Introduction
The landscape of clinical research in Mexico is rapidly evolving, propelled by an increasing demand for innovative solutions and local expertise. This market is projected to grow significantly, presenting stakeholders with a unique opportunity to leverage local representative services that enhance efficiency and compliance in research operations.
However, navigating the complexities of this dynamic environment raises critical questions about how to effectively harness these resources while addressing potential challenges. As we delve deeper into this topic, it becomes essential to explore the Medtech landscape and the pivotal role of bioaccess in overcoming key obstacles faced in clinical research.
bioaccess: Accelerate Clinical Research with Local Expertise in Mexico
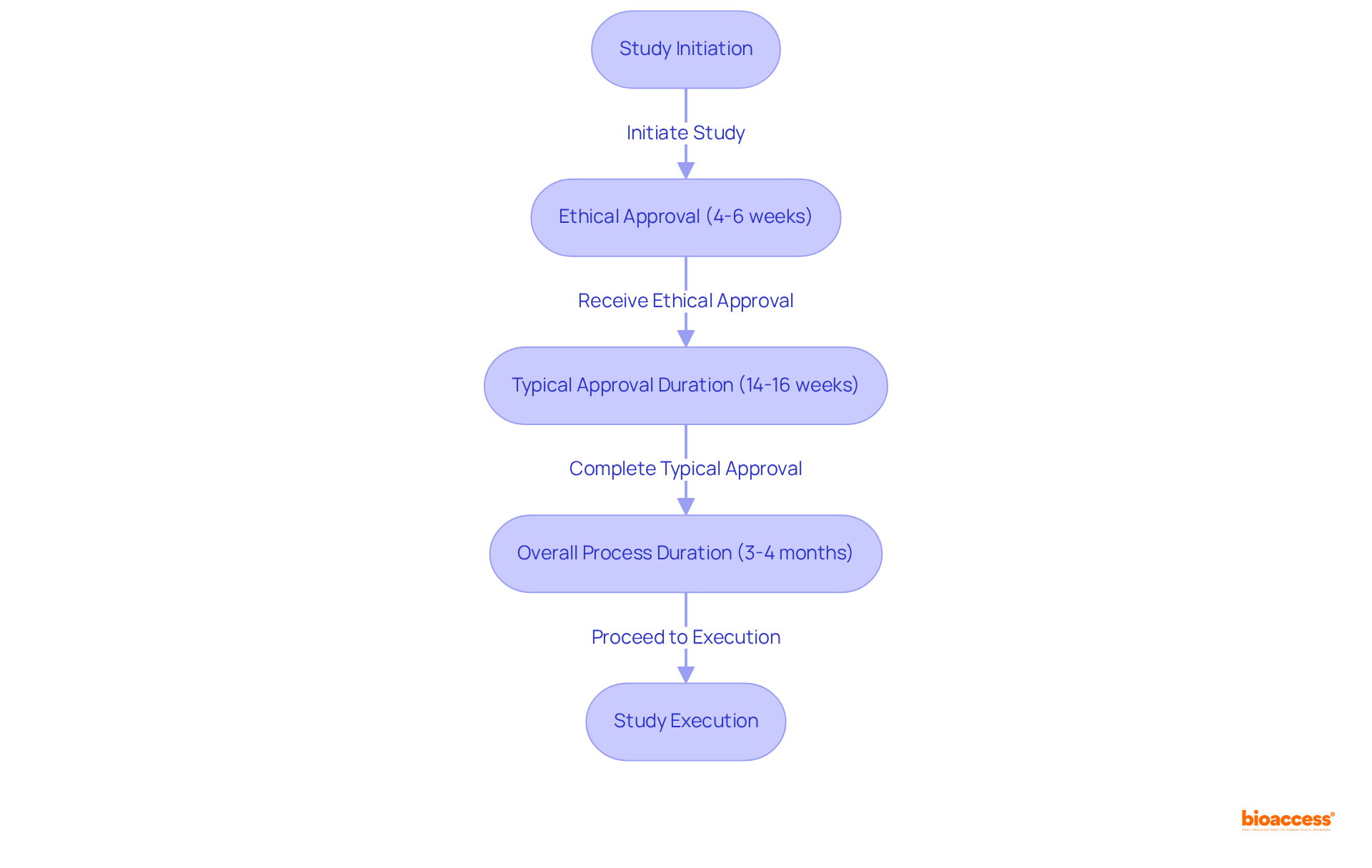
bioaccess® leverages over in Latin America to deliver exceptional research services for Medtech, Biopharma, and Radiopharma innovators. With ethical approvals secured in an impressive 4-6 weeks, bioaccess® enables a swift start to studies, fully capitalizing on Mexico’s advantageous .
Typically, the ranges from 14 to 16 weeks, while the overall approval and setup process generally spans 3-4 months, positioning it competitively with other Latin American countries. Moreover, the organization’s significantly enhances and study execution, ensuring high retention rates and fostering trust in the physician-patient relationship.
This combination of rapid regulatory processes and extensive local knowledge establishes bioaccess® as a preferred partner for , a market projected to reach USD 510.4 million by 2030, .
Nonetheless, it is crucial to acknowledge the challenges within the , including complexities that may lead to potential delays.
