Introduction
Selecting the right clinical trial site is crucial for the success of a study. It impacts participant recruitment, data integrity, and research pace. The use of electronic health records (EHR) in trials poses unique challenges, requiring sites to adapt and integrate EHR data effectively.
A case study exemplifies the meticulous planning required for successful site selection. Patient access and equity are also vital considerations, as logistical hurdles can hinder participation. The evolving clinical trial landscape demands sites that can handle increasing study volumes and technological demands.
Ultimately, site selection is a multifaceted decision that requires balancing technical requirements, participant needs, and operational capabilities. Each site chosen is a step towards advancing medical knowledge and improving patient outcomes.
Importance of Clinical Trial Site Selection
Selecting an optimal is pivotal for the seamless execution and success of a . The location impacts not just , but also the integrity and pace of the research. For instance, the utilization of electronic health records (EHR) in trials poses unique challenges; existing sites must adapt to integrate EHR data effectively into the study design to ensure high-quality data collection.
The demonstration project highlighted in Raman et al. Trials (2023) 24:566 exemplifies the meticulous planning required. It underscores the importance of a in providing technical and operational support to sites, ensuring that the EHR data complements the multi-center pharmaceutical industry outcomes trial data.
This case study illustrates the level of detail and consideration that must go into site selection to achieve the study’s aims.
Furthermore, site selection can have profound implications for participant access and equity. A scenario described by Etienne Nichols echoes this sentiment, where a patient from rural Pennsylvania with an ultra-rare disease faces logistical hurdles to participate in a trial abroad. Such complexities demonstrate the need for that considers patient convenience and the potential to leverage technology in alleviating these barriers.
With the evolving, reports by key industry leaders and reveal that sites are managing an increasing volume of studies and sponsor-provided technology. These changes necessitate that sites are chosen not only for their capacity to recruit participants but also for their ability to handle the technological demands of modern trials.
Ultimately, the selection of a is a multifaceted decision that requires balancing the technical requirements of the study, participant needs, and the operational capabilities of the site. Each site chosen is a step towards the collective goal of advancing medical knowledge and improving patient outcomes through rigorous and well-controlled research studies.
Pre-Selection Considerations
When preparing for in , it’s imperative to incorporate various critical factors that will shape the study’s trajectory and potential for success. Key elements include aligning with the study objectives, understanding the nuances of the target population, evaluating the , and abiding by stringent . For example, recent guidance has highlighted the importance of in trials, especially for multinational studies, where the FDA stipulates enrollment goals and metrics to ensure a representative participant demographic.
The selection process also benefits from innovative patient assignment methods, such as Equal Randomisation and Thompson Sampling, which prioritize both statistical power and . Furthermore, integrating , especially from electronic health records, into the trial design can improve data quality and operational efficiency, as demonstrated in recent multi-center pharmaceutical trials.
Real-world implications of these factors are evident in cases where patients with rare diseases face logistical challenges to participate in international trials. This underscores the necessity for careful consideration of patient accessibility and support in the site selection process. Ultimately, these pre-selection considerations are crucial in paving the way for a strategic and informed site selection, ensuring the trial is poised for success from the outset.
Assessing Feasibility and Resources
Navigating the complexities of for requires meticulous scrutiny of several pivotal factors. In this endeavor, understanding the is paramount. For instance, in cases where patients are imbued with comprehensive information about their involvement in trials, as seen in the context of radical prostatectomy studies, their consent and engagement can be more seamlessly integrated into the trial process.
This is not only about informing the patient but also about incorporating their health metrics directly into their medical records, optimizing both patient care and data accuracy.
Moreover, the infrastructure and resources at a site are crucial to meet the demands of a clinical trial. The burgeoning field of , such as HINT and SPOT algorithms, stands as a testament to the potential of leveraging computational tools to forecast trial outcomes. These innovative approaches can significantly influence the decision-making process regarding trial design and patient eligibility criteria, ensuring that sites are aptly equipped to address the multifaceted needs of a study.
Patient accessibility to trials also necessitates careful consideration. The narrative of a rural Pennsylvania resident requiring travel to Turkey for a trial participation underscores the significance of and the overarching need for . Matching patients with trials using AI and other technological aids exemplifies the industry’s shift towards more nuanced and personalized approaches to and trial accessibility.
The statistics paint a telling picture: the lengthening of and the high rate of delays highlight the urgency for . This efficiency is not only a matter of regulatory compliance but also a race against time for patients awaiting novel therapies. Companies are thus propelled by both competitive forces and the moral imperative to expedite the path to market for life-saving treatments.
In conclusion, the process is far from a mere administrative step; it is a strategic endeavor that influences the trajectory of clinical research and the lives of patients worldwide. As we integrate patient perspectives, harness predictive technologies, and streamline logistics, the clinical trial landscape can be transformed into a more effective and compassionate system.
