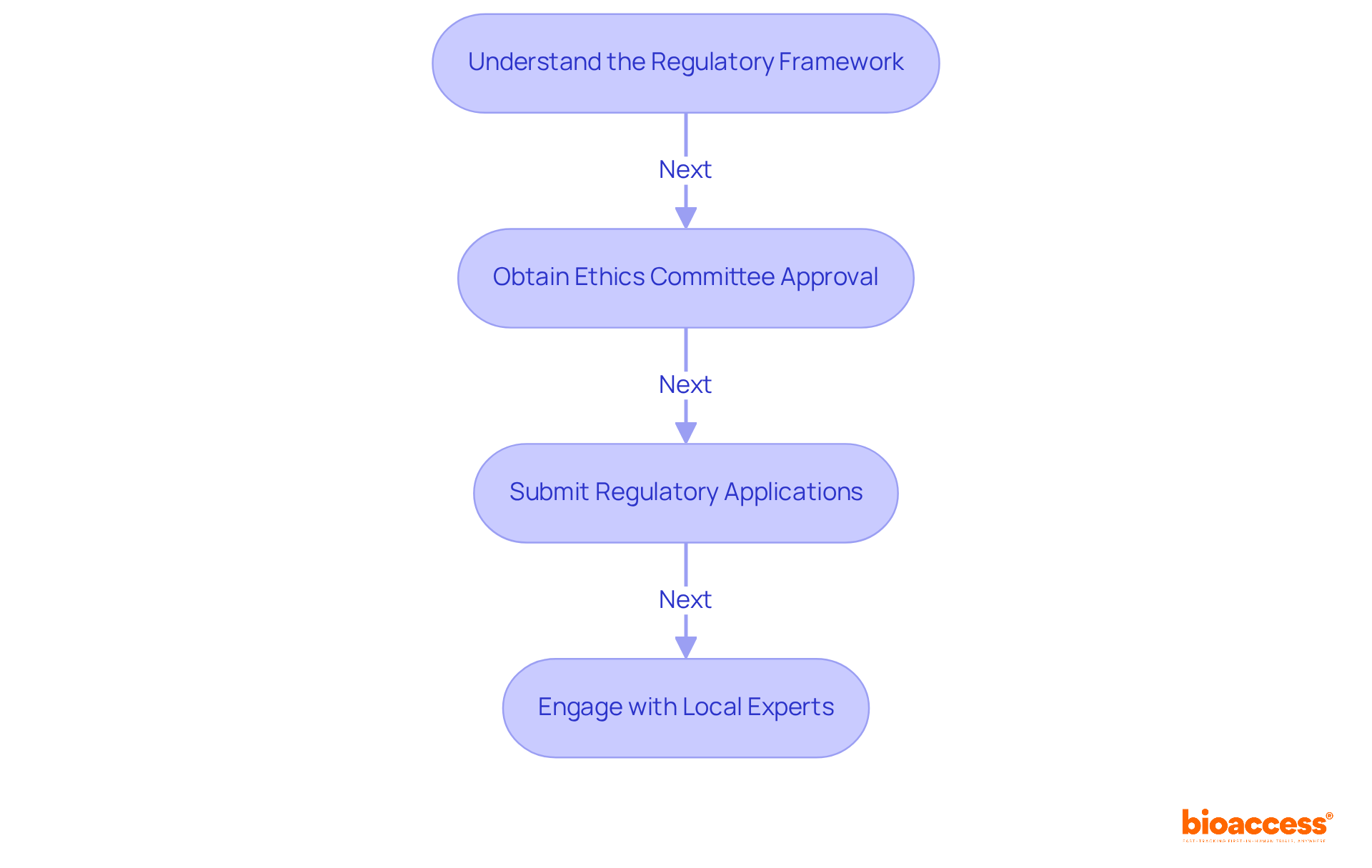
- Experience with Medical Devices: Choose a CRO with a proven track record in conducting medical device trials, including familiarity with local regulatory requirements such as Law 20,120 and clinical protocols specific to your product. This experience is essential for navigating the complexities of the Chilean market, which can be daunting without the right expertise.
- Regulatory Knowledge: The CRO must possess comprehensive knowledge of Chilean regulations, including compliance with ICH-GCP standards and the submission processes to the Instituto de Salud Pública (ISP). This expertise within a clinical research organization in Chile for medical devices helps you navigate the regulatory landscape smoothly, ensuring you get those approvals on time.
