Introduction
The Mexican healthcare landscape is undergoing a significant transformation, marked by a decisive shift towards private medical services within a complex interplay of public and private sectors. This evolution not only creates new opportunities for medical device companies but also presents unique challenges in navigating regulatory requirements and engaging key stakeholders. As the demand for innovative healthcare solutions escalates, companies must strategically optimize their approaches to enhance patient access while ensuring compliance in this dynamic environment.
Understand the Mexican Healthcare System and Market Dynamics
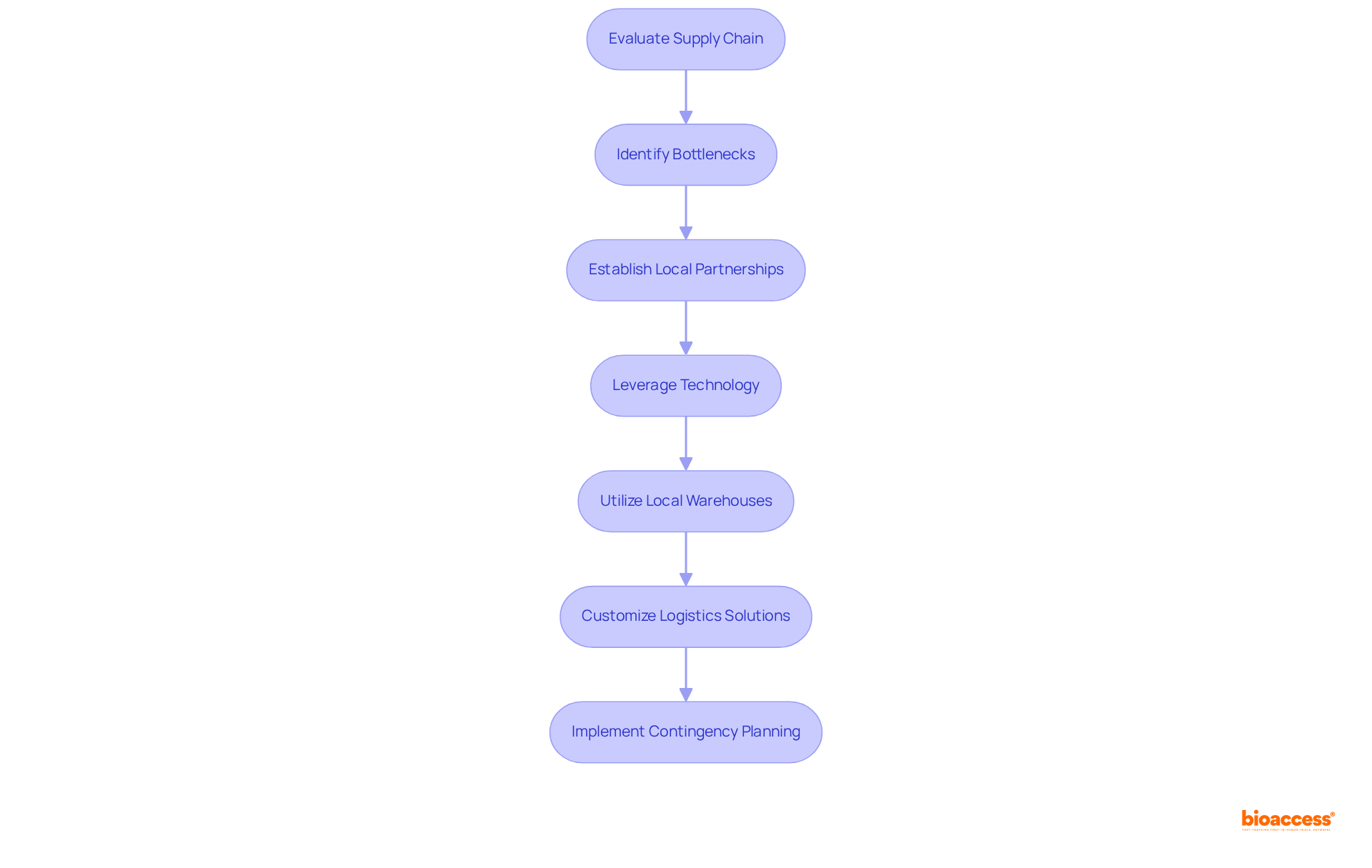
The Mexican medical landscape presents a complex interplay between public and private sectors, characterized by significant disparities in access and quality. Prominent organizations, such as the Mexican Institute of Social Security (IMSS), operate alongside a burgeoning that is increasingly favored by individuals. Recent trends indicate a notable , primarily driven by seeking enhanced medical options. This transformation offers a to delve into these dynamics, enabling them to identify target demographics and customize their offerings to address specific needs.
