Introduction
Navigating Brazil’s regulatory landscape is crucial for sponsors seeking success in clinical trials within this dynamic market. With the National Health Surveillance Agency (ANVISA) at the helm, navigating the complexities of regulatory submission presents significant challenges for sponsors. These challenges can significantly impact the success of clinical research initiatives. As Brazil positions itself as a leading destination for innovative studies, failure to navigate these complexities can jeopardize the success of clinical trials. Without a strategic approach to compliance, sponsors risk delays and setbacks that could undermine their research objectives.
Understand Brazil’s Regulatory Framework for Clinical Trials
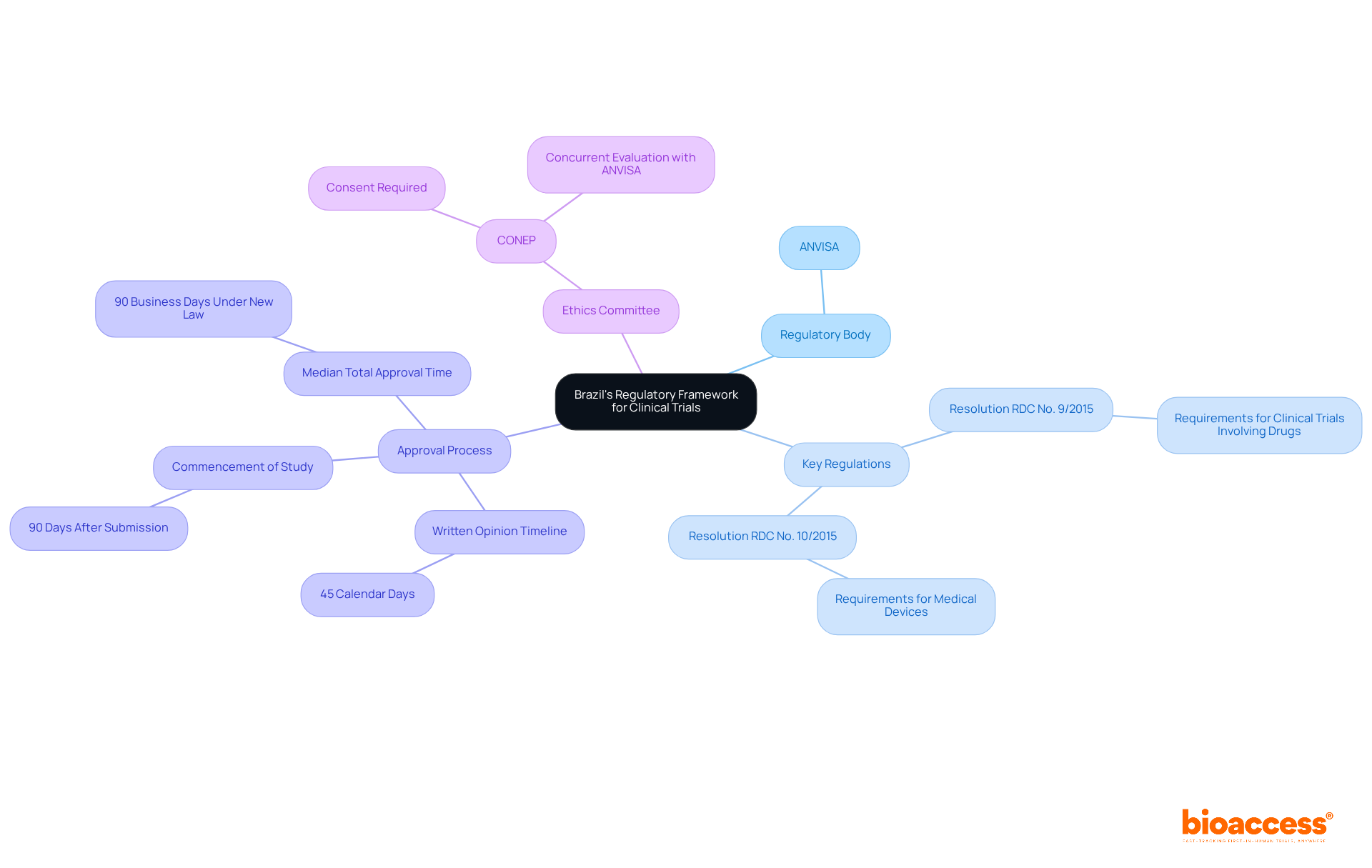
Understanding Brazil’s regulatory framework is crucial for organizations aiming to conduct clinical research in the country. The National Health Surveillance Agency (ANVISA) oversees this framework, making it essential for sponsors to grasp key regulations. Notable regulations include:
- Resolution RDC No. 9/2015: This outlines the requirements for clinical trials involving drugs.
