Introduction
The Humanitarian Device Exemption (HDE) program offers an expedited regulatory pathway for medical devices aimed at treating or diagnosing rare diseases or conditions, which affect fewer than 8,000 individuals in the United States annually. This program is pivotal in facilitating the availability of innovative medical solutions to patients with limited treatment options by allowing device manufacturers to bring their products to market with reduced clinical trial requirements. While streamlining the regulatory process, the program ensures that devices still undergo rigorous evaluation to ensure compliance with quality standards.
This article explores the criteria for HDE eligibility, the key elements of an HDE application, IRB approval and oversight requirements, emergency use of HDE devices, profit and use restrictions, the annual distribution number for profitable sales, a case study on navigating the HDE application process, and the challenges and considerations for HDE devices. Through these insights, we gain a comprehensive understanding of the complex landscape surrounding HDE devices and the importance of balancing innovation with patient safety.
Definition and Criteria for HUDs
To qualify for the , healthcare instruments must meet strict criteria. They should be designed for the diagnosis or treatment of conditions that affect fewer than 8,000 individuals annually in the United States. Importantly, there should be a lack of any comparable product on the market that deals with the same health problem. A case in point is the , which includes components like the Impella Automated Controller (AIC) for , and its web-based user portal and hardware for remote monitoring. The system’s capacity to provide timely, qualifies it as an instrument under Section 201(h) of the Act, highlighting the HDE’s emphasis on innovative solutions for unmet health requirements. Furthermore, as the landscape of healthcare equipment evolves, the FDA’s classification, coding systems, and guidance documents—such as those detailing the —provide essential regulatory clarity to support the advancement and proper use of .
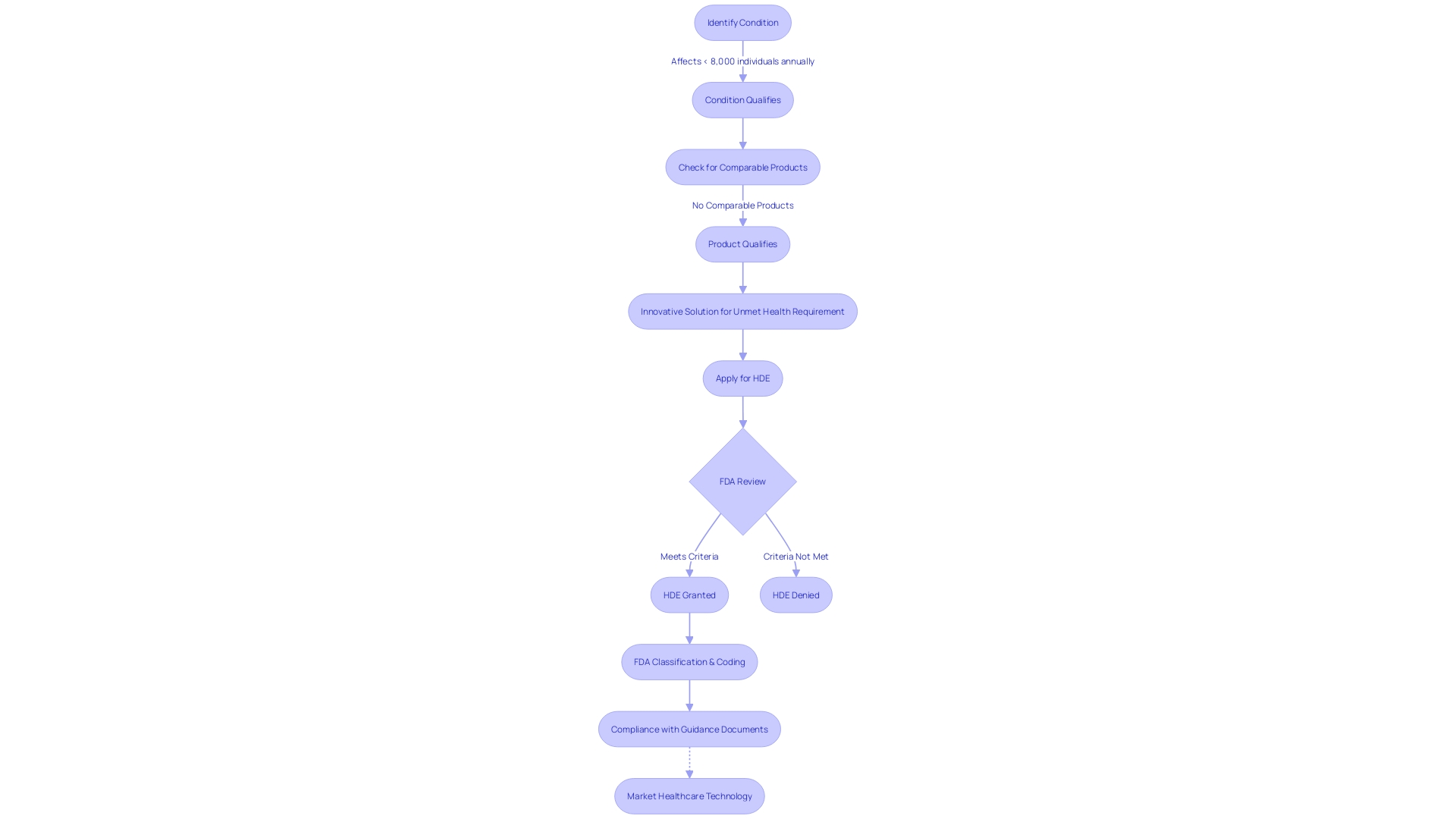
Humanitarian Device Exemption (HDE) Program Overview
The humanitarian Device Exemption (HDE) program offers an for healthcare instruments aimed at treating or diagnosing rare diseases or conditions, which affect fewer than 8,000 individuals in the United States annually. This program is crucial in enabling the availability of for patients with limited treatment options by permitting manufacturers to bring their products to market with reduced . This not only accelerates the advancement of specialized medical equipment but also aligns with the to ensure the safety, effectiveness, and security of . It’s crucial to highlight that while the HDE program simplifies the regulatory process, it does not compromise on the quality standards that products must meet. These instruments, despite being for niche applications, still undergo rigorous evaluation to ensure they comply with current (CGMP) requirements, as mandated by the FDA’s quality system regulation. The program highlights the FDA’s dedication to promoting the progress of while guaranteeing patient safety and the efficiency of medical equipment.