Furthermore, it is essential to remain informed about , as these factors are pivotal in revealing potential . For example, the current emphasis on digitalization and preventive care underscores the vital role that innovative can play in improving . As the private sector continues to expand, understanding these trends will be crucial for successful market entry and sustained growth within the country’s .
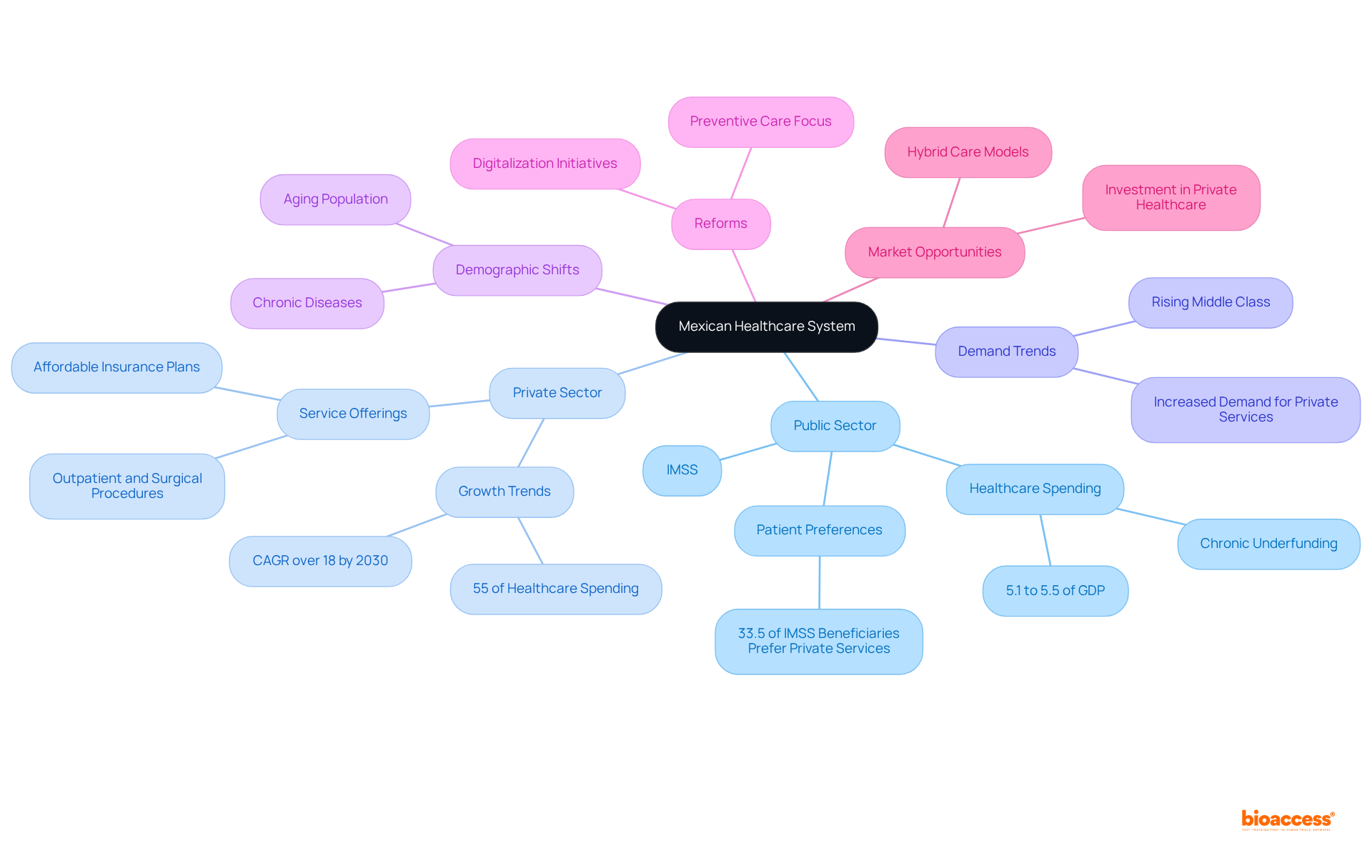
Navigate Regulatory Requirements for Medical Devices in Mexico
Navigating the necessitates a comprehensive understanding of the requirements set forth by COFEPRIS, particularly the imperative of obtaining a . This entails:
- A meticulous risk classification
- The submission of
- Unwavering adherence to
Recent updates introduced by NOM-241-SSA1-2025 underscore the vital role of and throughout the product lifecycle, ensuring that products meet established safety and effectiveness standards.
Furthermore, companies can benefit from leveraging , which facilitate the importation of products already approved in other jurisdictions. Engaging can yield valuable insights and streamline the navigation of the intricate regulatory environment, thereby enhancing the likelihood of successful registration.
