Introduction
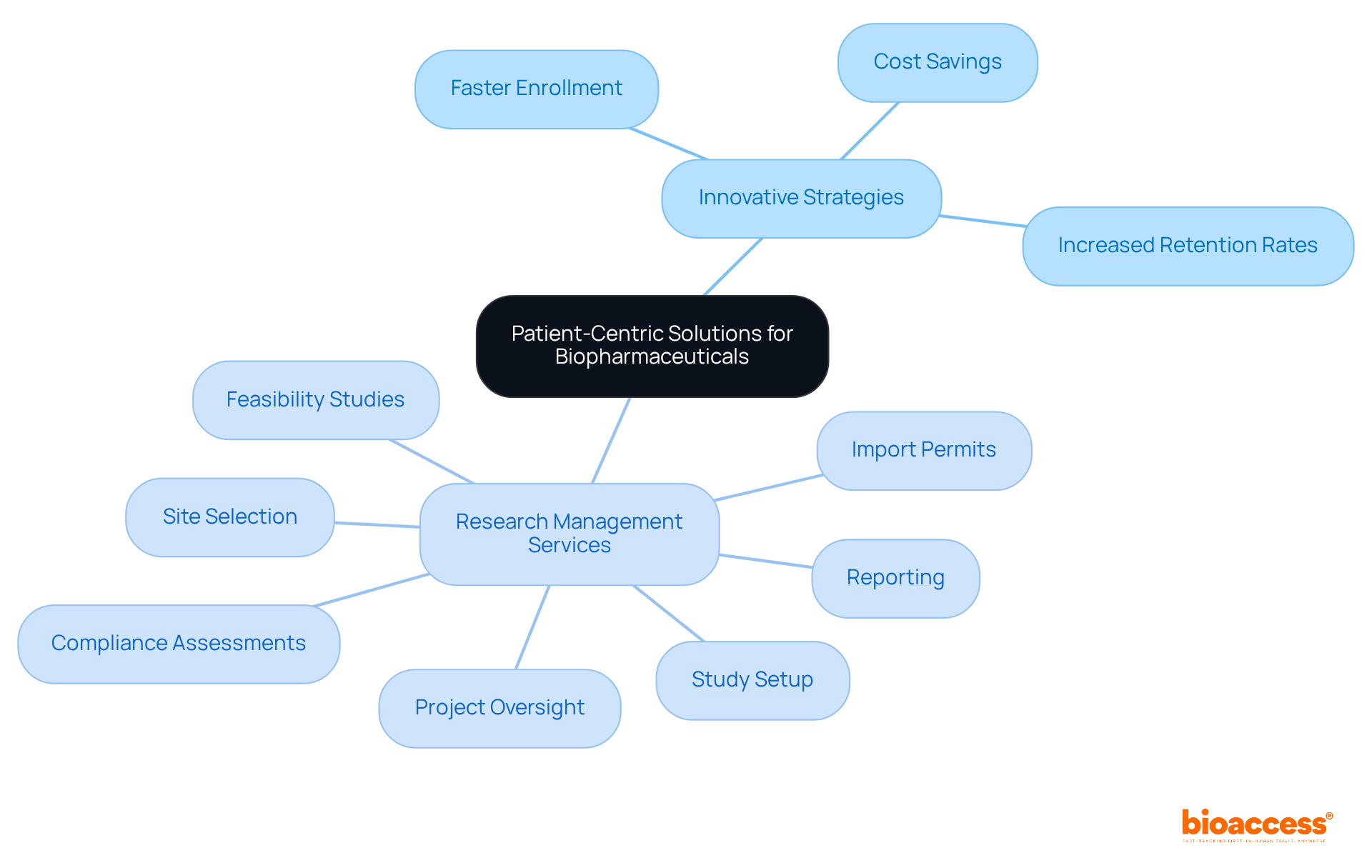
ICON plc stands as a paragon in the clinical trial landscape, bringing over three decades of expertise to the intricate process of medical research. Renowned for their impeccable standards of excellence, ICON has carved out a niche in various therapeutic domains, notably oncology, cardiovascular disorders, and neurosciences.