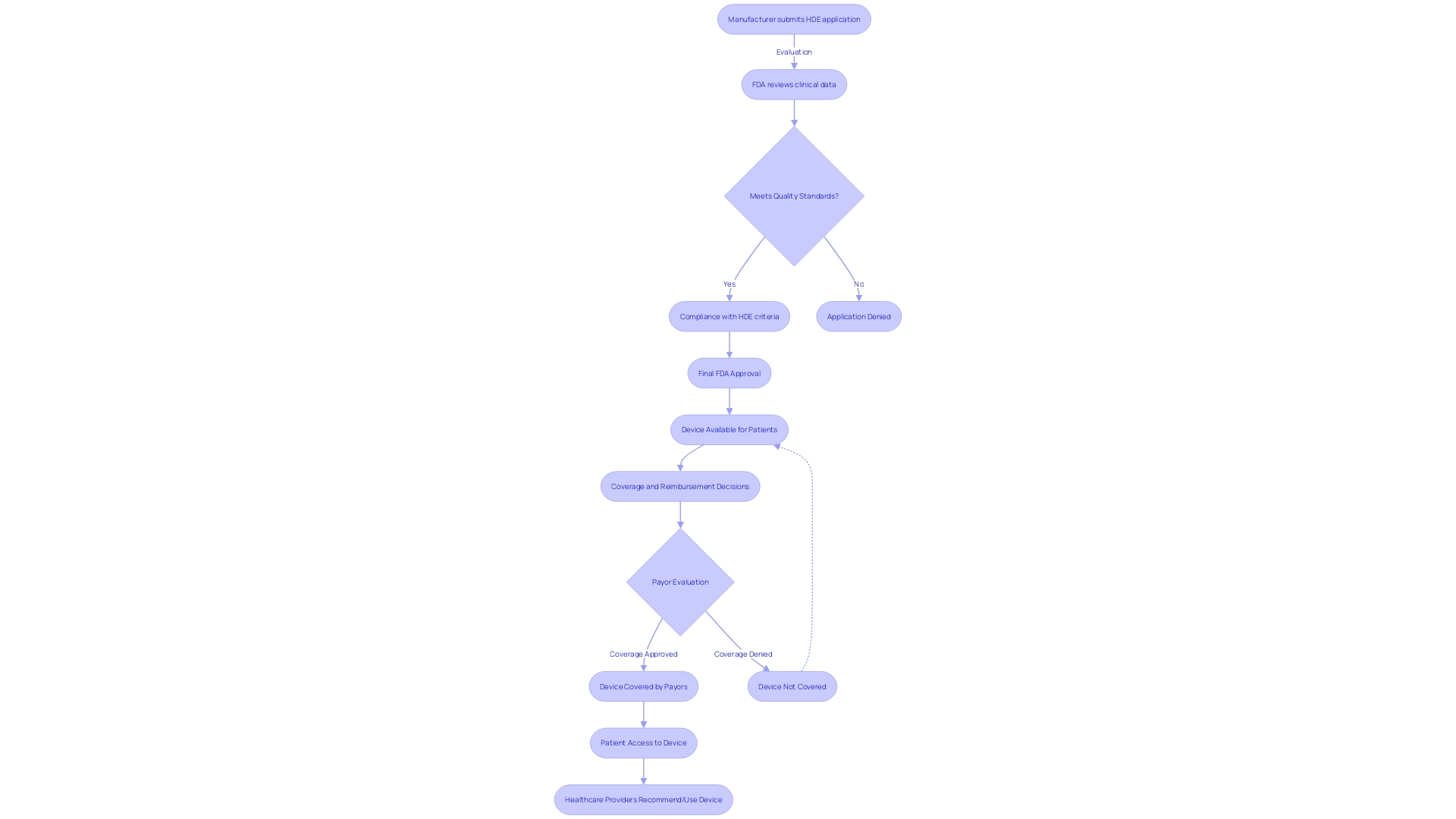
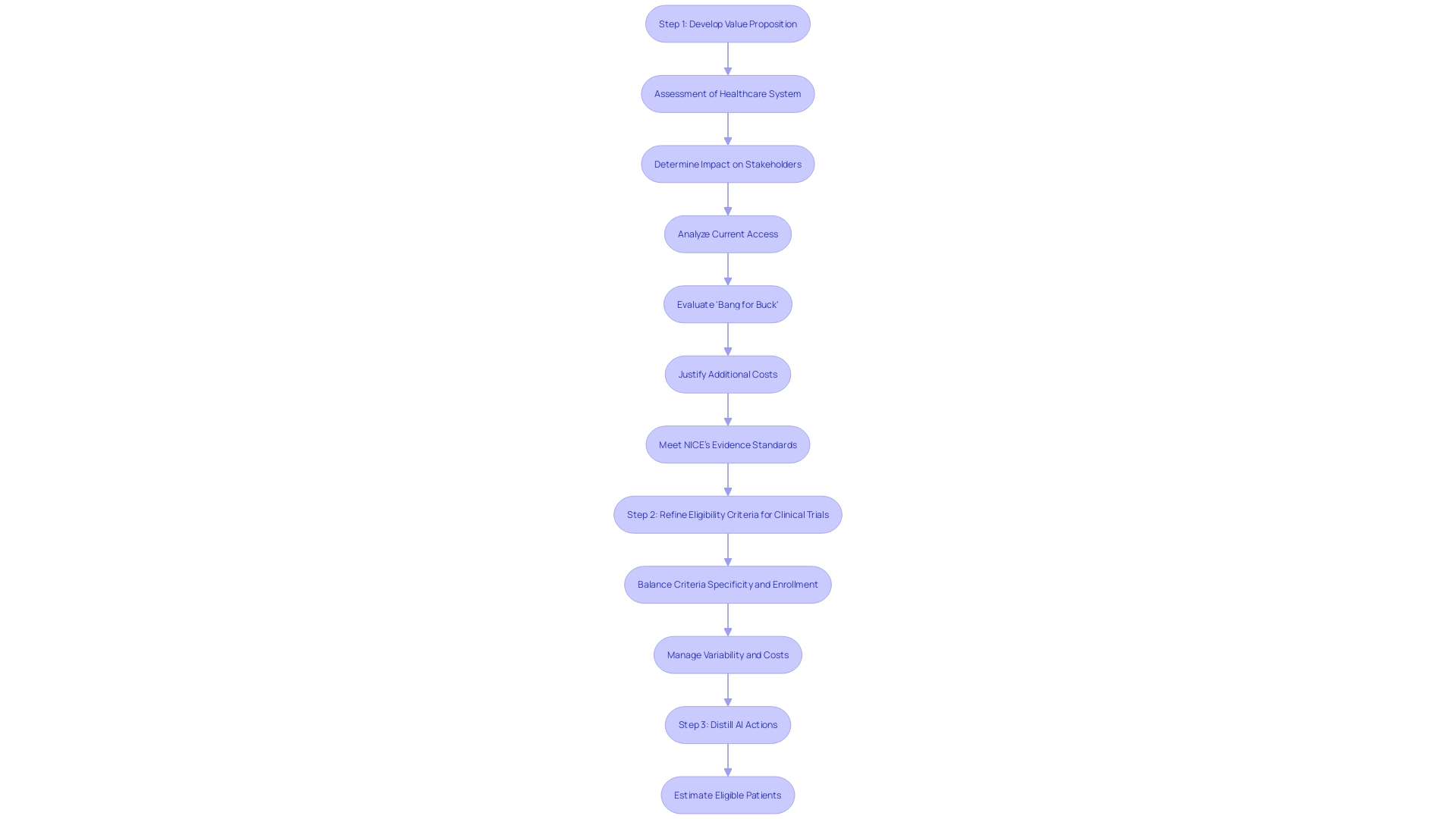
Key Elements of an HDE Application
An application for (HDE) requires a comprehensive compilation of details to show the safety and potential benefits of the equipment. The application should include a comprehensive description of the equipment, accompanied by visual aids such as diagrams and specifications if applicable. It must outline the functional components or ingredients of the apparatus, especially if it comprises multiple physical entities.
