Introduction
Navigating the regulatory landscape for clinical trials in Chile can be daunting, yet it holds the key to unlocking swift approvals for researchers. To achieve fast clinical trial approval, understanding the intricate framework established by the Instituto de Salud Pública (ISP) is essential. Under optimal conditions, this process can take as little as 30 days.
However, how can sponsors effectively streamline their processes and enhance patient recruitment to ensure compliance while maximizing efficiency? Let’s explore a step-by-step guide to mastering the approval process, preparing necessary documentation, and implementing effective recruitment strategies in the dynamic landscape of clinical research in Chile.
Understand Regulatory Framework for Clinical Trials in Chile
Navigating the complex regulatory landscape for fast clinical trial approval in Chile can be daunting, yet it is essential for successful research outcomes. To effectively carry out clinical trials in Chile, it is crucial to navigate the regulatory framework established by the Instituto de Salud Pública (ISP) to ensure fast clinical trial approval in Chile. This procedure usually requires fast clinical trial approval in Chile, which typically takes between 30 to 90 days. Here are key regulations that can simplify your approval process:
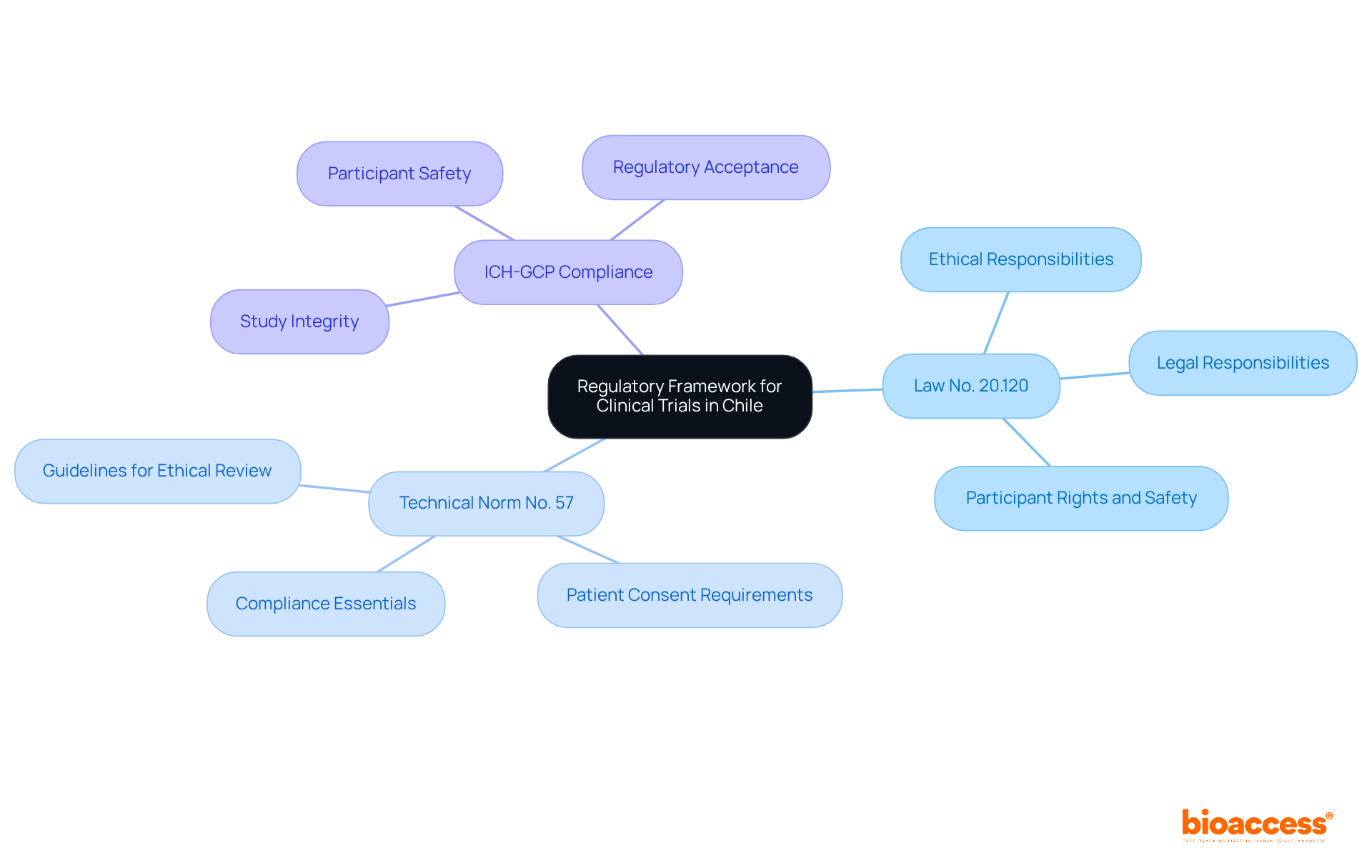
- Law No. 20.120: Regulates studies in Chile, detailing the ethical and legal responsibilities for carrying out research involving human participants, safeguarding their rights and safety.
