Introduction
In the rapidly evolving field of neurology clinical trials, Chile presents unique opportunities that demand attention from MedTech and Biopharma innovators. With its streamlined regulatory processes, notably rapid approval timelines from authorities like the Instituto de Salud Pública (ISP), and a cost-effective environment-studies can be up to 30% cheaper than in North America-Chile offers a compelling case for conducting first-in-human trials.
However, organizations often struggle with patient recruitment and ethical dilemmas that can hinder trial outcomes. Without addressing these challenges, the potential benefits of conducting trials in Chile may remain untapped. Organizations must implement effective strategies to meet regulatory demands and engage diverse patient populations successfully.
bioaccess®: Pioneering First-in-Human Trials in Chile’s Neurology Sector
In the rapidly evolving field of neurology, the organization emerges as a leader in conducting neurology clinical trial Chile, setting new standards for excellence in first-in-human (FIH) studies. Leveraging its specialized focus and vast expertise, the organization has optimized its processes to enable rapid initiation and execution of studies, adhering strictly to ICH-GCP standards and local regulations established by authorities such as the Instituto de Salud Pública (ISP) and the Agencia Nacional de Medicamentos (ANAMED).
By utilizing a network of over 50 pre-qualified research sites, bioaccess® improves patient enrollment and accelerates data gathering, significantly shortening the time to market for innovative neurological therapies. This strategic positioning enhances the credibility of medical data and attracts international sponsors eager for efficient approval pathways.
In Chile, the average duration for regulatory approval of medical device studies is remarkably quick, taking only one to one and a half months, which is significantly faster than in neighboring nations such as Brazil and Colombia. This effectiveness, paired with a robust healthcare infrastructure and commitment to high-quality data, firmly establishes us as a pivotal player in advancing neurology clinical trial Chile and neurology research in Latin America.
Additionally, studies in Chile offer a cost advantage, being up to 30% less expensive than those in North America or Europe, emphasizing the strategic benefits of this site for research. Furthermore, this organization provides comprehensive consulting for study compliance strategy and medical device adherence throughout LATAM markets, guaranteeing that MedTech and Biopharma innovators can effectively navigate the intricacies of the compliance landscape.
As part of the Global Trial Accelerators™, bioaccess® is committed to facilitating specialized studies for radiopharmaceuticals and theranostics, further enhancing its role in the region. By choosing to conduct studies in Chile, innovators can not only save costs but also expedite their journey to market, ultimately benefiting patients in need.
Understanding Regulatory Requirements for Neurology Trials in Chile
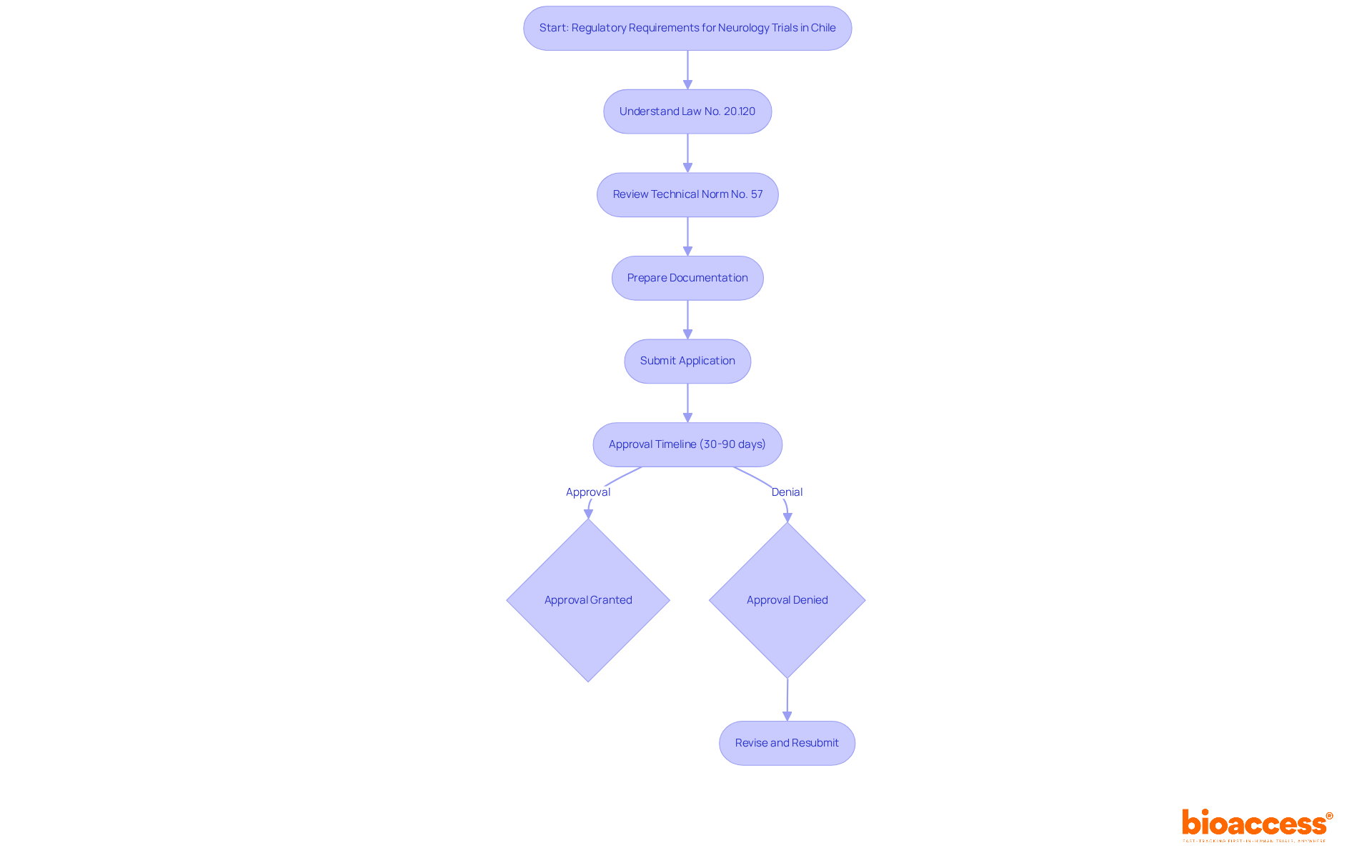
Navigating the regulatory landscape for neurology research in Chile presents unique challenges that require careful attention to detail. Conducting studies here demands strict compliance with the frameworks set by the Instituto de Salud Pública (ISP). Central to these regulations is Law No. 20.120, governing human research, alongside Technical Norm No. 57, which outlines application requirements for research studies. Approval timelines can vary significantly, ranging from 30 to 90 days based on study complexity, with an average of about 3-4 months for completion. However, bioaccess® achieves ethics/IRB approvals in as little as 4-8 weeks, significantly faster than typical timelines in North America or Europe.
