Introduction
Understanding the intricate landscape of regulatory submissions is essential for any organization aiming to navigate the complexities of medical device and pharmaceutical approvals in Colombia. The National Food and Drug Surveillance Institute (INVIMA) plays a crucial role in this process. Effective pre-submission teleconference planning can significantly enhance the chances of a successful application. However, many organizations face challenges in aligning their objectives with INVIMA’s expectations. This leads to a critical question: what are the key steps to ensure a productive teleconference that not only meets regulatory requirements but also fosters clear communication and effective follow-up?
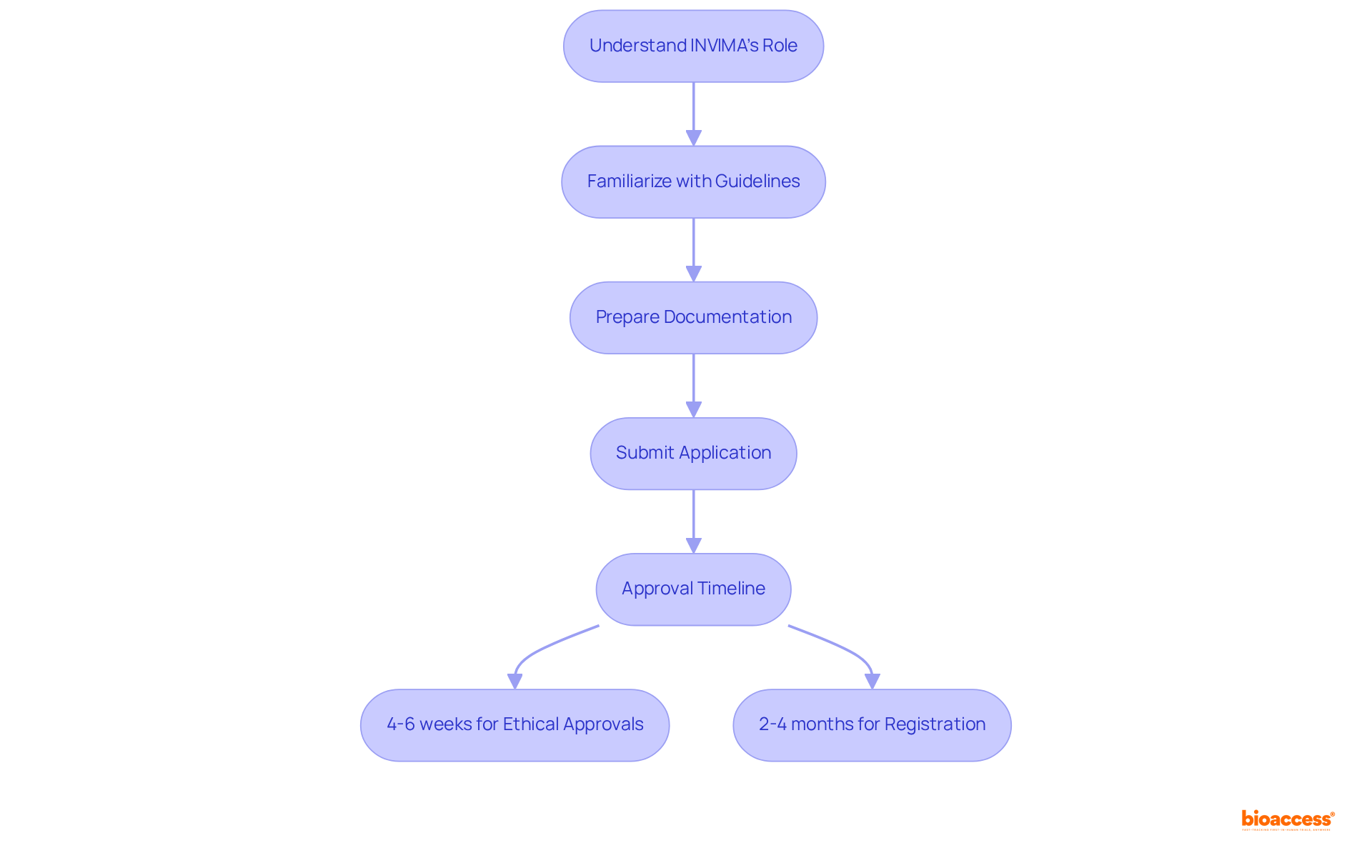
Understand INVIMA’s Role in Regulatory Submissions
The plays a pivotal role in the , pharmaceuticals, and . Understanding its functions is essential for effective . This regulatory body oversees the , ensuring that all applications adhere to stringent safety, efficacy, and quality standards.