- Technical Norm No. 57: Offers extensive guidelines on the conduct of research studies, outlining requirements for ethical review and patient consent, which are essential for compliance.
- ICH-GCP Compliance: Adherence to the International Council for Harmonisation Good Clinical Practice (ICH-GCP) guidelines is critical for maintaining study integrity and participant safety, essential for regulatory acceptance.
Partnering with bioaccess® ensures fast clinical trial approval in Chile, allowing you to secure ethics approvals in just 4-8 weeks, much quicker than the usual timelines in the US and EU. Moreover, bioaccess® provides cost efficiencies and access to treatment-naïve patient populations, boosting your study’s success in Latin America. By understanding these regulations and leveraging bioaccess®, you position your study for unparalleled success in the Latin American market.
Prepare Required Documentation and Preclinical Data
Navigating the regulatory landscape in clinical research requires meticulous preparation and attention to detail. To ensure a smooth approval process with the Instituto de Salud Pública (ISP) in Chile, follow these essential steps:
-
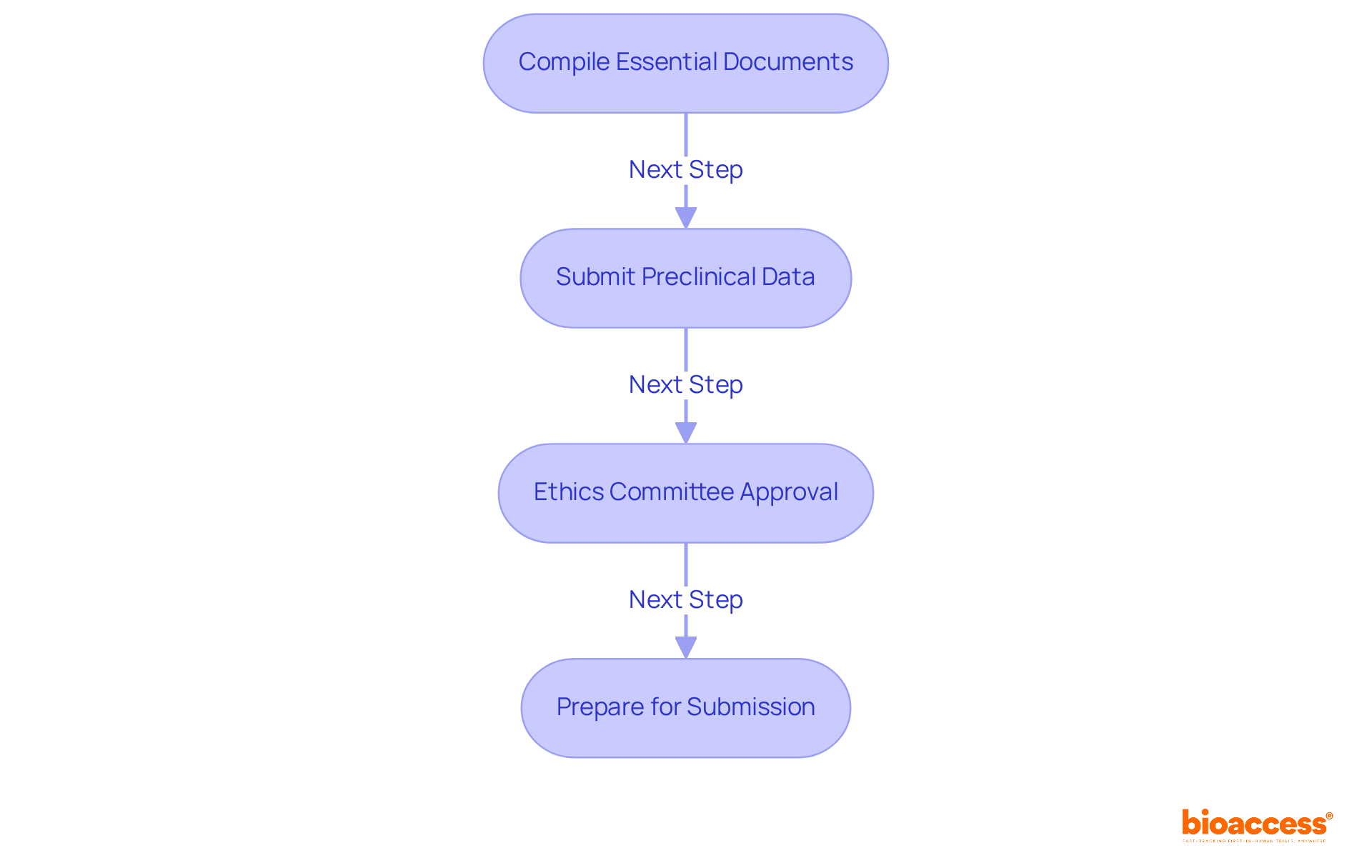
Compile Essential Documents: Gather the following key documents:
- A clinical study protocol that outlines the trial design, objectives, and methodology.
- Informed consent forms that adhere to local regulations and ethical standards.
- Investigator qualifications and detailed site information.
- Comprehensive preclinical data demonstrating the safety and efficacy of the investigational product.
-
Submit Preclinical Data: Have you considered how crucial this data is for establishing the safety profile of your investigational product before moving to human studies? Ensure that all relevant preclinical findings are included in your submission to the ISP.
-
Ethics Committee Approval: Obtaining endorsement from a recognized Ethics Committee (Comité de Ética) linked to the research site is vital. This approval is a prerequisite for submitting your application to the ISP, and failure to secure it could lead to application rejection.
-
Prepare for Submission: All documents must be translated into Spanish and formatted according to ISP requirements. A well-organized submission package not only expedites the review process but also boosts your chances of securing timely approval.