Their comprehensive suite of services caters to the entire spectrum of new therapy development, echoing the pioneering spirit of CMIC Group, which established the Contract Research Organization model in Japan. CMIC has redefined industry standards, offering an array of integrated services from contract development to Japan market entry solutions.
ICON shares this ethos of innovation and adaptability, ensuring they meet the unique demands of their clients, whether pharmaceutical firms, medical device producers, or academic institutions. This dedication is exemplified by the logistical challenges faced by patients, like the one from rural Pennsylvania who confronts the daunting task of participating in a trial abroad. ICON’s commitment to facilitating such complex scenarios underscores their role as a crucial ally in the quest to advance medical science and deliver lifesaving treatments.
ICON plc: Leading Clinical Research Organisation
ICON plc stands as a paragon in the landscape, bringing over three decades of expertise to the intricate process of . Renowned for their impeccable standards of excellence, ICON has carved out a niche in various , notably oncology, cardiovascular disorders, and neurosciences.
Their comprehensive suite of services caters to the entire spectrum of , echoing the pioneering spirit of CMIC Group, which established the model in Japan. CMIC has redefined industry standards, offering an array of integrated services from contract development to Japan market entry solutions.
ICON shares this ethos of innovation and adaptability, ensuring they meet the unique demands of their clients, whether pharmaceutical firms, medical device producers, or academic institutions. This dedication is exemplified by the faced by patients, like the one from rural Pennsylvania who confronts the daunting task of participating in a trial abroad. ICON’s commitment to facilitating such complex scenarios underscores their role as a crucial ally in the quest to advance medical science and deliver lifesaving treatments.
Duke Clinical Research Institute: Innovative Research for Better Health
The Duke Clinical Research Institute (DCRI) is a renowned academic research organization dedicated to conducting innovative and studies. With a focus on improving patient health outcomes, DCRI collaborates with leading experts from around the world to drive advancements in . Their multidisciplinary approach and state-of-the-art facilities make them a top choice for conducting cutting-edge .