Cigna: Comprehensive Health Insurance for Clinical Trial Participants
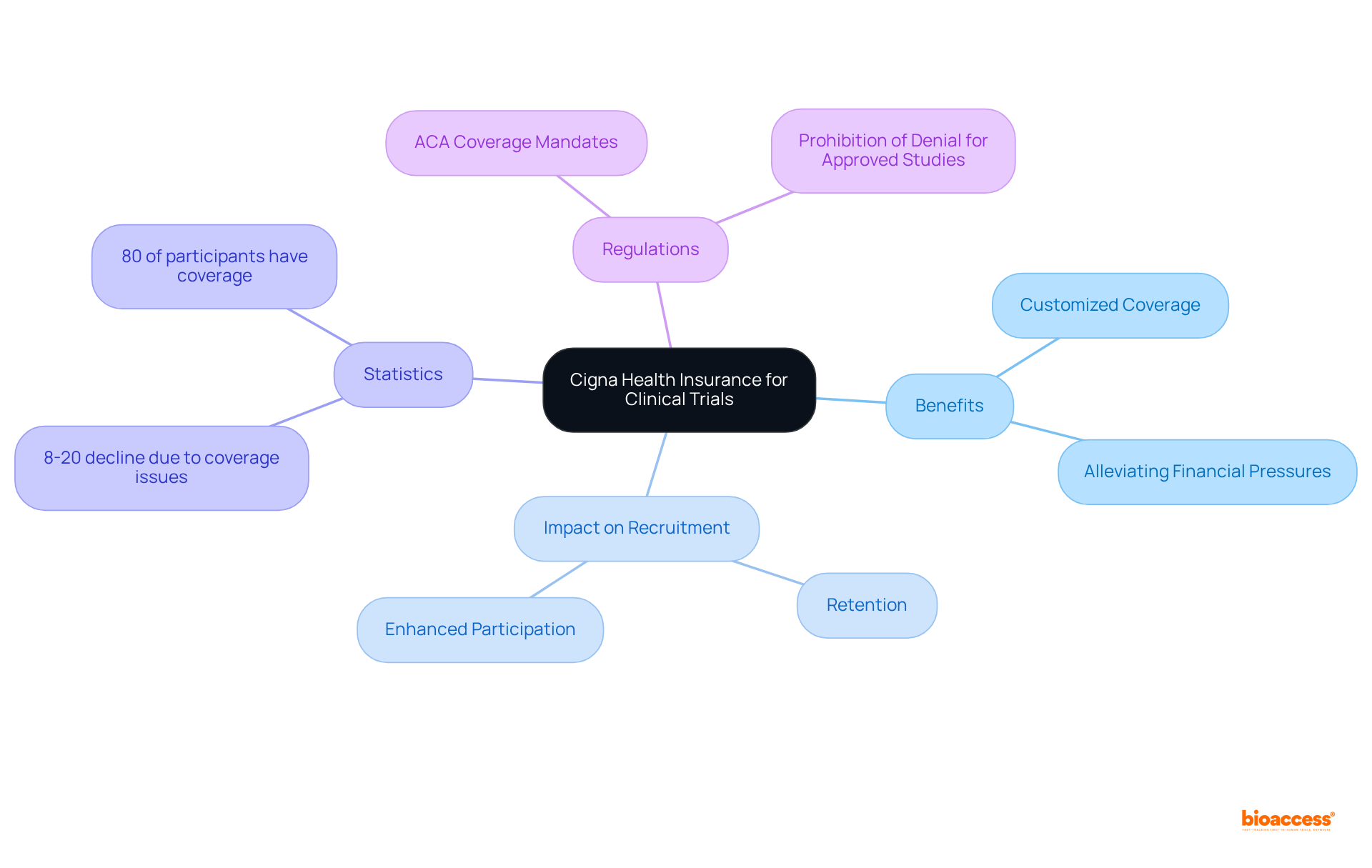
Cigna offers customized plans specifically designed for research participants, encompassing standard care alongside any additional medical costs incurred during the study. This extensive coverage significantly alleviates , thereby enhancing —two pivotal elements for the success of .
Research indicates that , with numerous patients citing . In fact, studies reveal that 8 to 20% of eligible patients decline participation due to potential denial of coverage. Furthermore, 80% of cancer research study participants possess that contributes to at least part of their care, underscoring the overall positive impact of coverage on participation rates.
By addressing these concerns, Cigna not only streamlines recruitment processes but also cultivates a supportive environment that encourages sustained participation, ultimately . According to the ACA, for cancer or another life-threatening illness, highlighting the crucial significance of coverage in medical investigations.
Progressive: Tailored Insurance Solutions for Clinical Research Operations
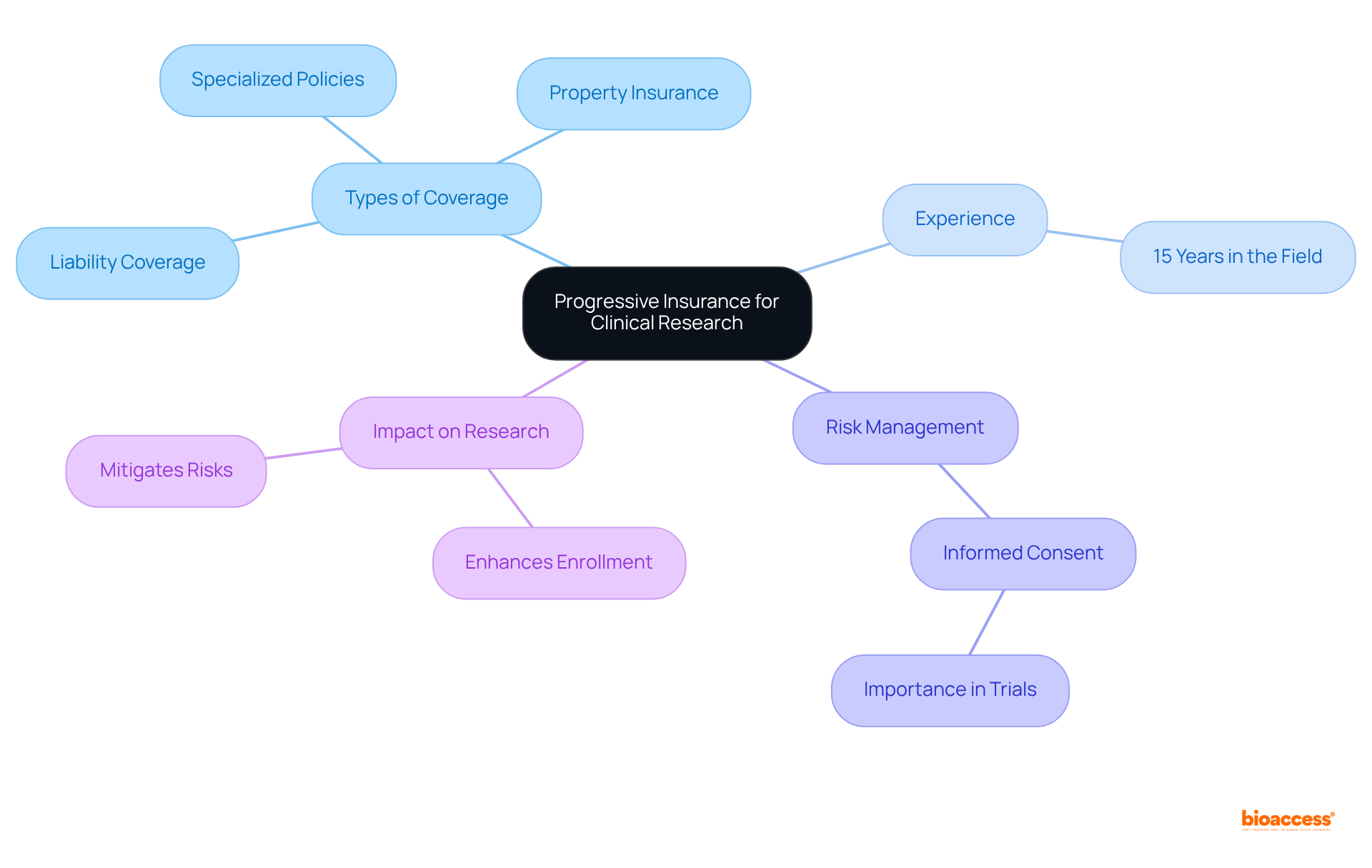