Implement Effective Patient Recruitment Strategies
Navigating the complexities of patient recruitment in Chile is crucial for the success of clinical studies, yet many sponsors face significant challenges in this area. Here are several strategies to enhance your recruitment efforts:
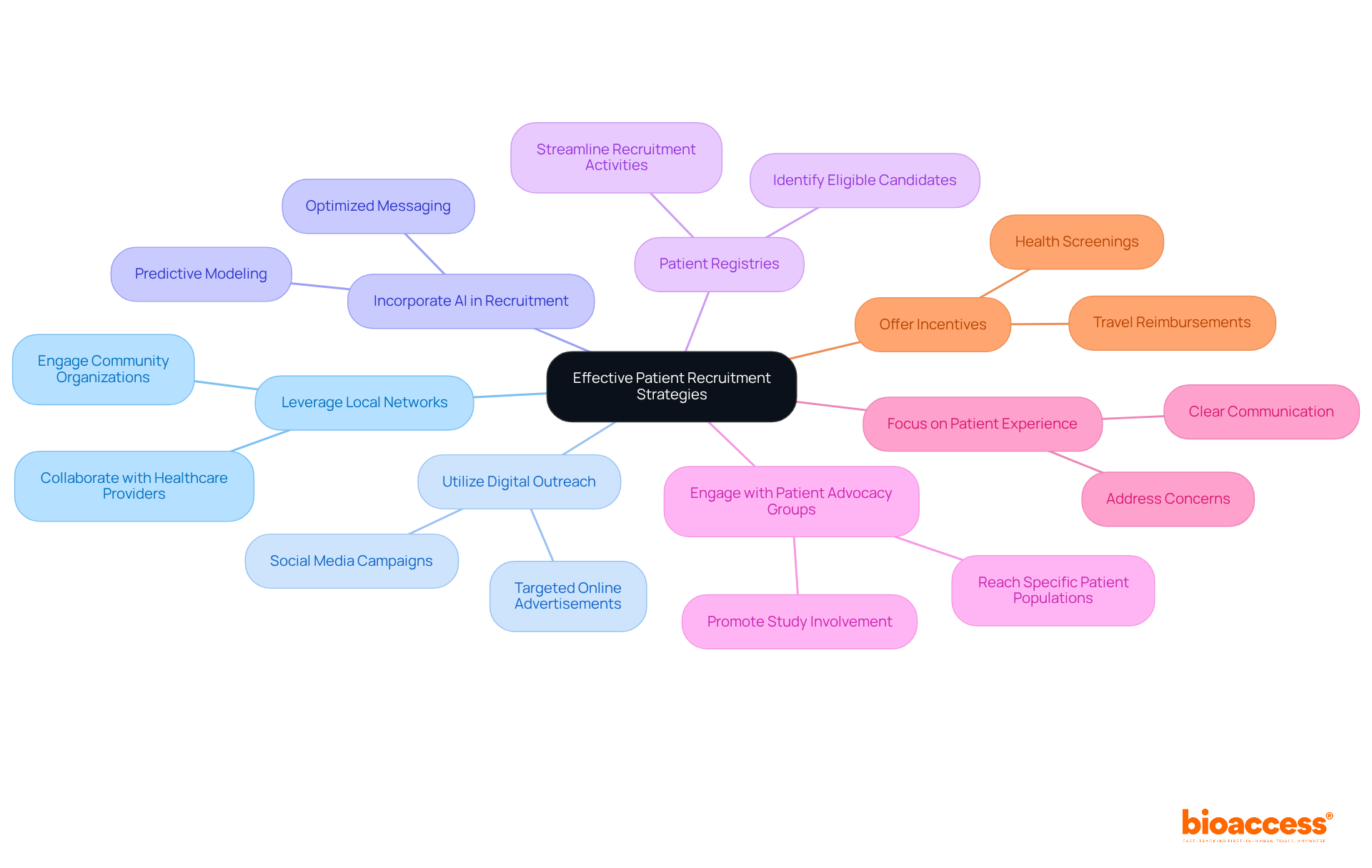
- Leverage Local Networks: Collaborate with local healthcare providers and community organizations to raise awareness about the study. Interacting with the community fosters trust and promotes involvement, which is vital for fulfilling compliance standards established by authorities such as the Instituto de Salud Pública (ISP).
- Utilize Digital Outreach: Implement digital marketing strategies, including social media campaigns and targeted online advertisements, to reach potential participants. How can you emphasize the advantages of involvement and supply clear details regarding the study procedure? This strategy can really boost visibility and engagement, particularly in urban areas where digital access is widespread.
- Incorporate AI in Recruitment: Utilize AI-driven predictive modeling to enhance recruitment strategies. AI can help identify eligible participants more efficiently and optimize messaging to resonate with potential recruits, thereby improving enrollment rates.
- Patient Registries: Utilize existing patient registries to identify eligible candidates efficiently. This can streamline recruitment activities and reduce the time needed to find suitable participants, aligning with the fast clinical trial approval Chile offers, which can be as short as 30 days under the right conditions.