- Resolution RDC No. 10/2015: This pertains to medical devices, detailing the essential documentation and ethical considerations needed for filing.
Typically, the regulatory body will get back to you with a written opinion within 45 calendar days after you submit your application. If you don’t hear back within 90 days, you can commence the study, provided all ethical approvals are in place. Importantly, the median total approval time for clinical studies under ANVISA has improved significantly, with a firm 90-business-day limit on review processes established under the new Brazilian Clinical Research Law (Lei 14.874), effective January 1, 2025.
Before presenting, studies must obtain consent from a local ethics committee (CONEP), ensuring compliance with ethical standards in research. The new regulations allow for concurrent evaluation by ANVISA and ethics committees, optimizing the process and decreasing the time to commence studies.
Familiarizing with these regulations helps sponsors prepare their applications. It also allows them to anticipate potential challenges in the approval process. With these changes, Brazil is poised to become a leading destination for innovative clinical research.
Develop a Strategic Submission Plan for Regulatory Compliance
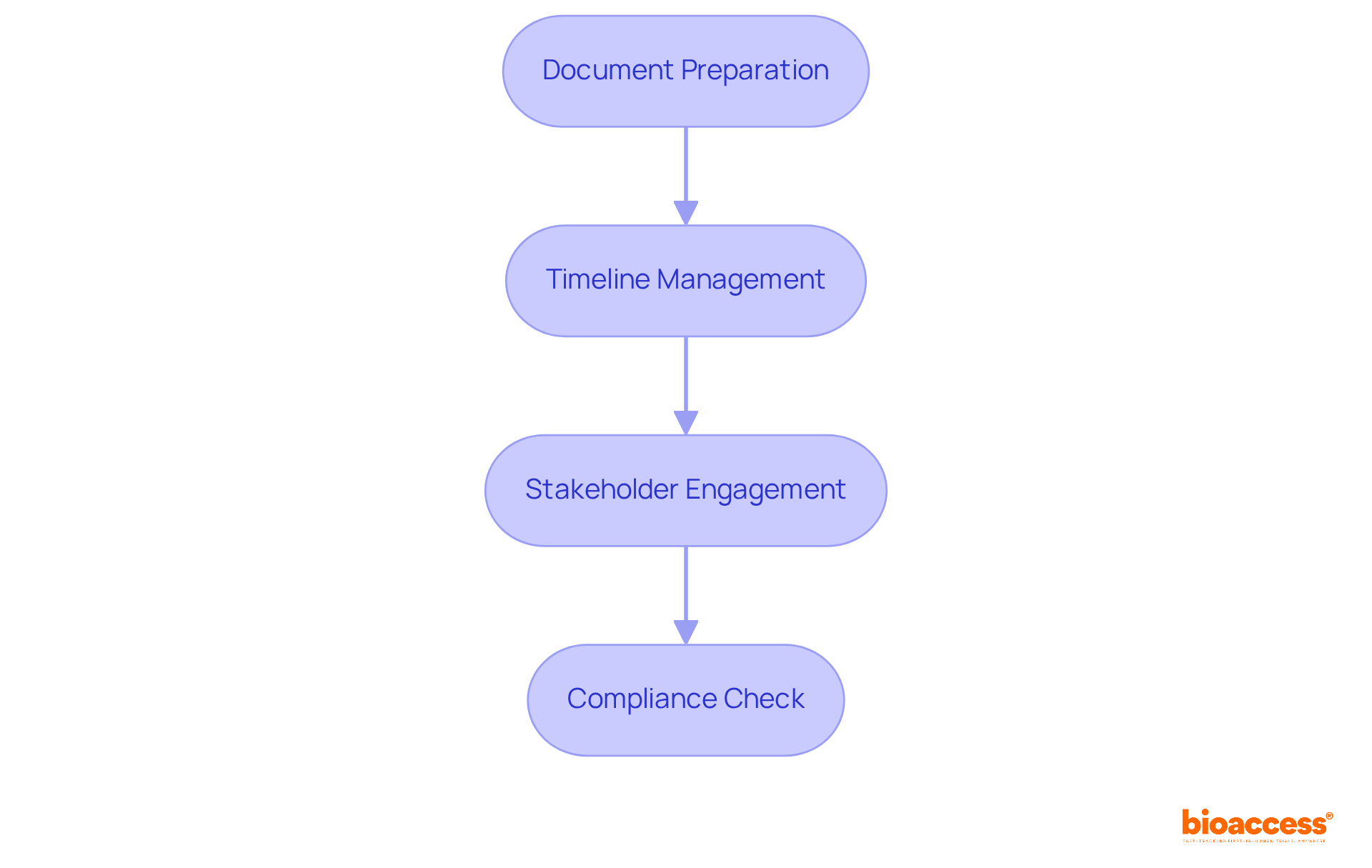
Navigating the regulatory landscape for regulatory submission Brazil clinical trial requires a strategic approach to ensure compliance and efficiency. Creating a strategic submission plan for regulatory compliance in clinical trials involves several critical steps:
- Document Preparation: Meticulously prepare all required documents, including the Clinical Trial Application (CTA), preclinical data, study protocols, and informed consent forms. Ensuring completeness and accuracy in these documents is essential for a seamless process.