The application also needs to encapsulate the conditions or diseases the apparatus intends to diagnose, treat, or mitigate, clarifying the apparatus’s impact on the body’s structure or function. This should be detailed along with a intended for its use. Moreover, the application should mention any for related legal medical instruments.
, which confirms the safety and probable benefit of the apparatus, is a critical component of the submission. The data should be robust, possibly including real-time feeds or shared health information system data, as well as any modernized data exchange practices that contribute to public health, a crucial consideration as noted by Dr. Liz Kwo, Chief Commercial Officer at Everly Health.
Furthermore, the application should include a summary of alternative practices, offering a framework against which the benefits of the apparatus can be evaluated. A plan for ongoing is essential to ensure the continued safety and effectiveness of the product after it enters the market. This plan should be efficient and secure to facilitate timely and accurate public health data exchange, aligning with the innovative approaches to data management across government and healthcare systems.
IRB Approval and Oversight Requirements
Securing (IRB) approval is the first critical step in the journey of a (HDE) application. The pivotal role of the IRB cannot be overstated as it safeguards the ethical treatment of human subjects in clinical research. Apart from this, the FDA remains watchful, with manufacturers obligated to adhere to a collection of reporting and monitoring requirements to guarantee the ongoing safety and efficacy of these products.
The governing environment for healthcare equipment is intricate and multi-tiered, with the into three categories according to the potential harm they may cause to patients. High-risk class three products, such as life-sustaining implantables, are subject to the most stringent regulatory scrutiny and lengthy approval processes, reflecting their critical nature. Despite the robust regulatory framework, challenges such as navigating the nuances of different classifications and registration pathways—be it 510(k), PMA, or De Novo—persist.
Moreover, the FDA’s role extends to overseeing the myriad ethical, legal, and social dimensions emerging from the technology’s use. Through , the agency not only examines the governance of these technologies but also the market incentives and intellectual property considerations that shape their evolution. These real-world examples highlight the intricacy of ethical dilemmas encountered in today’s healthcare technology industry.
