Introduction
The landscape of medical device research is evolving rapidly, driven by the need for innovative strategies and the integration of digital marketing techniques. In this article, we will explore the importance of these strategies and how they are shaping the future of medical device marketing.
From developing targeted marketing campaigns to enhancing customer experience and leveraging artificial intelligence, we will delve into the key factors that contribute to the success of medical device research and marketing efforts. Additionally, we will discuss the significance of creating detailed buyer personas and optimizing demand generation and lead nurturing. Ultimately, this article aims to provide a comprehensive understanding of the intricacies involved in marketing medical devices and the role it plays in advancing healthcare technology.
The Importance of Innovative Strategies
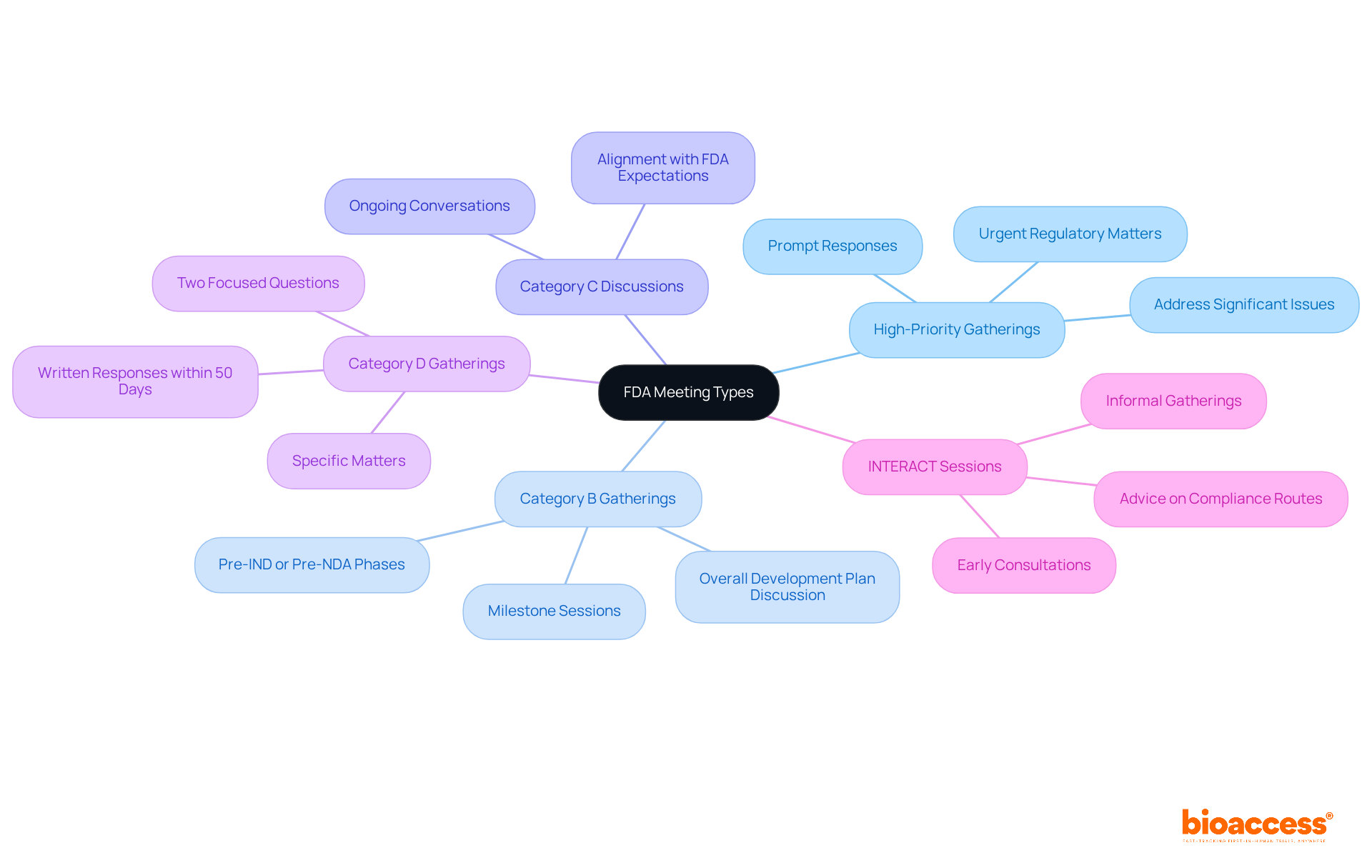
The exploration of innovative strategies in research is pivotal for the advancement of . These strategies are not a one-size-fits-all solution; they are as diverse as the global markets they aim to serve. A nuanced approach is required, one that considers the unique medical and technological needs of various regions.
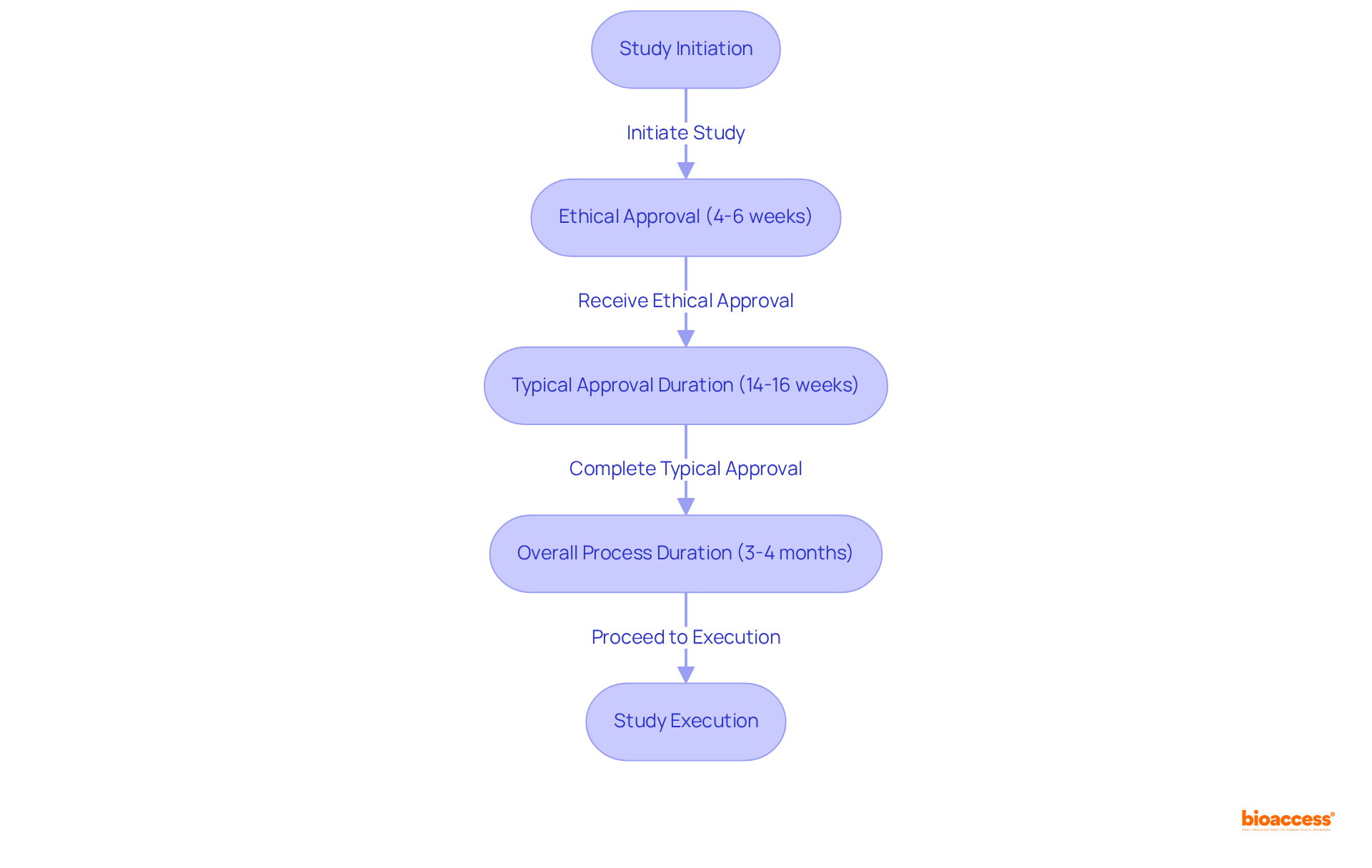
For instance, the strategy for a targeting low- and middle-income countries (LMICs) must be tailored to their specific contexts. With over 128 LMICs, including burgeoning economies like China, India, and Brazil, researchers must pinpoint key target markets based on disease incidence and prevalence data. By focusing on a select few representative locations, valuable insights can be drawn despite the inherent differences between countries.
Collaboration is also key. Identifying the right local partners can streamline the process of gathering and facilitate stakeholder site visits. The initial team meeting is crucial for aligning stakeholders on project priorities, which typically revolve around budget, time, and quality.