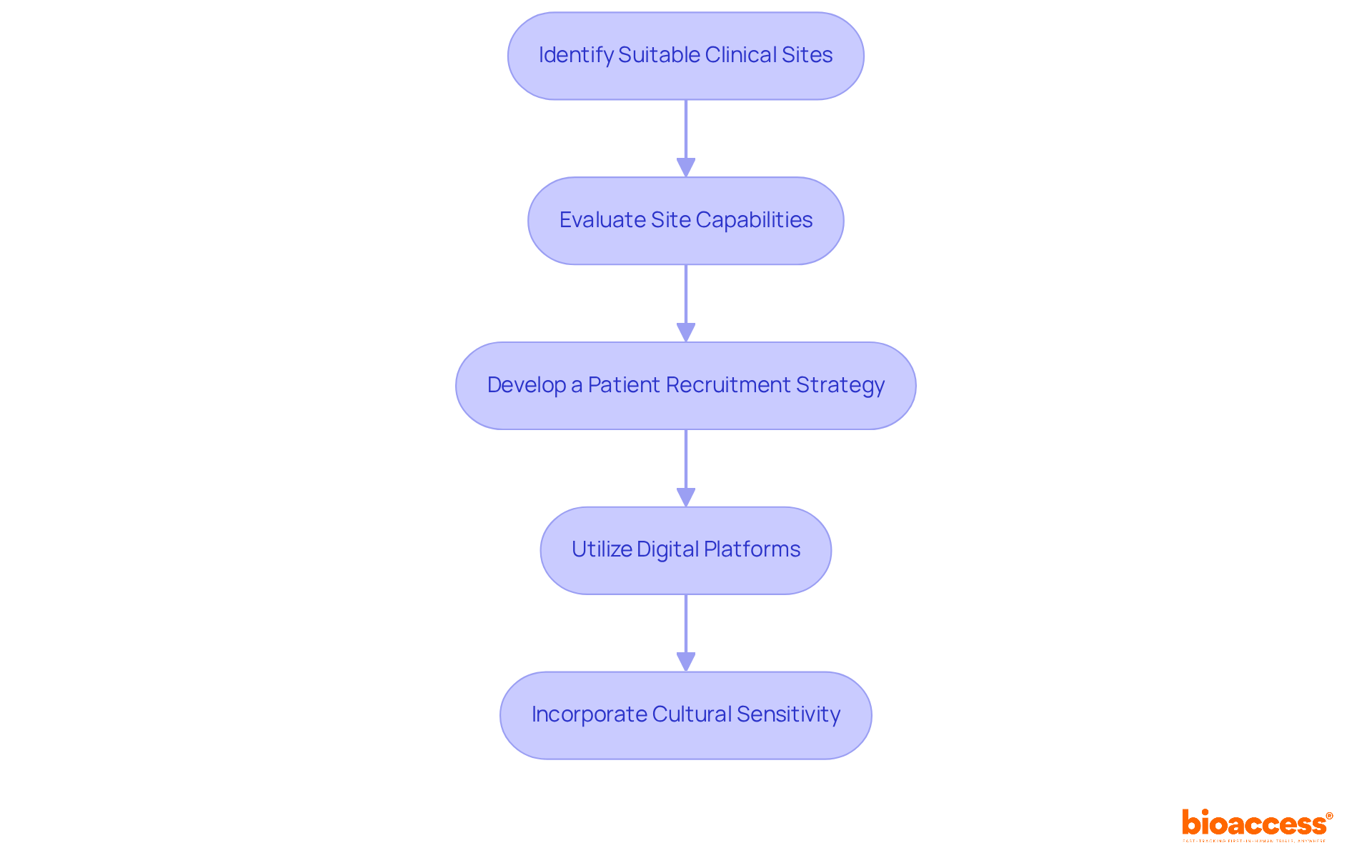
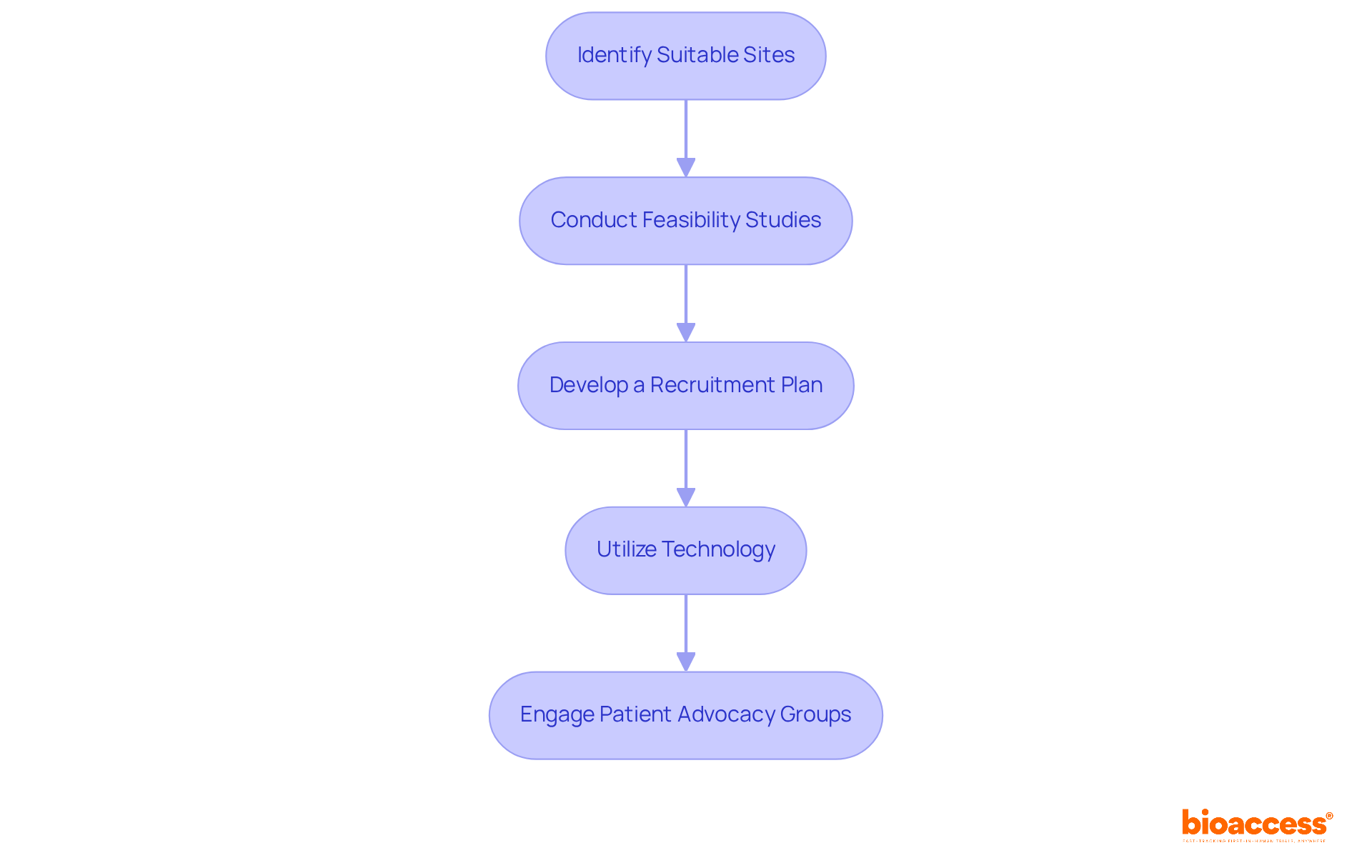
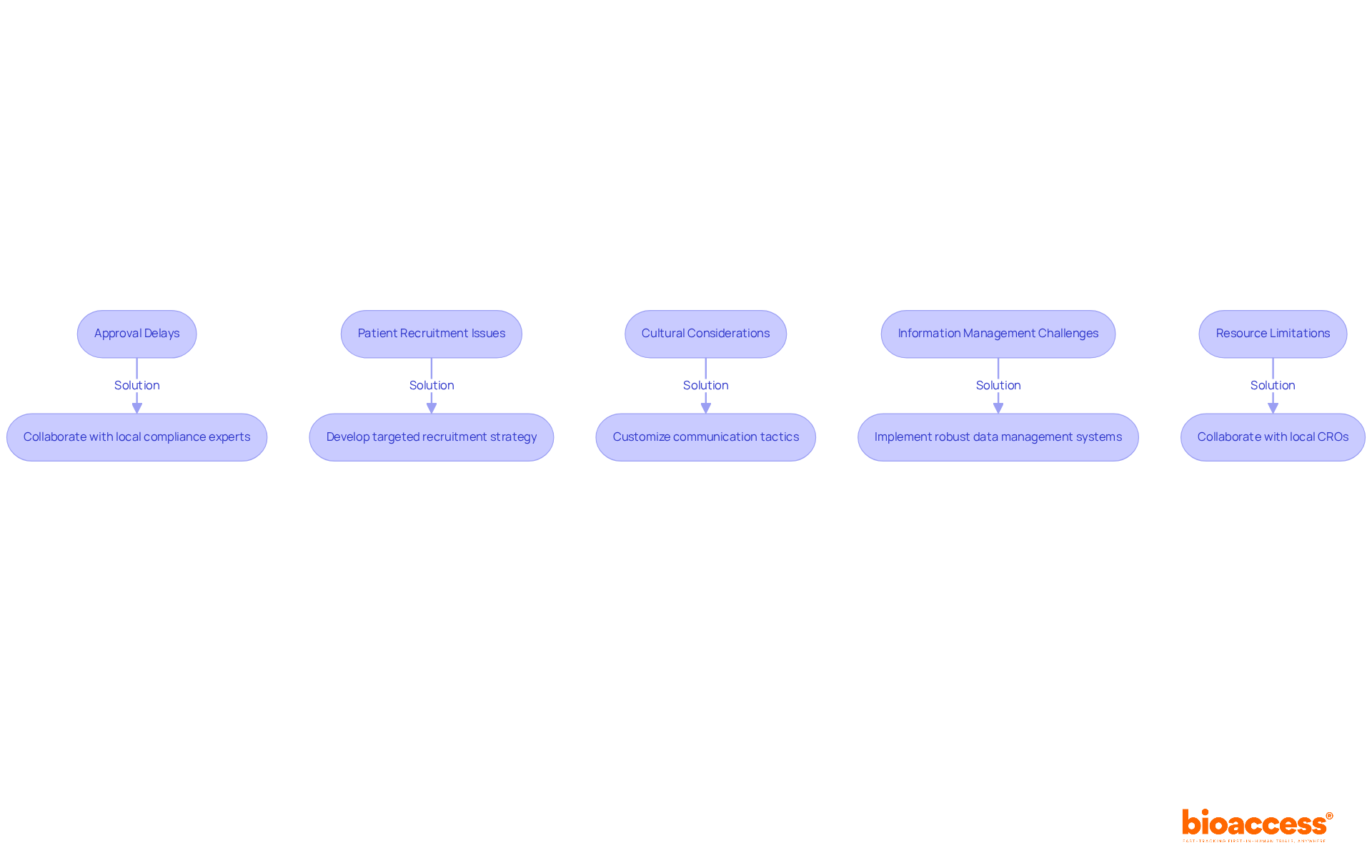
- Patient Recruitment Capabilities: Evaluate the CRO’s strategies for patient recruitment and retention. A strong recruitment strategy is crucial for adhering to timelines and guaranteeing a diverse participant demographic, which is essential for the credibility of clinical outcomes. Effective recruitment strategies customized for the local community can significantly improve study success.
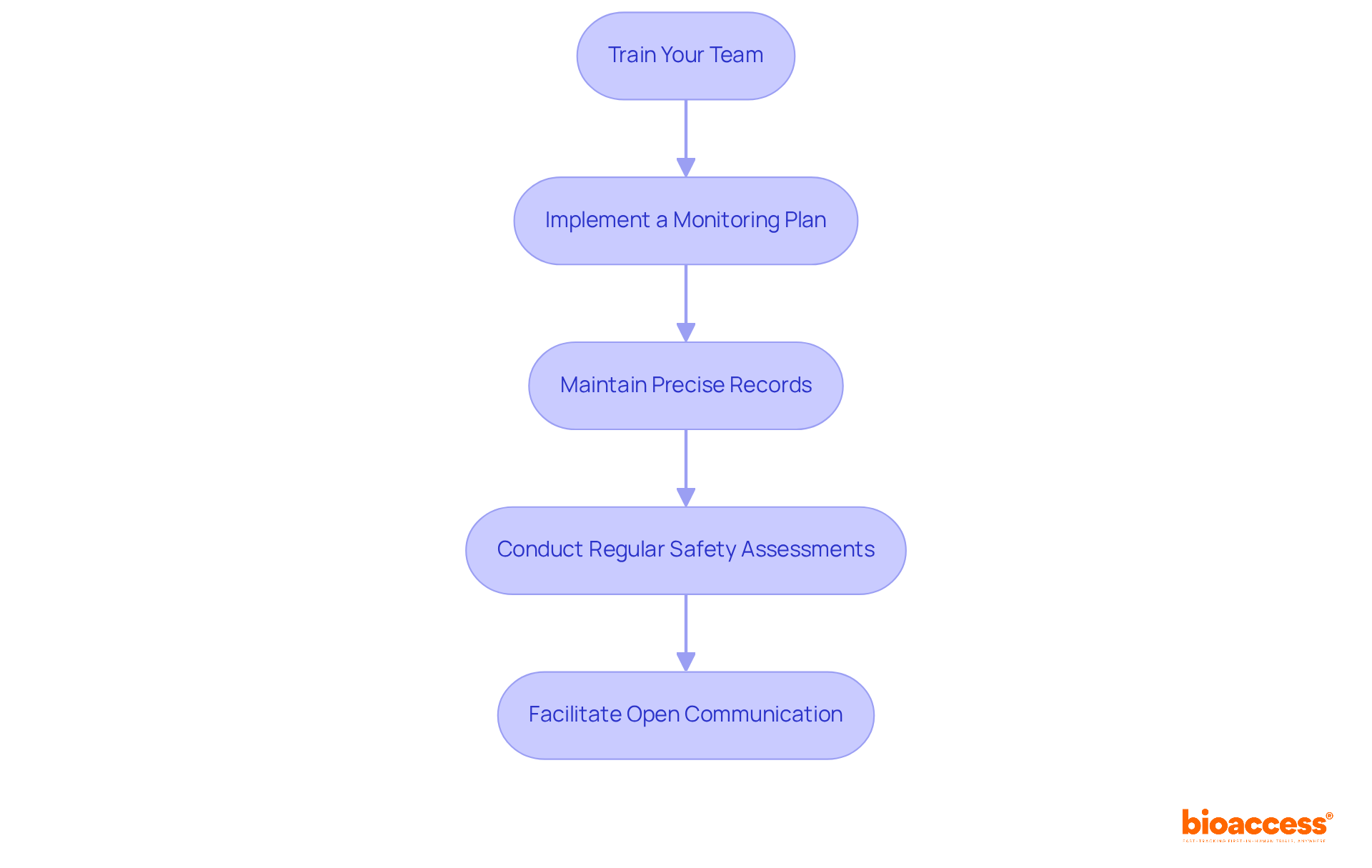
- Quality Assurance Processes: Investigate the CRO’s quality management systems to ensure adherence to high standards of data