Sponsors must ensure that all protocols align with ICH-GCP guidelines to maintain research integrity. Comprehensive documentation, including informed consent forms and safety monitoring plans, is essential for a smooth approval process. A 2022 survey indicated that 85% of international sponsors rated the data quality from Chilean studies as ‘excellent’ or ‘very good.’ Moreover, conducting medical studies in Chile can be up to 30% more affordable than in North America or Europe, with per-patient expenses significantly reduced due to pre-negotiated site agreements and streamlined compliance timelines. Understanding these regulations is not just a necessity; it’s a strategic advantage that can significantly enhance the success of your neurology clinical trial in Chile.
Effective Patient Recruitment Strategies for Neurology Trials in Chile
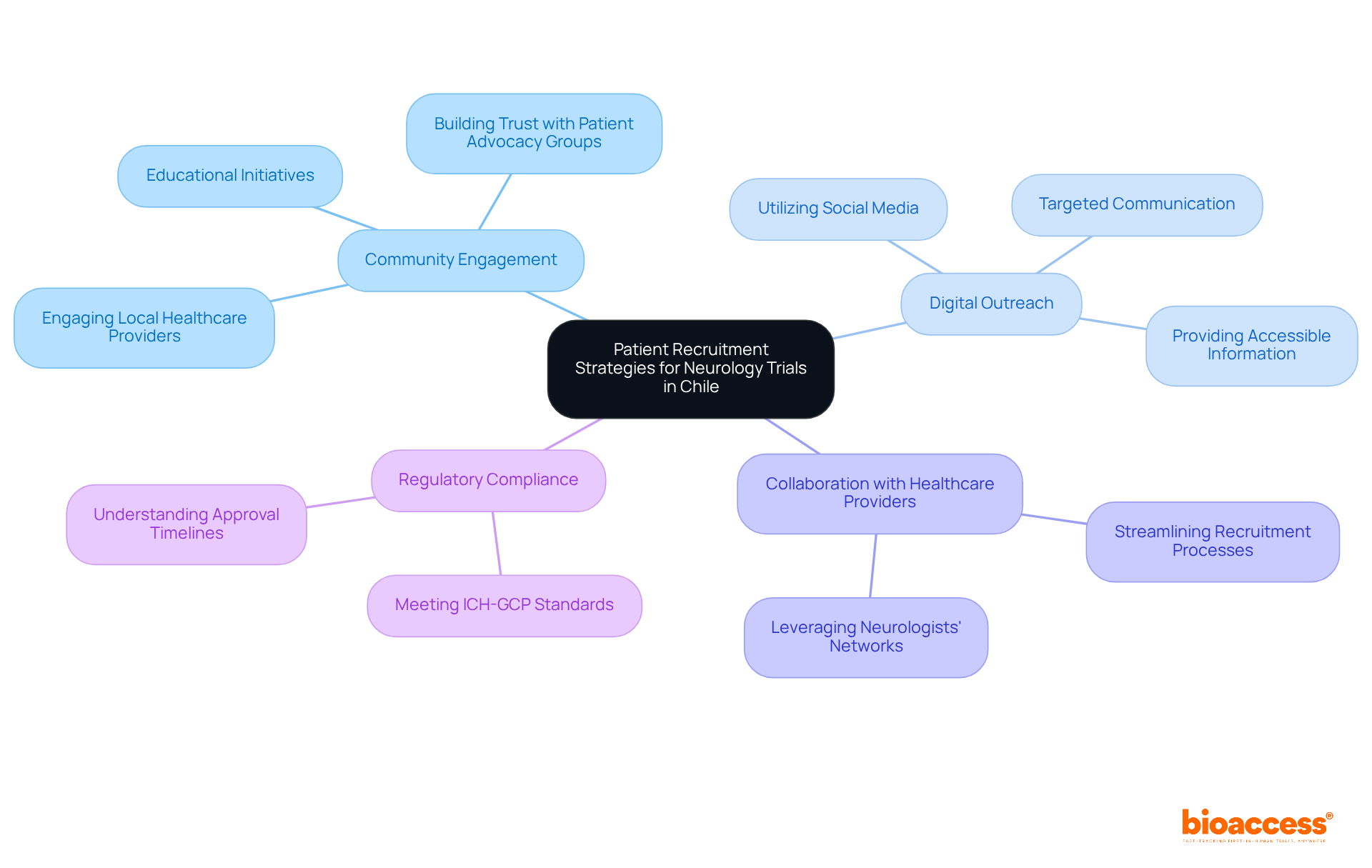
Recruiting patients for a neurology clinical trial in Chile is a complex endeavor, often hindered by the underrepresentation of neurological disorders in the neurology clinical trial in Chile. To boost recruitment, bioaccess® uses community-based strategies that engage local healthcare providers and patient advocacy groups. This method not only nurtures trust but also enables outreach to potential participants who may benefit from the studies.