- Timeline Management: Establish a clear schedule for each phase of the process, encompassing document preparation, delivery, and anticipated feedback from the regulatory authority. This structured approach aids in managing expectations and optimizing resource allocation.
- Stakeholder Engagement: Engage with all relevant stakeholders, including regulatory consultants and local ethics committees, early in the process. This collaboration can provide valuable insights and facilitate a more efficient approval process.
- Compliance Check: Regularly review the documentation against ANVISA’s guidelines to ensure all requirements are met. Following ICH-GCP standards is essential for preserving data integrity and ensuring participant safety throughout the study.
Navigating the regulatory landscape can often feel overwhelming for sponsors, leading to potential setbacks in trial timelines. By implementing a robust submission strategy, sponsors can not only streamline their processes but also enhance the overall success of their clinical trials in Latin America.
Navigate Logistics and Build Strategic Partnerships for Success
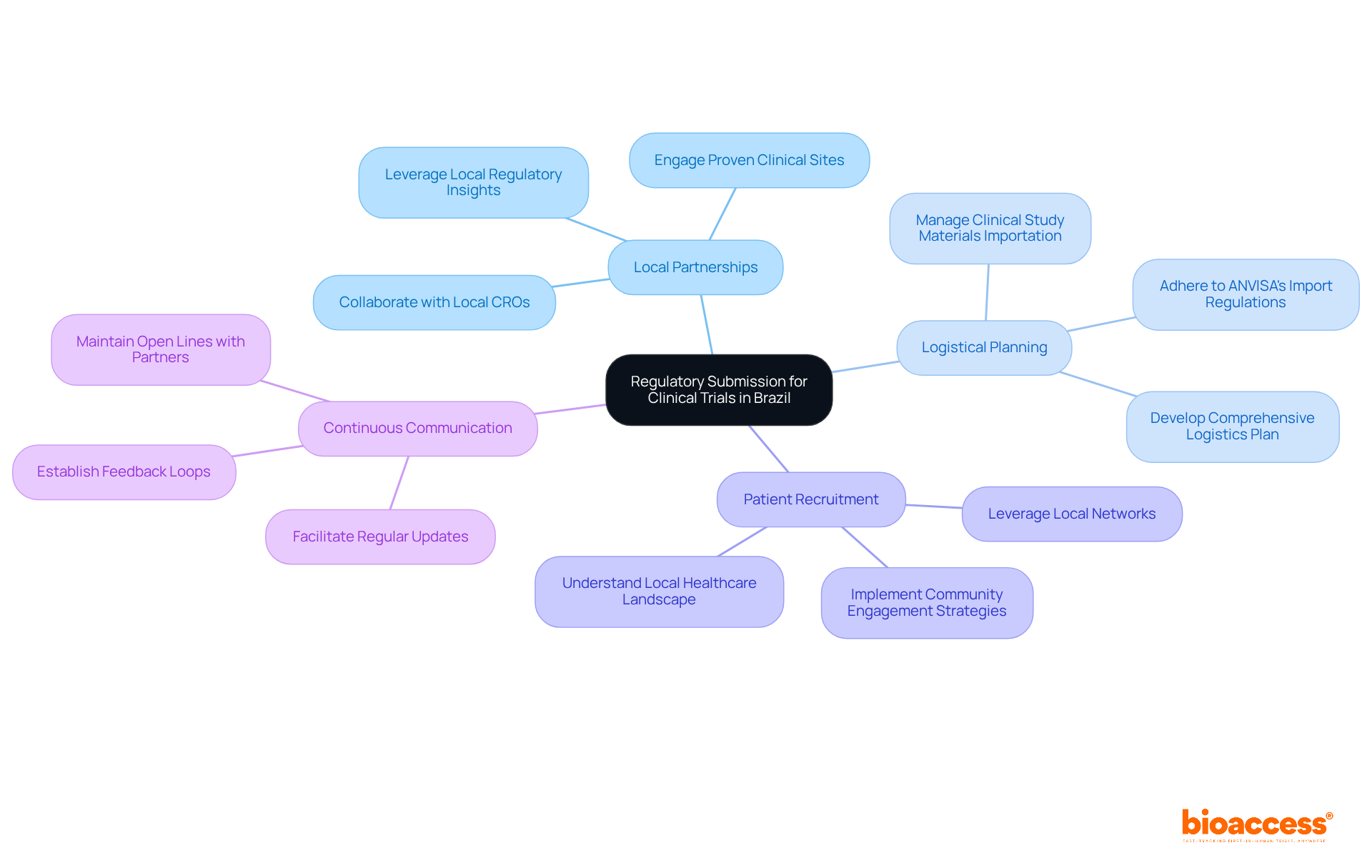
Navigating the complexities of regulatory submission for Brazil clinical trial requires more than just expertise; it demands strategic foresight and collaboration. Successful medical studies in Brazil require effective logistics management and strategic alliances, particularly for regulatory submission in a Brazil clinical trial. Key considerations include:
- Local Partnerships: Collaborating with local Contract Research Organizations (CROs) and clinical sites that have a proven track record in conducting trials in Brazil is essential. These partnerships provide invaluable insights into local regulations and effective patient recruitment strategies that are crucial for the regulatory submission Brazil clinical trial success.
- Logistical Planning: A comprehensive logistics plan must be developed to address the importation of clinical study materials, including medical devices and pharmaceuticals. Adhering to ANVISA’s import regulations is vital for the regulatory submission Brazil clinical trial to prevent delays that can jeopardize the entire study timeline, leading to costly setbacks.