integrity and patient safety throughout the study process. This includes meticulous management of informed consent documentation, as required by local laws, to uphold ethical standards.
- Communication and Collaboration: Assess the CRO’s approach to communication and partnership. A CRO that promotes open dialogue and teamwork can greatly improve the study experience and results, ensuring alignment with your project objectives and compliance expectations.
By focusing on these criteria and leveraging insights from bioaccess®, sponsors can select a clinical research organization in Chile specializing in medical devices that meets compliance needs while enhancing the efficiency and effectiveness of their studies.
Explore Available Services and Support from CROs
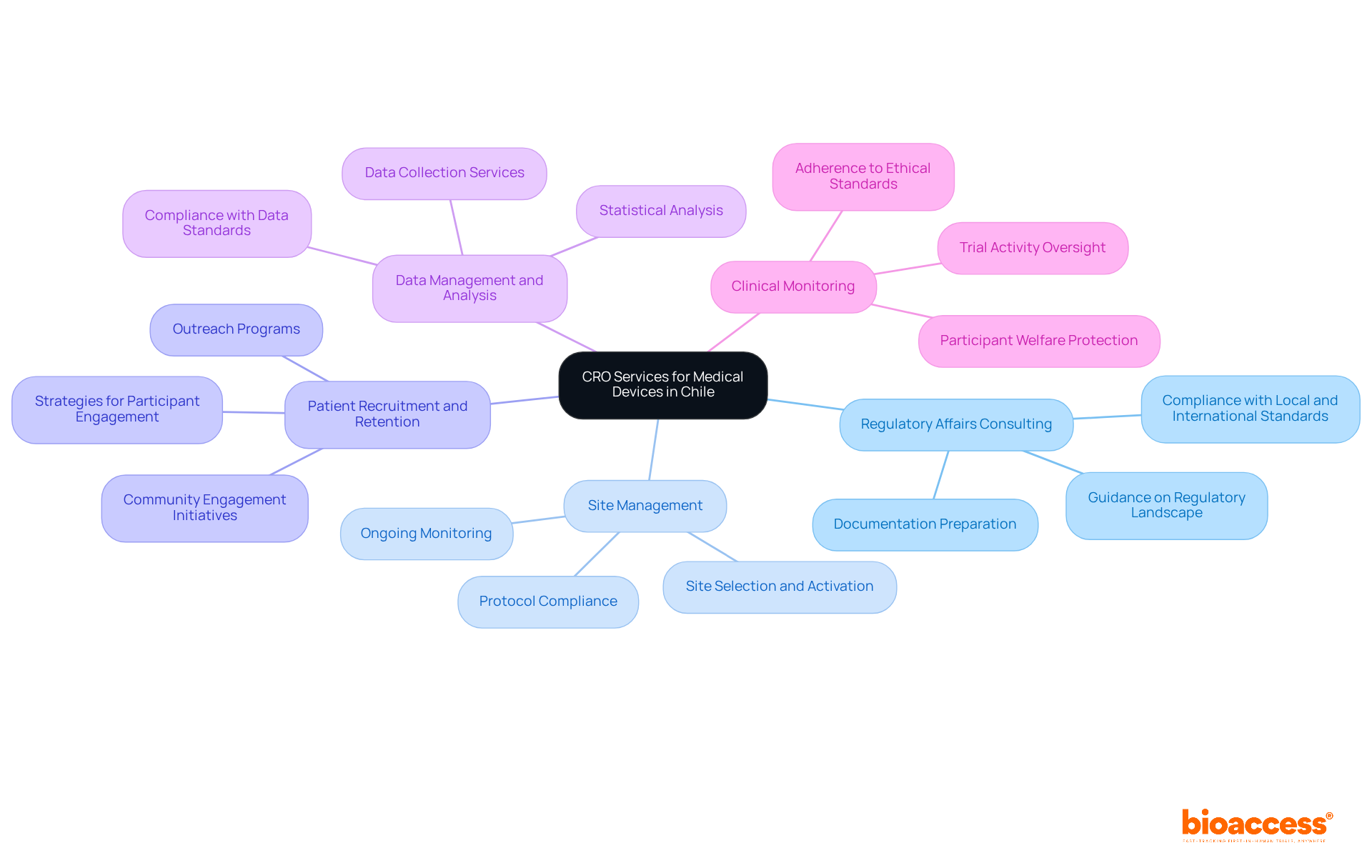
Navigating the complexities of clinical studies for medical devices in Chile necessitates the expertise of a clinical research organization Chile medical device for guidance and strategic collaboration. A clinical research organization in Chile focuses on providing an extensive range of services for medical device studies, ensuring adherence to regulations and efficiency throughout the process. Key services include:
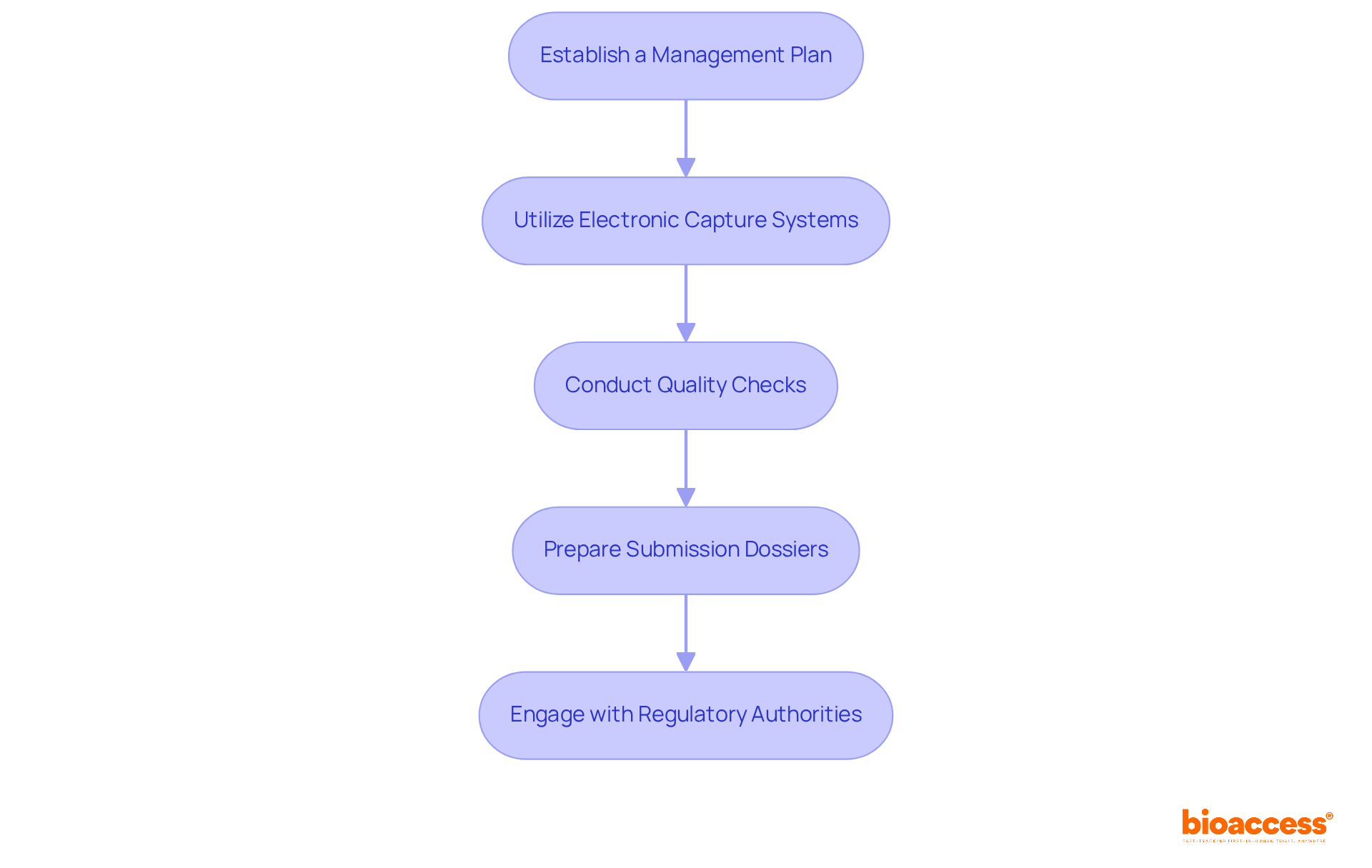
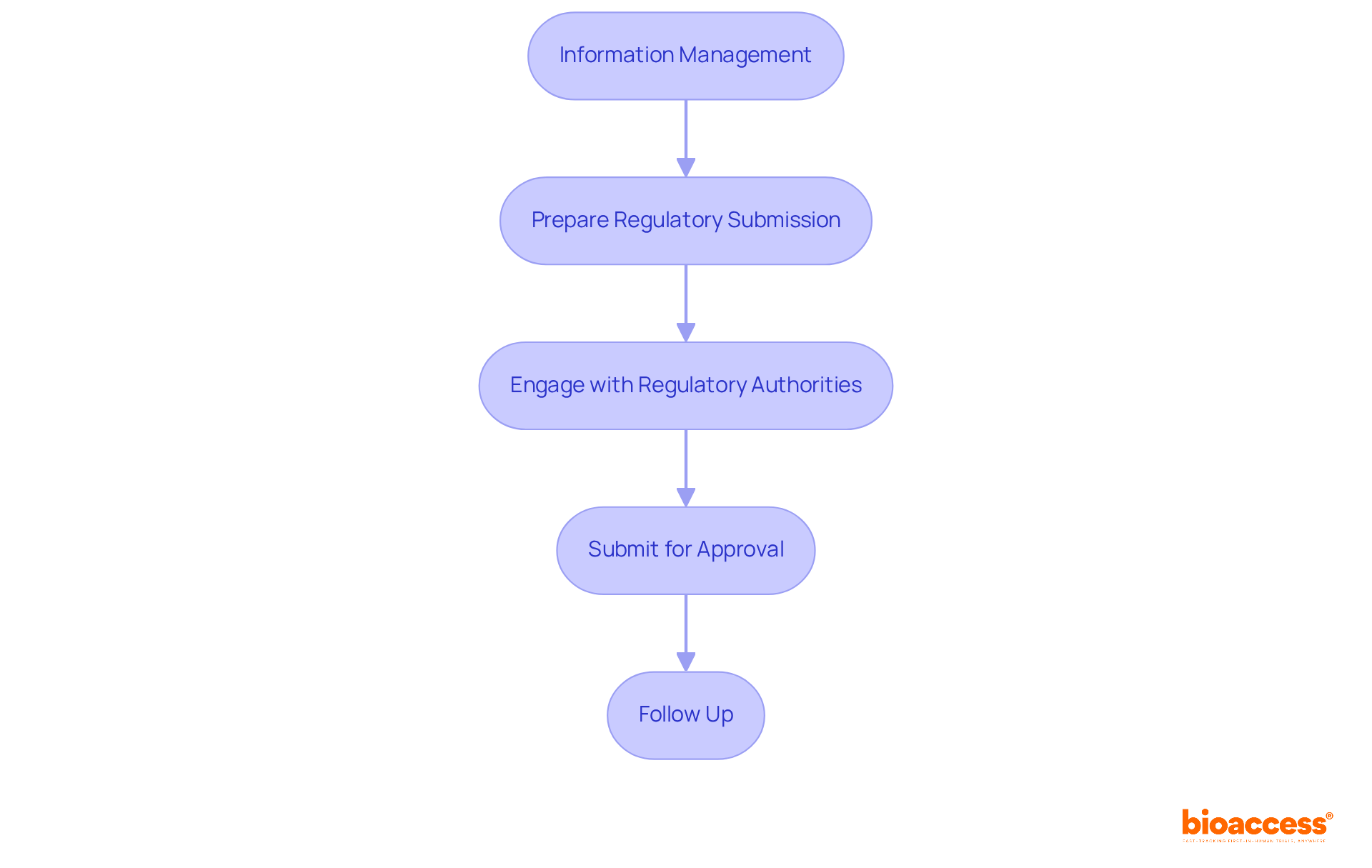