- Engage with Patient Advocacy Groups: Partner with patient advocacy organizations to reach specific patient populations. These groups can assist in spreading information about the study and promoting involvement among their members, ensuring adherence to ethical standards and improving recruitment efficiency.
- Focus on Patient Experience: Prioritize improving patient experiences throughout the recruitment process. This includes clear communication, addressing concerns, and providing support to potential participants, which can lead to higher retention rates and overall satisfaction.
- Offer Incentives: Consider providing incentives for participation, such as travel reimbursements or health screenings, to motivate potential participants to enroll in the study. This not only improves recruitment rates but also aligns with patient-centered approaches that are increasingly emphasized in regulatory frameworks.
Embracing these strategies not only streamlines recruitment but also fosters a more patient-centered approach, ultimately enhancing the integrity of clinical research in Chile.
Execute the Clinical Trial with Compliance and Efficiency
Executing a clinical study effectively is not just important; it’s essential for ensuring compliance and safeguarding patient welfare. Follow these steps to ensure compliance and efficiency:
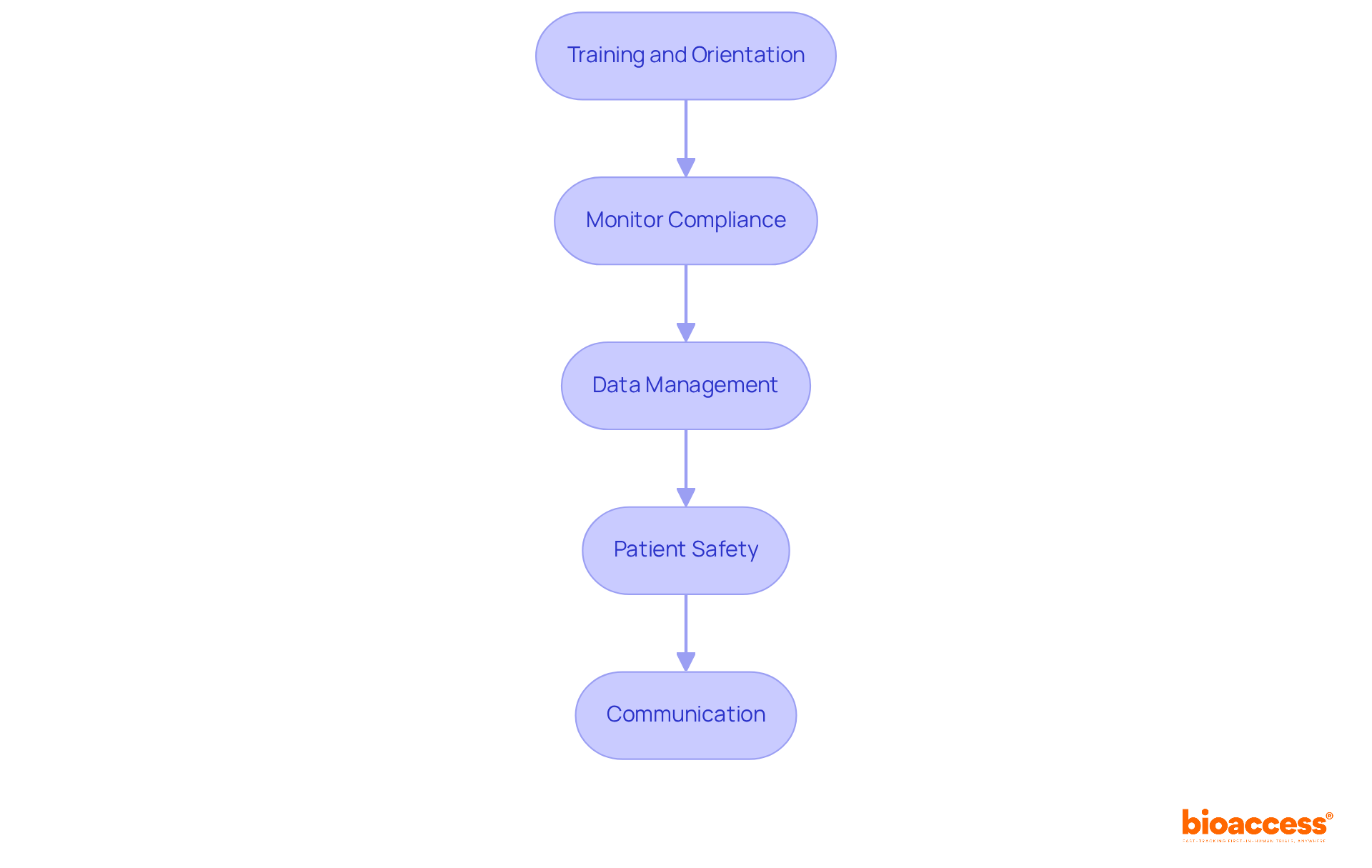
- Training and Orientation: Have you conducted comprehensive training sessions for all research staff? This ensures everyone understands the protocol and compliance requirements, which is vital for upholding ICH-GCP standards.