Understanding each team member’s role and responsibility, as outlined in the , lays the groundwork for successful project execution. Furthermore, as Dr. Thomas Fogarty insightfully remarked, the success of a hinges not only on the idea but on its implementation and acceptance within the healthcare ecosystem. This underscores the importance of innovators understanding and addressing the multifaceted needs of patients, physicians, providers, payors, regulators, investors, and acquirors from the outset.
The is witnessing a surge in activity due to the growing need for , preventative treatments, early diagnosis, and technologies like machine learning and digitalization. With over 710,000 patents filed and granted in the past three years, it’s crucial to recognize that innovations evolve along an from emergence to maturity. Identifying where an innovation stands in this life cycle is vital for gauging its adoption rate and future impact.
Embracing Digital Marketing for Medical Devices
In the realm of , which encompass a vast array of apparatus, machines, and software designed for diagnostic and therapeutic purposes, has become a cornerstone for promotion and advancement. By leveraging the interconnectedness of digital platforms, researchers and manufacturers can communicate the significance and benefits of their medical innovations to a broader audience.
The strategic use of techniques like SEO, social media, and content marketing enables them to highlight the value of that improve and quality of life. Telehealth, a sector that has seen a remarkable uptake during the recent global health crisis, stands as a testament to the synergy between healthcare and digital innovation.
With gaining traction, marketing opportunities in this space have expanded. The , driven by Big Data, mobile apps, and AI, is reshaping not only but also the distribution of medical supplies and services, making it crucial for to adapt and innovate. As the industry navigates through this digital revolution, it’s imperative to connect with the right professionals who can amplify the reach of these medical advancements. For those seeking to navigate the complexities of marketing in the medical device sector, reaching out to a team of experts can streamline the process of identifying the appropriate audience and crafting a message that resonates, ultimately driving forward the mission of enhancing healthcare through technology.

Developing Targeted Marketing Campaigns
At the core of lies the with the intricacies of . The initial step is to forge a clear and well-defined research question, which acts as the bedrock for constructing a robust study design.
Researchers must judiciously select variables to manipulate and outcomes to measure, drawing on a meticulously planned experimental design to curtail biases and confounding factors. By paralleling these scientific considerations with marketing efforts, researchers can craft messages that resonate deeply with the medical community, particularly emphasizing how —like machine learning, augmented reality, and digitalization—can revolutionize homecare, early diagnosis, and patient recovery.