In the pursuit of a cross-sectoral governance framework, the FDA’s responsibilities also involve ensuring public health by confirming the safety, security, and efficacy of healthcare equipment. This commitment is reflected in efforts to simplify , a movement that gained momentum during the COVID-19 pandemic, to address urgent healthcare needs swiftly. As the industry evolves, particularly in digital health and personalized medicine, regulatory processes are being recalibrated to keep pace with innovation while safeguarding patient safety.
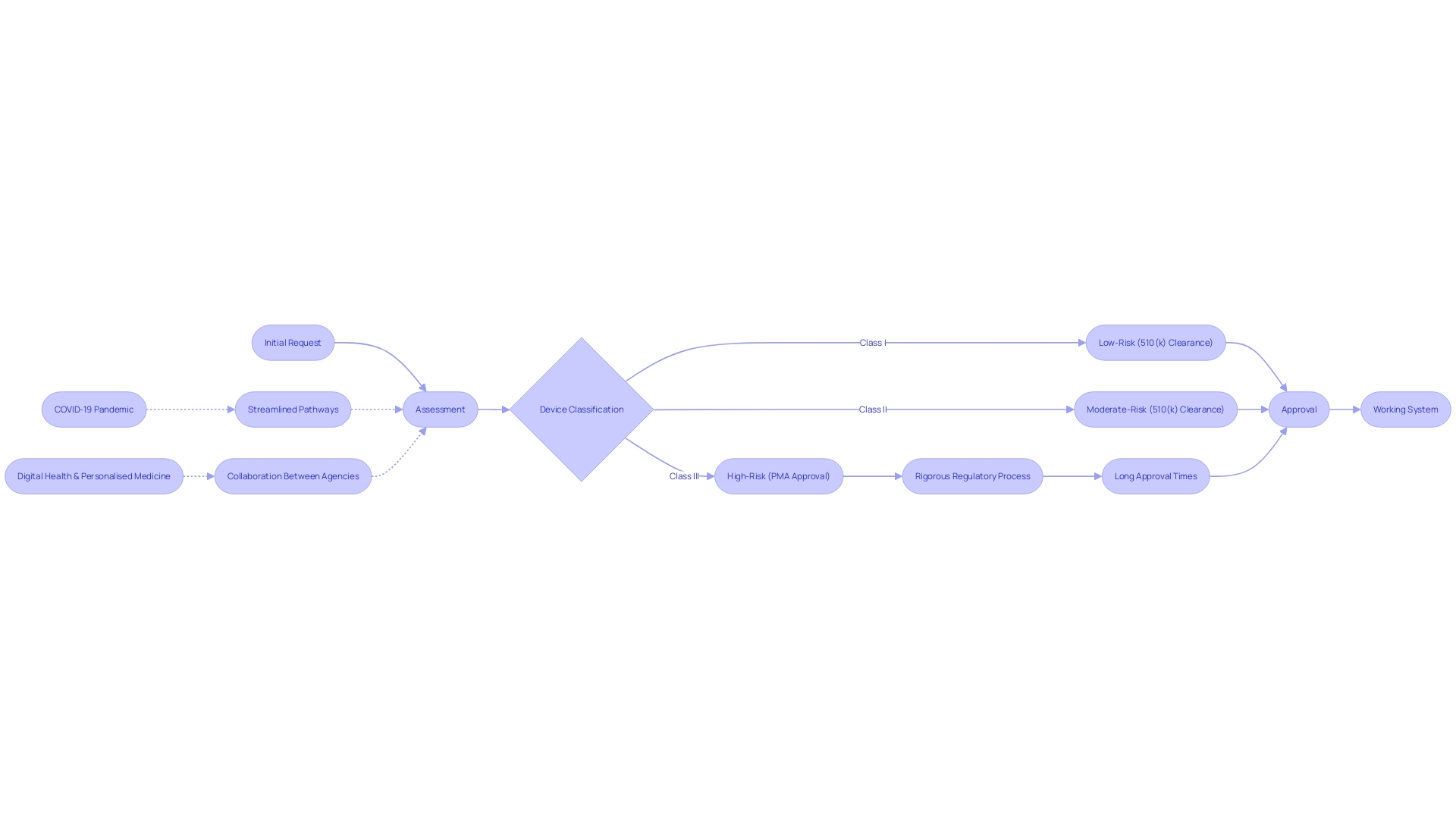
 Flowchart: Journey of a Humanitarian Device Exemption (HDE) Application](https://tely.blob.core.windows.net/telyai/flowchart-journey-of-a-humanitarian-device-exemption-hde-application.jpg.jpg)
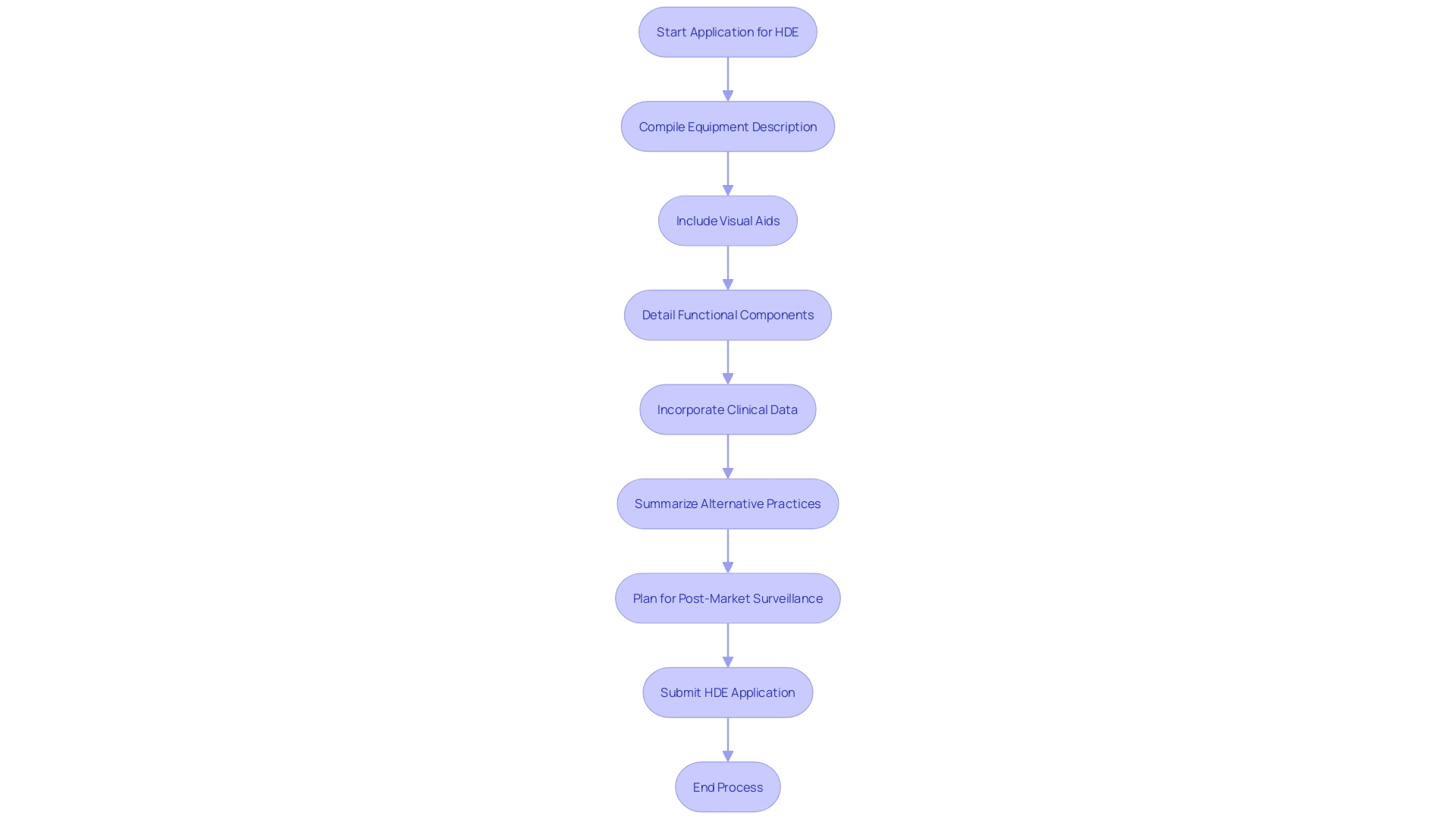
Emergency Use of HUDs
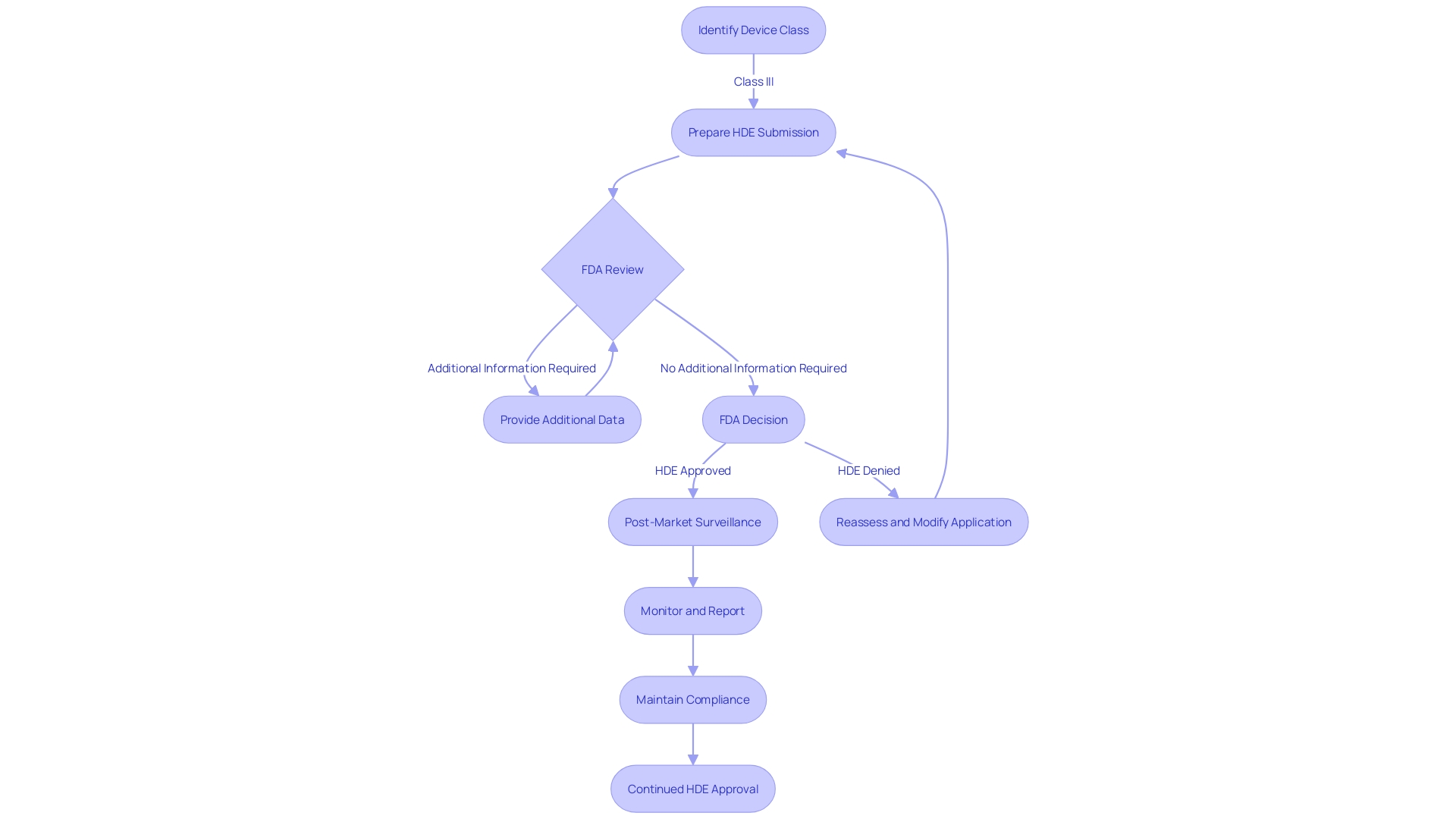
In urgent scenarios where patients face serious or life-threatening conditions and no alternative treatments are available, the of (HDE) devices. This crucial provision ensures that healthcare practitioners can access and utilize to provide the best possible care when every second counts. To start the , healthcare providers must comply with strict conditions established by to ensure patient safety and tool efficacy. As an example, the advanced Impella Connect System, created for crucial cardiac support, is an instance of an HDE apparatus that enables remote monitoring of the heart’s performance, offering essential, timely information to healthcare professionals. It’s crucial to mention that , as a division of the U.S. Department of Health and Human Services, is dedicated to ensuring the safety and efficacy of such healthcare instruments. As regulations and product information can vary widely from country to country, it’s crucial for healthcare providers to follow the guidelines specific to the United States. Healthcare professionals and consumers are encouraged to report any issues encountered with healthcare tools under an EUA to ’s MedWatch program. These reports are invaluable for the continuous assessment of equipment safety. for manufacturers under an EUA is also mandated, with specific requirements detailed within each EUA, following the regulatory guidelines outlined in 21 CFR Part 803.