Utilizing digital platforms and social media significantly broadens the outreach, allowing for targeted communication that effectively reaches diverse populations. Providing clear, accessible information about the study’s benefits and addressing common concerns can substantially improve enrollment rates. Furthermore, collaborating with neurologists and leveraging their established networks can streamline patient recruitment, ensuring a diverse and representative participant pool.
Incorporating these strategies also meets the regulatory requirements from local authorities like the Chilean Ministry of Health, ensuring compliance with ICH-GCP standards. For example, the approval timeline for studies in Chile generally varies from 30 to 90 days, based on the complexity of the investigation. By concentrating on community involvement and efficient communication, the organization increases the chances of successful patient recruitment for the neurology clinical trial in Chile, ultimately aiding the progress of medical knowledge and enhancing patient outcomes.
The leadership at the organization, including co-founders with extensive medical experience, emphasizes the significance of closing the gap between medical innovation and research potential in Latin America. Testimonials from clients emphasize the effectiveness of these strategies: ‘bioaccess®’s community engagement approach has significantly boosted our enrollment rates for trials,’ states a director from a partnering biopharma company. How often do you assess and adapt your recruitment strategies based on feedback from participants and stakeholders? Interacting with local communities through educational initiatives and outreach programs can also aid in clarifying the research process, making it more accessible for potential participants.
Leveraging Technology for Data Management in Neurology Clinical Trials
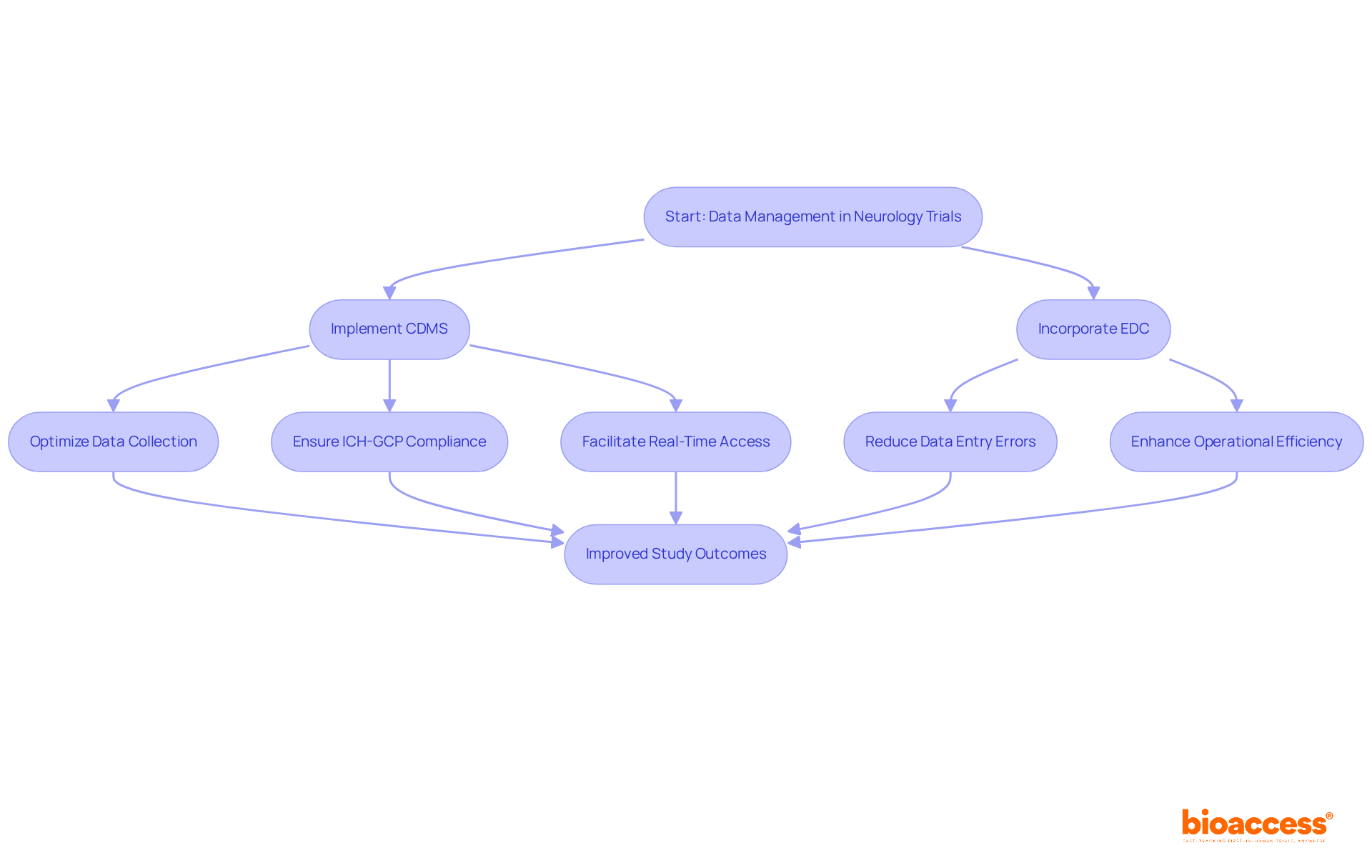
In the context of neurology clinical trial Chile, the stakes are high, and effective data management is non-negotiable for reliable outcomes. bioaccess® employs advanced clinical data management systems (CDMS) to optimize data collection, storage, and analysis. This ensures compliance with ICH-GCP standards and facilitates real-time access for monitoring and decision-making. Implementing electronic data capture (EDC) solutions significantly reduces data entry errors. Studies show that manual data entry can have error rates as high as 2.8%. By transitioning to EDC, studies can enhance operational efficiency and reduce the risk of inaccuracies that could compromise results.