With over in the last three years, as reported by GlobalData, the landscape is ripe with innovations at various stages of the adoption lifecycle. Marketing campaigns must, therefore, be nimble and informed, targeting who are attuned to the S-shaped curve of technology adoption—from early emergence to maturity.
As one experienced business development leader notes, the success of medical startups often hinges on grit and perseverance, but also on strategic business moves such as acquisitions, IPOs, or forming alliances. To effectively capture the attention of discerning and stakeholders, a proactive approach to public relations is crucial. As highlighted by industry experts, influencing decision-makers requires early and sustained engagement, addressing their risk aversion with well-timed, strategic communications. By integrating these insights into marketing campaigns, medical device researchers can ensure that their innovations are not only groundbreaking but also widely adopted and successful in improving .
Enhancing Customer Experience in Medical Device Marketing
Excellence in hinges on crafting a that resonates on a personal level with all stakeholders involved in healthcare—from patients to clinicians and maintenance staff. By embracing a holistic approach to design, focusing on the nuances of , researchers can develop that not only meet the unique needs of each user group but also create a positive emotional response upon interaction. This approach, rooted in empathy and understanding, leads to greater adoption, improved usability, and ultimately, .
Indeed, the emotional interaction a user has when they see or touch a medical device—often occurring instantaneously and before any practical use—can significantly shape perceptions of quality and performance. Addressing this by understanding users’ needs and desires through field research and collaboration with Human Factors Engineers is pivotal. It ensures that the design process is informed by real-world experiences and expectations, which is crucial for fostering a sense of confidence and ease among users.
The success of a medical device also extends into the business realm, where strategic decisions can lead to . A combination of grit, perseverance, and strategic business factors—such as acquisitions, IPOs, licensing agreements, and strategic alliances—can culminate in a profitable conclusion of operations or ownership changes. Such outcomes are a testament to the importance of aligning with business acumen in the medical device industry.
The Role of Artificial Intelligence in Medical Device Research
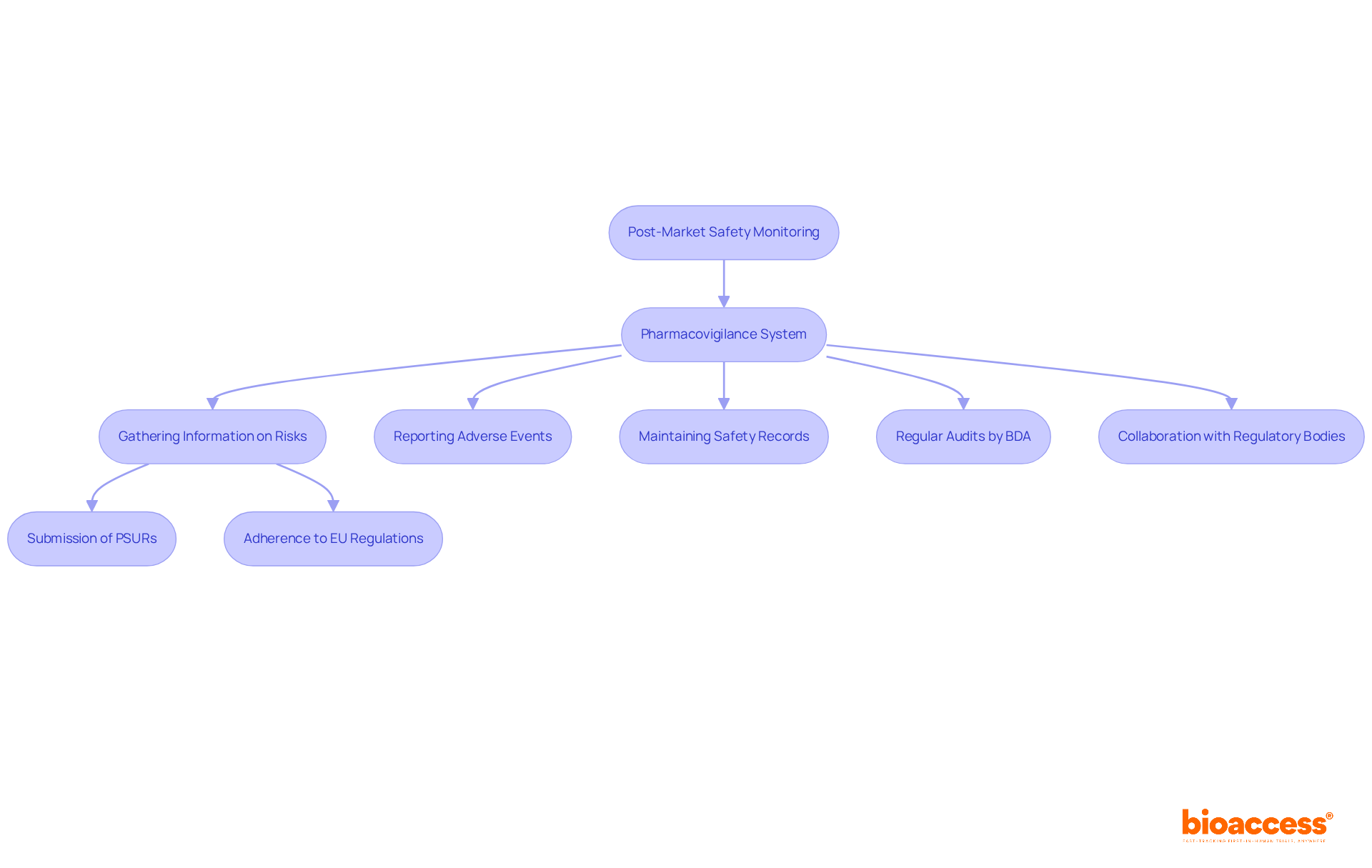
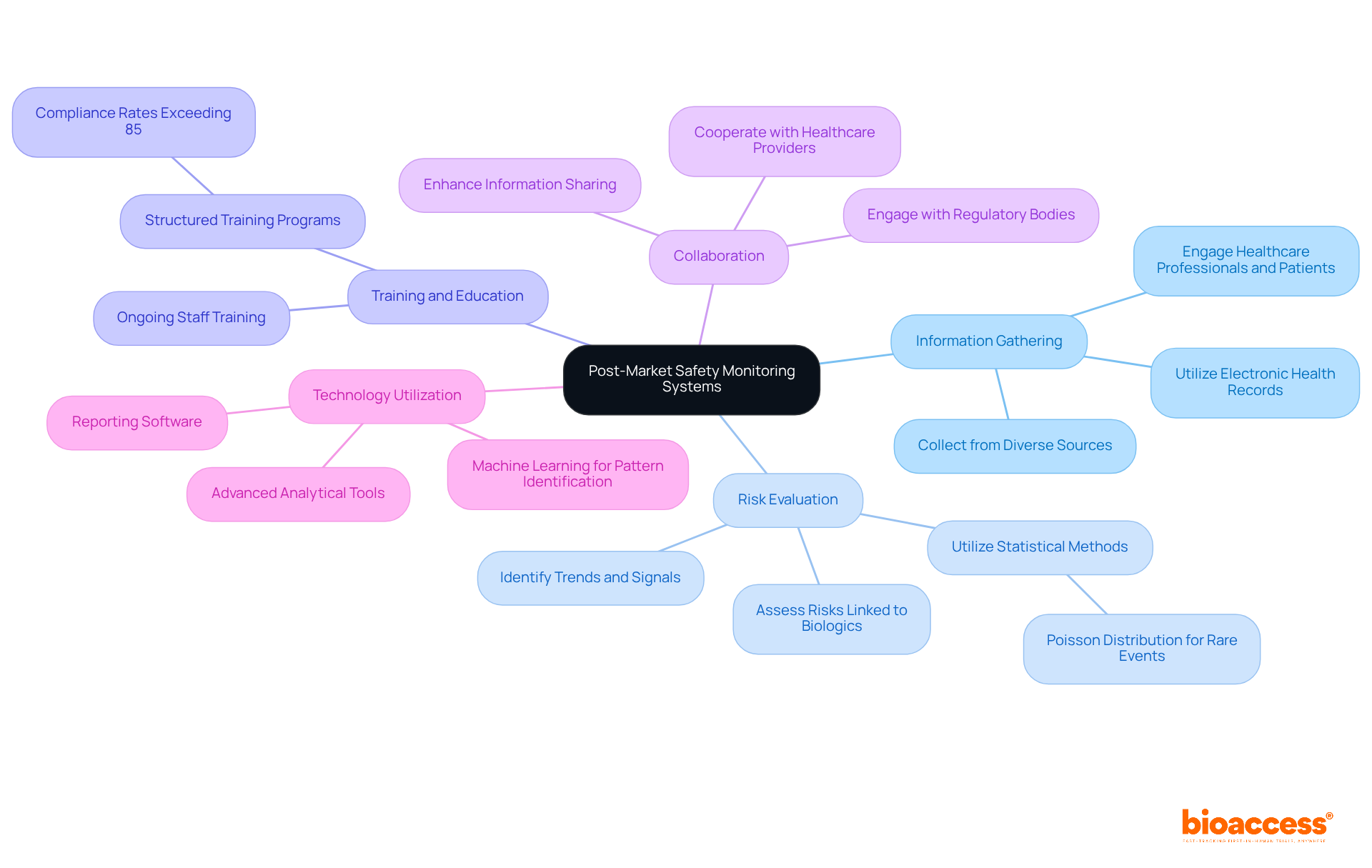
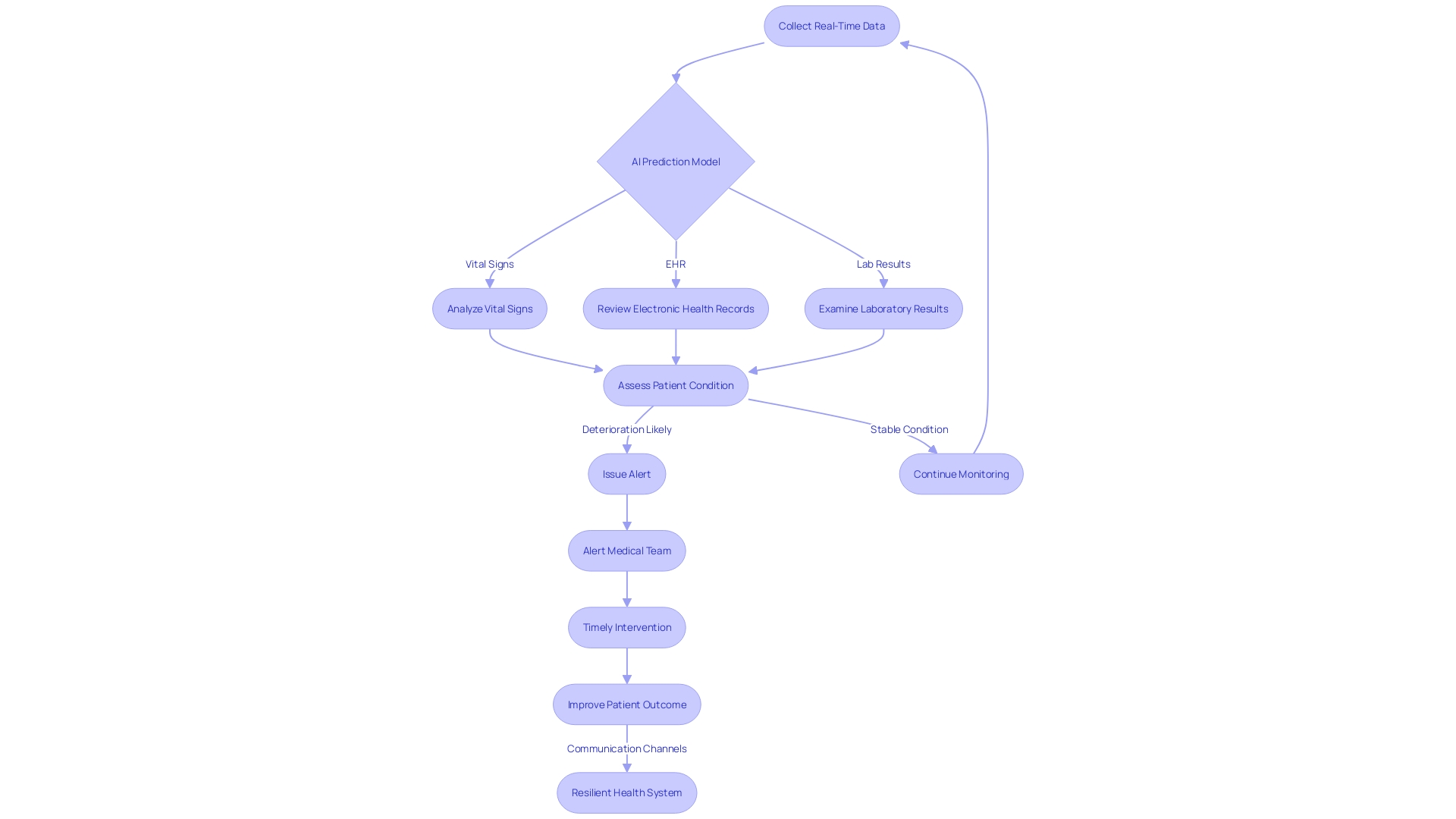
The integration of Artificial Intelligence (AI) into heralds a new era in healthcare innovation. One exemplary case is an that utilizes real-time data from various sources, such as vital signs, electronic health records, and laboratory results. This model scrutinizes data approximately every 15 minutes to forecast potential patient health deterioration.