 Devices Flowchart: Emergency Use of Humanitarian Device Exemption (HDE) Devices](https://tely.blob.core.windows.net/telyai/flowchart-emergency-use-of-humanitarian-device-exemption-hde-devices.jpg.jpg)
Profit and Use Restrictions for HDE Devices
Humanitarian Exemptions (HDE) are different from traditional medical tools in that their distribution and pricing are strictly regulated. In particular, HDE products cannot be sold at a price that exceeds the costs associated with their research, development, and fabrication. Moreover, healthcare facilities or professionals must either obtain an (IDE) or have an approved HDE application to gain access to these instruments. This regulatory framework ensures that such technology is used ethically and in a manner that prioritizes patient care over profit.
The concept of Health Institution Exemptions (HIE), also referred to as ‘in-house manufacturing,’ allows healthcare organizations to create and use customized to specific clinical needs without complying with the complete range of regulatory requirements that commercial manufacturers encounter. This tradition developed over time to enable clinicians to invent and modify tools within the boundaries of their practice. HIEs are applicable within entities primarily engaged in patient care, such as hospitals, public health institutes, and laboratories, but do not extend to wellness-focused businesses like spas or gyms.
In accordance with His, the MHRA Northern Ireland guidelines allow the transfer of between health institutions, each of which must independently assert their exemption. It’s significant to observe, however, that such products cannot be sold or transferred to other legal entities. Furthermore, any healthcare device—even those produced under HIE—must comply with the as indicated in Annex I of the MDR. While the specific criteria for an ‘appropriate’ may vary based on the instrument and its intended use, ISO 13485 is generally considered a suitable standard.
While the pricing and distribution of HDE instruments are controlled, the broader is influenced by a myriad of factors including therapeutic area, procedural complexity, reimbursement levels, and the competitive landscape. Market intelligence from GlobalData reveals that pricing strategies, sales targets, and regional factors also significantly impact the final selling price of . For instance, large healthcare institutions may negotiate discounts based on volume, which can lead to price variations between hospital settings and other healthcare facilities. These insights highlight the intricacy of pricing for and the significance of strategic financial planning within the healthcare sector.
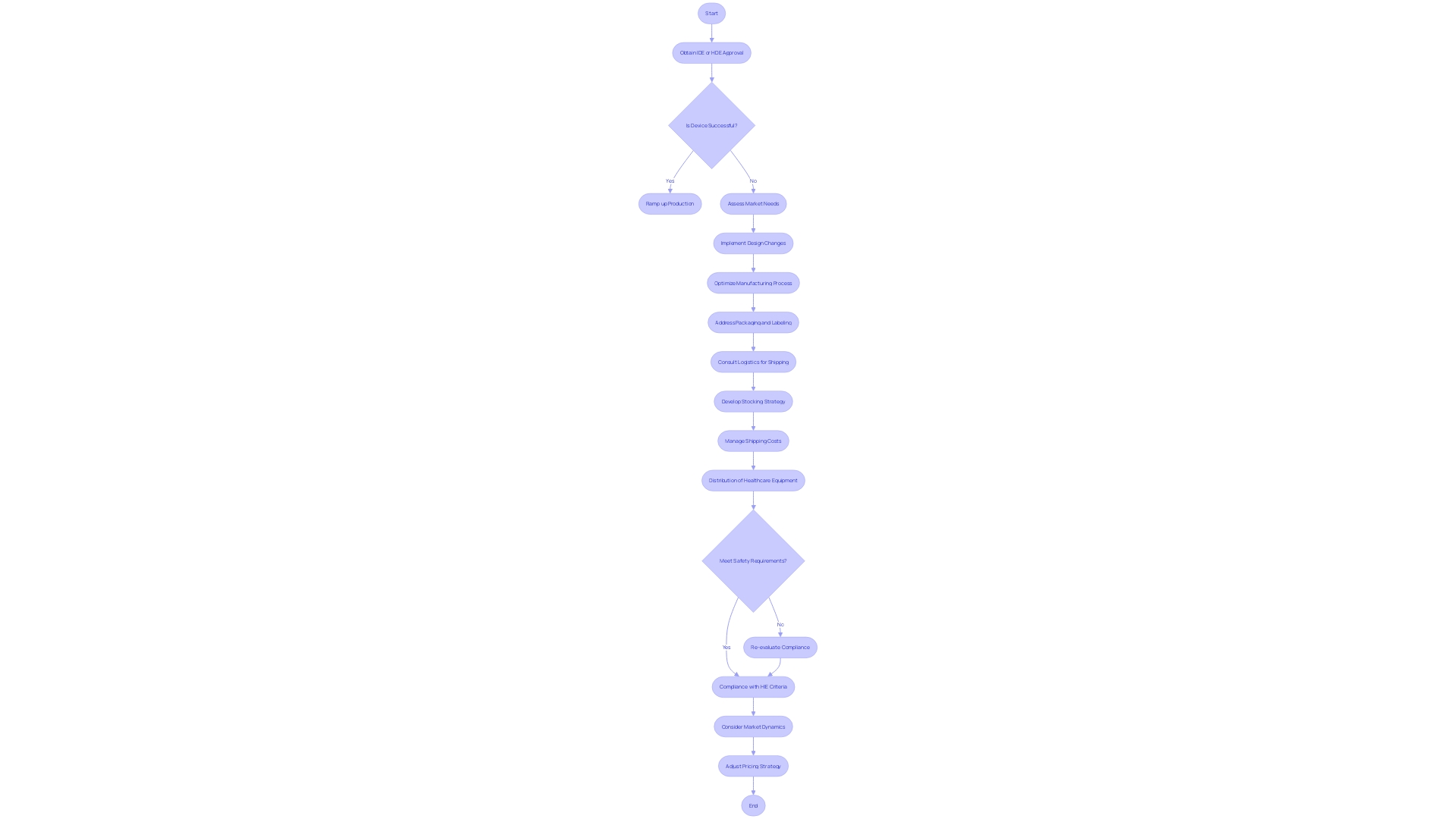
Annual Distribution Number (ADN) for Profitable Sales
The Food and Drug Administration (FDA) implements proactive measures to oversee the distribution of , particularly those that come under the (HDE). When a piece of equipment under HDE becomes profitable, the FDA determines an (ADN), which caps the quantity of equipment that can be distributed annually. Adherence to this cap is crucial for manufacturers to retain their HDE status. The creation of the ADN is impacted by a range of factors including the intricacy of the procedure the tool is designed for, the level of market competition, and the lifecycle of the product. Furthermore, the market dynamics of healthcare equipment are greatly influenced by and discount strategies, similar to those implemented by companies such as Stryker, which are based on internal strategies and sales targets. These factors, together with , emphasize the intricacy of the .