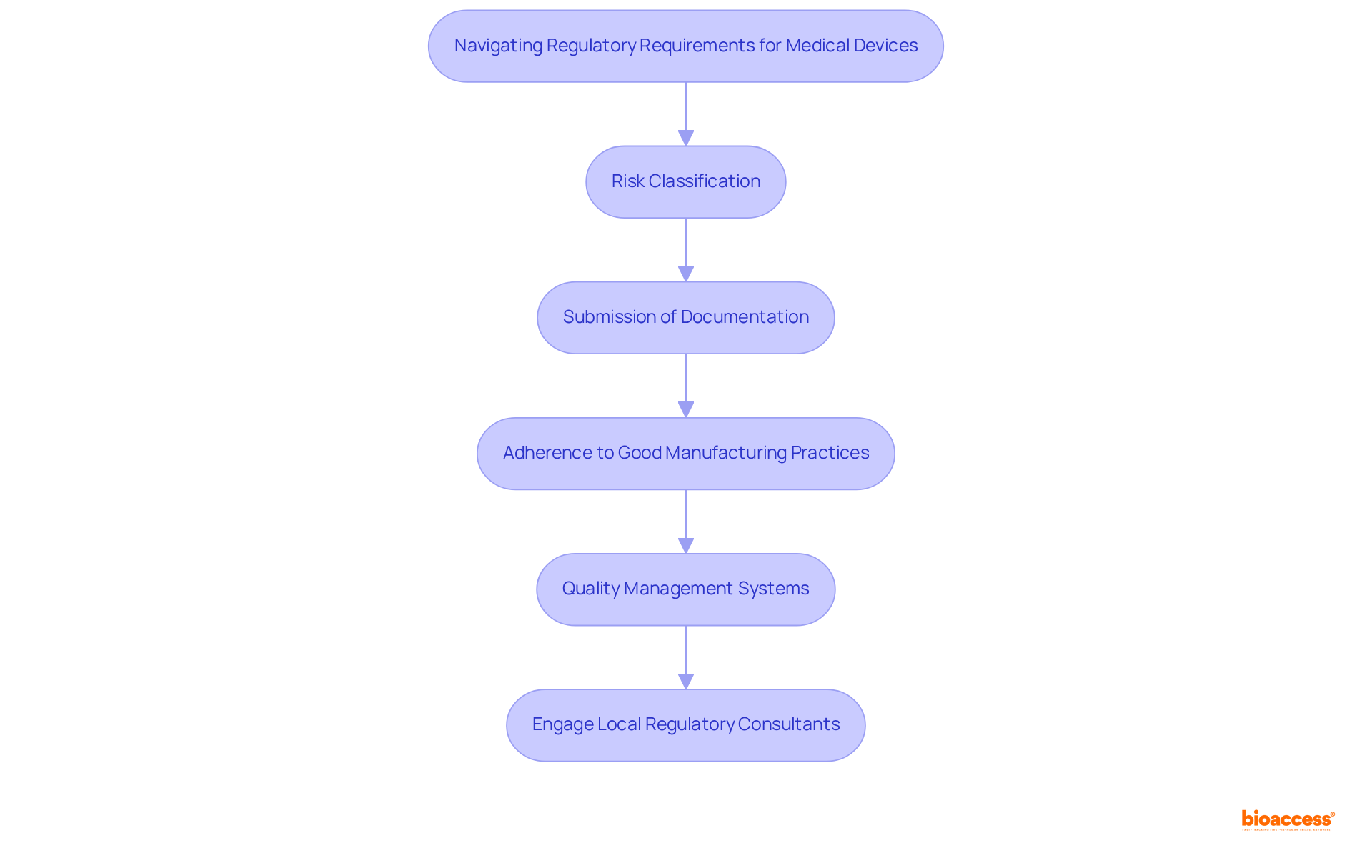
Engage Key Stakeholders to Enhance Patient Access
To improve , companies must actively , including:
- Medical professionals
- Hospital managers
- Advocacy organizations
Establishing robust connections with these entities yields valuable insights into individual needs and preferences, significantly guiding product development and marketing strategies. Collaborating with local medical providers not only facilitates the seamless integration of devices into but also fosters a culture of shared knowledge and innovation.
For instance, can elevate awareness and understanding of new technologies, ultimately leading to . By 2025, over 60% of Medtech firms in Mexico are expected to partner with academic institutions to enhance their trials, highlighting the critical nature of such collaborations. Furthermore, engaging user advocacy organizations in the development process ensures that solutions are designed with the end-user in mind, effectively addressing specific challenges faced by patients. This cooperative approach is essential for navigating the complexities of the healthcare environment and ensuring that align with the needs of both practitioners and patients alike.