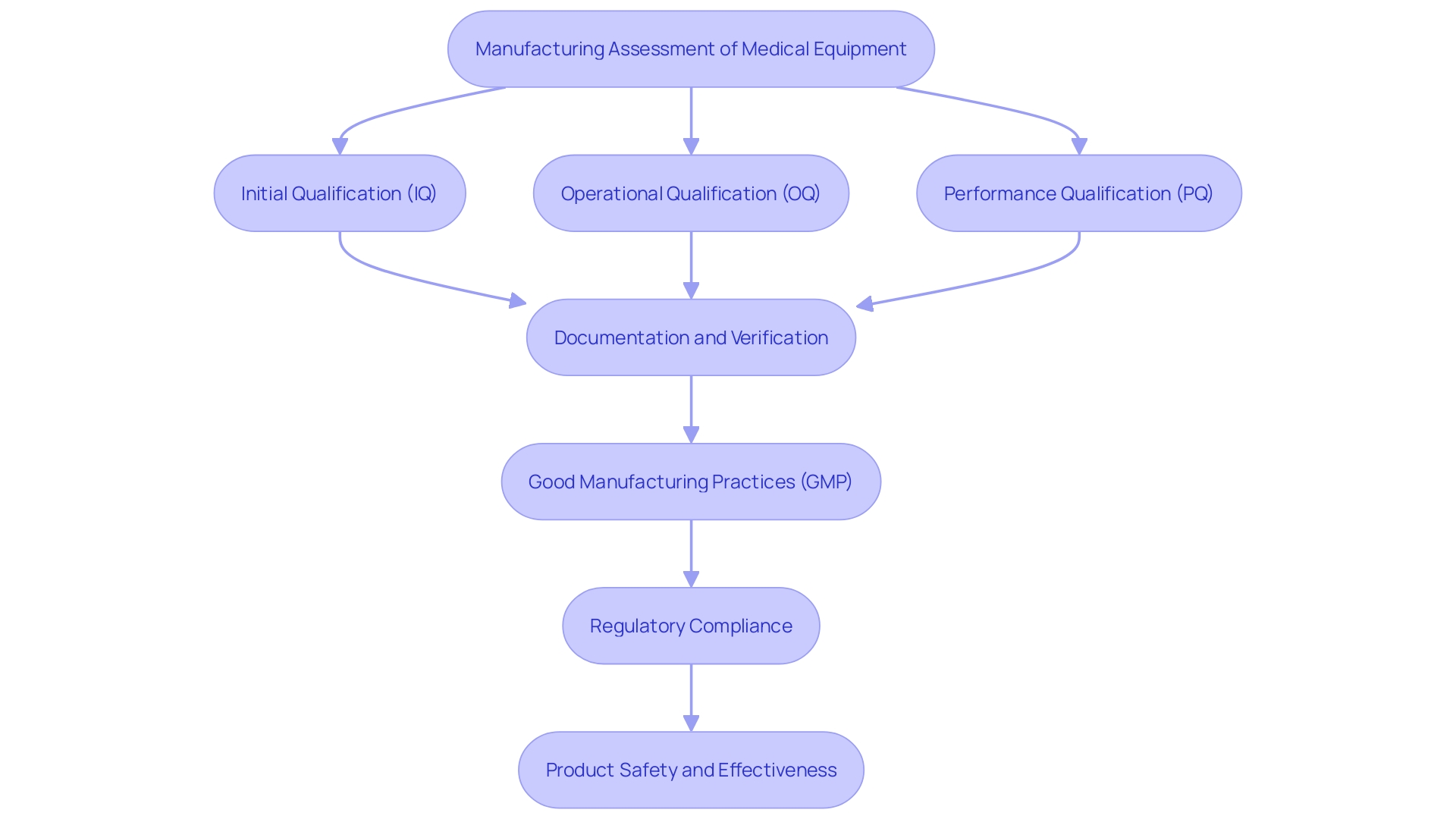
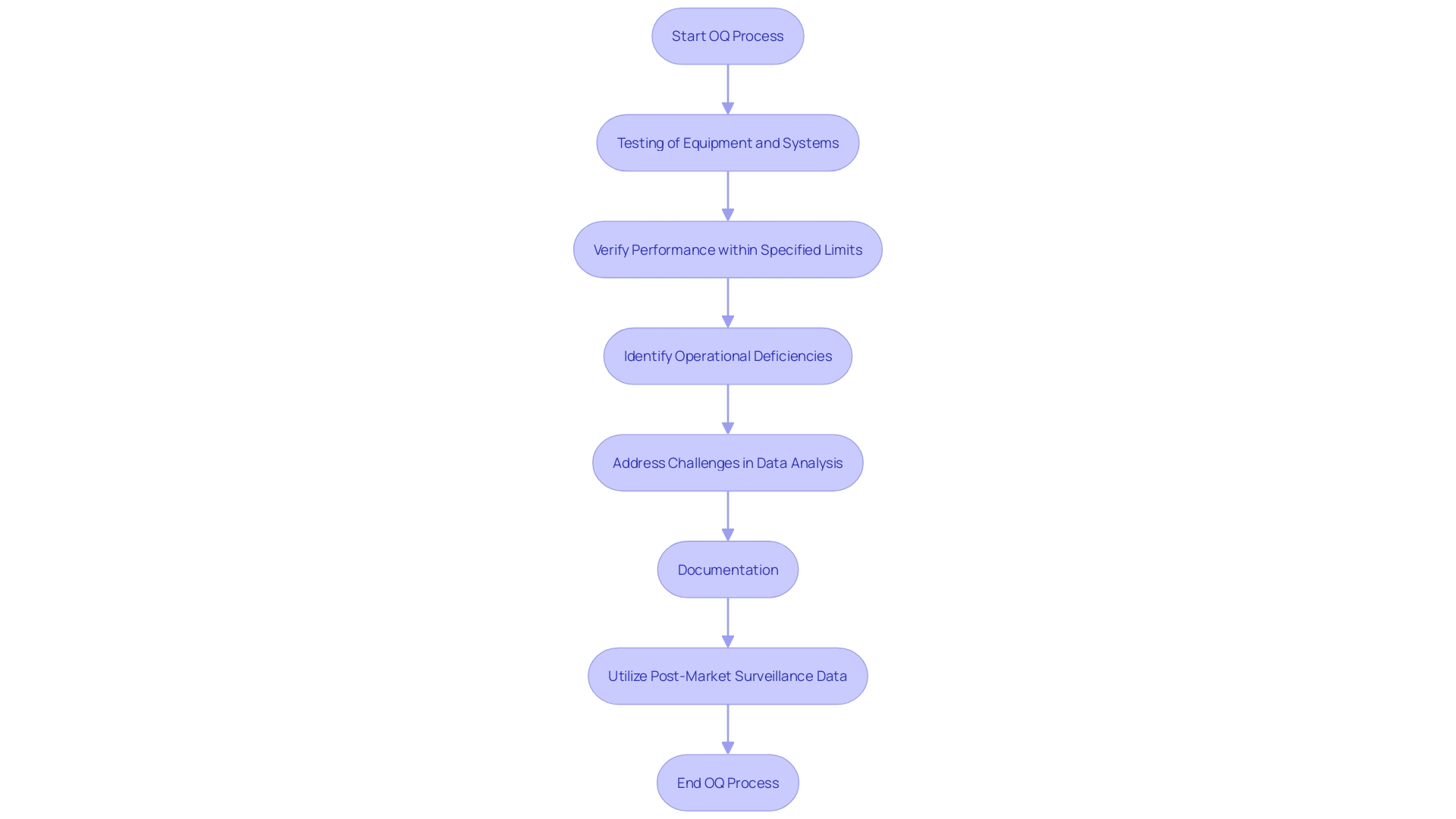
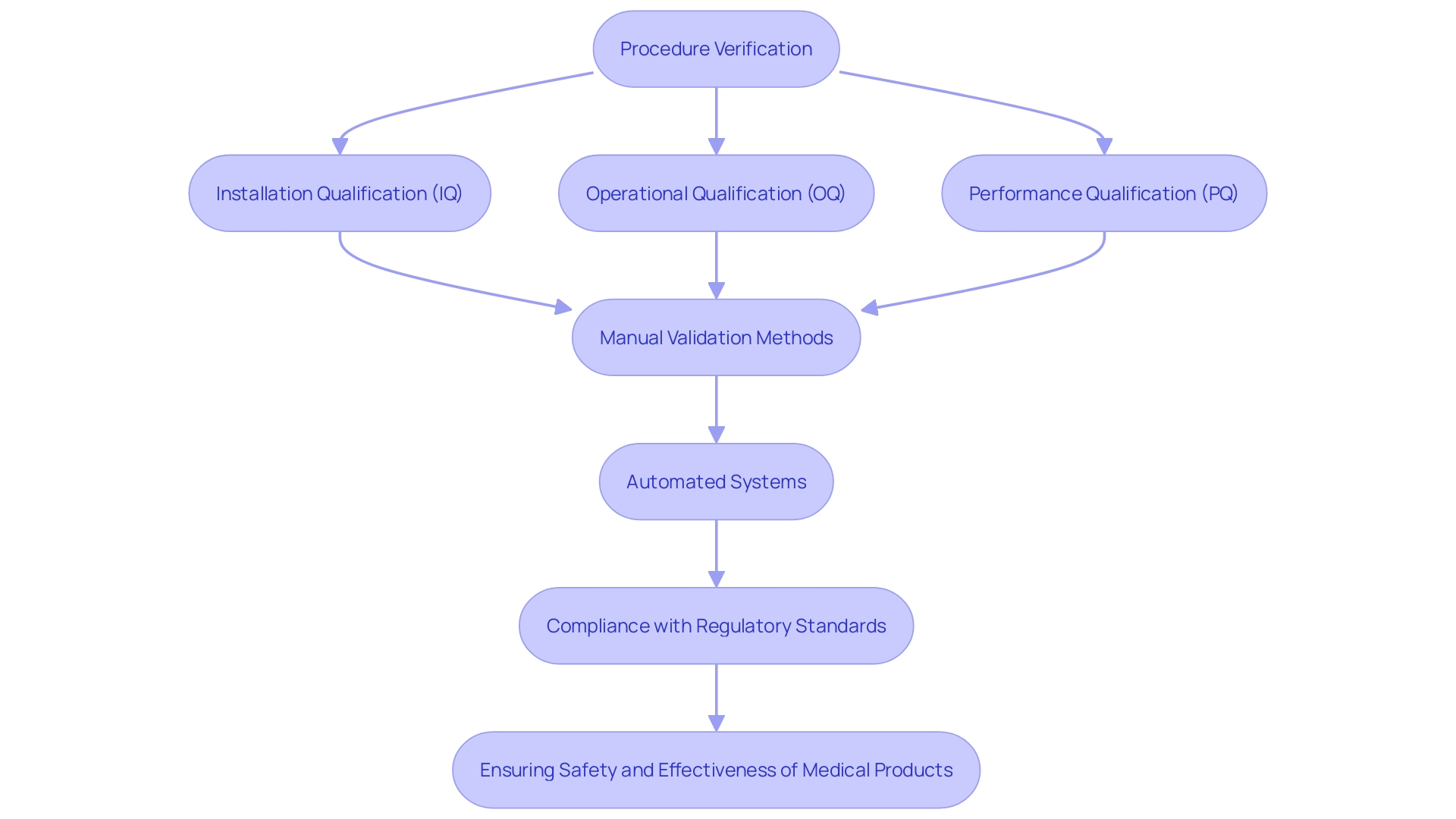
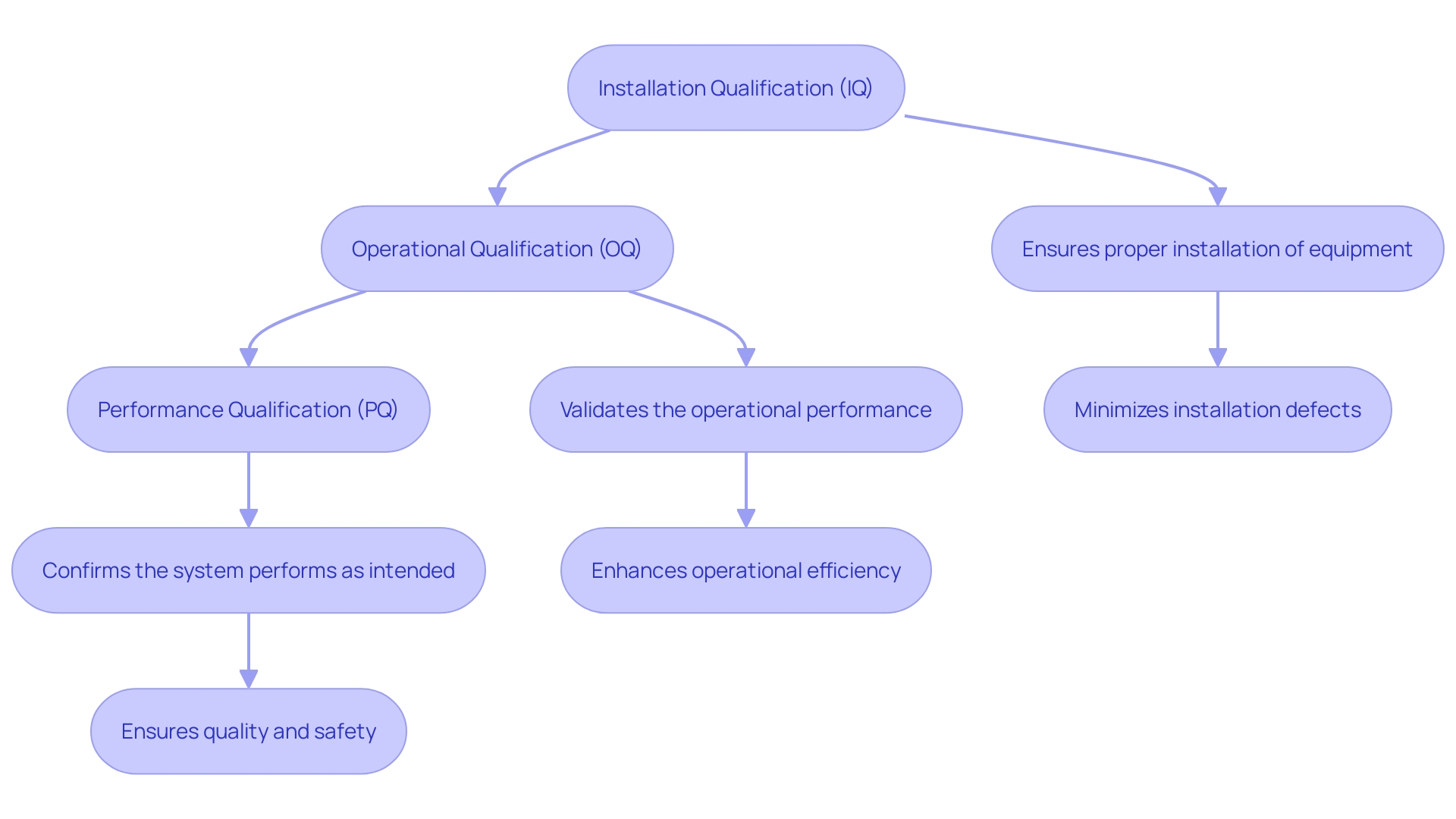
In instances where a patient’s condition is likely to worsen, the model issues alerts to the medical team, prompting timely interventions and fostering a resilient through improved communication channels. In the broader scope of medical device design, AI’s role is increasingly pivotal. The is crucial, ensuring that the device’s performance aligns with its intended design.
This phase involves rigorous testing, as seen with Renishaw’s neuroinfuse drug delivery system, which delivers medication directly to the brain, bypassing the blood-brain barrier. Such intricate systems underscore the importance of AI in enhancing . Aengus Ó Curraidhín, a senior medical design/development engineer at Renishaw, emphasizes the significance of AI in the future of MedTech.
With Ai’s ascendancy, the industry is poised to . However, these advancements necessitate robust design and validation processes to guarantee patient safety. As the industry trends towards