- Patient Recruitment: Leveraging local networks and community engagement strategies can significantly enhance patient recruitment efforts. Understanding the local healthcare landscape, including its cultural nuances and access points, can significantly boost enrollment rates.
- Continuous Communication: Maintaining open lines of communication with all partners throughout the study process is vital. Regular updates and feedback loops facilitate the prompt identification and resolution of issues, ensuring that the study progresses smoothly.
By addressing these challenges, sponsors can not only avoid setbacks but also enhance the regulatory submission for their clinical trial in Brazil, positioning their studies for greater success. Embracing these strategies will not only streamline operations but also unlock the full potential of clinical research in Brazil.
Implement Post-Submission Strategies for Ongoing Compliance
Once a clinical trial submission is made, the journey toward compliance becomes paramount:
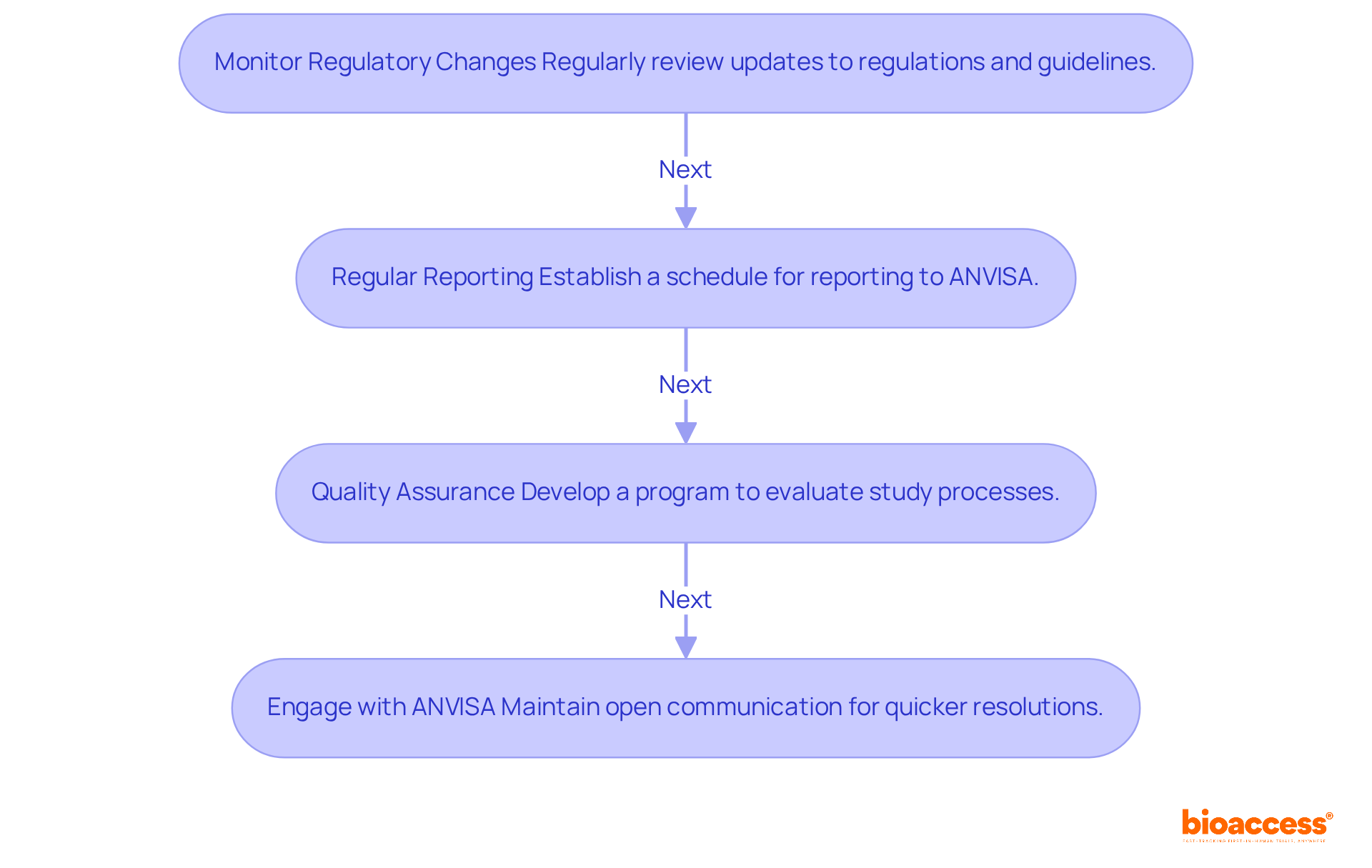
- Monitor Regulatory Changes: Regularly review updates to relevant regulations and guidelines that may impact your study. Track changes in approval timelines, which can now be completed within 90 working days, and modifications to documentation requirements. For example, Brazil documented 254 clinical studies in 2024, underscoring the expanding environment for clinical research and the importance of regulatory submission for Brazil clinical trial compliance.
- Regular Reporting: Establish a systematic schedule for reporting to ANVISA, including progress updates and documentation of any adverse events during the study. Timely and accurate reporting is essential for maintaining compliance and ensuring participant safety. Note that Research Ethics Committees (CEPs) now have a maximum period of 30 days to issue an opinion, which can significantly impact study timelines.