Rho: Changing the Face of Clinical Trials
like Rho are at the forefront of transforming the landscape of by integrating cutting-edge technologies and . They are adept at managing the deluge of data that modern clinical trials produce—an average of 3.6 million data points in Phase 3 trials, which is a threefold increase from a decade ago.
The rapid doubling of medical data every 70 days, compared to 50 years in 1950 and 7 years in 1980, necessitates sophisticated data analytics and real-time monitoring systems. Rho capitalizes on these technologies to refine study protocols and improve the processes of .
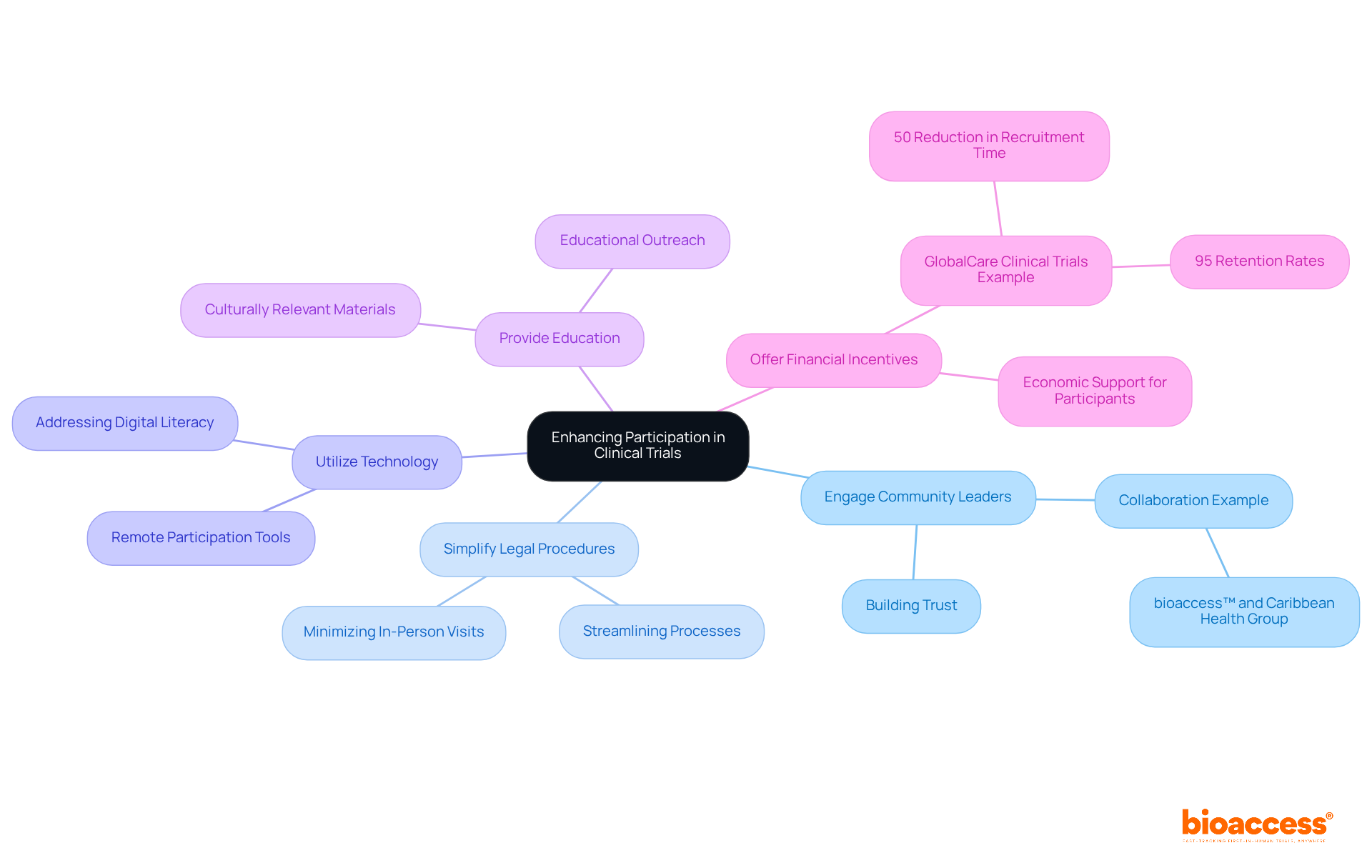
Their approach not only enhances the efficacy of the trials but also addresses the complexities faced by patients, such as those in remote locations with limited access to trial sites. By optimizing the clinical trial framework, Rho is making strides in ensuring that even patients with ultra-rare diseases have the opportunity to participate in , regardless of geographical barriers. This is an innovative leap forward in making life-saving treatments more accessible.
Medpace: Accelerating Drug Development
Medpace stands out as a that not only accelerates drug development but also navigates the intricate landscape of . Their comprehensive suite of services encompasses , regulatory support, and medical affairs, addressing the multifaceted challenges of bringing new therapies to market. Medpace’s dedication to collaboration and quality has solidified their reputation within the pharmaceutical sector.
Integral to their success is their understanding of the critical role such as the play in the drug development continuum. The FDA’s exhaustive evaluation of , manufacturing standards, and market analysis is a testament to the rigorous path that Medpace adeptly guides its clients through. Their ability to adapt to the varying requirements of regulatory authorities across global markets, like the in Europe, further exemplifies their expertise and commitment to advancing healthcare innovations safely and effectively.
PAREXEL International: Driving Innovation in Clinical Research
PAREXEL International is a global biopharmaceutical services company that specializes in providing comprehensive . With their extensive experience and expertise, PAREXEL helps clients navigate the complex and accelerate the development of . Their innovative technologies and data-driven insights drive efficiency and improve study outcomes.