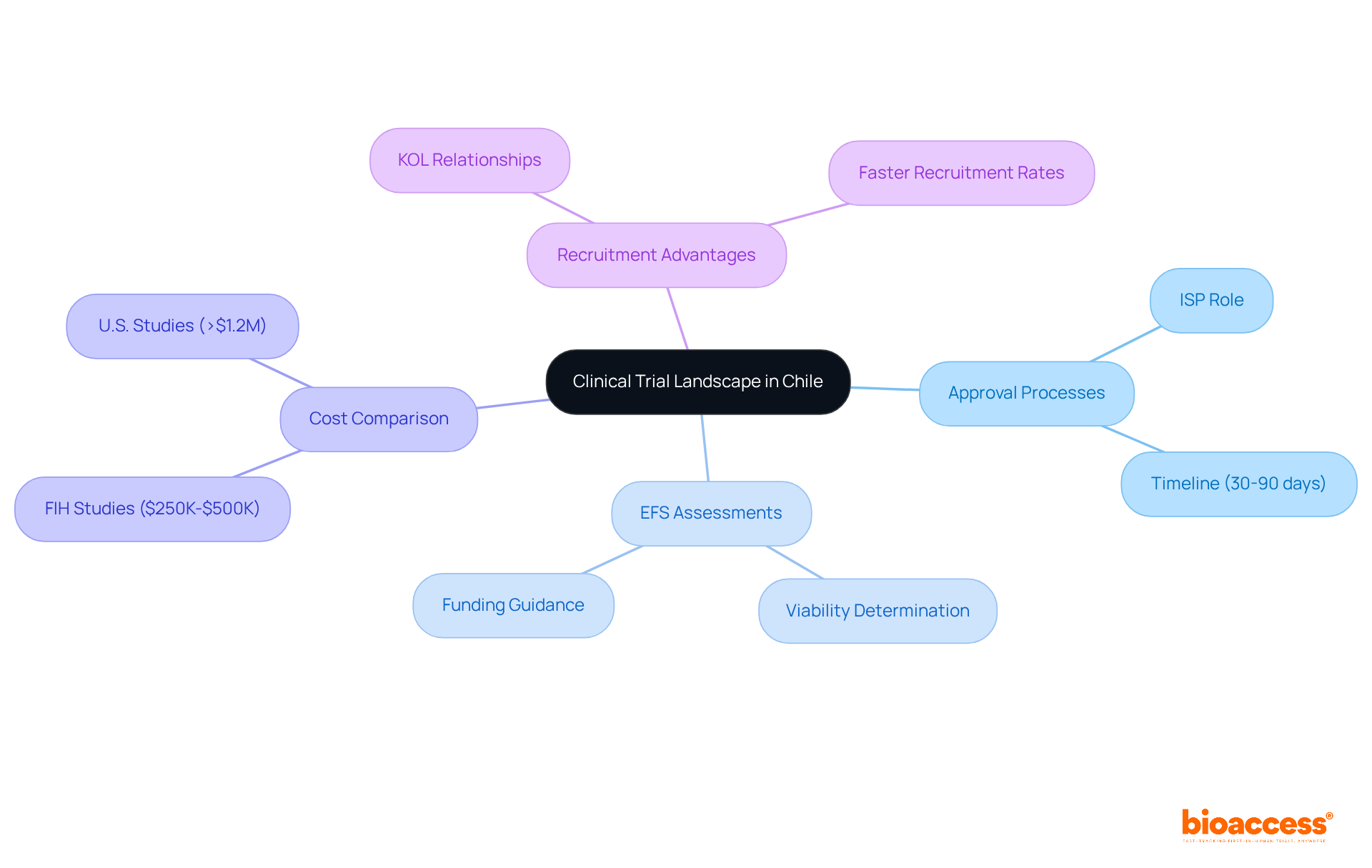
Site Selection Criteria
When embarking on a , the selection of is a pivotal step that can significantly influence the success and efficiency of the study. It is essential to evaluate potential sites through a detailed lens, considering the qualifications and experience of investigators and staff. This is particularly true for trials that utilize electronic health records (), where the capability to manage and operationalize EHR data is paramount.
A site’s infrastructure must be equipped to handle the specific requirements of , ensuring that the integration of electronic data aligns with the trial’s objectives.
are also pivotal, as the demographic and the ability to maintain participant numbers can greatly affect a trial’s power and statistical validity. A clear understanding of the site’s strategies to engage and retain participants is necessary. This includes considering the various , such as Equal Randomisation or Thompson Sampling, which could impact the outcome of the trial.
Additionally, it’s crucial that sites strictly adhere to . In the context of contemporary multi-center pharmaceutical industry outcomes trials, a central coordinating center can be instrumental in guiding sites through the technical, governance, and operational aspects of such regulatory adherence.
In light of recent discussions at the Outsourcing in s conference, it is evident that there is an ongoing need to prioritize data integration and streamline research timelines in clinical research spaces such as the UK. Such considerations underscore the importance of site selection in preparing a robust foundation for a successful . By establishing , sponsors are empowered to make informed decisions that enhance the quality and efficacy of clinical research, ultimately driving the field towards better patient outcomes and a more effective healthcare system.
Evaluating Site Capabilities and Experience
Selecting the right is a cornerstone of a successful study, as it ensures the reliability of data and the smooth progression of the trial. A demonstrated track record in conducting similar research is one of the foundational aspects when evaluating a site’s capabilities. One must consider not only the site’s previous experience but also the adequacy of its facilities, the presence of specialized equipment, and its proficiency in .
For instance, the integration of electronic health records (EHR) into , known as “EHR-sourced” trials, offers promising advancements but also presents significant challenges. A recent demonstration project described in Trials (2023) 24:566 illustrated how EHR data could complement traditional data collection methods in a multi-center pharmaceutical trial. The study highlighted the integral role of a in assisting sites with the technical, governance, and operational challenges of such trials.
It is essential for sponsors to consider the complexities of modern clinical research environments. Research sites often grapple with the use of numerous systems; a survey indicated that 60% of these sites utilize over 20 different systems regularly. This technological overload can lead to increased staff burnout and diminish a site’s capacity to effectively manage trials.
Moreover, the clinical trial landscape is evolving with a greater emphasis on . The deployment of connected devices and wearables, for example, has widened the scope of data available. This necessitates a well-defined that prioritizes patient safety and data quality, which should be established before the trial protocol is designed.
Such strategy should outline the optimal data collection methods and manage the flow of information from both traditional and digital sources.
Ultimately, the aim is to select sites that not only possess the necessary experience but also the capacity to handle the . This includes managing a vast array of data sources and maintaining the highest standards of data quality, all while navigating the operational intricacies that modern entail.
Assessing Site Infrastructure and Resources
The integration of electronic health records (EHR) into , known as ‘EHR-sourced’ trials, is an evolving practice that promises to enhance the efficiency of study operations. A demonstration project described in Trials (2023) 24:566 illustrated how EHR data could complement traditional data collection methods in a multi-center pharmaceutical trial. The project underscored the importance of robust site infrastructure to support technical, governance, and operational challenges.
Operational considerations such as , , and quality review became focal points, with a central coordinating center playing a pivotal role in assisting sites with these activities.
A recent initiative led by the White House Office of Science and Technology Policy aims to strengthen the U.S. infrastructure, which is critical to advancing health outcomes and driving forward initiatives like the Biden Cancer Moonshot. This is set to make trials more efficient and inclusive, thus enhancing the capacity to address health crises and expedite treatment development.
Furthermore, with nearly 60% of sites reporting an increase in study volume, as per Advarra’s 2023 Study Activation Survey, the burden of managing sponsor-provided technology has grown. Technologies such as electronic data capture (EDC), interactive voice response (IVR) systems, and safety letter distribution systems are now integral to site operations. These developments highlight the increasing complexity of and the necessity for sites to have comprehensive infrastructure and resources to handle these demands.
The Federal Drug Administration’s Oncology Center of Excellence’s Project Community initiative has emphasized the value of , facilitated by the availability of diverse data through wearables, electronic diaries, and decentralized trial solutions. However, the vast data volumes necessitate a strategic approach to collection, monitoring, and analysis, reinforcing the significance of a well-defined data strategy prior to protocol design.
In essence, ensuring that possess the requisite infrastructure and resources is not merely a matter of operational efficiency; it is a critical component of contemporary trial design that influences the quality of data, the speed of drug development, and ultimately, patient health outcomes.