Moreover, leveraging comprehensive , such as those provided by bioaccess®, can streamline the execution of early-feasibility studies, , and post-market follow-up studies. By concentrating on trial setup, compliance reviews, and project management, bioaccess® ensures that not only adhere to regulatory standards but also contribute to job creation and economic growth in the region. This holistic strategy ultimately transforms lives in Latin America through advanced Medtech solutions.
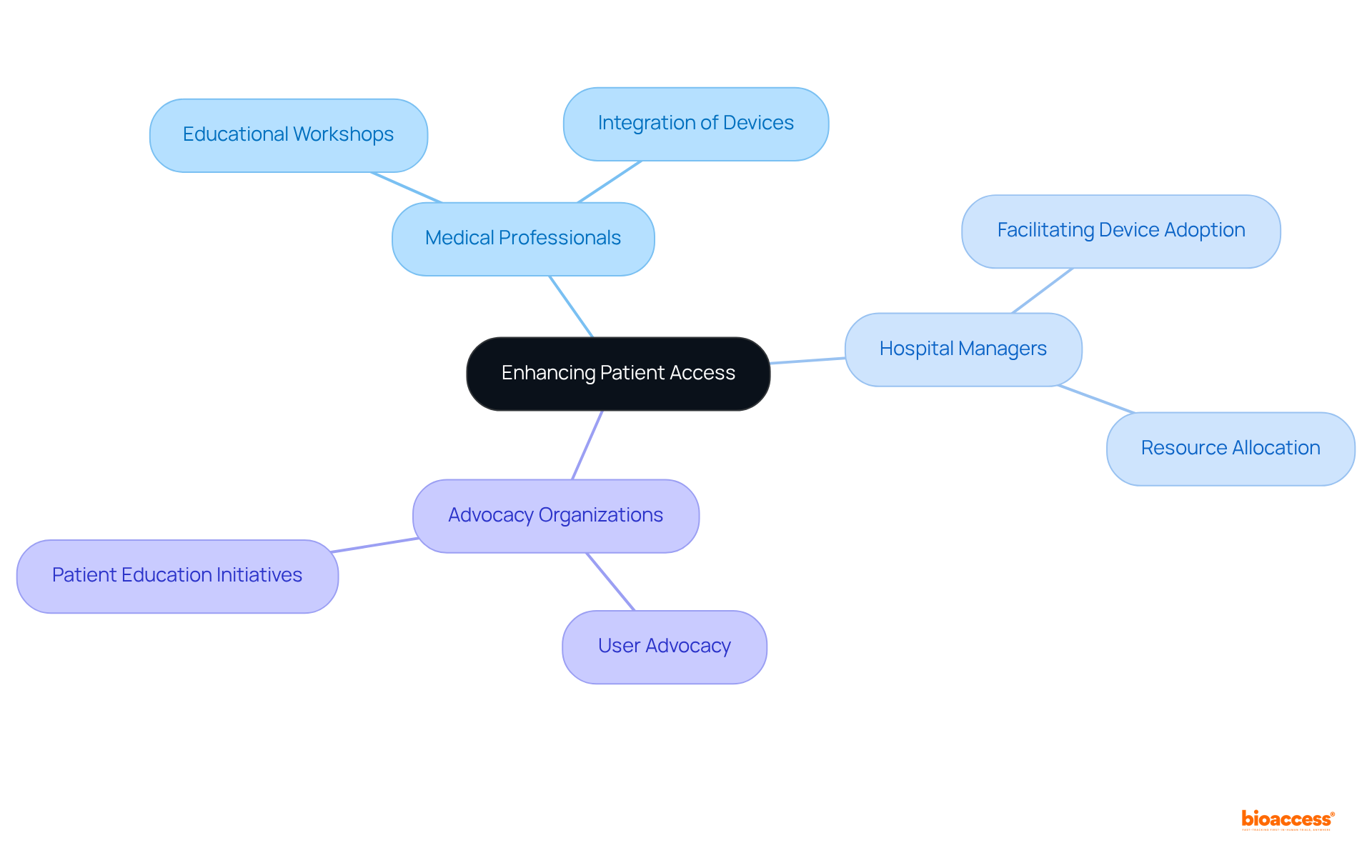
Optimize Distribution and Logistics for Medical Device Access
To enhance the distribution and logistics of medical products in the country, companies must critically evaluate their and identify potential bottlenecks. Establishing partnerships with is essential; these local entities can offer valuable insights into cultural sensitivities and , thereby enhancing efficiency and reducing lead times.
As Steve Garchow noted in the LATAM Medtech Leaders Podcast, significantly improves visibility throughout the supply chain, enabling better demand forecasting and responsiveness to market fluctuations. For instance, utilizing local warehouses can facilitate quicker response times and lower transportation costs, which are important components of for .
Furthermore, companies must consider Mexico’s geographical diversity, which may require to both urban and rural medical facilities. Implementing a robust that includes contingency planning for potential disruptions can further enhance reliability and customer satisfaction.
This proactive approach, combined with a comprehensive understanding of local healthcare systems and market dynamics, is crucial for success in the .
Conclusion
The Mexican healthcare market is teeming with potential for those who are proactive and informed. By embracing a collaborative approach that includes stakeholder engagement and innovative logistics strategies, companies can not only facilitate market penetration but also enhance healthcare access for patients. As the healthcare landscape continues to evolve, it is imperative for organizations to remain agile, adapting their strategies to align with the changing needs of the market and the communities they serve.
Frequently Asked Questions
What are the main characteristics of the Mexican healthcare system?