Have you considered how incorporating artificial intelligence (AI) tools into your data analysis could provide deeper insights into patient outcomes and study effectiveness? Recent advancements in AI have demonstrated the potential to lower error rates in data extraction, achieving as low as 7.0% compared to 14.2% for manual methods. This capability is especially advantageous in the context of a neurology clinical trial in Chile, where accurate data interpretation is crucial for assessing treatment effects.
As oversight bodies in Latin America, such as ANVISA and COFEPRIS, continue to refine approval procedures, utilizing these technological innovations not only speeds up study timelines – often securing ethics approvals in just 4-8 weeks – but also improves the quality of evidence generated. By adopting robust data management practices, the organization ensures that its clients can navigate the complexities of regulatory submissions efficiently, ultimately supporting faster pathways to market for innovative therapies. Plus, using specialized services can cut study costs by as much as 30%, facilitating reinvestment into essential sectors such as research and development. By prioritizing advanced data management solutions, research directors can ensure their studies yield trustworthy results that pave the way for groundbreaking therapies.
Fostering Collaboration in Neurology Clinical Trials: Local and International Partnerships
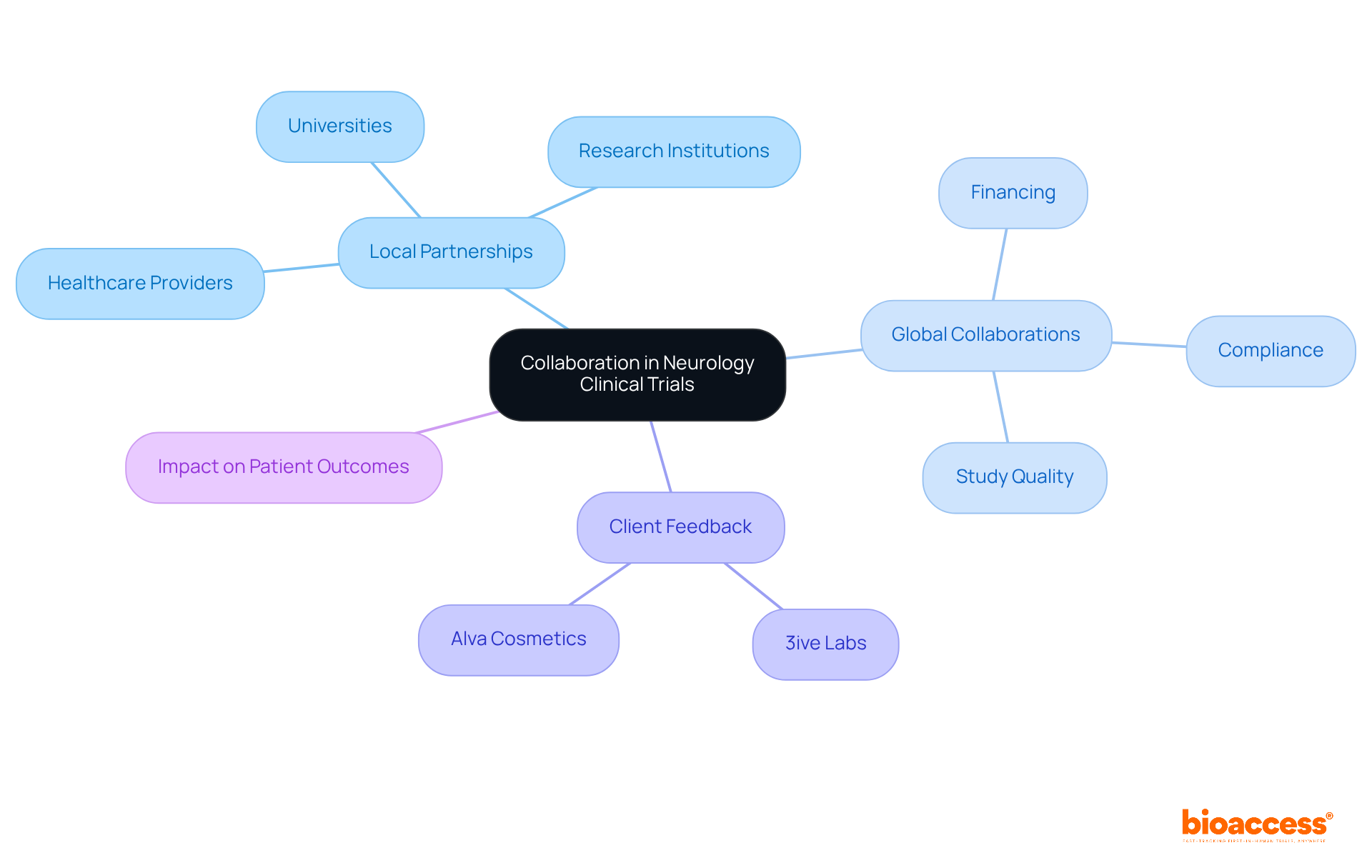
In the competitive landscape of neurology clinical trial Chile, collaboration is not just beneficial; it’s essential for success. The organization actively develops partnerships with local universities and research institutions, enhancing study capabilities through shared knowledge and resources. These collaborations facilitate access to specialized expertise and strengthen ties with local healthcare providers, and are vital for enhancing patient recruitment and retention.