Progressive offers tailored coverage options designed to address the distinct hazards associated with . Their extensive offerings include:
- Property insurance
With over in the field, Progressive understands the intricacies of medical studies and provides customized coverage that not only protects against potential liabilities but also fosters trust among participants, ultimately . This approach is particularly crucial as the landscape of evolves, introducing new risks, such as those linked to decentralized studies and , a reality underscored during the COVID-19 pandemic.
Furthermore, remains a critical component in ensuring that participants understand their involvement in studies, an aspect Progressive emphasizes in their . By effectively mitigating these risks, Progressive bolsters the viability of experimental operations, facilitating smoother project execution and addressing essential elements like that may arise during trials.
GEICO: Reliable Auto Insurance for Clinical Research Teams
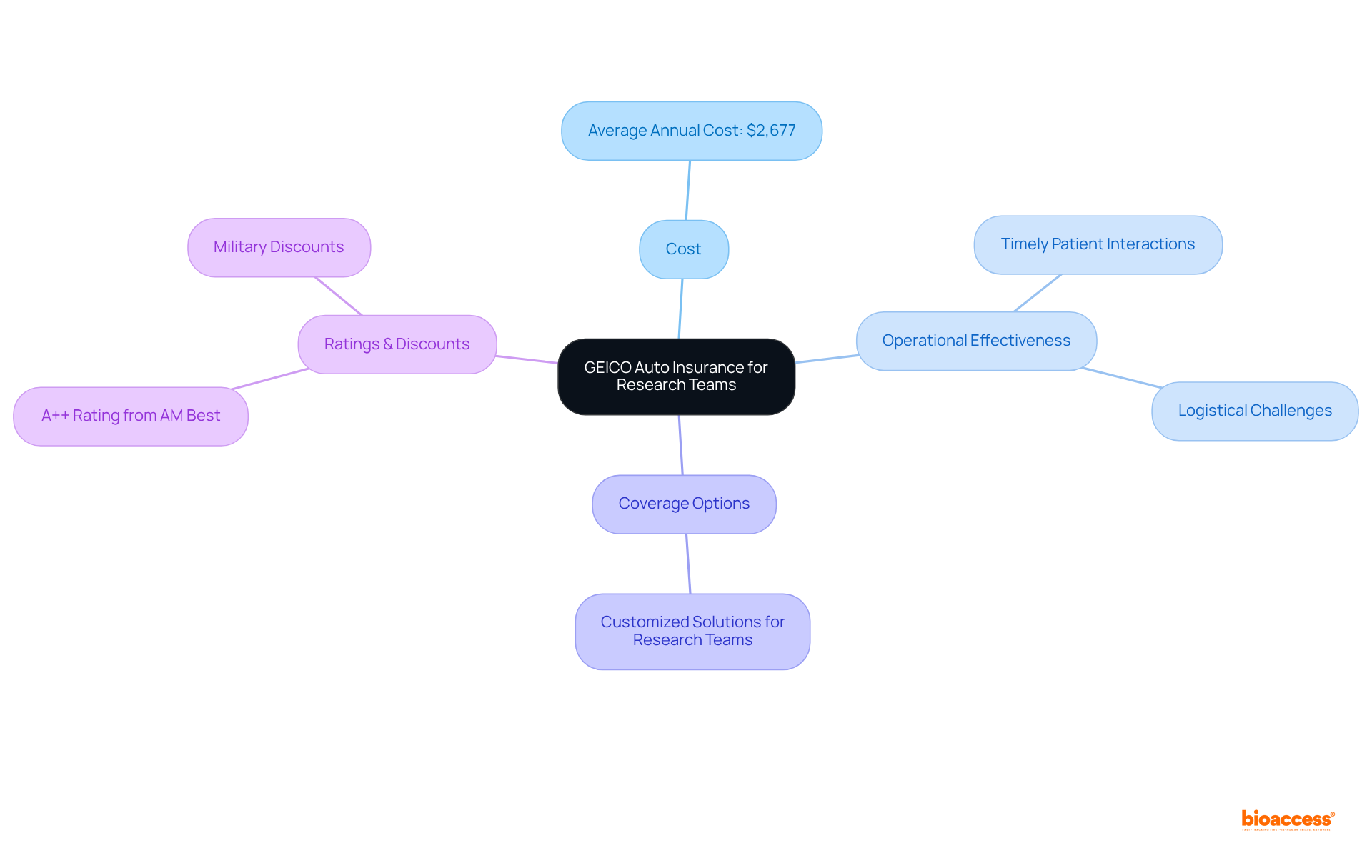
Dependable vehicle coverage is imperative for , particularly in 2025, as it directly influences . GEICO offers specifically designed for research teams, ensuring that team members can travel to various study locations without the burden of unexpected vehicle-related costs.