integrating AI, the thematic intelligence report by GlobalData outlines the technological, macroeconomic, regulatory, and industry trends that are shaping the theme over the next 12 to 24 months, highlighting the transformative potential of AI across the medical value chain.
Creating Detailed Buyer Personas for Medical Device Marketing
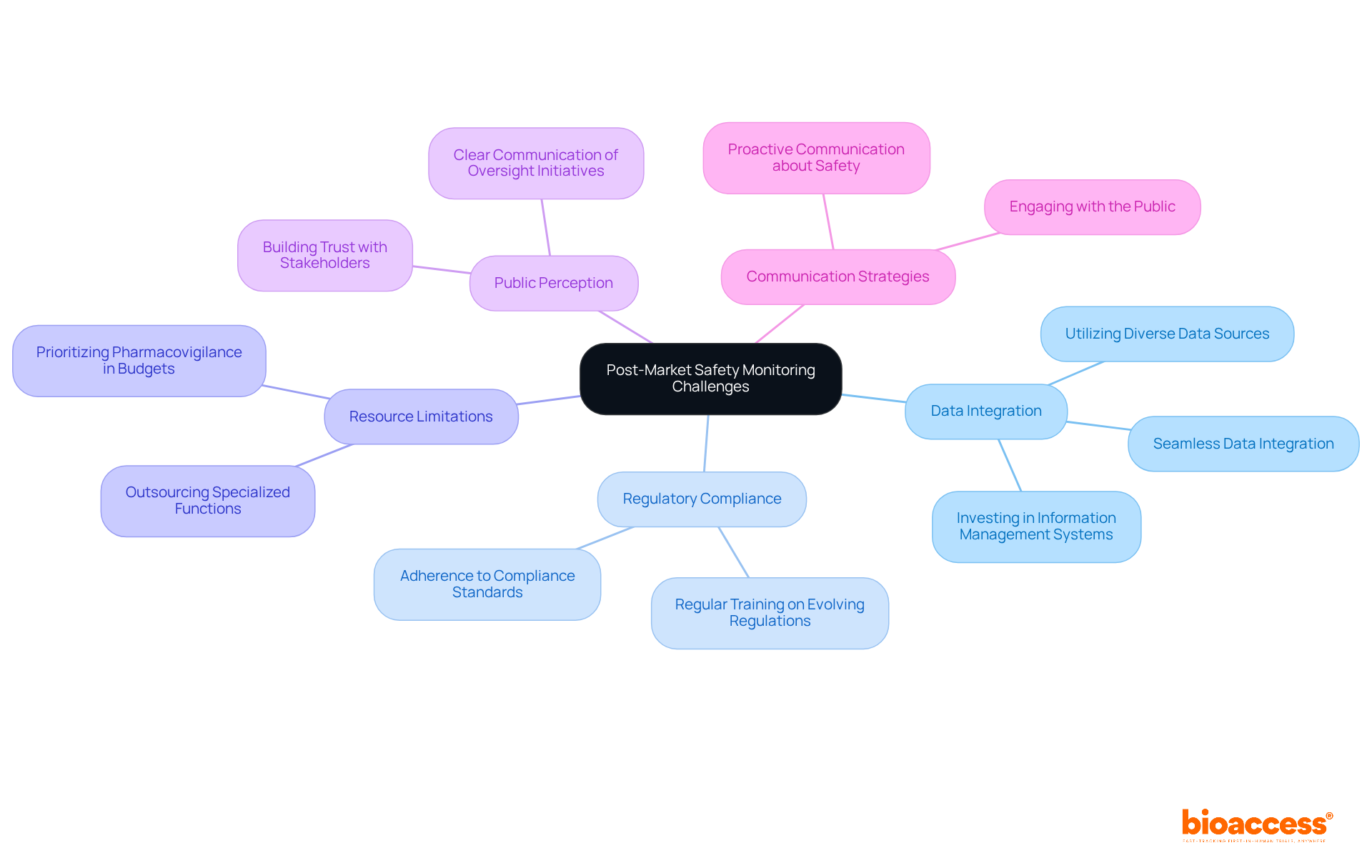
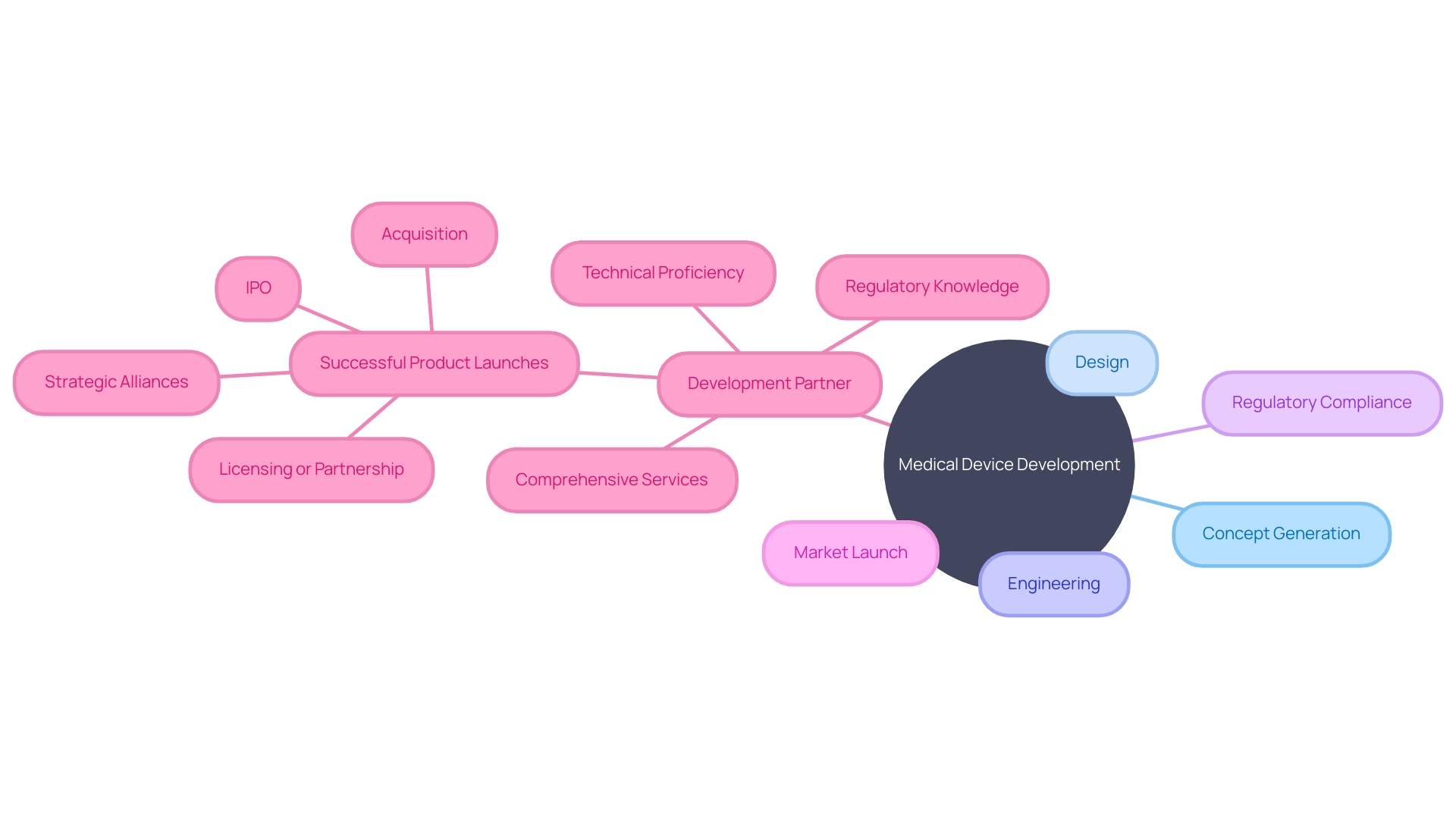
The landscape of is intricate and multifaceted, encompassing concept generation, design, engineering, , and successful market launch. Stakeholders in the prioritize finding a that offers a comprehensive suite of services. This holistic approach not only streamlines the transition between development phases but also mitigates costs and accelerates the , which is imperative for the success of any medical device.
The ideal partner is one that merges technical proficiency with an in-depth grasp of the regulatory environment and market tendencies. Indeed, the World Health Organization (WHO) defines medical devices broadly, ranging from simple tongue depressors to complex diagnostic software, each