Investigator and Staff Qualifications
The caliber of investigators and site personnel is pivotal to the success of . Their proficiency in understanding , therapeutic area expertise, and past research involvement is essential for ensuring the rigorous standards needed for patient safety and treatment efficacy. are tiered into phases, with each stage designed to meticulously test new treatments and discern any improvements over current options.
Phase one primarily assesses safety with a few healthy volunteers, while phase two expands to a larger cohort, including affected individuals, to appraise both safety and efficacy.
Diverse and inclusive participation is vital. Research indicates stark ; for instance, there’s a notable gap in life expectancy between the most and least socioeconomically disadvantaged groups. Recognizing such disparities, major research funders have launched initiatives like the ‘INCLUDE’ project to foster greater equality in research participation.
is evident in their efforts to harmonize human subject protection regulations and facilitate evidence generation for medical product safety and effectiveness. They underscore the significance of well-designed studies and reliable data in their decision-making processes. In line with this, are offered to stakeholders, including healthcare professionals and researchers, to deepen their understanding of safety concerns and address specific populations in drug development.
Moreover, the importance of (HEIs) is noteworthy. Their role, regardless of being on a research or professional services pathway, is crucial, with each offering distinct recognition and professional development opportunities. The intricacies of trial management and the need for experienced personnel highlight the imperative of selecting trial sites with competent investigators and staff to enhance study quality and reliability.
The insights and experiences shared by individuals like Barbara, who discovered a life-threatening heart condition through a study found on The New Normal platform, exemplify the personal and societal benefits of well-conducted research.
Patient Recruitment and Retention
Selecting clinical trial sites with robust is paramount to the success and efficiency of . One critical aspect involves the site’s ability to access a , ensuring the inclusion of underrepresented groups who may have unique responses to treatments, such as older patients often overlooked in hospital-based research. Incorporating patient-centric approaches, where trial participants are actively involved in planning and informed in a comprehensible manner, can significantly enhance their experience and willingness to engage.
This engagement is especially crucial when trials involve , such as international travel, which may deter participation.
The integration of that leverage electronic health records (EHR) can facilitate the inclusion of diverse demographic groups. These trials, by operationalizing study goals through existing healthcare infrastructure, can . For instance, harnessing data from EHR-sourced trials and employing AI-driven methodologies can yield richer insights into patient behavior, aiding in the development of more effective drugs.
However, the success of these strategies also hinges on clear communication and the removal of barriers to participation. For example, older patients in acute hospital settings may face challenges in engaging with research due to stress, urgency of treatment, or cognitive impairments. Persistence and flexibility on the part of researchers, coupled with a supportive healthcare environment, are essential to facilitate their involvement.
By prioritizing and leveraging technology, clinical trial companies can improve patient outcomes and foster a more inclusive research ecosystem.
Regulatory Compliance and Ethical Considerations
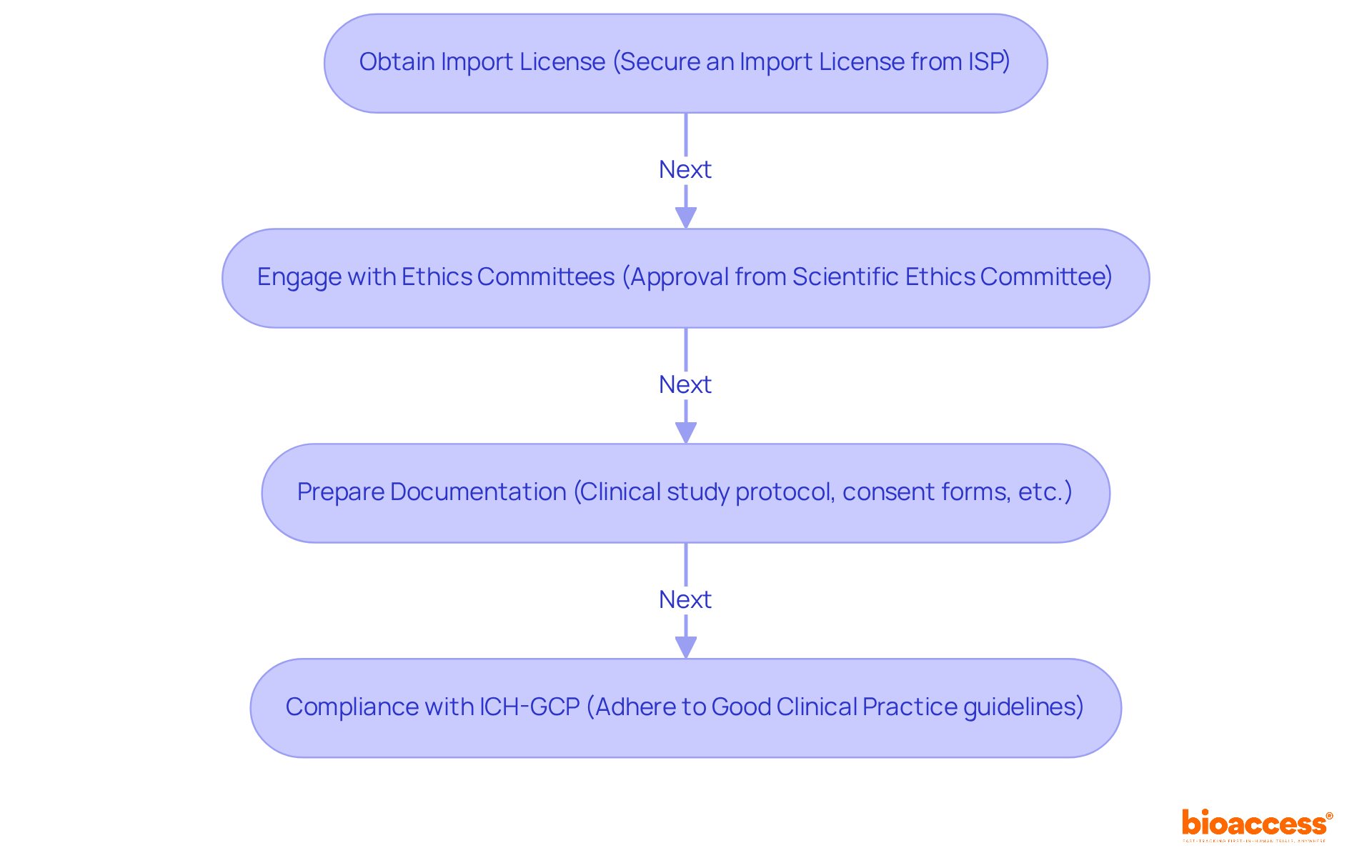
Upholding the highest standards of is vital in to ensure the integrity and validity of research. Such diligence in adhering to guidelines, obtaining informed consent, and respecting ethical considerations is not merely a legal requirement but a cornerstone in safeguarding participant rights and advancing medical knowledge.