With the being $2,677, having reliable insurance is essential for effectively managing operational expenses. This coverage not only provides but also extensive options that address the specific logistical challenges faced during research trials.
Experts emphasize that is vital for maintaining and facilitating seamless operations. Furthermore, GEICO’s A++ rating from AM Best and its competitive military discounts further establish it as a valuable partner in the healthcare sector.
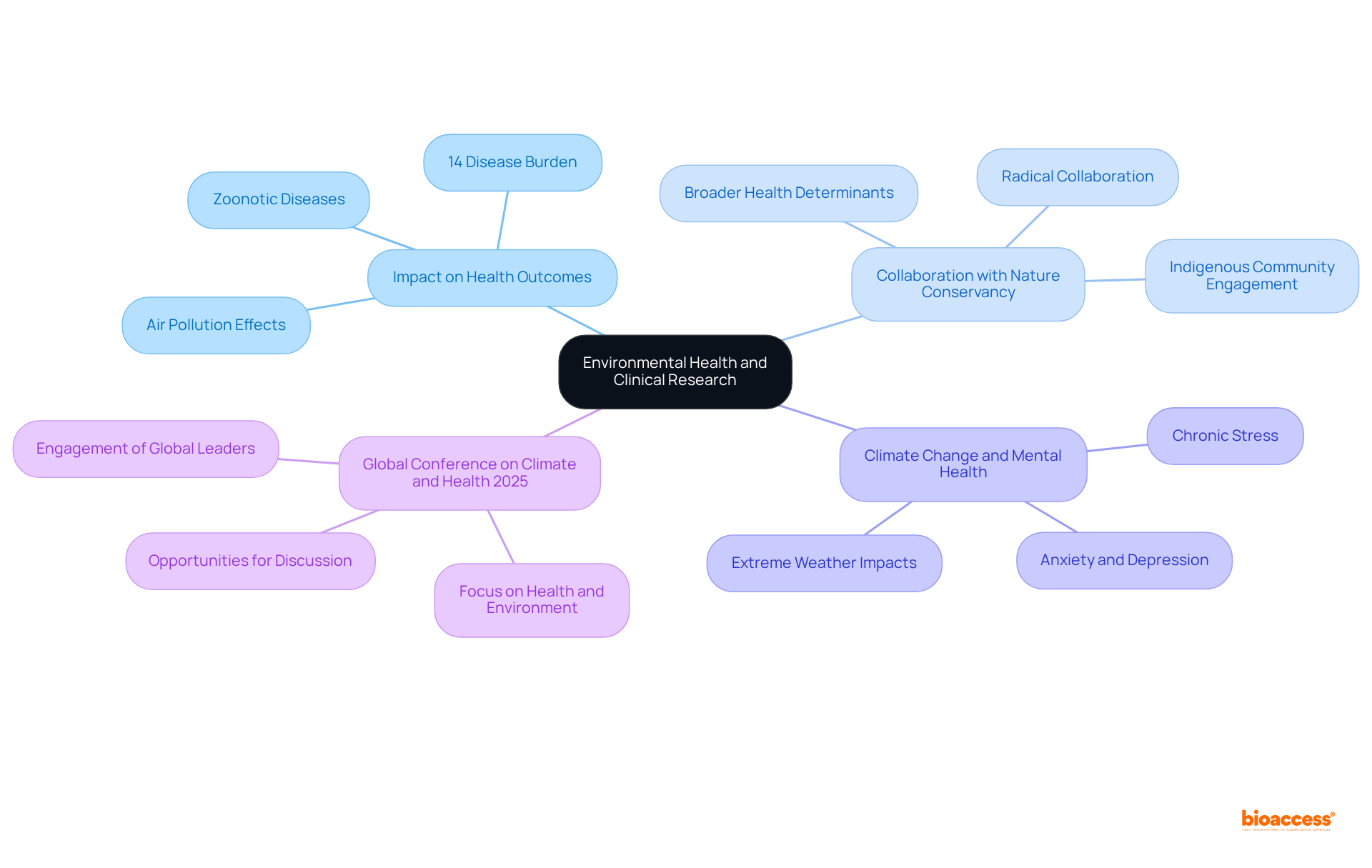
Nature Conservancy: Environmental Health Insights for Clinical Research
The Nature Conservancy provides crucial insights into environmental well-being that can significantly influence medical study methodologies. Understanding the —accounting for approximately 14% of the total burden of disease in the UK, as reported by the World Health Organization—enables researchers to develop more .
Collaborating with organizations like the Nature Conservancy allows research to address broader health determinants, thereby amplifying the overall significance of findings. Dr. Margaret Chan emphasizes that a , underscoring the necessity of .
Additionally, the , including anxiety and depression, underscore the urgency of this approach. Effective partnerships, exemplified in various case studies, demonstrate the tangible benefits of incorporating environmental factors into research.
With the forthcoming , there lies a .