The Mexican healthcare system features a complex interplay between public and private sectors, with significant disparities in access and quality.
What role does the Mexican Institute of Social Security (IMSS) play in the healthcare system?
The IMSS is a prominent organization within the public sector that provides medical services and operates alongside a growing private medical sector.
Why is there an increasing demand for private medical services in Mexico?
The surge in demand for private medical services is primarily driven by demographic shifts and a rising middle class seeking better medical options.
How can companies benefit from understanding the Mexican healthcare market dynamics?
Companies can identify target demographics and customize their offerings to meet specific needs by understanding the complex dynamics of the healthcare market.
What factors should be monitored to identify market opportunities in the Mexican healthcare system?
Ongoing reforms and budget allocations are crucial factors to monitor, as they can reveal potential market opportunities and risks.
What current trends are influencing the Mexican healthcare market?
Current trends include an emphasis on digitalization and preventive care, highlighting the importance of innovative medical solutions for improving patient outcomes.
How important is it to understand market trends for companies looking to enter the Mexican healthcare sector?
Understanding market trends is crucial for successful market entry and sustained growth within the Mexican healthcare domain.
List of Sources
- Understand the Mexican Healthcare System and Market Dynamics
- mexicobusiness.news (https://mexicobusiness.news/health/news/mexicos-healthcare-shift-private-sector-opportunity-knocks)
- linkedin.com (https://linkedin.com/pulse/opportunity-crisis-private-healthcare-rises-mexico-xavier-tello-2wiyc)
- practiceguides.chambers.com (https://practiceguides.chambers.com/practice-guides/healthcare-ma-2025/mexico/trends-and-developments)
- fitchsolutions.com (https://fitchsolutions.com/bmi/infrastructure/mexicos-public-and-private-healthcare-sectors-will-be-strengthened-medium-term-14-03-2024)
- mexicobusiness.news (https://mexicobusiness.news/health/news/health-services-cut-mexico-misses-2025-budget-goals)
- Navigate Regulatory Requirements for Medical Devices in Mexico
- COFEPRIS medical device and IVD registration and approval in Mexico (https://emergobyul.com/services/cofepris-medical-device-and-ivd-registration-and-approval-mexico)
- bioaccessla.com (https://bioaccessla.com/blog/9-key-class-ii-device-rules-cofepris-for-successful-registration)
- Ultimate Guide for Regulatory Affairs in Mexico (https://veraqueconsulting.com/mx/ultimate-guide-for-regulatory-affairs-in-mexico)
- QMS Compliance Requirements for Mexico Medical Devices (https://freyrsolutions.com/blog/qms-requirements-for-medical-device-compliance-in-mexico)
- Engage Key Stakeholders to Enhance Patient Access
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC11443951)
- researchgate.net (https://researchgate.net/publication/279301288_The_Impact_of_CME_on_Physician_Performance_and_Patient_Health_Outcomes_An_Updated_Synthesis_of_Systematic_Reviews)
- journals.sagepub.com (https://journals.sagepub.com/doi/10.1177/14779714221147297)
- bioaccessla.com (https://bioaccessla.com/blog/best-practices-for-medtech-innovation-through-mexican-research-proven-strategies-for-success)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC12090590)
- Optimize Distribution and Logistics for Medical Device Access
- ey.com (https://ey.com/en_us/insights/health/how-generative-ai-can-optimize-health-care-supply-chains)
- researchgate.net (https://researchgate.net/publication/366897800_Optimization_of_a_Government_Medical_Warehouse_Using_Lean_Logistics_Methodology)
- americanindustriesgroup.com (https://americanindustriesgroup.com/manufacturing-in-mexico)
- prodensa.com (https://prodensa.com/insights/blog/doing-business-in-mexico-a-manufacturers-guide)
- bakerinstitute.org (https://bakerinstitute.org/research/nearshoring-mexico-seizing-opportunities-and-facing-challenges)

Leave a Reply