The scrutiny of a site’s compliance history is crucial, as any deviation from established protocols can jeopardize the credibility of the data and, consequently, the entire study. This is exemplified by the implementation of advanced technologies like AI and ML in , which, while promising, introduce complex regulatory challenges. The FDA, EU, and EMA have issued guidelines necessitating a risk-based approach to AI, demanding transparency, and underscoring the need for robust data governance.
Moreover, the connection between a site and ethics committees and regulatory bodies is indicative of the site’s commitment to ethical practices. This relationship is especially important when considering the compensatory aspects of , where recent ethical discussions have highlighted the importance of fair treatment of participants, including appropriate reimbursement for their involvement.
The importance of maintaining in clinical research cannot be overstated, as evidenced by recent cases where lapses have had severe implications. For example, image is a particularly vulnerable area, with 20 to 35 percent of manuscripts being flagged for image-related problems, often due to accidental duplication.
In summary, selecting sites with a proven track record of and ethical behavior is essential. It ensures not only the protection of but also the generation of reliable data that informs the FDA’s decision-making and the development of safe, effective medical products for patients. As the landscape of clinical research evolves with emerging technologies, maintaining these standards becomes even more imperative to uphold the trust and effectiveness of .
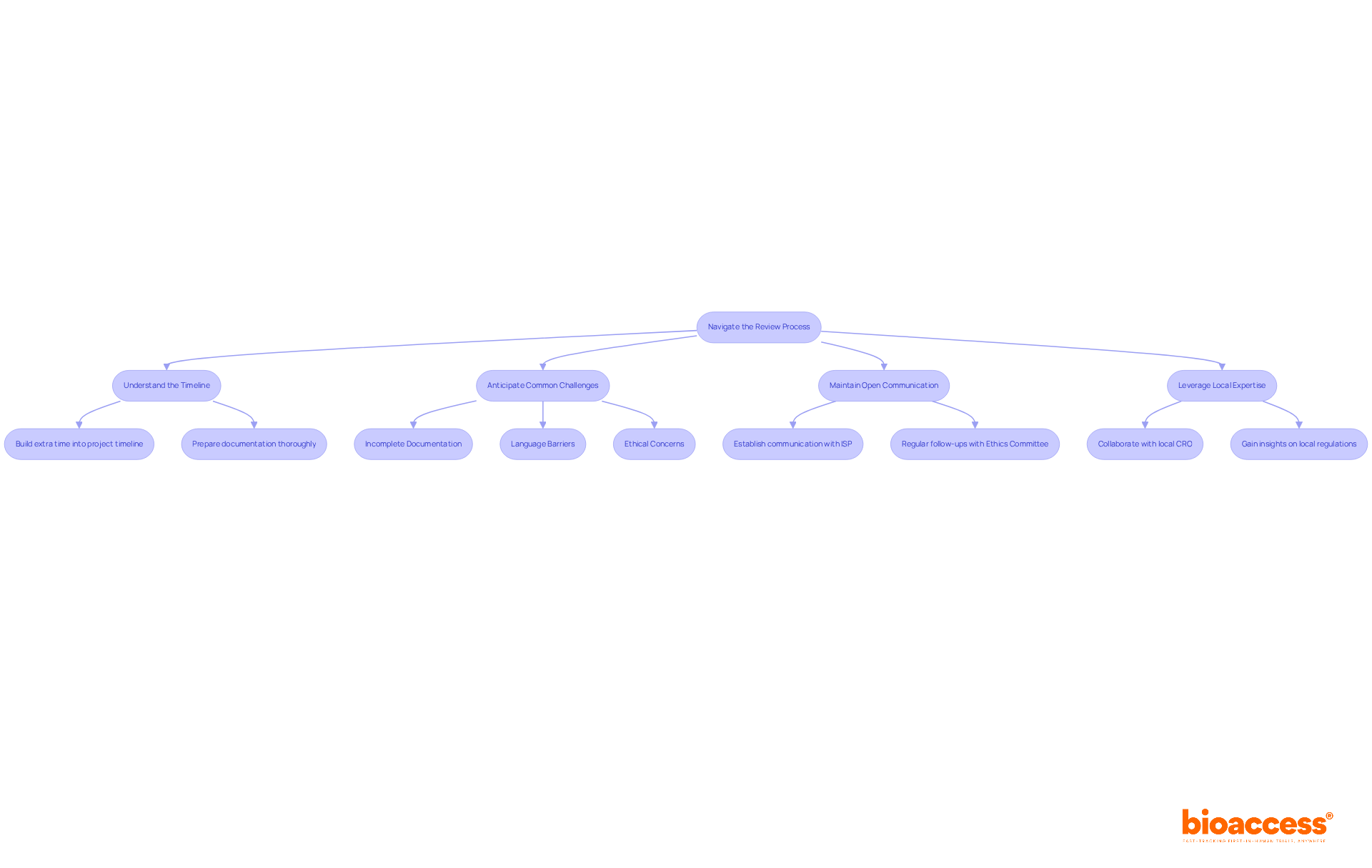
Stakeholder Involvement and Communication
The pivotal role of in the for cannot be overstated. It is a collaborative endeavor that demands active participation from all parties involved, including investigators, sponsors, ethics committees, and . In the intricate dance of site selection, each stakeholder’s voice is critical to align expectations and address potential concerns, ensuring the trial’s infrastructure is primed for success.
Raman et al. in their 2023 study, published in Trials, underscore the need for a of existing sites with new trial goals, especially when harnessing electronic health records (EHR) for data collection. The study highlights the operational challenges and emphasizes the importance of a central coordinating center in guiding sites through technical and governance aspects, thereby reinforcing the vital nature of and coordinated efforts.
Inclusion and diversity in are not only about adherence to ethical standards, but they also enhance the robustness of trial outcomes. Herron from RWS notes that patient-centric approaches, encompassing a broad spectrum of demographics, are essential for capturing the diversity of patient experiences. This sentiment is echoed by the positive responses of patient partners and research staff in a project focused on reducing low-value care practices, as they recognize the valued impact of patient involvement in research design and execution.
Moreover, is paramount, as evidenced by Regeneron Pharmaceuticals’ query on FDA’s definition of non-interventional studies, which highlights the need for harmonization across regions. Similarly, the National Organization for Rare Disorders (NORD) praises innovative informed consent approaches that cater to participant-specific needs, further exemplifying the importance of clear and accessible stakeholder communication.
The collective efforts to involve stakeholders not only build a foundation for successful but also ensure ethical integrity and public trust. As such, fostering transparent relationships and maintaining open dialogue throughout the is not a mere formality, but a strategic imperative that underpins the effective execution of clinical studies.