with its unique set of development challenges. Furthermore, the integration of software with medical hardware is becoming increasingly vital, especially in the digital health domain, providing a substantial competitive advantage.
According to industry experts, a company’s history of is indicative of its capability to navigate the intricate journey from an idea to market presence. This proficiency is a critical factor for and should be a primary consideration. As the medical devices industry evolves, staying informed and making strategic connections with the right s is more crucial than ever, especially for stakeholders aiming to navigate the competitive and regulated medtech landscape.
Optimizing Demand Generation and Lead Nurturing
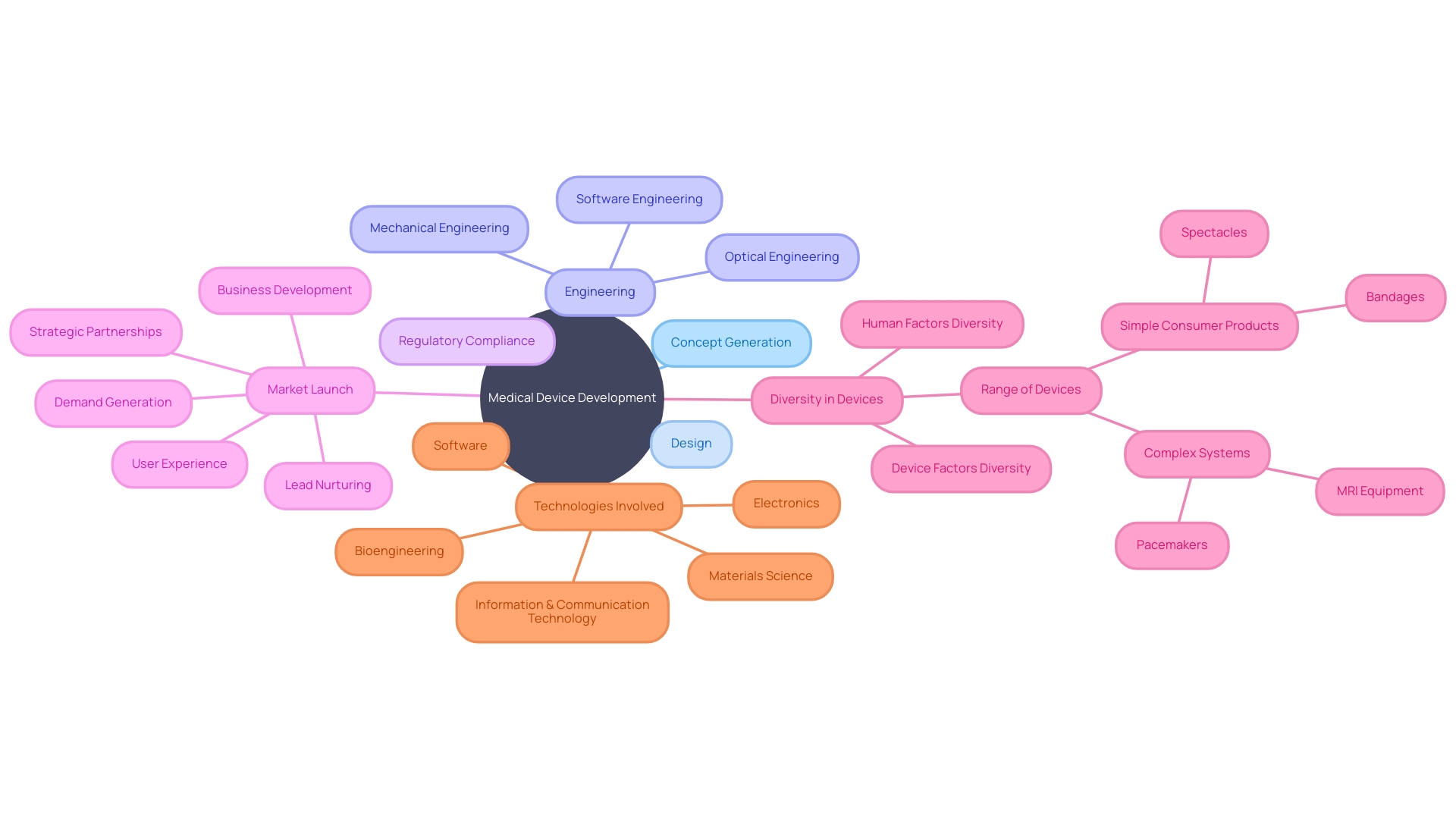
In the dynamic realm of , the journey from concept generation to market launch is intricate and multifaceted, involving a diverse array of technologies and disciplines. As the healthcare landscape evolves, so too does the complexity of , ranging from the simplicity of spectacles to the sophistication of MRI equipment and pacemakers.