- Regulatory Affairs Consulting: Expert guidance in navigating the complex regulatory landscape, including meticulous preparation and submission of documentation to the Instituto de Salud Pública (ISP), ANVISA, INVIMA, and COFEPRIS. This guarantees compliance with local and international standards, essential for prompt approvals, particularly for First-in-Human (FIH) studies.
- Site Management: Effective oversight of research sites includes site selection, activation, and ongoing monitoring. This guarantees strict compliance with protocols and schedules, which is essential for preserving the integrity of the study.
- Patient Recruitment and Retention: Implementing robust strategies to identify and engage potential participants is essential. Outreach programs and community engagement initiatives improve recruitment efforts, tackling one of the major challenges in clinical studies.
- Data Management and Analysis: Comprehensive services for data collection, management, and statistical analysis ensure high-quality, reliable results that meet compliance standards. This is critical for the successful evaluation of medical devices.
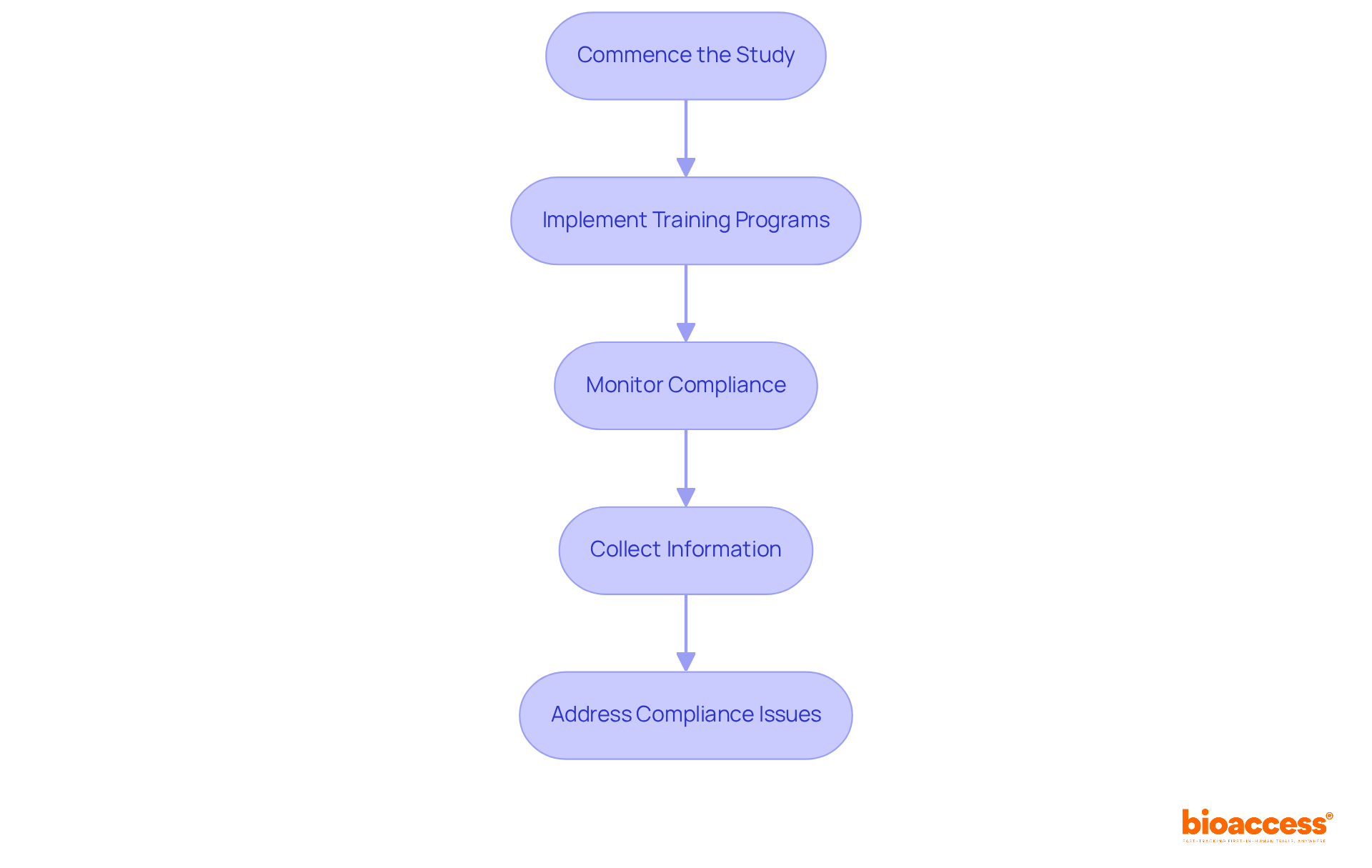
- Clinical Monitoring: Continuous oversight of trial activities ensures adherence to protocols, legal requirements, and ethical standards. This protects participant welfare and ensures data integrity, which is crucial in research trials.