Case Study: Navigating the HDE Application Process
Exploring the journey of healthcare innovation approval is a complex process that highlights the challenges and successes of innovation in healthcare. One illustrative example is the pathway to obtaining . Through the lens of a case study, we delve into the experiences of a company as they navigate the labyrinth of . The initial steps involve a request submitted by a clinician or department to the Digital Service Team, who conducts a thorough assessment to determine if the technology is secure, compliant, and not redundant within the trust. An interesting revelation from this process, as noted by an NHS representative, is the discovery of existing technologies within the organization which were previously unknown, thus underscoring the importance of internal communication and resource awareness.
When considering the wider healthcare framework, it is essential to acknowledge the significant function of , especially in rural regions where populations face substantial health challenges in affluent nations. For instance, the United States stands 29th among OECD nations, with life expectancy at its lowest in two decades, highlighting the urgent need for quality healthcare solutions. This fact speeds up the importance of for instruments that can deliver lifesaving care to these underserved communities.
The case study further reveals the multi-staged journey of a project from conception to a working system. Soeren, a project leader, shares their strategic approach which included initial analysis, design customization, and the development of alternatives, demonstrating a comprehensive and tailored pathway to innovation.
Furthermore, the dynamic advancements in the industry, like the development of Renewstable® hydrogen power plants and digital twin technologies, demonstrate the progress in health and technological fields. Such innovations hold the potential to revolutionize energy production and maritime operations, respectively, showcasing the intersection of healthcare and technology.
In terms of regulatory understanding, the FDA classifies instruments into three categories, with class three instruments requiring the most rigorous approval due to their critical role in sustaining life. Roughly 10% of healthcare equipment belong to this category, emphasizing the intricacy of HDE approval for tools that are vital for patient care.
The emphasis on , particularly considering the COVID-19 pandemic, highlights the changing approval landscape for . Organizations such as the FDA and EMA are leading the way in these transformations, impacting the time it takes to approve products based on their intricacy and the health conditions they target. The cooperative endeavors between regulatory agencies and industry stakeholders are crucial in accelerating the availability of technologies that meet unfulfilled healthcare requirements, with a notable emphasis on digital well-being and individualized treatment.
Challenges and Considerations for HDE Devices
Securing a Humanitarian Device Exemption (HDE) demands meticulous attention to a number of stringent requirements. One must consider the comprehensive collection of , adept navigation through the intricate , and the establishment of robust mechanisms. To illustrate the complexities involved, consider the comparison to California utilities’ efforts in mitigating . They encountered uncertainties in risk levels, costs, and effectiveness of mitigation strategies, similar to the uncertainties manufacturers face when seeking .
For instance, as expressed by Dr. Liz Kwo of Everly Health Solutions, the significance of early detection in healthcare demonstrates the crucial requirement for thorough of healthcare tools to safeguard public health. Likewise, the instance of Medtronic’s Symplicity blood pressure procedure, which obtained approval in more than 70 countries, illustrates the worldwide magnitude at which regulatory processes for healthcare equipment operate and the importance of acquiring in broadening a device’s scope.
The healthcare equipment sector, as emphasized by the World Health Organization, covers more than 10,000 varieties of appliances. This diversity requires a nuanced understanding of both human aspects and equipment factors. Taking into account the classification of healthcare tools by the FDA into three categories based on risk, category three tools—which encompass life-sustaining implants such as pacemakers—demand a particularly stringent procedure due to their high stakes.
The FDA’s main responsibility is to evaluate the safety and efficiency of healthcare equipment for the US market, however, the information submitted for FDA approval may not match the information payors require for coverage determinations, which can result in possible delays or rejections in accessing the equipment, as indicated by the FDA. This underscores the need for strategic planning and comprehensive data analysis to meet the criteria for .
To conclude, the pursuit of is a challenging yet crucial endeavor for , demanding a multifaceted approach that considers , regulatory navigation, post-market oversight, and the intricacies of medical device diversity.
Conclusion
In conclusion, the Humanitarian Device Exemption (HDE) program provides an expedited regulatory pathway for medical devices treating rare diseases or conditions. It allows manufacturers to bring innovative products to market with reduced clinical trial requirements while ensuring compliance with quality standards. The key elements of an HDE application include a comprehensive device description, clinical data, alternative practices, and post-market surveillance plans.
Securing Institutional Review Board (IRB) approval is crucial for ethical treatment of human subjects in clinical research. In urgent medical scenarios, the FDA authorizes the emergency use of HDE devices, ensuring timely access to advanced medical solutions. HDE devices are subject to profit and use restrictions, with pricing strictly regulated to prioritize patient care over profit.
Navigating the HDE application process requires assessments, communication, and strategic planning. The evolving regulatory landscape, particularly during the COVID-19 pandemic, emphasizes the need for efficient approval processes to address urgent medical needs swiftly.
Securing HDE approval presents challenges that demand attention to clinical evidence, regulatory navigation, and post-market surveillance. The complex landscape of medical devices, the significance of class three devices, and the need for comprehensive data analysis contribute to the complexity of the process.
In summary, the HDE program balances innovation and patient safety, enabling the availability of innovative medical solutions for rare diseases or conditions. Its criteria, application process, oversight requirements, emergency use provisions, and profit and use restrictions provide a comprehensive understanding of the complex landscape surrounding HDE devices.
Frequently Asked Questions
What is the Humanitarian Device Exemption (HDE) program?
The HDE program is an expedited regulatory pathway established by the FDA for healthcare instruments designed to diagnose or treat rare diseases or conditions affecting fewer than 8,000 individuals annually in the U.S. It enables faster access to innovative solutions for patients with limited treatment options.
What criteria must be met for a device to qualify for the HDE program?
To qualify, a device must be intended for conditions affecting fewer than 8,000 individuals annually and must not have any comparable products available on the market. Additionally, the device must meet strict safety and effectiveness standards.
Can you give an example of a device approved under the HDE program?
An example is the Impella Connect System, which provides critical care heart support. It includes components like the Impella Automated Controller (AIC) and offers remote monitoring to provide timely, patient-specific notifications.