These devices are not only varied in function but also in the human and device factors they encompass, catering to a wide spectrum of individual differences and needs. To navigate this diversity and complexity, researchers must adopt a holistic approach to development, integrating optical, mechanical, and software engineering with user experience.
This systems-oriented perspective ensures that all aspects of the device’s lifecycle are considered, from initial design to patient interaction. With over 10,000 types of identified by the World Health Organization, the significance of a cannot be overstated.
In this context, demand generation and lead nurturing transcend mere marketing tactics; they become instrumental in bridging the gap between innovation and implementation. By engaging potential users early and guiding them through the decision-making process with valuable information and trust-building interactions, researchers can optimize the adoption of groundbreaking . The success of these endeavors is often marked by strategic exits, be it through acquisition, IPO, or alliances, underscoring the importance of robust business development alongside technical expertise. The landscape is indeed a testament to the intertwined nature of science, engineering, and market dynamics. It is a sector where the successful commercialization of innovations demands not only a deep understanding of the but also an ability to foster that can propel a medical device from concept to clinical use.
Tailoring Product Messaging to the Target Audience
Effective communication of a ‘s value is paramount for its adoption and utilization. This necessitates a deep understanding of the emotional and practical needs of all users, which includes not only patients but also clinicians, caregivers, and hospital staff.
Crafting a message that resonates with these diverse stakeholders starts with recognizing the initial, often unconscious, emotional reaction users have when encountering the device. This can significantly influence perceptions of a device’s quality and performance, as well as the user’s mental state during its operation or application.
To foster a positive emotional connection, it’s essential to engage in . Through facilitated discussions and activities, we can glean insights into users’ emotional states and expectations related to medical procedures or conditions.
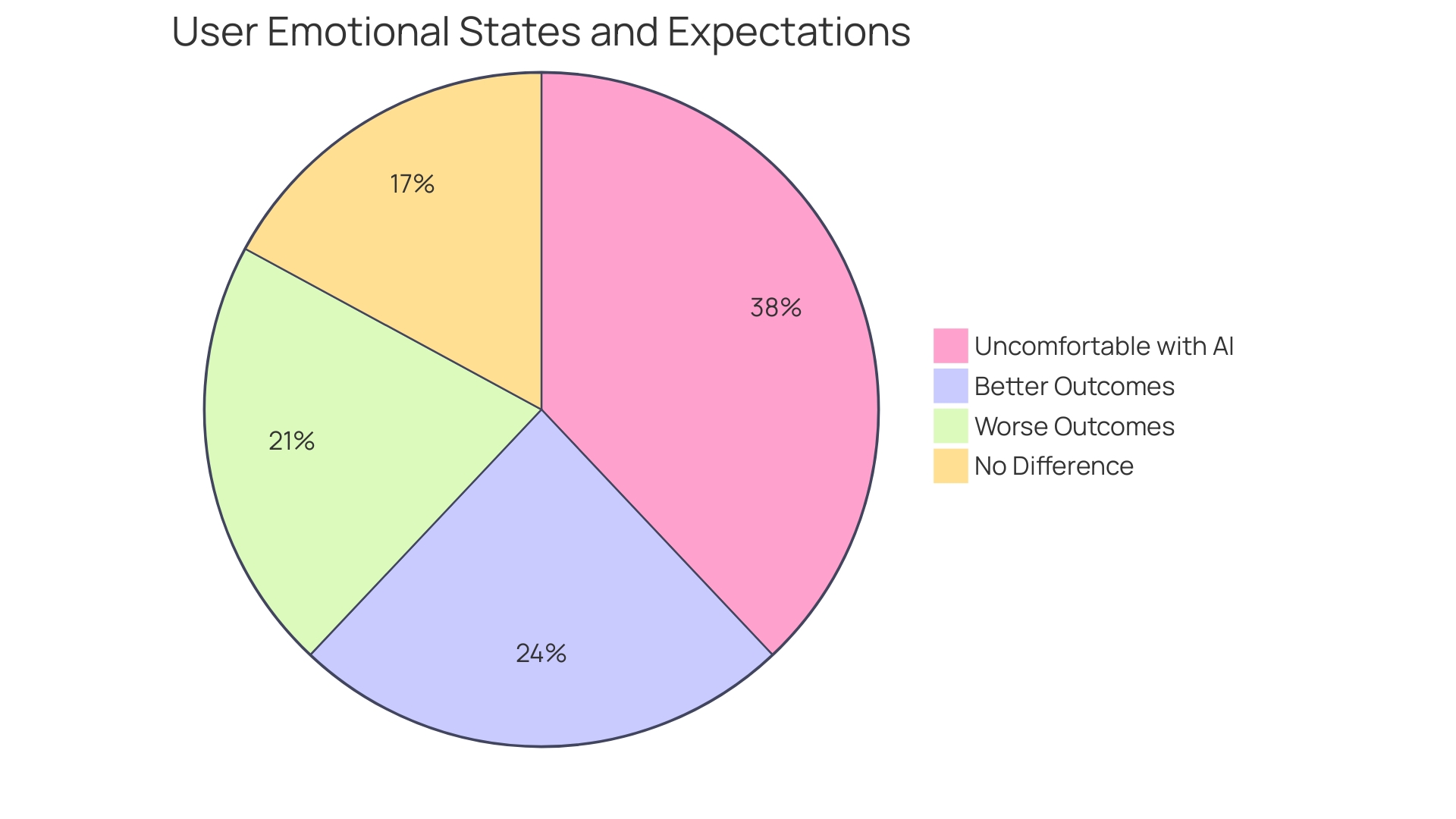
For instance, understanding the anxiety a patient feels before an MRI can inform a more empathetic approach to device design and communication. Moreover, about 50% of older adults face challenges with , often due to complex prescriptions or incorrect dosages.
Digital health solutions offer a promising avenue to improve adherence and patient outcomes, yet they must be designed with user-friendliness in mind, as some older adults find app settings and reminders cumbersome. Despite these hurdles, a significant majority (83%) of older adults are willing to learn new ways to enhance their well-being. In conclusion, s are not just tools for diagnosis or treatment; they are part of a broader ecosystem that includes the environment in which they are used. The design and messaging must therefore account for the setting and context of use, ensuring that the device is distinguishable and functional within its intended space. By adopting a comprehensive approach to device communication that encompasses , we can achieve not only better adoption rates but also improved compliance, usability, and .
Conclusion
In conclusion, the rapid evolution of medical device research is driven by innovative strategies and digital marketing techniques. Targeted campaigns aligned with controlled studies are crucial for promoting medical innovations, while digital marketing plays a pivotal role in reaching a broader audience. Enhancing customer experience through user-centered design is essential for improving clinical outcomes.
The integration of Artificial Intelligence (AI) revolutionizes healthcare innovation, from predicting patient health deterioration to enhancing device safety and efficacy. Creating detailed buyer personas streamlines development by finding the right partners offering comprehensive services. Optimizing demand generation and lead nurturing maximizes adoption rates for groundbreaking devices.
Tailoring product messaging to diverse stakeholders is paramount for effective communication. Understanding users’ emotional reactions and collaborating with Human Factors Engineers helps craft messages that resonate with target audiences. By embracing these strategies, we shape the future of medical device marketing and advance healthcare technology to improve patient outcomes.
Contact bioaccess™ today to learn how our targeted campaigns and digital marketing strategies can help promote your medical innovations and reach a broader audience for maximum impact.
Frequently Asked Questions
Why are innovative strategies important in medical device research?
Innovative strategies are essential for advancing healthcare technology. They cater to the diverse medical and technological needs of various global markets, especially in low- and middle-income countries (LMICs).
How should strategies be tailored for LMICs?
Strategies for medical devices targeting LMICs must consider specific regional contexts, including disease incidence and prevalence. Researchers should focus on a few representative locations to gain valuable insights.
What role does collaboration play in medical device research?
Collaboration is crucial for gathering Voice of the Customer (VoC) research and facilitating stakeholder engagement. Identifying local partners can streamline processes and enhance project success.
What is the RACI model, and why is it important?
The RACI model outlines team roles and responsibilities, which helps align stakeholders on project priorities such as budget, time, and quality. This clarity is vital for successful project execution.