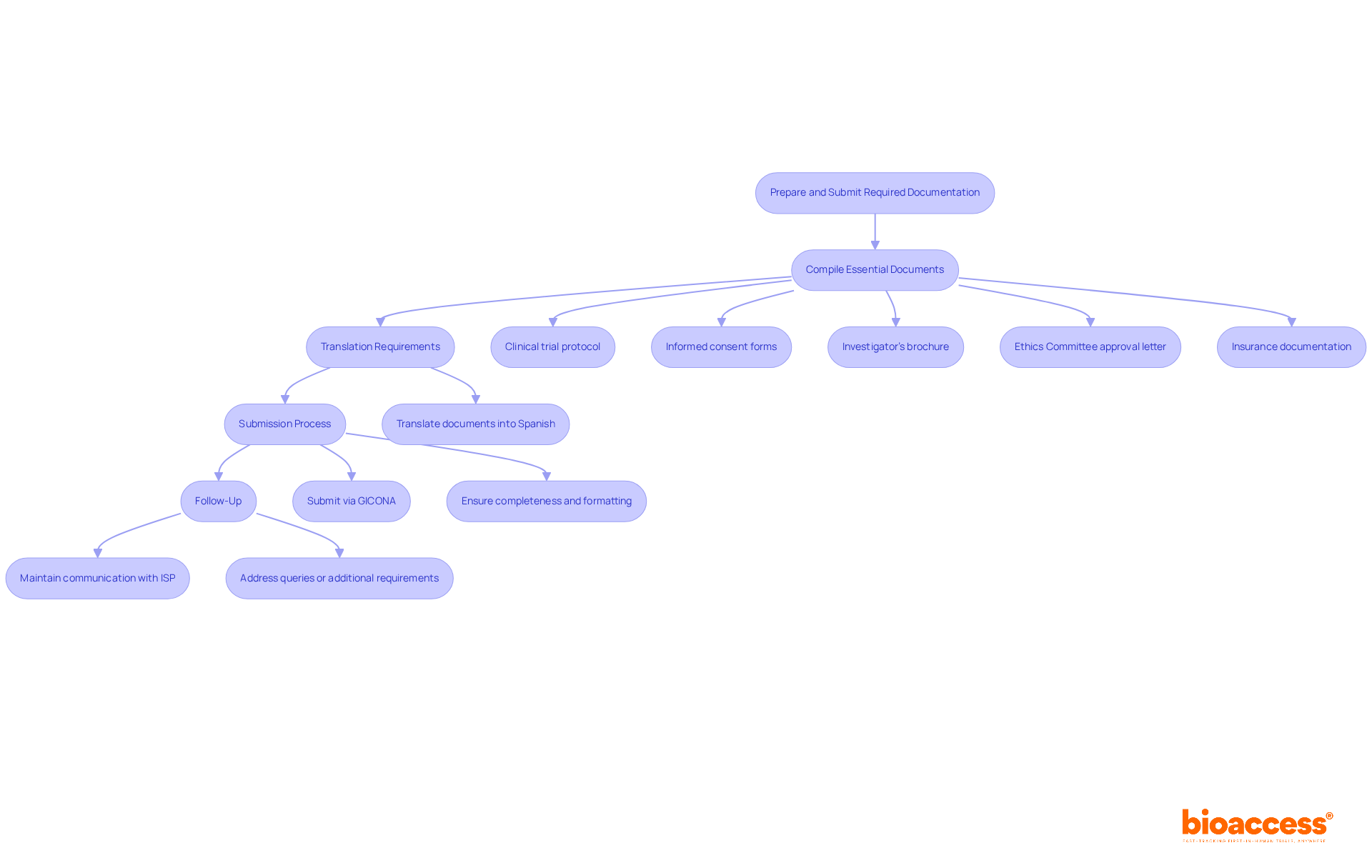
Documentation and Transparency
For , the integrity of research hinges on a meticulously documented and transparent . This practice ensures accountability and enhances the quality of the . A study by Raman et al.
underscores the significance of a clear methodology in site selection, particularly with trials that utilize electronic health records (EHRs) to achieve research objectives. EHR-sourced trials, while promising, are often hampered by uncertainties regarding the optimal use of existing sites and infrastructure to meet study goals. To combat these uncertainties, a , complete with , is paramount.
Furthermore, the variable levels of commitment to transparency observed across companies highlight the need for consistent and statistical analysis plans. As noted in research published in major journals, even registration on platforms like ClinicalTrials. Gov may not suffice to ensure the .
The ultimate goal is to make full protocols, analysis plans, and raw data widely available, thus fostering a more open research environment.
Recent news also reflects the evolving landscape of transparency. Regeneron Pharmaceuticals has raised concerns regarding the FDA’s definition of non-interventional studies, seeking greater harmonization with the EMA’s definition. Similarly, the acquisition of CancerLinQ by ConcertAI signals a move towards leveraging real-world data and AI to enhance the quality and integrity of s.
To encapsulate the sentiment, a quote from Osipenko, a leader in clinical research quality, illustrates the profound impact of patient participation in trials: for many patients, the hope lies not in their own benefit from the research but in the potential for future generations to avoid their suffering. This perspective reinforces the necessity of clear communication and documentation to respect and honor the contributions of trial participants.
In conclusion, sponsors are called to maintain rigorous standards by ensuring the is thoroughly documented and transparent. This approach not only honors the trust placed in them by patients and the public but also upholds the integrity of the research that paves the way for medical advancements.
Implementation and Dissemination
Effective site selection and the subsequent dissemination of outcomes are pivotal steps in the operationalization of . Central to this is the utilization of , which have shown immense promise in enhancing the efficiency and quality of data collection. A demonstration project highlighted in a recent publication in ‘Trials’ illustrates the value of EHR data in complementing traditional data collection methods for a multi-center pharmaceutical industry outcomes trial.
This project showcased the essential support provided by a central coordinating center in addressing the technical, governance, and operational aspects vital for site involvement.
The project underscored several operational considerations crucial for site selection. These included assessing , determining site capabilities, and strategizing on data extraction. This approach facilitated the evaluation of site performance and ensured the integrity of data transfer and quality review.
The challenges and lessons learned from this initiative offer valuable insights into the best practices for planning and implementing site initiation, training, support for site personnel, and ongoing monitoring.
Furthermore, recent FDA guidance has emphasized the importance of including to ensure broad applicability of research findings and to address health disparities. This guidance has been well-received by the industry and reflects a cohesive strategy to improve trial diversity through careful site selection and .
In light of these advancements, it is imperative to share with stakeholders and the research community at large. This practice not only fosters knowledge sharing but also encourages future collaborations that can leverage the to streamline clinical trial processes, enhancing the overall impact of the study.