Navigating the regulatory landscape in Chile can be a daunting task for many sponsors, often leading to delays and frustration. However, with the expertise of a clinical research organization Chile medical device, sponsors can significantly reduce these delays, ensuring a smoother path to market entry. The ISP typically assesses applications within 30 business days, and CROs optimize this process, utilizing their knowledge to improve the effectiveness of studies. By focusing on meticulous documentation and adherence to established guidelines, these organizations help sponsors navigate the certification and approval process effectively. Notably, the ISP has reduced bureaucratic delays in research approval by over 30%, further facilitating timely access to the market. This strategic advantage positions Latin America as an ideal region for early-stage studies, providing speed and cost efficiency that are crucial for MedTech startups. By leveraging the expertise of CROs, sponsors can not only overcome regulatory hurdles but also accelerate their journey to market success.
Initiate Partnership and Communication with Your Chosen CRO
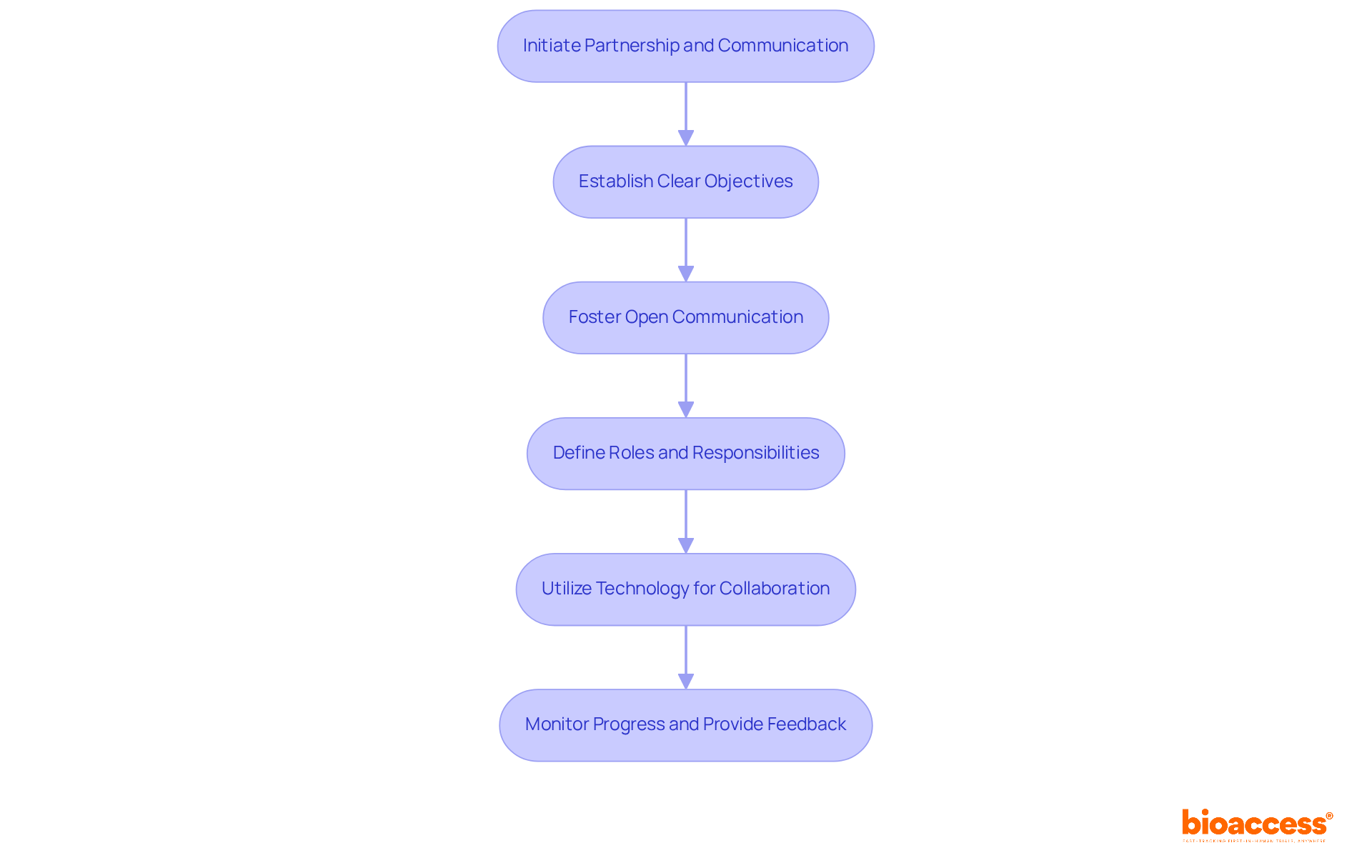
Choosing the right clinical research organization in Chile for a medical device is just the beginning; establishing a robust collaboration is essential for your clinical study’s success. Follow these steps:
- Establish Clear Objectives: Clearly define the goals and expectations for the trial. Are both your team and bioaccess® aligned on the project scope, timelines, and deliverables? Their expertise in First-in-Human (FIH) studies means you can tap into their knowledge of regulatory pathways, including INVIMA, ANVISA, and COFEPRIS, to set realistic objectives.