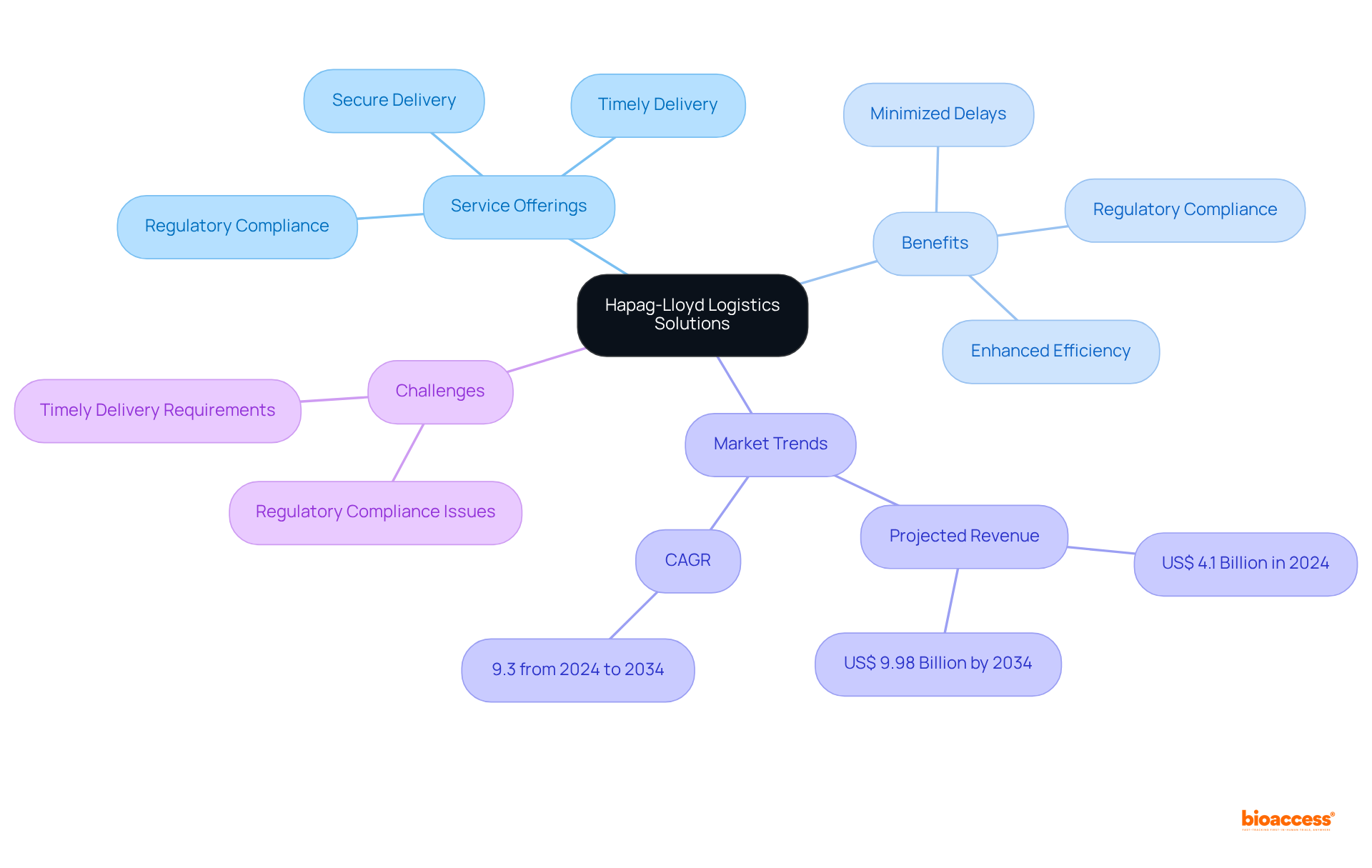
Hapag-Lloyd: Logistics Solutions for Clinical Trial Material Transport
Hapag-Lloyd excels in delivering . Their services are designed to ensure the of , including sensitive biological specimens and investigational products. By leveraging Hapag-Lloyd’s expertise, research teams can effectively minimize delays and comply with stringent regulatory requirements, thereby enhancing overall study efficiency.
As we look toward 2025, the logistics landscape is transforming, with an increasing focus on real-time tracking and automation to tackle transportation challenges. The global healthcare research logistics market is projected to generate a revenue of US$ 4.1 billion in 2024, with a compound annual growth rate (CAGR) of 9.3%, underscoring the growing demand for efficient logistics services.
The integration of advanced technologies is becoming increasingly essential for maintaining compliance and ensuring the integrity of throughout the supply chain. Hapag-Lloyd’s case studies illustrate successful implementations of these logistics solutions, showcasing their commitment to supporting research initiatives with precision and reliability.
Furthermore, addressing the challenges faced in , such as and the need for , is critical for the success of .
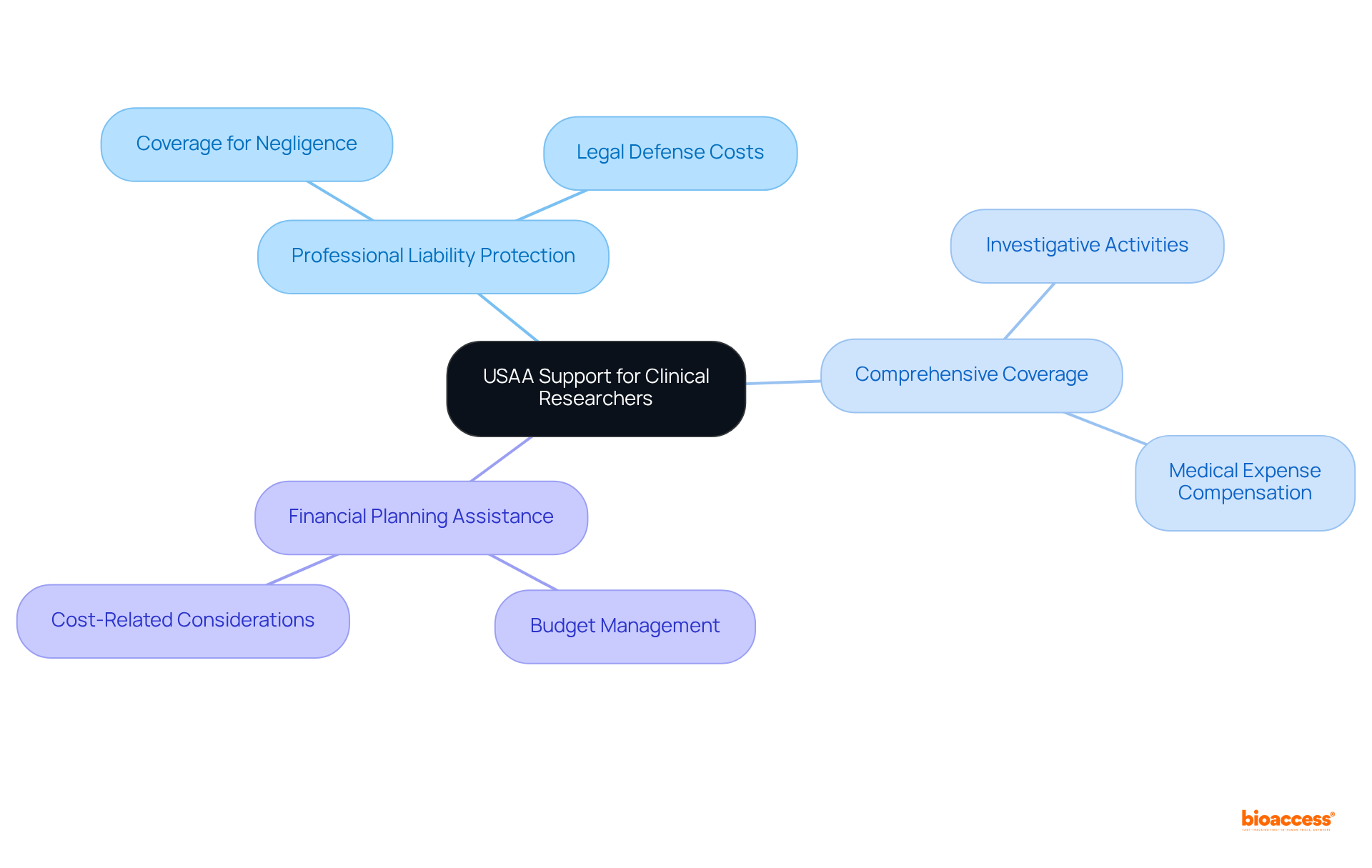
USAA: Financial and Insurance Support for Clinical Researchers