Best Practices and Future Directions
The landscape of is evolving with the integration of innovative models and data-driven strategies. One such transformative approach is the utilization of established community-based infrastructure, like Walgreens’ retail footprint, to bridge the accessibility gap. This initiative is poised to address healthcare disparities by offering within familiar community stores and pharmacies, thus enhancing participant diversity and representation.
Additionally, the advent of EHR-sourced trials promises to streamline the use of patient data, although it introduces complexities regarding trial site readiness and . A pivotal demonstration project highlighted the potential of EHR data to augment traditional data collection, with a central coordinating center playing a key role in navigating technical and operational challenges.
To further enhance , the industry is harnessing . This necessitates a robust data strategy to manage the deluge of information effectively. Critical considerations include predefined data management expectations, risk mitigation through data strategies, and the impact on trial sites.
Moreover, contemporary involves ethical and responsible use of artificial intelligence and machine learning. The NIH Pragmatic Trials Collaboratory’s recent workshop and Living Textbook chapter are invaluable resources for navigating the IRB approval process, data procurement, and consent considerations, all aimed at building equity-enhancing models.
, such as Equal Randomisation and Thompson Sampling, are also under the spotlight. These strategies influence the assignment of treatments to participants, highlighting the continuous need for methodological advancements to maximize statistical power and trial effectiveness.
In summary, the future of site selection for clinical trials lies in the convergence of community engagement, streamlined data utilization, advanced technologies, and ethical research practices. By embracing these directions, sponsors can significantly elevate the efficiency and outcome of clinical research.
Conclusion
Selecting the right clinical trial site is crucial for successful research. It impacts participant recruitment, data integrity, and research pace. The use of electronic health records (EHR) in trials poses unique challenges that sites must adapt to effectively integrate EHR data.
Patient access and equity are also vital considerations, as logistical hurdles can hinder participation. The evolving clinical trial landscape demands sites that can handle increasing study volumes and technological demands. Ultimately, site selection requires balancing technical requirements, participant needs, and operational capabilities.
Preparing for site selection involves aligning with study objectives, understanding the target population, evaluating geographic location, and abiding by regulatory requirements. Innovative patient assignment methods and integrating EHR data improve data quality and operational efficiency. Navigating site selection complexities requires assessing site feasibility, understanding patient populations, and ensuring adequate infrastructure and resources.
Patient accessibility and leveraging technology play key roles in site selection and trial participation.
Investigator and staff qualifications are crucial for upholding rigorous standards and ensuring participant safety. Patient recruitment and retention strategies significantly impact a trial’s power and validity. Regulatory compliance is essential to maintain integrity.
Upholding ethical conduct and data integrity safeguards participant rights and advances medical knowledge. Stakeholder engagement and transparent communication throughout the site selection process are imperative.
The landscape of clinical trial site selection is evolving with innovative models and data-driven strategies. Utilizing established community-based infrastructure and integrating EHR data enhance efficiency and data quality. Future site selection focuses on community engagement, streamlined data utilization, advanced technologies, and ethical research practices.
Selecting the right site is a critical step towards advancing medical knowledge and improving patient outcomes.
Choose bioaccess™ for expert clinical trial site selection in Latin America and accelerate your research success.
Frequently Asked Questions
Why is clinical trial site selection important?
Clinical trial site selection is crucial for the seamless execution and success of a clinical study. It affects participant recruitment and retention, the integrity and pace of the research, and ensures the ability to handle technological demands of modern trials.
What are some pre-selection considerations for choosing a clinical trial site?
Before selecting a site, it’s important to align with study objectives, understand the target population, evaluate geographic location, adhere to regulatory requirements, and consider diversity action plans (DAPs) for representative demographics, among other factors.
How do patient accessibility and technology impact site selection?
Patient convenience and the potential to use technology to alleviate barriers to participation are key. Sites must be chosen for their capacity to recruit participants and handle technological demands, such as managing electronic health records and sponsor-provided technology.
What is the role of a central coordinating center?
A central coordinating center provides technical and operational support to sites, ensuring that data, such as from electronic health records, is effectively integrated into the study design for high-quality data collection.
How do trials involving electronic health records (EHR) influence site selection?
Trials utilizing EHR data require sites that can integrate this data effectively into the study design. This involves considering the site’s infrastructure, resources, and experience with EHR-sourced trials.
What is the significance of infrastructure and resources in site selection?
A site’s infrastructure and resources are critical to meet the demands of a clinical trial. This includes the site’s ability to manage various systems and technologies integral to modern trials, such as electronic data capture and safety letter distribution systems.
How important are investigator and staff qualifications in site selection?
Highly. Investigators and staff should have expertise in the therapeutic area, understand study protocols, and have experience in research, ensuring patient safety and treatment efficacy.
What strategies can improve patient recruitment and retention?
Strategies include accessing a diverse patient population, incorporating patient-centric approaches, and leveraging technology like electronic health records and AI to facilitate inclusion and improve engagement.
Why is regulatory compliance and ethical conduct crucial in site selection?
Upholding regulatory compliance and ethical standards ensures the integrity of the research, the protection of participant rights, and the validity of the study’s data. This includes adherence to Good Clinical Practice (GCP) guidelines and informed consent processes.
How does stakeholder involvement and communication factor into site selection?
Effective stakeholder involvement ensures that expectations are aligned, concerns are addressed, and the trial infrastructure is set up for success. It includes collaboration between investigators, sponsors, ethics committees, and patient advocacy groups.
What role does documentation and transparency play in site selection?
Rigorous documentation and transparency in the site selection process ensure accountability and uphold the integrity of the research. It involves making protocols, analysis plans, and data available to maintain public trust.
How does the site selection process influence implementation and dissemination of trial outcomes?
Site selection affects the trial’s operationalization, including data extraction and quality review. Effective site selection and outcome dissemination share best practices and lessons learned, enhancing the impact of the study.
What are the best practices and future directions for clinical trial site selection?
Best practices include using community-based infrastructure for better accessibility, creating robust data strategies for managing information from digital sources, and ethically using advanced technologies like AI and ML. Future directions point towards increasing community engagement, streamlining data utilization, and embracing ethical research practices.