Familiarizing yourself with the organization’s guidelines—including the , timelines, and necessary documentation—will significantly enhance your preparedness for the . This crucial knowledge aligns your objectives with the agency’s expectations, thereby increasing the likelihood of successful regulatory filings.
As of 2025, the has been optimized, with within just 4 to 6 weeks, and the overall timeline for medical device registration typically spanning 2 to 4 months. Grasping these dynamics is vital for effectively navigating the complexities of the .
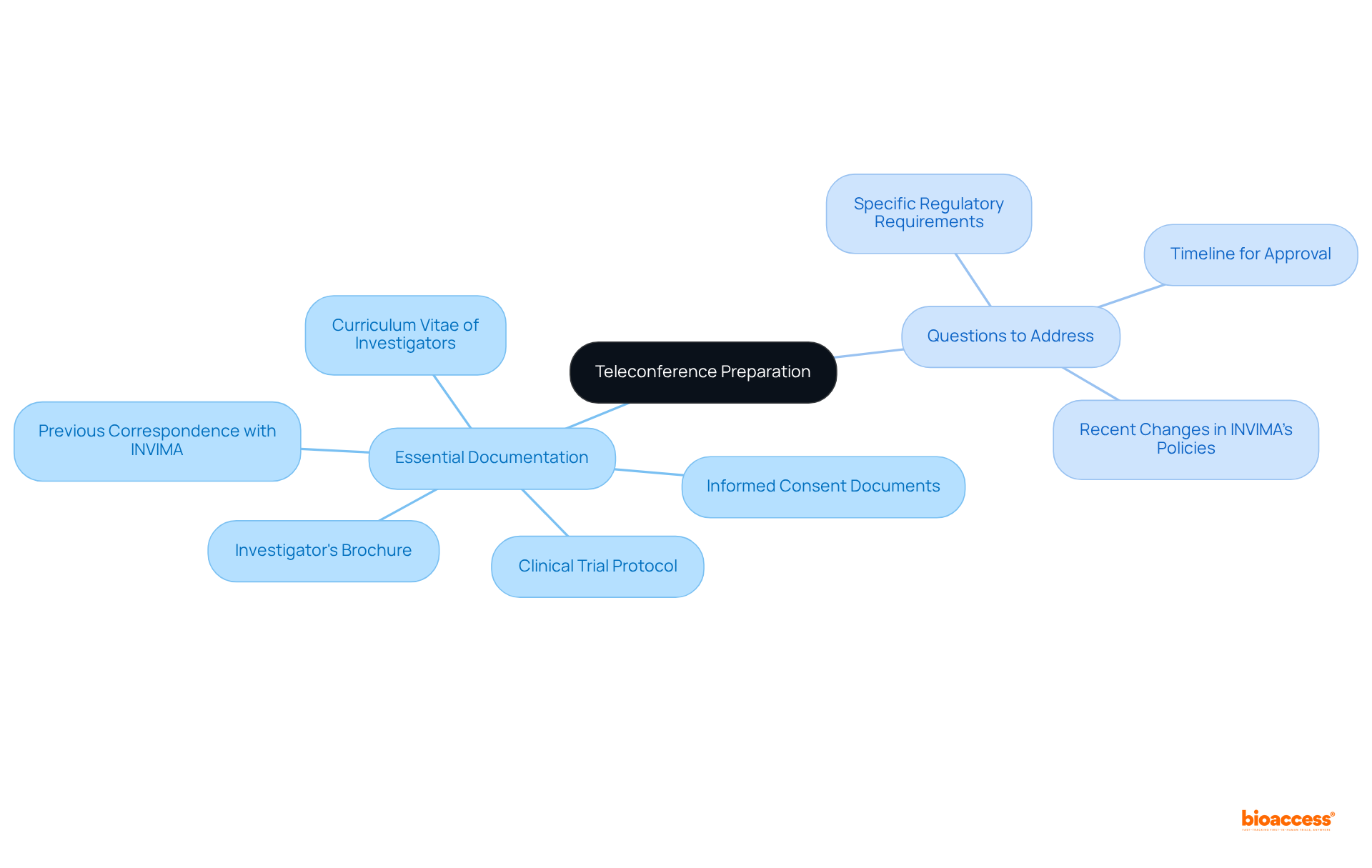
Prepare Key Documentation and Questions for the Teleconference
Thorough planning for the pre-submission teleconference is essential for a successful application process. Begin by gathering all essential documentation, which includes:
- Informed Consent Documents
- Investigator’s Brochure
- Curriculum Vitae of Investigators
- Any previous correspondence with
In addition to the documentation, it is vital to formulate or uncertainties regarding the . Organize these questions by topic to facilitate a structured discussion. For instance, consider inquiring about:
- Recent changes in ‘s policies