USAA delivers tailored specifically designed for medical researchers, addressing the critical need for efficient management of trial-related expenses. Their services encompass:
- Professional liability protection
- Comprehensive coverage for investigative activities
- Expert financial planning assistance
By collaborating with USAA, medical researchers can concentrate on advancing their studies while effectively managing . This strategic partnership not only enhances operational efficiency but also aligns with contemporary trends emphasizing the necessity for robust protection solutions within the research environment.
As bioaccess® states, “,” underlining the ethical imperatives of . Furthermore, with 55% of respondents indicating that at least one , the urgency of addressing these financial barriers becomes increasingly apparent. Bioaccess®’s focus on and their further underscores the vital role of financial solutions in facilitating successful trials.
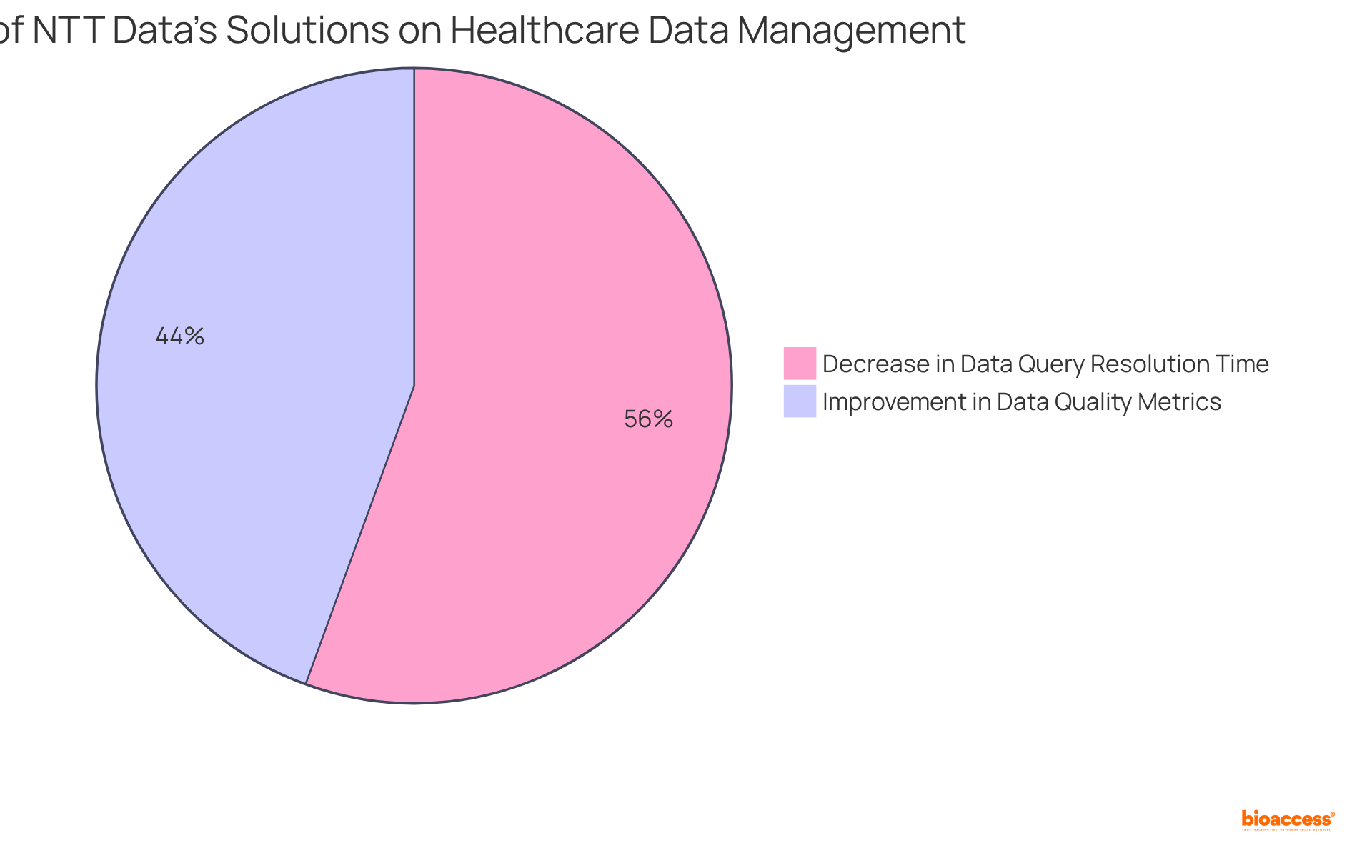
NTT Data: Technology Solutions for Efficient Clinical Data Management
NTT Data stands at the forefront of transforming healthcare through innovative technology solutions. Their platforms , analysis, and reporting, enabling researchers to efficiently handle large datasets while adhering to stringent . By utilizing NTT Data’s advanced solutions, researchers can significantly enhance , achieving a 28% improvement in , and expedite the overall .