However, many studies struggle with patient recruitment and retention, hindering their success. Global collaborations provide additional financing and support for complex studies, ensuring effective navigation of the compliance environment. By establishing a robust network of partners, the organization improves the quality and efficiency of its studies. This alignment with ICH-GCP standards ensures compliance with local regulatory bodies, including the Chilean Institute of Public Health.
Feedback from clients like 3ive Labs and Alva Cosmetics underscores the effectiveness of this method in expediting research studies. This strategic approach positions the company as a leader in first-in-human studies, meeting both local health needs and international research standards. Through the Innovation Runway, bioaccess® accelerates development, allowing startups to achieve critical milestones 40% faster and boosting their chances for successful funding and market entry.
By fostering these partnerships, organizations can not only enhance their research capabilities but also significantly impact patient outcomes in the region.
Cultural Considerations in Patient Participation for Neurology Trials
In Chile, cultural elements significantly influence patient involvement in neurology studies, presenting both challenges and opportunities. Understanding local customs, beliefs, and health perceptions is essential for fostering effective communication and engagement. Statistics reveal that cultural factors significantly impact patient trust in clinical studies. Varied populations often require tailored strategies to enhance their willingness to engage.
At bioaccess®, we focus on recruitment strategies that truly connect with diverse patient groups. Offering information about studies in local languages and addressing cultural sensitivities can significantly enhance trust and participation rates. Moreover, engaging community leaders and local healthcare providers in outreach initiatives promotes a sense of ownership among potential participants, fostering increased involvement in research studies.
Successful strategies include:
- Creating culturally appropriate materials
- Building trust through community initiatives
These approaches have effectively increased enrollment in the neurology clinical trial Chile across the region. Adherence to local regulatory bodies such as INVIMA and compliance with ICH-GCP standards are essential for ensuring the integrity and acceptance of research data. This further emphasizes the strategic benefits of conducting studies in Latin America, including accelerated approval timelines and cost-effectiveness, which our organization utilizes to assist innovative startups in generating the data they require to secure funding and attract strategic buyers.
Navigating Funding Challenges in Neurology Clinical Trials
Securing funding for neurology clinical trial Chile is not just a challenge; it’s a critical hurdle that can determine the success of innovative research. To navigate these challenges effectively, bioaccess® suggests a strategic approach to diversifying funding sources. This includes:
- Leveraging insights from the Global Trial Accelerators™ initiative
- Utilizing regulatory updates
- Implementing market access strategies tailored for Medtech startups in Latin America
Engaging with local agencies such as ANID, which supports scientific research across various fields, forming partnerships with pharmaceutical companies, and seeking venture capital investments can provide essential financial support crucial for executing studies.
A well-crafted budget that highlights the project’s potential impact is key to attracting investors. By demonstrating the significance of the research and its implications for patient care, sponsors can enhance their appeal to funding sources. Additionally, utilizing programs like CORFO’s Early Stage Tech Fund, which aims to assist high-potential tech startups in the health sector, can offer tailored financial backing. As CORFO states, “Our goal is to help founders develop products, validate their business models, and reach early customers, making it a great stepping stone for future growth.”
To ensure the financial sustainability of research studies, it is essential to actively manage funding opportunities and maintain open communication with stakeholders. Tackling the recruitment challenge is also vital, as active patient involvement in studies is crucial for guaranteeing representative and applicable outcomes. By implementing these approaches, sponsors can enhance their ability to obtain the essential resources for a successful neurology clinical trial Chile. Addressing these funding challenges is essential for advancing research in the neurology clinical trial Chile and improving patient outcomes.
Ethical Considerations in Neurology Clinical Trials: Ensuring Patient Safety
In the realm of neurology clinical trial Chile, ethical considerations are not just guidelines; they are the bedrock of patient safety and research integrity. bioaccess® strictly adheres to ethical guidelines, which include obtaining informed consent and safeguarding participant confidentiality. The informed consent process should be an ongoing dialogue. This allows participants to fully grasp the implications of their involvement. Regular evaluations by accredited ethics committees, such as those mandated by the Instituto de Salud Pública (ISP), are conducted to assess study protocols and ensure adherence to ethical standards.
Navigating the regulatory environment is essential for successful research studies in Latin America. The company offers crucial guidance on medical device registration and clearance pathways through ANVISA, INVIMA, and COFEPRIS, facilitating rapid market access. With access to over 50 pre-qualified research sites and rapid patient recruitment capabilities, bioaccess® enhances the efficiency of studies. Implementing robust safety monitoring plans is essential for promptly identifying and addressing any adverse events. By taking this proactive approach, we not only protect participants but also enhance the credibility of our studies. By prioritizing ethical considerations and fostering transparent communication, sponsors can build trust with participants and stakeholders, ultimately contributing to the success of their research initiatives in the region.