- Monitor Compliance: Implement robust monitoring processes to ensure adherence to ICH-GCP guidelines and local regulations, such as those set by INVIMA in Colombia or ANVISA in Brazil. Regular audits and compliance checks can help identify and address any issues early, minimizing risks associated with mismanagement of the study. Mismanagement can lead to costly delays and jeopardize patient safety.
- Data Management: Establish a comprehensive data management plan to ensure the accuracy and integrity of collected data. Utilizing electronic data capture systems can streamline data collection and reporting, facilitating compliance with ICH-GCP documentation standards.
- Patient Safety: Prioritize patient safety throughout the study. Diligently observe participants for adverse events and ensure that any concerns are reported swiftly to the Institutional Review Board (IRB) and Ethics Committee, following local compliance requirements. Neglecting patient safety can result in severe ethical and legal repercussions.
- Communication: Maintain open channels of dialogue with all stakeholders, including sponsors, authorities, and study participants. Frequent updates and clear reporting build trust and enable smoother study operations, which is crucial for achieving fast clinical trial approval in Chile.
With bioaccess®’s expertise, you can achieve cost-effective clinical research solutions, including $25K savings per patient, 30% lower overall study costs, and 50% faster enrollment through pre-qualified networks and pre-negotiated site contracts. Additionally, our Global Trial Accelerators™ provide essential insights into regulatory updates and market access strategies, ensuring your trial is positioned for success. With bioaccess® at your side, you’re not just conducting a study; you’re paving the way for groundbreaking advancements in clinical research.
Conclusion
Navigating the regulatory landscape in Chile can be daunting, yet it holds the key to unlocking rapid clinical trial approvals in a burgeoning market. By understanding the regulatory framework, preparing the necessary documentation, and implementing effective patient recruitment strategies, researchers can significantly expedite the approval process and enhance the overall quality of their clinical trials.
Key insights from this guide highlight the importance of adhering to local regulations, such as:
- Law No. 20.120
- Technical Norm No. 57
- ICH-GCP compliance
Thorough preparation of essential documents and preclinical data, coupled with proactive engagement in patient recruitment, can streamline the approval process and ensure ethical standards are met. Utilizing local networks, digital outreach, and patient advocacy groups can further enhance recruitment efforts, ultimately leading to a more efficient and effective clinical trial execution.
By adopting these strategies, researchers can not only expedite their trials but also drive significant advancements in patient care and treatment options.
Frequently Asked Questions
What is the regulatory framework for clinical trials in Chile?
The regulatory framework for clinical trials in Chile is primarily established by the Instituto de Salud Pública (ISP) and includes key regulations such as Law No. 20.120, which outlines ethical and legal responsibilities for research involving human participants, and Technical Norm No. 57, which provides guidelines on ethical review and patient consent.
How long does it typically take to get clinical trial approval in Chile?
The typical timeline for fast clinical trial approval in Chile ranges from 30 to 90 days.
What is Law No. 20.120?