How does the success of a medical device depend on its implementation?
According to Dr. Thomas Fogarty, a device’s success relies not just on the idea but also on its acceptance within the healthcare ecosystem. Innovators must consider the needs of patients, providers, and other stakeholders from the beginning.
What is the current trend in the medical devices industry?
There is a surge in activity driven by the demand for home care, preventative treatments, and early diagnosis, alongside advancements in technologies like machine learning and digitalization. Over 710,000 patents have been filed in the past three years.
How can digital marketing benefit medical device promotion?
Digital marketing techniques, such as SEO and social media, enable researchers and manufacturers to communicate the value of their medical innovations effectively. This is particularly important in the context of telehealth and remote monitoring services.
What is the significance of developing targeted marketing campaigns in medical device research?
Targeted marketing campaigns align with controlled medical studies, ensuring that the messaging resonates with the medical community. A clear research question is crucial for designing these campaigns effectively.
How can user experience impact the adoption of medical devices?
Creating a user experience that resonates with all stakeholders leads to greater adoption and improved clinical outcomes. Understanding emotional interactions with devices is essential for fostering positive perceptions.
What role does Artificial Intelligence (AI) play in medical device research?
AI enhances healthcare innovation by enabling real-time data analysis for predicting patient health deterioration. It also plays a critical role in the design verification phase, ensuring device safety and efficacy.
Why is it important to create detailed buyer personas for medical device marketing?
Developing buyer personas helps identify the unique needs of potential users, informing marketing strategies that effectively communicate the device’s value and improve adoption rates.
How can demand generation and lead nurturing optimize medical device adoption?
Demand generation and lead nurturing involve engaging potential users early in the decision-making process. This builds trust and encourages the adoption of innovative medical devices.
What strategies should be employed for effective communication of a medical device’s value?
Effective communication should address both the emotional and practical needs of users. Engaging in field research and collaborating with Human Factors Engineers can provide insights to enhance messaging and design.
How do emotional interactions with medical devices influence user perceptions?
Emotional interactions can significantly shape users’ perceptions of quality and performance. Understanding these emotional states is crucial for improving the overall user experience and device effectiveness.
What is the broader ecosystem surrounding medical devices?
Medical devices are part of a larger system that includes the environment in which they are used. Design and messaging must consider this context to ensure functionality and distinguishability within the intended setting.
List of Sources
- The Importance of Innovative Strategies
- infomeddnews.com (https://infomeddnews.com/solving-problems-in-healthcare/)
- starfishmedical.com (https://starfishmedical.com/blog/voice-of-the-customer-medical-device-research-in-low-and-middle-income-countries/)
- globaldata.com (https://www.globaldata.com:443/store/report/medical-devices-industry-m-and-a-deals-by-theme-quarterly-analysis/)
- medicaldevice-network.com (https://www.medicaldevice-network.com/data-insights/innovators-cpr-assistance-devices-medical-devices/)
- medicaldevice-network.com (https://www.medicaldevice-network.com/data-insights/innovators-cardiac-stimulation-implants-medical-devices/)
- starfishmedical.com (https://starfishmedical.com/blog/starting-medical-device-projects-on-the-right-foot/)
- Embracing Digital Marketing for Medical Devices
- infomeddnews.com (https://infomeddnews.com/the-role-of-digital-transformation-in-healthcare-distribution/)
- infomeddnews.com (https://infomeddnews.com/advertise-with-medical-device-news-magazine/)
- smartinsights.com (https://www.smartinsights.com/digital-marketing-strategy/healthcare-marketing-strategy-trends/)
- infomeddnews.com (https://infomeddnews.com/about-medical-device-news-magazine-2024/)
- globaldata.com (https://www.globaldata.com:443/store/report/medical-devices-industry-m-and-a-deals-by-theme-quarterly-analysis/)
- smartinsights.com (https://www.smartinsights.com/digital-marketing-strategy/healthcare-marketing-strategy-trends/)
- globaldata.com (https://www.globaldata.com:443/store/report/medical-devices-industry-m-and-a-deals-by-theme-quarterly-analysis/)
- Developing Targeted Marketing Campaigns
- infomeddnews.com (https://infomeddnews.com/how-to-conduct-controlled-medical-research-in-a-lab/)
- infomeddnews.com (https://infomeddnews.com/advertise-with-medical-device-news-magazine/)
- medicaldevice-network.com (https://www.medicaldevice-network.com/data-insights/innovators-targeted-neurostimulation-medical-devices/)
- starfishmedical.com (https://starfishmedical.com/blog/18-business-factors-that-determine-successful-medical-device-exits/)
- starfishmedical.com (https://starfishmedical.com/blog/starting-medical-device-projects-on-the-right-foot/)
- med-technews.com (https://www.med-technews.com/medtech-insights/latest-medtech-insights/what-are-you-waiting-for/)
- globaldata.com (https://www.globaldata.com:443/store/report/medical-devices-industry-m-and-a-deals-by-theme-quarterly-analysis/)
- starfishmedical.com (https://starfishmedical.com/blog/18-business-factors-that-determine-successful-medical-device-exits/)
- globaldata.com (https://www.globaldata.com:443/store/report/medical-devices-industry-m-and-a-deals-by-theme-quarterly-analysis/)
- Enhancing Customer Experience in Medical Device Marketing
- starfishmedical.com (https://starfishmedical.com/blog/18-business-factors-that-determine-successful-medical-device-exits/)
- starfishmedical.com (https://starfishmedical.com/blog/how-to-improve-emotional-interaction-with-a-medical-device/)
- starfishmedical.com (https://starfishmedical.com/blog/service-design-in-medical-device-development/)
- infomeddnews.com (https://infomeddnews.com/advertise-with-medical-device-news-magazine/)
- globaldata.com (https://www.globaldata.com:443/store/report/medical-devices-industry-m-and-a-deals-by-theme-quarterly-analysis/)
- The Role of Artificial Intelligence in Medical Device Research
- globaldata.com (https://www.globaldata.com:443/store/report/ai-in-medical-theme-analysis/)
- How AI improves physician and nurse collaboration (https://med.stanford.edu/news/all-news/2024/04/ai-patient-care.html)
- med-technews.com (https://www.med-technews.com/medtech-insights/ai-in-healthcare-insights/the-future-of-medtech-with-ai/)
- Creating Detailed Buyer Personas for Medical Device Marketing
- starfishmedical.com (https://starfishmedical.com/blog/18-business-factors-that-determine-successful-medical-device-exits/)
- medicaldevice-network.com (https://www.medicaldevice-network.com/buyers-guide/medical-device-development-companies/)
- globaldata.com (https://www.globaldata.com:443/store/report/medical-devices-industry-m-and-a-deals-by-theme-quarterly-analysis/)
- medicaldevice-network.com (https://www.medicaldevice-network.com/buyers-guide/medical-devices-development/)
- infomeddnews.com (https://infomeddnews.com/about-medical-device-news-magazine-2024/)
- starfishmedical.com (https://starfishmedical.com/blog/voice-of-the-customer-medical-device-research-in-low-and-middle-income-countries/)
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-commercialization-vision/)
- starfishmedical.com (https://starfishmedical.com/blog/18-business-factors-that-determine-successful-medical-device-exits/)
- medicaldevice-network.com (https://www.medicaldevice-network.com/buyers-guide/medical-device-development-companies/)
- Optimizing Demand Generation and Lead Nurturing
- octopart.com (https://octopart.com/pulse/p/ensuring-reliable-sourcing-medical-device-supply-chains)
- medicaldevice-network.com (https://www.medicaldevice-network.com/buyers-guide/medical-device-development-companies/)
- globaldata.com (https://www.globaldata.com:443/store/report/medical-devices-industry-m-and-a-deals-by-theme-quarterly-analysis/)
- starfishmedical.com (https://starfishmedical.com/blog/commercializing-medical-devices-with-optics/)
- starfishmedical.com (https://starfishmedical.com/blog/18-business-factors-that-determine-successful-medical-device-exits/)
- medicaldevice-network.com (https://www.medicaldevice-network.com/buyers-guide/medical-device-development-companies/)
- octopart.com (https://octopart.com/pulse/p/ensuring-reliable-sourcing-medical-device-supply-chains)
- Tailoring Product Messaging to the Target Audience
- infomeddnews.com (https://infomeddnews.com/about-medical-device-news-magazine-2024/)
- starfishmedical.com (https://starfishmedical.com/blog/how-to-improve-emotional-interaction-with-a-medical-device/)
- sciencedirect.com (https://www.sciencedirect.com/science/article/pii/S2949761223000913)
- med-technews.com (https://www.med-technews.com/medtech-insights/latest-medtech-insights/tips-and-tricks-for-user-centric-design/)
- starfishmedical.com (https://starfishmedical.com/blog/service-design-in-medical-device-development/)
- pewresearch.org (https://www.pewresearch.org/short-reads/2023/11/21/what-the-data-says-about-americans-views-of-artificial-intelligence/)