Have you ever considered that only 50% of participants in research studies truly understand the elements of informed consent? This highlights the need for effective communication strategies. Addressing challenges such as cognitive impairments among participants and providing educational materials can further improve the informed consent process, ensuring that all participants are adequately informed and protected. By enhancing communication and understanding, we can ensure that every participant is not only informed but also empowered, paving the way for more successful research outcomes.
Defining Outcome Measures for Neurology Clinical Trials
In the realm of neurology clinical trial Chile, the choice of outcome measures can make or break a trial’s success. It’s essential to choose outcome measures that align with the study’s goals and the specific neurological conditions relevant to the neurology clinical trial Chile. Employing validated scales and metrics not only boosts the reliability of outcomes but also streamlines approval processes, especially under ICH-GCP standards and with acceptance from authorities like ANVISA and COFEPRIS.
Bringing in patient-reported outcomes (PROs) is crucial because they shed light on how interventions truly affect patients’ quality of life. Did you know that only 16% of neuro-oncology trials have tapped into COAs? This reveals a huge opportunity for better PRO adoption. By establishing robust outcome measures, sponsors can clearly show how effective their treatments are, which is key for getting funding and approvals. This strategic emphasis on PROs not only aligns with the growing trend in research but also positions sponsors to better meet the needs of patients and stakeholders alike.
Despite the critical role of outcome measures, many trials fall short in their adoption, leaving a gap in effective patient assessment. This gap not only hinders trial success but also limits the potential for innovative treatments to reach patients. By prioritizing the right outcome measures, sponsors can not only enhance trial success but also ensure that patient voices are heard in the research process.
Future Trends in Neurology Clinical Trials in Chile: Opportunities Ahead
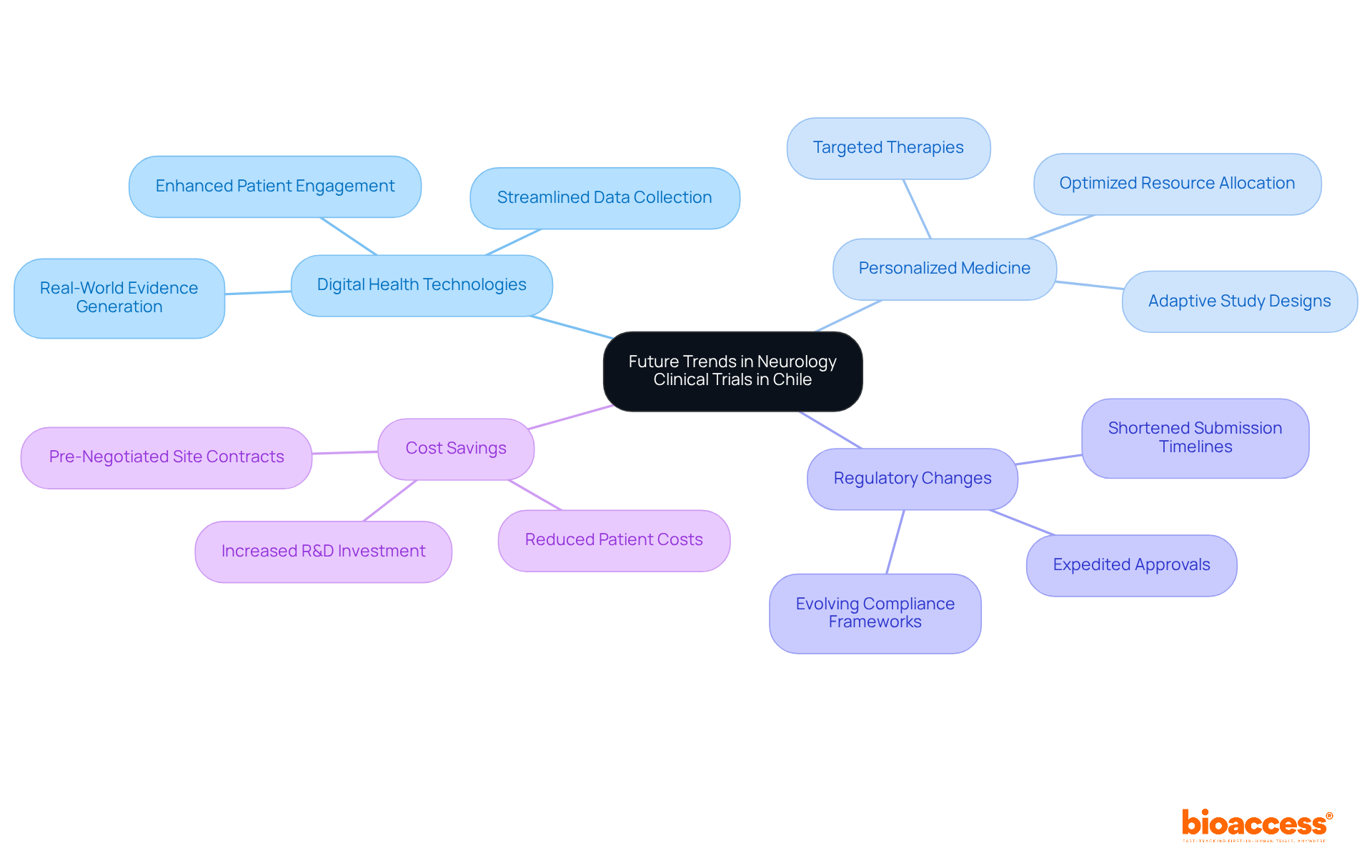
As the landscape of neurology clinical trial Chile transforms, sponsors face both challenges and opportunities. The growing adoption of digital health technologies is significantly enhancing patient engagement and streamlining data collection processes. This transition enhances data quality. It also aids in adhering to ICH-GCP standards, which are essential for acceptance by authorities like ANVISA and COFEPRIS.