Moreover, studies utilizing indicate a 35% decrease in data query resolution time, underscoring the effectiveness of these technologies. As the environment evolves, particularly with the anticipated incorporation of AI and machine learning technologies by 2025—which is projected to reach $4.9 billion by 2028—NTT Data’s contributions are essential for ensuring compliance and enhancing efficiency in medical studies.
As Sarah Lee observes, ‘ in the healthcare field includes the gathering, purification, and oversight of information from trials and studies—a procedure that has changed significantly with technological progress.
Air Canada: Travel Services for Clinical Research Teams in Mexico
Air Canada provides a comprehensive suite of travel services specifically designed for . Their offerings encompass flexible booking options, , and dedicated support for managing . By collaborating with Air Canada, research teams can , ensuring that all logistical needs are thoroughly addressed. This partnership allows researchers to focus on their primary objectives, thereby in their studies.
Given that the is projected to grow at a CAGR of 7.9% by 2032, the importance of effective is paramount. As Mariam Faizullabhoy noted, ‘ are essential for ensuring the .
Canada Post: Secure Shipping Solutions for Clinical Trial Materials
Canada Post offers specialized options tailored for research materials, ensuring compliance with stringent regulatory standards. Their services are meticulously designed to during transit, significantly mitigating the risk of damage or loss. By leveraging Canada Post’s expertise, can guarantee prompt and secure delivery of their materials, a critical factor in preserving the integrity of research studies.
Notably, the is valued at $3.9 billion in 2023 and is projected to reach $6.3 billion by 2028, underscoring the necessity of in this burgeoning sector. Furthermore, highlights that IoT devices facilitate continuous monitoring of temperature and environmental conditions during transportation, thereby enhancing compliance and safety.
The integration of contemporary trends in solutions, such as the collaboration between SkyCell and Marken for , illustrates practical applications that can further bolster the . To ensure , researchers should consider implementing and engaging specialized logistics providers.
Conclusion
The landscape of clinical research in Mexico is significantly enhanced by a diverse array of local representative services that improve the efficiency and effectiveness of studies. By leveraging local expertise, organizations can adeptly navigate the complexities of regulatory approvals, participant recruitment, and logistical challenges, ultimately accelerating the research process. This article has highlighted key players such as bioaccess®, Cigna, and Hapag-Lloyd, showcasing how their specialized services cater to the unique needs of clinical trials.
Throughout this discussion, critical insights have emerged. The rapid approval times offered by bioaccess® and the comprehensive health coverage from Cigna exemplify how local representatives can mitigate barriers to participation and streamline processes. Furthermore, the tailored insurance solutions from Progressive and the logistics expertise of Hapag-Lloyd underscore the importance of addressing both operational risks and material transport needs. Each of these elements plays a pivotal role in ensuring that clinical trials are conducted smoothly and effectively.
As the demand for clinical research services continues to grow, particularly in emerging markets like Mexico, the significance of local partnerships cannot be overstated. Researchers and organizations are strongly encouraged to explore these local representative services to enhance their study designs and operational strategies. By doing so, they not only improve their chances of success but also contribute to the advancement of medical knowledge and patient care on a broader scale. Embracing local expertise is not merely beneficial; it is essential for the future of clinical research.
Frequently Asked Questions
What services does bioaccess® provide for clinical research in Mexico?
bioaccess® offers research services for Medtech, Biopharma, and Radiopharma innovators, leveraging over 15 years of experience in Latin America.
How quickly can bioaccess® secure ethical approvals for studies?
bioaccess® can secure ethical approvals in an impressive 4-6 weeks.
What is the typical duration for research study approvals in Mexico?
The typical approval duration for research studies in Mexico ranges from 14 to 16 weeks.
What is the overall approval and setup process duration for clinical studies in Mexico?
The overall approval and setup process generally spans 3-4 months.
What advantages does bioaccess® offer in terms of patient recruitment and study execution?
bioaccess®’s local expertise enhances patient recruitment and study execution, ensuring high retention rates and fostering trust in the physician-patient relationship.
What is the projected market size for clinical research in Mexico by 2030?
The market for clinical research in Mexico is projected to reach USD 510.4 million by 2030, growing at a CAGR of 6.9% from 2024 to 2030.
What challenges exist within the regulatory landscape for clinical research in Mexico?
The regulatory landscape has complexities that may lead to potential delays in the research process.
What health coverage does Cigna provide for clinical trial participants?
Cigna offers customized health coverage plans that encompass standard care and any additional medical costs incurred during the study.
How does health coverage impact participant recruitment in clinical trials?
Health coverage significantly alleviates financial pressures, enhancing participant recruitment and retention, as many eligible patients decline participation due to coverage issues.
What percentage of eligible patients decline participation in trials due to potential denial of coverage?
Studies reveal that 8 to 20% of eligible patients decline participation due to potential denial of coverage.
What does the Affordable Care Act (ACA) state regarding health coverage for trial participants?
According to the ACA, health providers are prohibited from denying coverage for involvement in an authorized study for cancer or another life-threatening illness.
What insurance solutions does Progressive offer for clinical research operations?
Progressive offers tailored coverage options including liability coverage, property insurance, and specialized policies for study sponsors and contract organizations.
How does Progressive address the evolving risks associated with clinical studies?