List of Sources
- Importance of Clinical Trial Site Selection
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- jamanetwork.com (https://jamanetwork.com/journals/jama/fullarticle/2819131)
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/counter/pdf/10.1186/s13063-023-07563-y)
- greenlight.guru (https://www.greenlight.guru/blog/selecting-a-site-for-your-clinical-trial)
- greenlight.guru (https://www.greenlight.guru/blog/how-to-set-up-clinical-studies-to-comply-with-us-fda-regulations)
- prnewswire.com (https://www.prnewswire.com/news-releases/wcg-announces-release-of-clinical-research-trends–insights-2024-report-302026408.html)
- hitconsultant.net (https://hitconsultant.net/2023/11/06/survey-shows-burdens-on-clinical-research-sites-sharply-increasing-since-2018/)
- biztoc.com (https://biztoc.com/t/biomedicalresearch)
- ncbi.nlm.nih.gov (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2656491/)
- Pre-Selection Considerations
- medtechintelligence.com (https://medtechintelligence.com/news_article/fda-publishes-long-awaited-clinical-trial-diversity-guidance/)
- forum.effectivealtruism.org (https://forum.effectivealtruism.org/posts/mZK974Whp6cPoyqLM/the-relative-ethicalness-of-clinical-trial-designs)
- ncbi.nlm.nih.gov (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2656491/)
- fortrea.com (https://www.fortrea.com)
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/counter/pdf/10.1186/s13063-023-07563-y)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- greenlight.guru (https://www.greenlight.guru/blog/how-to-set-up-clinical-studies-to-comply-with-us-fda-regulations)
- greenlight.guru (https://www.greenlight.guru/blog/selecting-a-site-for-your-clinical-trial)
- Assessing Feasibility and Resources
- ncbi.nlm.nih.gov (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2656491/)
- nature.com (https://www.nature.com/articles/d41586-024-00753-x)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- healthywomen.org (https://www.healthywomen.org/your-care/how-to-get-into-a-clinical-trial)
- ncats.nih.gov (https://ncats.nih.gov/research/research-resources/clinical-research-toolbox?utm_source=X&utm_medium=Social&utm_campaign=Research-Resources-Clinical)
- mckinsey.com (https://www.mckinsey.com/industries/life-sciences/our-insights/accelerating-clinical-trials-to-improve-biopharma-r-and-d-productivity)
- forum.effectivealtruism.org (https://forum.effectivealtruism.org/posts/mZK974Whp6cPoyqLM/the-relative-ethicalness-of-clinical-trial-designs)
- greenlight.guru (https://www.greenlight.guru/blog/selecting-a-site-for-your-clinical-trial)
- clinicaltrialsarena.com (https://www.clinicaltrialsarena.com/news/focus-clinical-research-uk-all-nhs/)
- clinicaltrialsarena.com (https://www.clinicaltrialsarena.com/news/uks-clinical-landscape-faces-new-reality-as-international-competition-heats-up/)
- medicaldevice-network.com (https://www.medicaldevice-network.com/news/clinical-trials-in-africa-where-there-is-a-challenge-there-is-an-opportunity/)
- greenlight.guru (https://www.greenlight.guru/blog/selecting-a-site-for-your-clinical-trial)
- Site Selection Criteria
- first10em.com (https://first10em.com/how-to-create-a-focused-and-answerable-research-question/)
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/counter/pdf/10.1186/s13063-023-07563-y)
- forum.effectivealtruism.org (https://forum.effectivealtruism.org/posts/mZK974Whp6cPoyqLM/the-relative-ethicalness-of-clinical-trial-designs)
- clinicaltrialsarena.com (https://www.clinicaltrialsarena.com/news/focus-clinical-research-uk-all-nhs/)
- clinicaltrialsarena.com (https://www.clinicaltrialsarena.com/news/uks-clinical-landscape-faces-new-reality-as-international-competition-heats-up/)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- ncbi.nlm.nih.gov (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC11017749/)
- journals.sagepub.com (https://journals.sagepub.com/author-instructions/CTJ#ArticleTypes)
- Evaluating Site Capabilities and Experience
- clinregs.niaid.nih.gov (https://clinregs.niaid.nih.gov/country/india#scope_of_assessment?utm_medium=social&utm_source=twitter&utm_campaign=clinregs_india_7122024)
- protoncenter.nm.org (https://www.protoncenter.nm.org/what-to-expect/clinical-trials?utm_source=twitter&utm_medium=social&utm_term=healthbeat&utm_campaign=non_brand_social_all_regions_fy20_consumer_organic)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- hitconsultant.net (https://hitconsultant.net/2024/07/10/solving-for-tech-overload-in-clinical-trials/)
- hitconsultant.net (https://hitconsultant.net/2023/11/21/clinical-trial-data-strategy-shifting-to-innovative-data-collection/)
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/counter/pdf/10.1186/s13063-023-07563-y)
- statnews.com (https://www.statnews.com/2024/06/14/walgreens-ramita-tandon-diversity-recruiting-customers-into-clinical-trials/?utm_campaign=rss)
- raps.org (https://www.raps.org/News-and-Articles/News-Articles/2024/6/Experts,-FDA-officials-discuss-future-of-clinical?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- rethinkingclinicaltrials.org (https://rethinkingclinicaltrials.org/news/january-8-2024-pragmatic-trials-researchers-share-lessons-from-collecting-patient-reported-outcomes-in-the-electronic-health-record/)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/counter/pdf/10.1186/s13063-023-07563-y)
- Assessing Site Infrastructure and Resources
- fortrea.com (https://www.fortrea.com)
- whitehouse.gov (https://www.whitehouse.gov/ostp/news-updates/2023/10/26/a-stronger-clinical-trial-infrastructure-for-better-health-outcomes/)
- nature.com (https://www.nature.com/articles/s41591-023-02665-1)
- hitconsultant.net (https://hitconsultant.net/2023/11/21/clinical-trial-data-strategy-shifting-to-innovative-data-collection/)
- ncats.nih.gov (https://ncats.nih.gov/research/research-resources/clinical-research-toolbox?utm_source=X&utm_medium=Social&utm_campaign=Research-Resources-Clinical)
- medtechintelligence.com (https://medtechintelligence.com/news_article/fda-publishes-long-awaited-clinical-trial-diversity-guidance/)
- hitconsultant.net (https://hitconsultant.net/2023/11/06/survey-shows-burdens-on-clinical-research-sites-sharply-increasing-since-2018/)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/counter/pdf/10.1186/s13063-023-07563-y)
- Investigator and Staff Qualifications
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/counter/pdf/10.1186/s13063-023-07598-1)
- nature.com (https://www.nature.com/articles/s41591-023-02665-1)
- scientia.global (https://www.scientia.global/mr-anthony-keyes-understanding-and-improving-clinical-trial-compliance/)
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/articles/10.1186/s13063-024-08216-4)