Furthermore, the focus on personalized medicine is driving the advancement of targeted therapies, requiring adaptive study designs that permit changes based on interim findings. This optimization enhances resource allocation and patient outcomes. However, sponsors may find navigating these changes daunting. As compliance frameworks evolve, opportunities for expedited approvals are becoming more common, with timelines for submissions potentially shortened to as little as 30-90 days in certain jurisdictions. Not embracing these advancements could lead to missed opportunities and longer timelines, providing a strategic advantage for sponsors compared to the lengthy processes in the US and EU.
Utilizing these advancements allows sponsors to position themselves strategically within the neurology clinical trial Chile research space. The integration of digital health technologies not only enhances operational efficiency but also aligns with the growing demand for real-world evidence generation, which is increasingly recognized as vital for successful regulatory submissions. Additionally, with pre-negotiated site contracts, sponsors can achieve significant cost savings – up to $25K per patient – allowing them to invest more into R&D or their next funding milestone. Embracing these advancements is not just beneficial; it’s essential for staying competitive in the evolving clinical research landscape.
Conclusion
In the rapidly evolving field of neurology clinical trials in Chile, opportunities for innovation are abundant. The organization bioaccess® stands at the forefront, leveraging its expertise to navigate the complexities of first-in-human trials while adhering to stringent ICH-GCP standards. This commitment accelerates research and bolsters data credibility, establishing Chile as a leading hub for neurology clinical studies.
The discussion underscores how efficient regulatory processes can significantly shorten approval timelines compared to neighboring countries, making Chile a cost-effective choice for conducting trials. Strategies for patient recruitment, including community engagement and digital outreach, are essential for overcoming challenges in participant enrollment. Additionally, the integration of advanced data management technologies ensures high-quality and reliable outcomes, further solidifying the region’s appeal to international sponsors.
Looking ahead, stakeholders must embrace advancements in digital health and personalized medicine. Collaborative partnerships will be essential for capitalizing on the evolving neurology clinical trial landscape in Chile. By prioritizing these strategies, sponsors can enhance their studies and contribute to the broader goal of improving patient outcomes across Latin America. Engaging with local communities, understanding cultural nuances, and navigating funding opportunities will pave the way for successful research initiatives that ultimately benefit patients in need. Ultimately, these efforts will not only advance clinical research but also lead to improved patient outcomes across Latin America.
Frequently Asked Questions
What is bioaccess® and its role in neurology clinical trials in Chile?
bioaccess® is a specialized contract research organization that leads first-in-human (FIH) studies in the neurology sector in Chile. It focuses on optimizing processes for rapid study initiation and execution while adhering to ICH-GCP standards and local regulations.
How does bioaccess® improve the efficiency of neurology trials?
bioaccess® utilizes a network of over 50 pre-qualified research sites to enhance patient enrollment and accelerate data gathering, significantly reducing the time to market for innovative neurological therapies.
What is the average duration for regulatory approval of medical device studies in Chile?
The average duration for regulatory approval of medical device studies in Chile is between one to one and a half months, which is notably faster than in neighboring countries like Brazil and Colombia.
What are the cost advantages of conducting studies in Chile?
Conducting studies in Chile can be up to 30% less expensive than in North America or Europe, making it a cost-effective location for research.
What regulatory requirements must be met for neurology trials in Chile?
Neurology trials in Chile must comply with regulations set by the Instituto de Salud Pública (ISP), including Law No. 20.120 governing human research and Technical Norm No. 57 outlining application requirements.
How quickly can bioaccess® achieve ethics/IRB approvals compared to typical timelines?
bioaccess® can achieve ethics/IRB approvals in as little as 4-8 weeks, which is significantly faster than the typical timelines of 3-4 months in North America or Europe.
What strategies does bioaccess® employ for patient recruitment in neurology trials?
bioaccess® employs community-based strategies that engage local healthcare providers and patient advocacy groups, along with digital platforms and social media, to effectively reach and recruit diverse populations for trials.
How does bioaccess® ensure compliance with local regulatory requirements during patient recruitment?
The organization ensures compliance with local regulatory requirements by adhering to ICH-GCP standards and collaborating with local authorities like the Chilean Ministry of Health.
What feedback have clients provided regarding bioaccess®’s recruitment strategies?
Clients have reported that bioaccess®’s community engagement approach significantly boosts enrollment rates for trials, demonstrating the effectiveness of their recruitment strategies.
Why is understanding the regulatory landscape important for sponsors conducting trials in Chile?
Understanding the regulatory landscape is crucial for sponsors as it provides a strategic advantage that can enhance the success of their neurology clinical trials, ensuring compliance and facilitating smoother approval processes.