QuintilesIMS: Advancing Healthcare through Research
IQVIA, formerly known as Quintiles IMS, stands at the forefront of , offering comprehensive services that extend from to and the generation of . Their expertise is particularly crucial in the era of , which informs in the United States by evaluating the clinical and economic implications of healthcare interventions.
IQVIA’s global presence and robust infrastructure are instrumental in executing research that aligns with HTA parameters—cost, safety, and efficacy—thereby influencing treatment decisions and healthcare costs. In parallel, CMIC Group, Japan’s pioneering Contract Research Organization (CRO), exemplifies industry innovation by providing end-to-end solutions across the pharmaceutical value chain. IQVIA’s and CMIC’s collaborative efforts with stakeholders underscore the evolving landscape of healthcare, where patient quality of life and meticulous research converge to shape future therapeutic directions.
Covance: Delivering Excellence in Clinical Development
Covance stands at the forefront of clinical development, with a commitment deeply rooted in delivering services that embody scientific acumen and operational finesse. Recognizing that decisions made early in the research process can profoundly affect outcomes, they invest significant time and energy into meticulous planning and execution of . This approach is informed by an understanding that nearly 80% of decisions could be optimized with .
Covance’s suite of services, from to , is tailored to address the complex challenges of . They interlink each aspect of a trial, akin to a chain’s links, to ensure seamless progression and integrity throughout the study duration. This comprehensive method not only streamlines the process but also aims to bridge the gap between and , a critical issue highlighted by experts.
According to a special communication in JAMA, the disconnection between trialists and clinicians often leads to inefficiencies, limiting the trials’ scope and impact. By aligning their trials with , Covance contributes to reducing the that persists despite thousands of registered RCTs. Their data-driven approach, leveraging statistics and user interaction analytics, further enables them to refine their services, ensuring they resonate with their intended audience and meet the highest standards of security, fraud prevention, and technical compatibility.
Syneos Health: Accelerating Biopharmaceutical Innovation
has emerged as a pivotal player in the , combining , advanced commercialization strategies, and insightful consulting services. Their commitment to expediting the journey from drug discovery to delivery is reshaping how the industry approaches the intricate . With a reputation for integrating services seamlessly and harnessing their extensive therapeutic knowledge, Syneos Health stands out as a strategic ally, propelling medical research forward.
The concerns raised by industry leaders about the potential conflicts of interest within large consulting firms underscore the necessity for an unbiased, holistic approach to . This perspective highlights the importance of academic involvement and a granular understanding of industry challenges to foster innovation and enhance the efficiency of . Syneos Health’s model appears to address these concerns by providing a balanced, client-focused partnership that is attentive to the nuanced needs of stakeholders.
Charles River Laboratories: Supporting Drug Discovery and Development
Charles River Laboratories exemplifies the fusion of scientific acumen with cutting-edge technology, serving as a pivotal ally in the realm of . Their extensive suite of offerings encompasses not only research models and preclinical services but also , facilitating pharmaceutical and biotech entities in their quest to forge new .
Echoing the innovative spirit of physicist Mario Krenn at the Max Planck Institute for the Science of Light, Charles River harnesses as a catalyst for scientific creativity. Krenn’s AI-driven approach, which led to the discovery of a quantum entanglement experiment, mirrors the forward-thinking methodologies employed by Charles River. Their commitment to leveraging advanced technologies ensures the realization of complex research endeavors and the continual progression of medical science.
PPD: Driving Clinical Excellence
PPD stands as a global beacon in the realm of clinical research, offering comprehensive services that extend from managing to navigating the intricacies of . Their expertise in is pivotal in ensuring the success of . PPD’s unwavering dedication to quality and innovation has not only earned them the trust of pharmaceutical and biotechnology companies across the globe but also cemented their status as a preferred partner in the industry.
plays a critical role in the services offered by PPD. HTA involves a thorough analysis of the clinical and economic implications of , considering factors such as cost, safety, and efficacy. This approach is integral to shaping treatment protocols and determining the allocation of healthcare costs, thereby influencing decisions made by a broad spectrum of stakeholders, including employers, payers, and policymakers.