What are the requirements for submitting an HDE application?
An HDE application must include: a detailed description of the device and its components, information about the conditions it treats, clinical data confirming safety and probable benefit, a summary of alternative treatments, and a plan for ongoing post-market surveillance.
What is the role of the Institutional Review Board (IRB) in the HDE application process?
The IRB is responsible for ensuring the ethical treatment of human subjects in clinical research. Securing IRB approval is a critical first step in the HDE application process.
How does the FDA classify healthcare devices?
The FDA classifies devices into three categories based on the potential harm they may cause, with Class III devices requiring the most stringent regulatory scrutiny. Class III devices typically include life-sustaining implants.
What happens if a device under HDE becomes profitable?
If an HDE device becomes profitable, the FDA determines an Annual Distribution Number (ADN), which limits the quantity that can be distributed each year. Compliance with this cap is essential for manufacturers to maintain their HDE status.
What is the difference between HDE and traditional medical devices?
HDE devices have stricter distribution and pricing regulations, ensuring they are not sold for more than the costs related to their research and development. Access to HDE devices also requires either an Investigational Device Exemption (IDE) or an approved HDE application.
Can healthcare institutions create and use customized devices?
Yes, through Health Institution Exemptions (HIE), healthcare organizations can produce customized devices for specific clinical needs without adhering to the full range of regulatory requirements imposed on commercial manufacturers.
What is the FDA’s role in ensuring the safety and efficacy of HDE devices?
The FDA oversees the approval process, ensuring that all devices meet safety and effectiveness standards. The agency also monitors post-market performance and requires ongoing reporting from manufacturers.
How can healthcare providers report issues with HDE devices?
Healthcare professionals and consumers can report any problems encountered with HDE devices to the FDA’s MedWatch program, which is essential for ongoing safety assessments.
What challenges do manufacturers face when seeking HDE approval?
Manufacturers must navigate a complex regulatory landscape, provide comprehensive clinical evidence, and establish robust post-market surveillance mechanisms to ensure compliance with FDA standards.
List of Sources
- Definition and Criteria for HUDs
- ecfr.gov (https://ecfr.gov/current/title-21/chapter-I/subchapter-H/part-807/subpart-A)
- fda.gov (https://fda.gov/about-fda/cdrh-innovation/medical-device-coverage-initiatives-connecting-payors-payor-communication-task-force)
- medicaldevice-network.com (https://medicaldevice-network.com/news/everly-health-at-home-test-kidney-disease)
- medicaldevice-network.com (https://medicaldevice-network.com/news/perfuze-enrols-first-patient-in-stroke-trial-with-its-millipede-system)
- medicaldevice-network.com (https://medicaldevice-network.com/news/recor-medical-hypertension-treatment-system)
- ecfr.gov (https://ecfr.gov/current/title-21/chapter-I/subchapter-H/part-807/subpart-D)
- medtechintelligence.com (https://medtechintelligence.com/feature_article/fda-cleared-samd-by-the-numbers)
- fda.gov (https://fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/abiomed-inc-663150-09192023)
- greenlight.guru (https://greenlight.guru/blog/bringing-a-self-funded-medical-device-to-market)
- federalregister.gov (https://federalregister.gov/documents/2023/11/17/2023-25458/notifying-the-food-and-drug-administration-of-a-permanent-discontinuance-or-interruption-in)
- uscode.house.gov (https://uscode.house.gov/view.xhtml?req=(title:21%20section:360bbb-3a%20edition:prelim))
- dhs.gov (https://dhs.gov/ohss)
- Humanitarian Device Exemption (HDE) Program Overview
- medicaldevice-network.com (https://medicaldevice-network.com/news/owlet-fda-infant-pulse-oximeter)
- medicaldevice-network.com (https://medicaldevice-network.com/news/perfuze-enrols-first-patient-in-stroke-trial-with-its-millipede-system)
- medicaldevice-network.com (https://medicaldevice-network.com/news/everly-health-at-home-test-kidney-disease)
- fda.gov (https://fda.gov/about-fda/cdrh-innovation/medical-device-coverage-initiatives-connecting-payors-payor-communication-task-force)
- ecfr.gov (https://ecfr.gov/current/title-21/chapter-I/subchapter-H/part-820/subpart-A/section-820.1)
- medtechintelligence.com (https://medtechintelligence.com/feature_article/fda-cleared-samd-by-the-numbers)
- gao.gov (https://gao.gov/products/gao-24-106699?utm_medium=social&utm_source=twitter&utm_campaign=usgao)
- ecfr.gov (https://ecfr.gov/current/title-21/chapter-I/subchapter-H/part-807/subpart-D)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-launches-pilot-program-help-further-accelerate-development-rare-disease-therapies)
- fda.gov (https://fda.gov/about-fda/cdrh-innovation/medical-device-coverage-initiatives-connecting-payors-payor-communication-task-force)
- Key Elements of an HDE Application
- medicaldevice-network.com (https://medicaldevice-network.com/sponsored/reducing-medical-device-approval-times-in-2023)
- medicaldevice-network.com (https://medicaldevice-network.com/news/owlet-fda-infant-pulse-oximeter)
- medicaldevice-network.com (https://medicaldevice-network.com/news/everly-health-at-home-test-kidney-disease)
- ecfr.gov (https://ecfr.gov/current/title-21/chapter-I/subchapter-H/part-860/subpart-D/section-860.220)
- dewberry.com (https://dewberry.com/insights-news/post/blog/2024/01/18/demystifying-technology-understanding-the-human-element)
- stories.cste.org (https://stories.cste.org)
- energy.gov (https://energy.gov/eere/buildings/building-america-technical-support#:~:text=Energy%20Savings%20in-,Chicago,-Homes:%20A%20Case)
- alirahealth.com (https://alirahealth.com/our-services/medical-device-regulation-mdr)
- medicaldevice.how (https://medicaldevice.how)
- IRB Approval and Oversight Requirements
- rimsys.io (https://rimsys.io/blog/fda-listed-cleared-approved-granted)
- fda.gov (https://fda.gov/about-fda/cdrh-innovation/medical-device-coverage-initiatives-connecting-payors-payor-communication-task-force)
- medicaldevice-network.com (https://medicaldevice-network.com/sponsored/reducing-medical-device-approval-times-in-2023)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-november-21-2023)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-october-20-2023)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-february-16-2024)
- nam.edu (https://nam.edu/neurotechnology-and-noninvasive-neuromodulation-case-study-for-understanding-and-anticipating-emerging-science-and-technology)
- fda.gov (https://fda.gov/about-fda/cdrh-innovation/medical-device-coverage-initiatives-connecting-payors-payor-communication-task-force)
- fda.gov (https://fda.gov/medical-devices/premarket-submissions-selecting-and-preparing-correct-submission/division-standards-and-conformity-assessment)
- Emergency Use of HUDs