Progressive provides customized coverage that protects against potential liabilities, especially in the context of decentralized studies and emerging technologies.
What role does informed consent play in clinical research according to Progressive?
Informed consent is emphasized as a critical component to ensure participants understand their involvement in studies, which is part of Progressive’s risk management strategies.
How does Progressive facilitate smoother project execution in clinical trials?
By effectively mitigating risks and addressing essential elements like medical expenses and rehabilitation costs, Progressive bolsters the viability of experimental operations.
List of Sources
- bioaccess: Accelerate Clinical Research with Local Expertise in Mexico
- Clinical trial regulation in Mexico – PMLiVE (https://pmlive.com/intelligence/clinical_trial_regulation_in_mexico_477081)
- linkedin.com (https://linkedin.com/posts/lrubin_clinicalresearch-publicpolicy-innovation-activity-7328978221146796032-Zy_u)
- Clinical Trials in Mexico: Addressing the Challenges (https://languageconnections.com/clinical-trials-in-mexico-addressing-the-challenges)
- pharmaboardroom.com (https://pharmaboardroom.com/articles/mexico-towards-a-clinical-trials-powerhouse)
- Mexico Clinical Trials Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trials-market/mexico)
- Cigna: Comprehensive Health Insurance for Clinical Trial Participants
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- numberanalytics.com (https://numberanalytics.com/blog/health-insurance-clinical-trial-participation)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC3408810)
- cigna.co.uk (https://cigna.co.uk/quote?elqTrackId=6c6742ae1d704645bcc5d9b3c652468e)
- fredhutch.org (https://fredhutch.org/en/research/clinical-trials/guide-clinical-trials/health-insurance.html)
- Progressive: Tailored Insurance Solutions for Clinical Research Operations
- alanboswell.com (https://alanboswell.com/business-insurance/science-technology-insurance/clinical-trial-insurance)
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- foundershield.com (https://foundershield.com/blog/clinical-trials-insurance-guide)
- tridentinsurance.com.au (https://tridentinsurance.com.au/the-role-of-insurance-in-safeguarding-clinical-trial-participants)
- laplayapartners.com (https://laplayapartners.com/science-technology/life-sciences-insurance/clinical-trials-liability-insurance)
- GEICO: Reliable Auto Insurance for Clinical Research Teams
- constructioncoverage.com (https://constructioncoverage.com/commercial-auto-insurance)
- policygenius.com (https://policygenius.com/auto-insurance/car-insurance-statistics)
- cnbc.com (https://cnbc.com/select/best-cheap-car-insurance-veterans-military)
- bankrate.com (https://bankrate.com/insurance/car/medical-professionals)
- Nature Conservancy: Environmental Health Insights for Clinical Research
- azquotes.com (https://azquotes.com/quotes/topics/environmental-health.html)
- nature.org (https://nature.org/en-us/about-us/who-we-are/our-people/jennifer-morris/time-to-act-climate-change)
- 7 Quotes on Climate Change and Health | unfoundation.org (https://unfoundation.org/blog/post/7-quotes-on-climate-change-and-health)
- gov.uk (https://gov.uk/government/publications/state-of-the-environment/state-of-the-environment-health-people-and-the-environment)
- who.int (https://who.int/health-topics/environmental-health)
- Hapag-Lloyd: Logistics Solutions for Clinical Trial Material Transport
- acumenresearchandconsulting.com (https://acumenresearchandconsulting.com/press-releases/clinical-trial-supply-and-logistics-market)
- factmr.com (https://factmr.com/report/clinical-trial-logistics-market)
- logisticsviewpoints.com (https://logisticsviewpoints.com/author/lveditor)
- grandviewresearch.com (https://grandviewresearch.com/industry-analysis/clinical-trial-packaging-market-report)
- USAA: Financial and Insurance Support for Clinical Researchers
- foundershield.com (https://foundershield.com/blog/clinical-trials-insurance-guide)
- howdengroup.com (https://howdengroup.com/ie-en/sector/life-sciences-and-pharmaceuticals/clinical-trials)
- Influence of Cost-Related Considerations on Clinical Trial Participation: Results from the 2020 Health Information National Trends Survey (HINTS) – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC9713084)
- newsroom.usaa360.com (https://newsroom.usaa360.com/news)
- statnews.com (https://statnews.com/2025/07/17/medical-research-trial-participants-payment-ethics-irbs)
- NTT Data: Technology Solutions for Efficient Clinical Data Management
- acldigital.com (https://acldigital.com/blogs/future-clinical-data-research-management-trends-and-predictions-2025)
- Clinical Data Management System Market Share, Report 2035 (https://marketresearchfuture.com/reports/clinical-data-management-system-market-32286)
- numberanalytics.com (https://numberanalytics.com/blog/clinical-data-management-pharma-7-stats)
- careerfoundry.com (https://careerfoundry.com/en/blog/data-analytics/inspirational-data-quotes)
- Air Canada: Travel Services for Clinical Research Teams in Mexico
- clinregs.niaid.nih.gov (https://clinregs.niaid.nih.gov/country/australia/mexico)
- clinregs.niaid.nih.gov (https://clinregs.niaid.nih.gov/country/mexico/thailand)
- gminsights.com (https://gminsights.com/industry-analysis/clinical-trial-supply-and-logistics-market)
- newswire.ca (https://newswire.ca/news-releases/air-canada-to-launch-into-summer-2025-with-new-non-stop-flights-to-europe-and-the-u-s–881877774.html)
- mordorintelligence.com (https://mordorintelligence.com/industry-reports/clinical-trials-market)
- Canada Post: Secure Shipping Solutions for Clinical Trial Materials
- towardshealthcare.com (https://towardshealthcare.com/insights/clinical-trial-supply-and-logistics-market-sizing)
- precedenceresearch.com (https://precedenceresearch.com/clinical-trial-supply-and-logistics-market)
- clinicalleader.com (https://clinicalleader.com/doc/clinical-trial-supply-market-outlook-developments-technology-trends-0001)

Leave a Reply