Reflecting on the evolution of the industry, it’s evident that despite a contraction in the sector over the last five years, the demand for has grown. This underscores the industry’s crucial function in managing risks for individuals, covering costs associated with illness, injury, and death. As the industry navigates these challenges, companies like PPD are at the forefront, leveraging their expansive services to meet the diverse needs of their clients and support the development of innovative healthcare solutions.
Conclusion
In conclusion, ICON plc stands as a leading clinical research organization with over three decades of expertise in the field. Renowned for their impeccable standards of excellence, ICON has carved out a niche in various therapeutic domains, catering to the entire spectrum of new therapy development.
Their commitment to innovation and adaptability ensures they meet the unique demands of their clients, facilitating complex scenarios and advancing medical science. The article highlights other key players in the clinical research landscape, such as the Duke Clinical Research Institute (DCRI), Rho, Medpace, PAREXEL International, QuintilesIMS (now IQVIA), Covance, Syneos Health, Charles River Laboratories, and PPD.
Each organization brings its own strengths and expertise to the table, driving advancements in medical research and accelerating drug development. These organizations employ cutting-edge technologies, data-driven methodologies, and multidisciplinary approaches to address the challenges faced by patients and researchers alike.
They navigate regulatory complexities and collaborate with stakeholders to ensure the safety and efficacy of healthcare interventions. The evolving landscape of healthcare requires a patient-centric approach to clinical trials. These organizations strive to make life-saving treatments more accessible by overcoming geographical barriers and incorporating real-world evidence into their research. Overall, these clinical research organizations play a crucial role in advancing medical science and delivering innovative healthcare solutions. Their dedication to excellence, collaboration, and patient-centricity propels the industry forward and paves the way for future therapeutic directions.
Frequently Asked Questions
What is a Clinical Research Organization (CRO)?
A Clinical Research Organization (CRO) is a company that provides support to the pharmaceutical, biotechnology, and medical device industries in the form of research services outsourced on a contract basis. CROs offer a range of services including clinical trial management, regulatory support, and data analysis.
What are the main services provided by CROs like ICON plc and Medpace?
CROs like ICON plc and Medpace provide comprehensive services that encompass the entire spectrum of new therapy development, clinical trial management, regulatory support, medical affairs, and market entry solutions.
What therapeutic areas does ICON plc specialize in?
ICON plc specializes in various therapeutic domains, notably oncology, cardiovascular disorders, and neurosciences.
How does ICON plc approach logistical challenges faced by patients in clinical trials?
ICON plc is committed to facilitating complex scenarios for patients, including those in remote locations, to ensure they can participate in trials and contribute to advancing medical science.
What is the focus of the Duke Clinical Research Institute (DCRI)?
DCRI focuses on conducting innovative clinical trials and studies with the goal of improving patient health outcomes. They collaborate with experts worldwide and employ a multidisciplinary approach.
What is unique about Rho’s approach to clinical trials?
Rho integrates cutting-edge technologies and data-driven methodologies to manage the large volume of data produced by modern clinical trials. They focus on refining study protocols and improving patient recruitment and retention processes.
How does Medpace handle the regulatory complexities of drug development?
Medpace navigates the regulatory landscape by offering regulatory support and adapting to the requirements of various regulatory authorities, such as the FDA and EMA, to advance healthcare innovations safely and effectively.
What role does PAREXEL International play in clinical research?
PAREXEL International specializes in providing comprehensive clinical research solutions and helps clients accelerate the development of new therapies with their innovative technologies and data-driven insights.
How does IQVIA contribute to healthcare research?