Having this information readily available will enable you to engage effectively during the meeting, ensuring that all critical aspects of your submission are addressed.
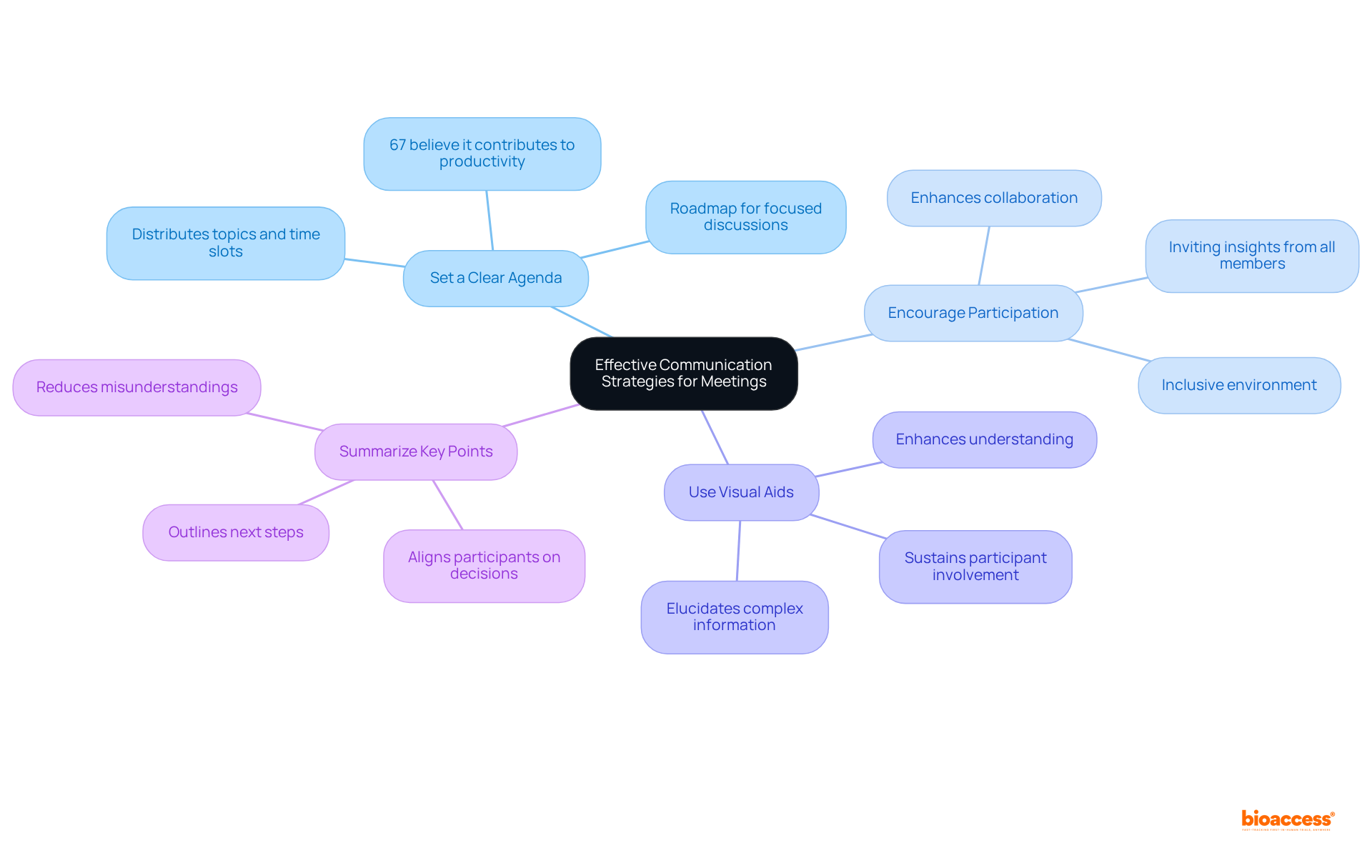
Develop Effective Communication Strategies for the Meeting
To enhance communication during the teleconference, it is imperative to implement the following strategies:
- Set a : Distributing a well-defined agenda prior to the meeting is crucial. This should outline the topics to be discussed and allocate specific time slots for each item. Research indicates that only , underscoring the necessity for organized discussions. A serves as a roadmap, ensuring conversations remain focused and relevant. In fact, .
- : Creating an inclusive environment is essential for . Actively inviting all participants to share their insights and questions, particularly encouraging quieter members to contribute, fosters collaboration and enhances the overall quality of the discussion.
- Use : Incorporating , such as slides or documents, can significantly enhance understanding. Visuals elucidate intricate information and , which is vital in a virtual meeting environment where focus can easily wane.
- : At the conclusion of each topic, and outlining any decisions made is vital. This practice ensures that all participants are aligned and informed about the next steps, reducing the likelihood of misunderstandings and enhancing the discussion’s effectiveness.