- Foster Open Communication: How often do you meet to discuss progress and challenges? Regular updates can make all the difference. Establishing open lines of communication with our organization helps foster trust and enhance problem-solving, particularly due to their dedication to swift ethics approvals in Latin America.
- Define Roles and Responsibilities: Make sure everyone knows their roles and responsibilities to keep things running smoothly. Their experienced team understands the urgency of medical innovation and can guide you effectively.
- Utilize Technology for Collaboration: Leverage project management tools and communication platforms to streamline collaboration. The organization utilizes advanced technologies to improve project efficiency and transparency, ensuring that all team members are aligned.
- Monitor Progress and Provide Feedback: Regularly assess the study’s progress and offer constructive feedback to bioaccess®. Their focus on early feasibility studies allows for ongoing evaluation and adjustments, ensuring that the trial remains on track and aligned with your objectives.
By prioritizing collaboration, you not only enhance the study’s efficiency but also pave the way for groundbreaking medical advancements.
Conclusion
Selecting the right clinical research organization (CRO) in Chile is not just important; it’s a strategic decision that can make or break medical device trials. Chile offers unique advantages – regulatory efficiency, cost-effectiveness, and a diverse patient population – that create fertile ground for successful early-stage studies. By understanding these benefits and the key criteria for choosing a CRO, sponsors can navigate the complexities of clinical research more effectively and accelerate their innovations to market.
The article highlights essential factors in selecting a CRO, including:
- Their experience with medical devices
- Regulatory knowledge
- Patient recruitment capabilities
- Quality assurance processes
- Communication strategies
These elements are crucial for ensuring compliance, enhancing the efficiency of studies, and ultimately achieving reliable outcomes. These insights guide sponsors in making informed decisions that align with their project goals and regulatory requirements.
In conclusion, by embracing these strategic opportunities, companies can ensure their innovations not only reach the market but also set new standards in healthcare. By prioritizing the selection of a capable CRO and fostering effective collaboration, sponsors can streamline their research processes and contribute to groundbreaking advancements in healthcare. Embracing these opportunities in Latin America positions companies for competitive advantage, ensuring that their innovations reach the market swiftly and efficiently.
Frequently Asked Questions
What are the key advantages of conducting clinical trials in Chile?
Chile offers regulatory efficiency with approval times of 30 to 90 days, cost-effectiveness with study costs up to 40% lower than in the U.S., a diverse patient population for robust data collection, a strong healthcare infrastructure, and a supportive oversight environment from the Instituto de Salud Pública (ISP).
How does the regulatory landscape in Chile benefit clinical trials?
The regulatory landscape in Chile is streamlined, with an average assessment period for medical studies of around 30 days, allowing for quicker market entry of new medical devices and therapies, especially beneficial for first-in-human phases.
What is the cost advantage of conducting studies in Chile compared to the United States?
Conducting studies in Chile can be up to 40% less costly than in the U.S., with costs related to EDC setup, data cleaning, query management, and biostatistical analysis being 40-50% lower in Latin America.
Why is the diverse patient population in Chile important for clinical trials?
The demographic variety in Chile allows for comprehensive data collection on the effectiveness and safety of medical devices across different population groups, enhancing the robustness and applicability of research findings.
What does Chile’s healthcare infrastructure provide for clinical trials?
Chile boasts a solid network of medical sites and skilled health professionals, ensuring high-quality data collection and patient care, with retention rates exceeding 85%, which is above global averages.
How does the Instituto de Salud Pública (ISP) contribute to the clinical trial process in Chile?
The ISP serves as the primary oversight authority, known for its supportive approach to research involving human subjects, facilitating an environment conducive to innovation and effective navigation of the approval process.
What compliance requirements must be met for conducting clinical trials in Chile?
Comprehensive documentation preparation is essential to ensure compliance with local regulations, including ICH-GCP standards, which are critical for navigating the approval process in Chile.
How can companies leverage Chile’s strengths for early-stage studies?
By utilizing Chile’s administrative efficiency, cost benefits, diverse demographics, and robust infrastructure, companies can enhance their research outcomes and position themselves for success in the competitive landscape of early-stage clinical trials.
List of Sources
- Understand the Benefits of Conducting Trials in Chile
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Clinical Research and Drug Development Case Studies | Novotech CRO (https://novotech-cro.com/case-studies)
- Costs & Timelines | bioaccess® (https://bioaccessla.com/costs-and-timelines)
- Clinical Trial Costs: Latin America vs US/EU | 2026 Benchmark | bioaccess® (https://bioaccessla.com/blog/clinical-trial-costs-latin-america-vs-us-eu-benchmark)
- First-in-Human Medical Device Trial in Chile | bioaccess® (https://bioaccessla.com/blog/first-in-human-medical-device-trial-chile)
- Evaluate Key Criteria for Selecting a CRO
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Medical Device CRO for EU MDR Clinical Investigations – Former Notified Body Expertise (https://eclevarmedtech.com/en/selecting-a-medical-device-diagnostic-clinical-research-organization)
- Key Factors in CRO Selection | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/key-factors-cro-selection)
- How To Choose The Right CRO For Clinical Trial… | bioaccess® (https://bioaccessla.com/blog/how-to-choose-the-right-cro-for-clinical-trials-in-latin-america-2026-guide)
- Key Factors in CRO Selection | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/key-factors-cro-selection-0)
- Explore Available Services and Support from CROs
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- 3 quotes about regulatory affairs that will make you smile | Regulatory Affairs Professionals Society (RAPS) (https://linkedin.com/posts/regulatory-affairs-professionals-society-raps-_3-quotes-about-regulatory-affairs-that-will-activity-7370109430329409536-T7xL)
- Clinical Research and Drug Development Case Studies | Novotech CRO (https://novotech-cro.com/case-studies)
- Initiate Partnership and Communication with Your Chosen CRO
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)