Law No. 20.120 regulates studies in Chile, detailing the ethical and legal responsibilities for conducting research with human participants, ensuring their rights and safety are protected.
What does Technical Norm No. 57 cover?
Technical Norm No. 57 offers extensive guidelines on the conduct of research studies, including requirements for ethical review and patient consent, which are essential for compliance during clinical trials.
Why is ICH-GCP compliance important in Chile?
Adherence to the International Council for Harmonisation Good Clinical Practice (ICH-GCP) guidelines is critical for maintaining the integrity of the study and ensuring participant safety, which is essential for regulatory acceptance in Chile.
How can partnering with bioaccess® benefit clinical trials in Chile?
Partnering with bioaccess® can expedite the clinical trial approval process in Chile, allowing for ethics approvals in just 4-8 weeks, which is significantly quicker than typical timelines in the US and EU. Additionally, bioaccess® provides cost efficiencies and access to treatment-naïve patient populations, enhancing the success of studies in Latin America.
What advantages does conducting clinical trials in Latin America offer?
Conducting clinical trials in Latin America offers advantages such as faster approval timelines, cost efficiency, better patient recruitment opportunities, and streamlined regulatory pathways, making it an attractive region for early-stage clinical trials.
List of Sources
- Understand Regulatory Framework for Clinical Trials in Chile
- Number of clinical trials registered in the Chilean Institute of Public… (https://researchgate.net/figure/Number-of-clinical-trials-registered-in-the-Chilean-Institute-of-Public-Health-ISP_tbl1_351504068)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOooyu85GFsjIf3kSx5Tkgbd9ylUbPQzVCWVX6U_AGJGQ7OlbqUnJ)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- 4 Best Practices for First in Human Biopharma Trials in Chile | bioaccess® (https://bioaccessla.com/blog/4-best-practices-for-first-in-human-biopharma-trials-in-chile)
- Prepare Required Documentation and Preclinical Data
- Preclinical Development: The Safety Hurdle Prior to Human Trials (https://americanpharmaceuticalreview.com/Featured-Articles/187349-Preclinical-Development-The-Safety-Hurdle-Prior-to-Human-Trials)
- Navigate Medical Device Clinical Trials in Chile: A Step-by-Step Guide – cms.bioaccessla.com (https://cms.bioaccessla.com/blog/navigate-medical-device-clinical-trials-in-chile-a-step-by-step-guide)
- Navigate Biopharma Clinical Trials in Chile: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/navigate-biopharma-clinical-trials-in-chile-a-step-by-step-guide)
- How to Conduct First-in-Human Trials in Chile: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/how-to-conduct-first-in-human-trials-in-chile-a-step-by-step-guide)
- Implement Effective Patient Recruitment Strategies
- Precision, Relevance and Prediction: The Future of Clinical Trial Recruitment (https://gcihealth.com/2026/01/22/precision-relevance-and-prediction-the-future-of-clinical-trial-recruitment)
- Digital Marketing Revolutionizes Patient Recruitment | ImageBloom (https://imagebloom.com/digital-marketing-revolutionizes-clinical-trial-recruitment)
- Looking Back at SCOPE 2026: Recruitment Trends | Citeline (https://citeline.com/en/resources/recap-of-recruitment-trends-at-scope)
- Patient Recruitment for Clinical Trials: Strategies That Actually Work (https://kapsuletech.com/blog/patient-recruitment-clinical-trials)
- Execute the Clinical Trial with Compliance and Efficiency
- Well-Trained Staff Can Improve Your Clinical Trial Outcomes (https://taskacademy.org/how-well-trained-staff-can-improve-your-clinical-trial-outcomes)
- Ensuring Quality Clinical Trials with ICH-GCP Compliance (https://statswork.com/insights/articles/statistical-programming-biostatistics/ich-gcp-compliance-clinical-trials)
- Managing Clinical Research Teams: Leadership Strategies for PIs (https://ccrps.org/clinical-research-blog/managing-clinical-research-teams-leadership-strategies-for-pis)

Leave a Reply