- Quality Assurance: Develop a comprehensive quality assurance program to routinely evaluate study processes and data management practices. Taking a proactive approach allows you to spot compliance issues before they become serious, helping you stick to ICH-GCP standards.
- Engage with ANVISA: Foster an open line of communication with ANVISA throughout the study. This engagement can facilitate quicker resolutions to any inquiries or concerns that may arise, enhancing the overall efficiency of the study process. As Anne McLellan highlights, ensuring compliance and being responsive to feedback is crucial for effective management of studies.
By implementing these post-submission strategies, sponsors can maintain compliance with regulatory requirements. This commitment leads to successful outcomes and a more efficient path to market.
Conclusion
Organizations face significant challenges in navigating Brazil’s complex regulatory submission landscape for clinical trials, which can impact their success in this dynamic environment. Understanding the framework set forth by ANVISA, including key regulations and streamlined processes, is crucial for conducting trials efficiently and ethically.
This article emphasizes the need for a strategic submission plan that includes careful document preparation, timeline management, and active engagement with stakeholders. By fostering local partnerships and maintaining open communication with regulatory bodies, sponsors can enhance their chances of successful trial outcomes. Additionally, implementing robust post-submission strategies ensures ongoing compliance and responsiveness to regulatory updates, ultimately facilitating smoother study progression.
As Brazil emerges as a top destination for innovative clinical research, adopting these best practices will streamline operations and create significant opportunities for MedTech, Biopharma, and Radiopharmaceutical companies. By prioritizing regulatory compliance, organizations can not only avoid setbacks but also position themselves for success in Brazil’s burgeoning clinical research market.