By concentrating on these strategies, organizations can elevate their virtual meeting outcomes with regulatory bodies through , ensuring that discussions are productive and aligned with their objectives.
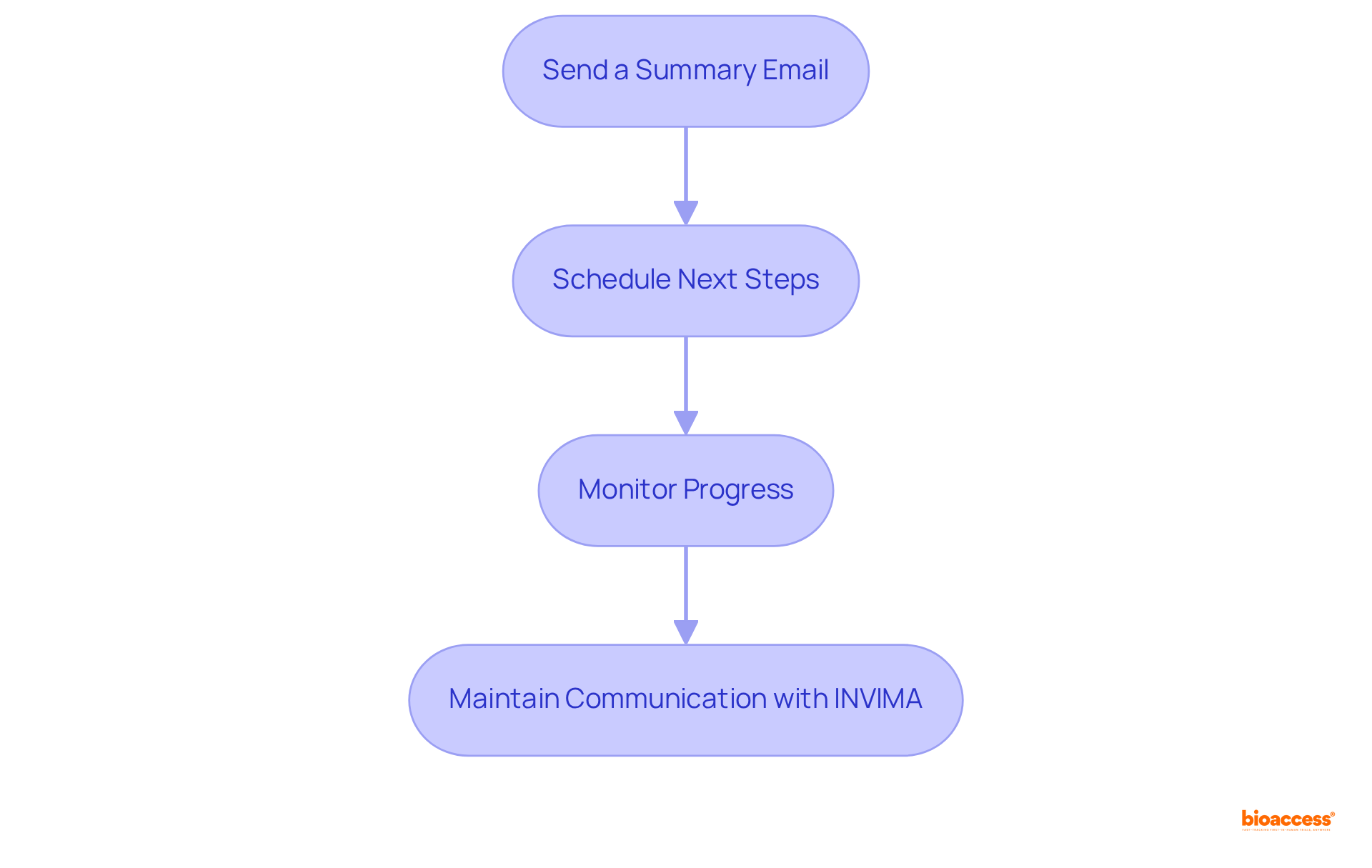
Implement Follow-Up Actions Post-Teleconference
To ensure effective follow-up after the teleconference with INVIMA, consider the following actions:
- Send a Summary Email: that captures key points discussed, decisions made, and action items assigned to specific individuals. This not only reinforces understanding but also serves as a reference for all participants.
- Schedule Next Steps: Promptly organize any required follow-up discussions or . Clearly outline deadlines for tasks assigned during the teleconference to ensure accountability.
- Monitor Progress: Regularly check in with team members regarding their assigned tasks. This can be achieved through brief updates or follow-up discussions, ensuring that everyone remains on track and any potential issues are addressed quickly.
through by sending any additional questions or clarifications that arise post-meeting. This proactive approach demonstrates your commitment to compliance and fosters a collaborative relationship.
Statistics indicate that significantly enhance the , with . Therefore, implementing these follow-up strategies is crucial for maintaining a competitive edge in the .
Conclusion
Effective pre-submission teleconference planning with INVIMA is crucial for navigating the complexities of regulatory submissions in Colombia. Understanding INVIMA’s role, preparing key documentation, developing effective communication strategies, and implementing follow-up actions significantly enhance the chances of a successful application process. This structured approach aligns objectives with regulatory expectations and fosters a collaborative relationship with INVIMA.
The article highlights essential steps, including:
- Familiarizing oneself with INVIMA’s guidelines
- Gathering necessary documents
- Crafting specific questions to ensure clarity during discussions
Furthermore, it emphasizes the importance of:
- A clear agenda
- Participant engagement
- The use of visual aids to facilitate effective communication
Following the teleconference, timely follow-up actions, such as sending summary emails and monitoring progress, are vital for maintaining momentum and accountability.