- nucats.northwestern.edu (https://www.nucats.northwestern.edu/about/news/2024/tnn-success.html)
- fda.gov (https://www.fda.gov/drugs/news-events-human-drugs/fda-clinical-investigator-training-course-citc-2023-12062023)
- fda.gov (https://www.fda.gov/regulatory-information/search-fda-guidance-documents/use-data-monitoring-committees-clinical-trials?utm_content=bufferf9e54&utm_medium=social&utm_source=twitter.com&utm_campaign=buffer)
- fda.gov (https://www.fda.gov/news-events/fda-voices/increasing-options-clinical-research-facilitate-medical-product-development)
- rethinkingclinicaltrials.org (https://rethinkingclinicaltrials.org/news/july-23-2024-article-from-ethics-and-regulatory-core-highlights-key-challenges-for-pragmatic-trials/)
- Patient Recruitment and Retention
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- pmlive.com (https://pmlive.com/blogs/how-patient-engagement-improves-outcomes-in-chronic-illness/)
- clinicaltrialsarena.com (https://www.clinicaltrialsarena.com/sponsored/why-patient-centricity-is-the-answer-to-the-clinical-trial-enrolment-gap/)
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/counter/pdf/10.1186/s13063-023-07563-y)
- nature.com (https://www.nature.com/articles/s41591-023-02665-1)
- hitconsultant.net (https://hitconsultant.net/2023/11/21/clinical-trial-data-strategy-shifting-to-innovative-data-collection/)
- journals.sagepub.com (https://journals.sagepub.com/author-instructions/CTJ#ArticleTypes)
- nihr.ac.uk (https://www.nihr.ac.uk/blog/how-weve-succeeded-in-recruiting-older-hospital-patients-onto-our-trial/36442)
- clinicaltrialsarena.com (https://www.clinicaltrialsarena.com/news/focus-clinical-research-uk-all-nhs/)
- Regulatory Compliance and Ethical Considerations
- rwmalonemd.substack.com (https://rwmalonemd.substack.com/p/indication-labeling-and-fraud)
- nature.com (https://www.nature.com/articles/d41573-024-00107-2)
- fda.gov (https://www.fda.gov/news-events/fda-voices/increasing-options-clinical-research-facilitate-medical-product-development)
- med-technews.com (https://www.med-technews.com/medtech-insights/medical-device-insights/improving-image-integrity-in-clinical-research/)
- pharmalive.com (https://www.pharmalive.com/balancing-innovation-with-patient-safety-navigating-regulatory-guidelines-in-clinical-research/)
- scientia.global (https://www.scientia.global/mr-anthony-keyes-understanding-and-improving-clinical-trial-compliance/)
- gao.gov (https://www.gao.gov/products/gao-24-106383?utm_medium=social&utm_source=twitter&utm_campaign=usgao)
- nam.edu (https://nam.edu/regenerative-medicine-case-study-for-understanding-and-anticipating-emerging-science-and-technology/)
- Stakeholder Involvement and Communication
- clinicaltrialsarena.com (https://www.clinicaltrialsarena.com/sponsored/why-patient-centricity-is-the-answer-to-the-clinical-trial-enrolment-gap/)
- greenlight.guru (https://www.greenlight.guru/blog/selecting-a-site-for-your-clinical-trial)
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/counter/pdf/10.1186/s13063-023-07563-y)
- researchinvolvement.biomedcentral.com (https://researchinvolvement.biomedcentral.com/counter/pdf/10.1186/s40900-023-00483-w)
- raps.org (https://www.raps.org/News-and-Articles/News-Articles/2024/6/Stakeholders-request-elaboration,-consistency-in-F?utm_campaign=regulatory-focus&utm_source=twitter&utm_medium=social)
- raps.org (https://www.raps.org/news-and-articles/news-articles/2024/5/stakeholders-welcome-fda-s-informed-consent-%E2%80%98key-i?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- jamanetwork.com (https://jamanetwork.com/journals/jama/article-abstract/2820595)
- jamanetwork.com (https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2811814)
- fda.gov (https://www.fda.gov/regulatory-information/search-fda-guidance-documents/use-data-monitoring-committees-clinical-trials?utm_content=bufferf9e54&utm_medium=social&utm_source=twitter.com&utm_campaign=buffer)
- researchinvolvement.biomedcentral.com (https://researchinvolvement.biomedcentral.com/counter/pdf/10.1186/s40900-023-00483-w)
- medtechintelligence.com (https://medtechintelligence.com/feature_article/from-concept-to-market-the-strategic-journey-from-ind-to-nda/)
- Documentation and Transparency
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/counter/pdf/10.1186/s13063-023-07563-y)
- medpagetoday.com (https://www.medpagetoday.com/opinion/second-opinions/107038)
- jamanetwork.com (https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2811814)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- clinicaltrialsarena.com (https://www.clinicaltrialsarena.com/sponsored/why-patient-centricity-is-the-answer-to-the-clinical-trial-enrolment-gap/)
- siteselection.com (https://siteselection.com/issues/2024/jan/24-for-24.cfm)
- raps.org (https://www.raps.org/News-and-Articles/News-Articles/2024/6/Stakeholders-request-elaboration,-consistency-in-F?utm_campaign=regulatory-focus&utm_source=twitter&utm_medium=social)
- cancerletter.com (https://cancerletter.com/clinical-roundup/20240112_8a/)
- Implementation and Dissemination
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/counter/pdf/10.1186/s13063-023-07563-y)
- clinicaltrialsarena.com (https://www.clinicaltrialsarena.com/interviews/diversity-fda-guidance-reaction/)
- cms.gov (https://www.cms.gov/medicare/quality/measures/national-impact-assessment)
- fda.gov (https://www.fda.gov/drugs/drug-approvals-and-databases/about-postmarketing-requirements-and-commitments-searchable-database)
- c-dias.org (https://www.c-dias.org/implementation-guides-and-measures/#partner-guide)
- nature.com (https://www.nature.com/articles/s41591-023-02665-1)
- forum.effectivealtruism.org (https://forum.effectivealtruism.org/posts/mZK974Whp6cPoyqLM/the-relative-ethicalness-of-clinical-trial-designs)
- cares-blog.gse.harvard.edu (https://cares-blog.gse.harvard.edu/post/plotting-distributions/)
- Best Practices and Future Directions
- myemail.constantcontact.com (https://myemail.constantcontact.com/NIH-Pragmatic-Trials-Collaboratory-Newsletter—November-2023.html?soid=1130073635641&aid=WuEh_IKNgJ4)
- forum.effectivealtruism.org (https://forum.effectivealtruism.org/posts/mZK974Whp6cPoyqLM/the-relative-ethicalness-of-clinical-trial-designs)
- walgreens.com (https://www.walgreens.com/health-services/clinical-trials)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- hitconsultant.net (https://hitconsultant.net/2023/11/21/clinical-trial-data-strategy-shifting-to-innovative-data-collection/)
- statnews.com (https://www.statnews.com/2024/06/14/walgreens-ramita-tandon-diversity-recruiting-customers-into-clinical-trials/?utm_campaign=rss)
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/counter/pdf/10.1186/s13063-023-07563-y)