Frequently Asked Questions
What is the role of ANVISA in Brazil’s clinical trial regulatory framework?
The National Health Surveillance Agency (ANVISA) oversees the regulatory framework for clinical trials in Brazil, ensuring compliance with key regulations for sponsors conducting research.
What are the key regulations related to clinical trials in Brazil?
Notable regulations include Resolution RDC No. 9/2015, which outlines requirements for clinical trials involving drugs, and Resolution RDC No. 10/2015, which pertains to medical devices, detailing necessary documentation and ethical considerations.
What is the typical approval timeline for clinical trial applications in Brazil?
ANVISA typically provides a written opinion within 45 calendar days after submission. If no response is received within 90 days, the study can commence, provided all ethical approvals are in place.
How has the approval process changed under the new Brazilian Clinical Research Law?
The median total approval time for clinical studies under ANVISA has improved, with a firm 90-business-day limit on review processes established by the new Brazilian Clinical Research Law (Lei 14.874), effective January 1, 2025.
What is required before presenting a clinical study in Brazil?
Studies must obtain consent from a local ethics committee (CONEP) to ensure compliance with ethical standards in research.
How do the new regulations affect the evaluation process for clinical trials?
The new regulations allow for concurrent evaluation by ANVISA and ethics committees, which optimizes the process and decreases the time needed to commence studies.
Why is it important for sponsors to familiarize themselves with Brazil’s regulatory framework?
Understanding the regulatory framework helps sponsors prepare their applications effectively and anticipate potential challenges in the approval process, positioning Brazil as a leading destination for innovative clinical research.
List of Sources
- Understand Brazil’s Regulatory Framework for Clinical Trials
- Brazil Just Changed The Rules For Clinical Trial Approvals — What Sponsors Need To Know | bioaccess® (https://bioaccessla.com/blog/brazil-anvisa-parallel-review-clinical-trial-approvals-2026)
- (PDF) A Baseline Analysis of Regulatory Review Timelines for ANVISA: 2013–2016 (https://researchgate.net/publication/342061566_A_Baseline_Analysis_of_Regulatory_Review_Timelines_for_ANVISA_2013-2016)
- Brazil Cuts Clinical Trial Approval Timeline to 90 Days | bioaccess®: Fast-Tracking First-in-Human Trials, Anywhere posted on the topic | LinkedIn (https://linkedin.com/posts/bioaccess_clinicaltrials-brazil-anvisa-activity-7447976698958106627-98MW)
- Develop a Strategic Submission Plan for Regulatory Compliance
- Why clinical trial success rates are declining (https://ideagen.com/thought-leadership/blog/why-clinical-trial-success-rates-are-declining)
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- Benchmarking R&D success rates of leading pharmaceutical companies: an empirical analysis of FDA approvals (2006–2022) (https://sciencedirect.com/science/article/pii/S1359644625000042)
- Clinical Trial Success Rates: How Many Drugs Make It to Market? (Latest Approval Stats) (https://patentpc.com/blog/clinical-trial-success-rates-how-many-drugs-make-it-to-market-latest-approval-stats)
- Navigate Logistics and Build Strategic Partnerships for Success
- Brazil Clinical Trials Market Size, Share | Analysis Report [2032] (https://fortunebusinessinsights.com/brazil-clinical-trials-market-112310)
- New regulations for clinical research in Brazil | Licks Attorneys (https://lickslegal.com/post/new-regulations-for-clinical-research-in-brazil)
- Brazil Clinical Trials Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trials-market/brazil)
- Unlocking Brazil’s Clinical Trial Opportunity: A Strategic Roadmap for Healthcare Leaders (https://lek.com/insights/life-sciences-pharma/unlocking-brazils-clinical-trial-opportunity-strategic-roadmap)
- CLINICAL TRIALS SCENARIO IN BRAZIL- STUDY AND SPONSOR PROFILES (https://ispor.org/heor-resources/presentations-database/presentation/ispor-19th-annual-european-congress/clinical-trials-scenario-in-brazil-study-and-sponsor-profiles)
- Implement Post-Submission Strategies for Ongoing Compliance
- New regulations for clinical research in Brazil | Licks Attorneys (https://lickslegal.com/post/new-regulations-for-clinical-research-in-brazil)
- TOP 25 COMPLIANCE QUOTES (of 106) | A-Z Quotes (https://azquotes.com/quotes/topics/compliance.html)

Leave a Reply