IQVIA advances healthcare research by offering services that range from clinical trial design to patient recruitment and generating real-world evidence. They also play a crucial role in Health Technology Assessment (HTA).
What is Covance’s approach to clinical development?
Covance invests in meticulous planning and execution of clinical trials, aiming to ensure seamless progression and integrity throughout the study, and aligning their trials with clinical practice.
How does Syneos Health accelerate biopharmaceutical innovation?
Syneos Health combines clinical research, advanced commercialization strategies, and consulting services to expedite the journey from drug discovery to delivery, focusing on integrating services and therapeutic knowledge.
What services does Charles River Laboratories provide?
Charles River Laboratories provides research models, preclinical services, clinical support, and leverages artificial intelligence to aid in drug discovery and development.
What expertise does PPD offer in clinical research?
PPD offers comprehensive services including clinical trial management and regulatory affairs expertise. They are also pivotal in site selection for clinical studies and are involved in Health Technology Assessment (HTA).
What is the significance of Health Technology Assessment (HTA) in clinical research?
HTA involves evaluating the clinical and economic implications of healthcare interventions, which influences treatment decisions and healthcare costs, shaping the allocation of resources across the healthcare sector.
List of Sources
- ICON plc: Leading Clinical Research Organisation
- en.cmicgroup.com (https://en.cmicgroup.com)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- Rho: Changing the Face of Clinical Trials
- medtechintelligence.com (https://medtechintelligence.com/column/the-power-of-ai-to-enhance-clinical-trials/)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- Medpace: Accelerating Drug Development
- forbes.com (https://www.forbes.com/sites/alexzhavoronkov/2023/08/09/616-billion-per-drug-approval-almost-half-of-big-pharma-companies-hit-negative-rd-productivity/)
- pharmaoffer.com (https://pharmaoffer.com/blog/how-long-does-it-take-to-bring-new-medicines-to-the-market/)
- QuintilesIMS: Advancing Healthcare through Research
- valueresearch.org (https://valueresearch.org)
- en.cmicgroup.com (https://en.cmicgroup.com)
- Covance: Delivering Excellence in Clinical Development
- biospace.com (https://www.biospace.com/article/treehill-partners-ali-pashazadeh-on-how-to-improve-clinical-trial-design/?utm_source=dlvr.it&utm_medium=twitter)
- raps.org (https://www.raps.org/News-and-Articles/News-Articles/2024/6/Experts,-FDA-officials-discuss-future-of-clinical?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- iqvia.com (https://www.iqvia.com/form-pages/institute-gated?redirectUrl=%2f-%2fmedia%2fiqvia%2fpdfs%2finstitute-reports%2fglobal-trends-in-r-and-d-2023%2fiqvia-institute-global-trends-in-rd-2023-forweb.pdf&title=IQVIA+Institute+Global+Trends+in+RD+2023+forWeb)
- Syneos Health: Accelerating Biopharmaceutical Innovation
- forbes.com (https://www.forbes.com/sites/alexzhavoronkov/2023/08/09/616-billion-per-drug-approval-almost-half-of-big-pharma-companies-hit-negative-rd-productivity/)
- Charles River Laboratories: Supporting Drug Discovery and Development
- nationalacademies.org (https://www.nationalacademies.org/news/2023/11/how-ai-is-shaping-scientific-discovery)
- PPD: Driving Clinical Excellence
- valueresearch.org (https://valueresearch.org)
- en.cmicgroup.com (https://en.cmicgroup.com)
- ibisworld.com (https://www.ibisworld.com/global/industry-trends/biggest-industries-by-revenue/)
- iqvia.com (https://www.iqvia.com/form-pages/institute-gated?redirectUrl=%2f-%2fmedia%2fiqvia%2fpdfs%2finstitute-reports%2fglobal-trends-in-r-and-d-2023%2fiqvia-institute-global-trends-in-rd-2023-forweb.pdf&title=IQVIA+Institute+Global+Trends+in+RD+2023+forWeb)