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-october-20-2023)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-november-21-2023)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-july-11-2023)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-approves-first-nasal-spray-treatment-anaphylaxis)
- federalregister.gov (https://federalregister.gov/documents/2023/09/29/2023-21412/technical-considerations-for-medical-devices-with-physiologic-closed-loop-control-technology)
- fda.gov (https://fda.gov/consumers/consumer-updates/10-facts-about-what-fda-does-and-does-not-approve)
- heartrecovery.com (https://heartrecovery.com/en-us/danger-shock-rct)
- fda.gov (https://fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/abiomed-inc-663150-09192023)
- fda.gov (https://fda.gov/medical-devices/coronavirus-covid-19-and-medical-devices/adverse-event-reporting-medical-devices-under-emergency-use-authorization-eua)
- amdr.org (https://amdr.org/resources-for-advocates)
- medicaldevice-network.com (https://medicaldevice-network.com/news/sechrist-hyperbaric-software)
- medicaldevice-network.com (https://medicaldevice-network.com/news/bd-ultrasound-clinicians)
- fda.gov (https://fda.gov/about-fda/cdrh-innovation/medical-device-coverage-initiatives-connecting-payors-payor-communication-task-force)
- federalregister.gov (https://federalregister.gov/documents/2023/11/17/2023-25458/notifying-the-food-and-drug-administration-of-a-permanent-discontinuance-or-interruption-in)
- Profit and Use Restrictions for HDE Devices
- hardianhealth.com (https://hardianhealth.com/insights/health-institution-exemptions-for-samd-in-great-britain)
- medicaldevice-network.com (https://medicaldevice-network.com/news/everly-health-at-home-test-kidney-disease)
- medicaldevice-network.com (https://medicaldevice-network.com/news/nyse-issues-sonendo-with-non-compliance-notice)
- medicaldevice-network.com (https://medicaldevice-network.com/news/hksh-medical-proton-therapy-hong-kong)
- ecfr.gov (https://ecfr.gov/current/title-21/chapter-I/subchapter-H/part-807/subpart-A)
- medicaldevice-network.com (https://medicaldevice-network.com/data-insights/medical-devices-reef-micro-pricing)
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-transfer-to-manufacturing)
- Annual Distribution Number (ADN) for Profitable Sales
- medicaldevice-network.com (https://medicaldevice-network.com/data-insights/medical-devices-synchron-lx-micro-pricing)
- medicaldevice-network.com (https://medicaldevice-network.com/data-insights/medical-devices-adm-mdm-micro-pricing)
- medicaldevice-network.com (https://medicaldevice-network.com/data-insights/medical-devices-accolade-micro-pricing)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-september-6-2024)
- medicaldevice-network.com (https://medicaldevice-network.com/news/fda-issues-guidance-to-combat-medical-misinformation-online)
- globenewswire.com (https://globenewswire.com/en/news-release/2023/12/04/2790172/0/en/Medical-Devices-Market-Size-Estimated-to-Reach-USD-996-93-Billion-By-2032.html)
- starfishmedical.com (https://starfishmedical.com/blog/18-business-factors-that-determine-successful-medical-device-exits)
- fda.gov (https://fda.gov/drugs/novel-drug-approvals-fda/novel-drug-approvals-2023)
- gao.gov (https://gao.gov/products/gao-24-106699?utm_medium=social&utm_source=twitter&utm_campaign=usgao)
- medtechintelligence.com (https://medtechintelligence.com/feature_article/fda-cleared-samd-by-the-numbers)
- gao.gov (https://gao.gov/products/gao-24-106699?utm_medium=social&utm_source=twitter&utm_campaign=usgao)
- Case Study: Navigating the HDE Application Process
- digitalregulations.innovation.nhs.uk (https://digitalregulations.innovation.nhs.uk/case-studies/using-a-chatbot-to-ease-staff-shortages-and-improve-patient-care)
- ednc.org (https://ednc.org/case-study-a-community-college-and-health-care-system-partnered-to-launch-the-first-registered-nursing-apprenticeship-program-in-north-carolina)
- dematic.com (https://dematic.com/en-au/insights/case-studies/john-dee-australia)
- gfoa.org (https://gfoa.org/materials/solving-labor-shortages-with-a-new-hiring-strategy)
- medicaldevice-network.com (https://medicaldevice-network.com/sponsored/reducing-medical-device-approval-times-in-2023)
- starfishmedical.com (https://starfishmedical.com/blog/esg-medical-device-impact)
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-transfer-to-manufacturing)
- fuelcellsworks.com (https://fuelcellsworks.com/2024/09/23/h2/hydrogene-de-france-prioritisation-of-projects-at-an-advanced-stage-of-development-launch-of-industrial-project)
- fuelcellsworks.com (https://fuelcellsworks.com/news/hydrogene-de-france-hdf-energy-poised-for-growth-set-to-surpass-e100-million-revenue-mark-by-2027?utm_source=rss&utm_medium=rss&utm_campaign=hydrogene-de-france-hdf-energy-poised-for-growth-set-to-surpass-e100-million-revenue-mark-by-2027)
- offshore-energy.biz (https://offshore-energy.biz/dnv-okays-hmds-60000-cbm-lpg-nh3-vcm-carrier-design)
- medicaldevice-network.com (https://medicaldevice-network.com/sponsored/reducing-medical-device-approval-times-in-2023)
- Challenges and Considerations for HDE Devices
- fda.gov (https://fda.gov/about-fda/cdrh-innovation/medical-device-coverage-initiatives-connecting-payors-payor-communication-task-force)
- medicaldevice-network.com (https://medicaldevice-network.com/sponsored/reducing-medical-device-approval-times-in-2023)
- forum.effectivealtruism.org (https://forum.effectivealtruism.org/posts/oSAbWDuxmFZnsGEvx/case-study-safety-standards-on-california-utilities-to)
- starfishmedical.com (https://starfishmedical.com/blog/18-business-factors-that-determine-successful-medical-device-exits)
- octopart.com (https://octopart.com/pulse/p/ensuring-reliable-sourcing-medical-device-supply-chains)
- medicaldevice-network.com (https://medicaldevice-network.com/news/everly-health-at-home-test-kidney-disease)
- medicaldevice-network.com (https://medicaldevice-network.com/news/medtronic-fda-hypertension-treatment-system)
- fda.gov (https://fda.gov/about-fda/cdrh-innovation/medical-device-coverage-initiatives-connecting-payors-payor-communication-task-force)
- medicaldevice-network.com (https://medicaldevice-network.com/sponsored/reducing-medical-device-approval-times-in-2023)
- schlafenderhase.com (https://schlafenderhase.com/ebooks/medical-device-report-how-are-compliance-strategies-evolving)




 concept. Starting at the center, you can follow branches to see how ISF relates to hormone therapy, calculation methods, biological roles, and practical applications in diabetes management. This mindmap shows the evolution of the Sensitivity Factor (ISF) concept. Starting at the center, you can follow branches to see how ISF relates to hormone therapy, calculation methods, biological roles, and practical applications in diabetes management.](https://images.tely.ai/telyai/cnbrdfbp-this-mindmap-shows-the-evolution-of-the-sensitivity-factor-isf-concept-starting-at-the-center-you-can-follow-branches-to-see-how-isf-relates-to-hormone-therapy-calculation-methods-biological-roles-and-practical-applications-in-diabetes-management.webp)