In summary, thorough preparation and strategic communication are paramount for successful interactions with INVIMA. By adopting these best practices, stakeholders can navigate the regulatory landscape more effectively, ensuring compliance and fostering positive relationships with regulatory bodies. Embracing these insights will streamline the submission process and position organizations for long-term success in the pharmaceutical and medical device sectors.
Frequently Asked Questions
What is the role of INVIMA in Colombia?
INVIMA, the National Food and Drug Surveillance Institute in Colombia, regulates medical devices, pharmaceuticals, and clinical trials, overseeing the approval process to ensure compliance with safety, efficacy, and quality standards.
Why is it important to understand INVIMA’s functions before a submission?
Understanding INVIMA’s functions is essential for effective pre-submission teleconference planning, as it helps align your objectives with the agency’s expectations, increasing the likelihood of successful regulatory filings.
What should applicants familiarize themselves with when preparing for INVIMA submissions?
Applicants should familiarize themselves with INVIMA’s guidelines, including the types of applications required, timelines, and necessary documentation to enhance their preparedness for the submission process.
What is the typical timeline for medical device registration in Colombia as of 2025?
As of 2025, the overall timeline for medical device registration typically spans 2 to 4 months, with ethical approvals attainable within 4 to 6 weeks.
How can understanding the regulatory timeline help applicants?
Grasping the regulatory timeline helps applicants effectively navigate the complexities of the Colombian regulatory landscape, allowing for better planning and management of their submissions.
List of Sources
- Understand INVIMA’s Role in Regulatory Submissions
- iaocr.com (https://iaocr.com/colombias-regulatory-authority-for-clinical-trials)
- Understanding INVIMA Requirements for Medtech … | bioaccess® (https://bioaccessla.com/blog/understanding-invima-requirements-for-medtech-clinical-trials-an-in-depth-tutorial)
- bioaccessla.com (https://bioaccessla.com/es/blog/10-essential-insights-on-invima-risk-classification-guide)
- Prepare Key Documentation and Questions for the Teleconference
- bioaccess® | MedTech, Biopharma & Radiopharma CRO in Latin America (https://bioaccessla.com/blog/how-to-navigate-the-invima-clinical-trial-submission-process-a-step-by-step-guide)
- Understanding INVIMA Clinical Trial Requiremen… | bioaccess® (https://bioaccessla.com/blog/understanding-invima-clinical-trial-requirements-an-in-depth-tutorial)
- 6 Best Practices in Regulatory Writing (https://blog.medpoint.com/6-best-practices-in-regulatory-writing)
- asphalion.com (https://asphalion.com/key-considerations-and-tips-for-submitting-good-submission-files-to-regulatory-authorities)
- Develop Effective Communication Strategies for the Meeting
- aiscreen.io (https://aiscreen.io/digital-signage-employee-communications/statistics-about-employee-communications)
- 100 Surprising Meeting Statistics for 2026 (https://flowtrace.co/collaboration-blog/50-meeting-statistics)
- 43 Remarkable Workplace Communication Statistics in 2026 (https://apollotechnical.com/workplace-communication-statistics)
- Business Communication Statistics 2026 | Project.co (https://project.co/communication-statistics)
- Workplace Communication Statistics for 2026 (https://pumble.com/learn/communication/communication-statistics)
- Implement Follow-Up Actions Post-Teleconference
- 74 Important Sales Follow-Up Statistics for 2025 (https://profitoutreach.app/blog/sales-follow-up-statistics)
- 130+ Compliance Statistics & Trends to Know for 2026 (https://secureframe.com/blog/compliance-statistics)
- mtdsalestraining.com (https://mtdsalestraining.com/mtdblog/sales-follow-up-statistics.html)
- 14 Sales Follow-Up Statistics for Pipeline Success in 2025 (https://qwilr.com/blog/sales-follow-up-statistics)
- woodpecker.co (https://woodpecker.co/blog/follow-up-statistics)

Leave a Reply