List of Sources
- bioaccess®: Pioneering First-in-Human Trials in Chile’s Neurology Sector
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOopx8BcUsUCrGPaZqj1fa0j6U68OV5Wwhm0nOGHu3lSDUE4CAVTz)
- How Chile Is Shaping Medical Device Clinical Trials In Latin America (https://meddeviceonline.com/doc/how-chile-is-shaping-medical-device-clinical-trials-in-latin-america-0001)
- Master Early Phase Clinical Trials in Chile: Key Strategies and Insights | bioaccess® (https://bioaccessla.com/blog/master-early-phase-clinical-trials-in-chile-key-strategies-and-insights)
- Understanding Regulatory Requirements for Neurology Trials in Chile
- How Chile Is Shaping Medical Device Clinical Trials In Latin America (https://meddeviceonline.com/doc/how-chile-is-shaping-medical-device-clinical-trials-in-latin-america-0001)
- Clinical Trials in Latin America (https://languageconnections.com/clinical-trials-in-latin-america)
- How to Conduct First-in-Human Trials in Chile: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/how-to-conduct-first-in-human-trials-in-chile-a-step-by-step-guide)
- Effective Patient Recruitment Strategies for Neurology Trials in Chile
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- Patient Engagement Quotes: For Every Purpose & Audience (https://nclusiv.co.uk/edi-consulting/f/patient-engagement-quotes-for-every-purpose-audience)
- Trends in clinical trials performed in Chile (https://scielo.cl/article_plus.php?pid=S0034-98872021000100110&tlng=en&lng=es)
- Clinical Trials Patient Recruitment in Latin America | H Clinical (https://hclinical.com/patient-recruitment)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOopYf5fgQrlHLZV_rIjHu2k2o34AUc8x0OnxaRwHV-Ur7rsLL5Hc)
- Leveraging Technology for Data Management in Neurology Clinical Trials
- Clinical Trial Data Integrity | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/clinical-trial-data-integrity)
- Error rates of data processing methods in clinical research: A systematic review and meta-analysis of manuscripts identified through PubMed (https://sciencedirect.com/science/article/abs/pii/S138650562400412X)
- Error rates in a clinical data repository: lessons from the transition to electronic data transfer—a descriptive study (https://bmjopen.bmj.com/content/3/5/e002406)
- Fostering Collaboration in Neurology Clinical Trials: Local and International Partnerships
- Trends in clinical trials performed in Chile (https://scielo.cl/article_plus.php?pid=S0034-98872021000100110&tlng=en&lng=es)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOooCzKSsX7IoRkPnm3yUx_B8ilof6DKUqS0W81jNFwQ-Bnp4qA4n)
- Clinical trial success depends on strong partnerships – here’s why – Siron Clinical (https://sironclinical.com/clinical-trial-success-depends-on-strong-partnerships-heres-why)
- Cultural Considerations in Patient Participation for Neurology Trials
- Diversity of Participation in Clinical Trials and Influencing Factors: Findings from the Health Information National Trends Survey 2020 – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10039144)
- Patient experience and satisfaction in Chilean hospitals: Insights for enhancing person-centered care (https://sciencedirect.com/science/article/abs/pii/S0738399125006998)
- How Cultural Competency Can Increase Clinical Trial Diversity (https://clinicalleader.com/doc/how-cultural-competency-can-increase-clinical-trial-diversity-0001)
- Improving diverse patient enrollment in clinical trials, focusing on Hispanic and Asian populations: recommendations from an interdisciplinary expert panel (https://sciencedirect.com/science/article/pii/S1048891X25009946)
- Navigating Funding Challenges in Neurology Clinical Trials
- Funding Opportunities — ACCESS (https://accesschilesweden.org/funding-opportunities)
- Clinical research in Chile: high standard, but there are still challenges in regulation, investment and patient participation – BOPAL (https://bopalcro.com/clinical-research-in-chile-high-standard-but-there-are-still-challenges-in-regulation-investment-and-patient-participation)
- For businesses | Government Grants That Can Help Fund Your Business in Chile in 2025 (https://joinadro.com/blog/government-grants-that-can-help-fund-your-business-in-chile-in-2025)
- Ethical Considerations in Neurology Clinical Trials: Ensuring Patient Safety
- Trends in clinical trials performed in Chile (https://scielo.cl/article_plus.php?pid=S0034-98872021000100110&tlng=en&lng=es)
- Participants’ understanding of informed consent in clinical trials: A systematic review and updated meta-analysis – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10760836)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Overcoming barriers to informed consent in neurological research: Perspectives from a national survey – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10609656)
- Considerations For Informed Consent In Clinical Trials Involving Neurologic Disorders – BRANY (https://brany.com/considerations-for-informed-consent-in-clinical-trials-involving-neurologic-disorders)
- Defining Outcome Measures for Neurology Clinical Trials
- Improving Outcome Measurements in Stroke Clinical Trials (https://signanthealth.com/resources/blog/improving-outcome-measurements-in-stroke-clinical-trials)
- Statistical advances in clinical trials and clinical research – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC6118095)
- Clinical outcome assessment trends in neuro-oncology trials (https://academic.oup.com/nop/article/12/6/998/8177193)
- The Spectrum of Functional Rating Scales in Neurology Clinical Trials (https://sciencedirect.com/science/article/pii/S1878747923015222)
- Future Trends in Neurology Clinical Trials in Chile: Opportunities Ahead
- Neurology Clinical Trials Market Size, Share & Drivers, 2033 (https://straitsresearch.com/report/neurology-clinical-trials-market)
- Chile Neurology Devices Market Size | Mordor Intelligence (https://mordorintelligence.com/industry-reports/chile-neurology-devices-market)
- Neurology Clinical Trials Market Report 2026 – Research and Markets (https://researchandmarkets.com/reports/6168642/neurology-clinical-trials-market-report?srsltid=AfmBOop1BIAZVIQbyUy1BRoFM-Nv9Zf-YRdlnuEF6iydCE9gP8ja0RpZ)
- Neurology Clinical Trials Market Share, Trends Analysis – 2034 (https://polarismarketresearch.com/industry-analysis/neurology-clinical-trials-market)
- Digital Health Trends 2024 (https://iqvia.com/insights/the-iqvia-institute/reports-and-publications/reports/digital-health-trends-2024)

Leave a Reply