Introduction
In an increasingly regulated landscape, receiving a warning letter from the USFDA represents a pivotal moment for any business. Such letters not only underscore regulatory violations but also present a critical opportunity for organizations to showcase their commitment to compliance and corrective action. The challenge, however, lies in navigating the complexities of an effective response.
How can companies transform a potential crisis into a stepping stone for improvement? This guide delineates five essential steps to address a warning letter, ensuring that businesses not only meet regulatory expectations but also fortify their operational integrity.
Understand the Importance of FDA Warning Letters
When the agency identifies during inspections, it issues from usfda. These documents underscore specific concerns and provide the recipient with an . Ignoring a can result in , including:
- Enforcement actions
- Product recalls
- Damage to the
Understanding the implications of these communications is the first step in effectively managing the situation and ensuring compliance with .
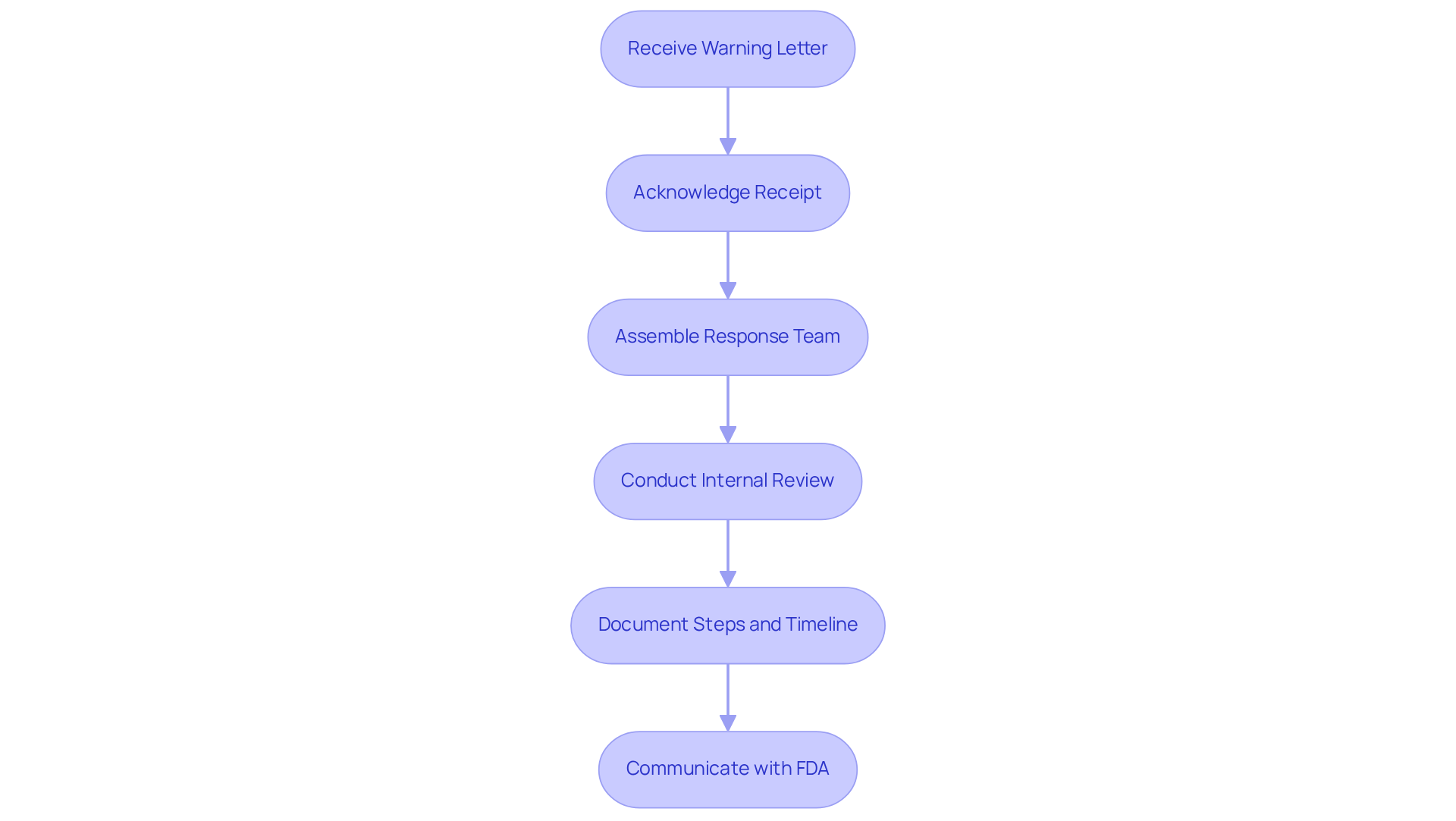
Initiate Immediate Response Actions
Upon receiving a warning letter from the US, the immediate priority is to formally acknowledge its receipt. Following this acknowledgment, an must be conducted.
- Assemble a , , and to thoroughly assess the findings.
- It is crucial to record all steps undertaken and create a mentioned in the letter.
- is critical; typically, you have 15 days to inform them of your intent to respond and outline your initial actions.
This proactive approach not only but also sets the stage for a .
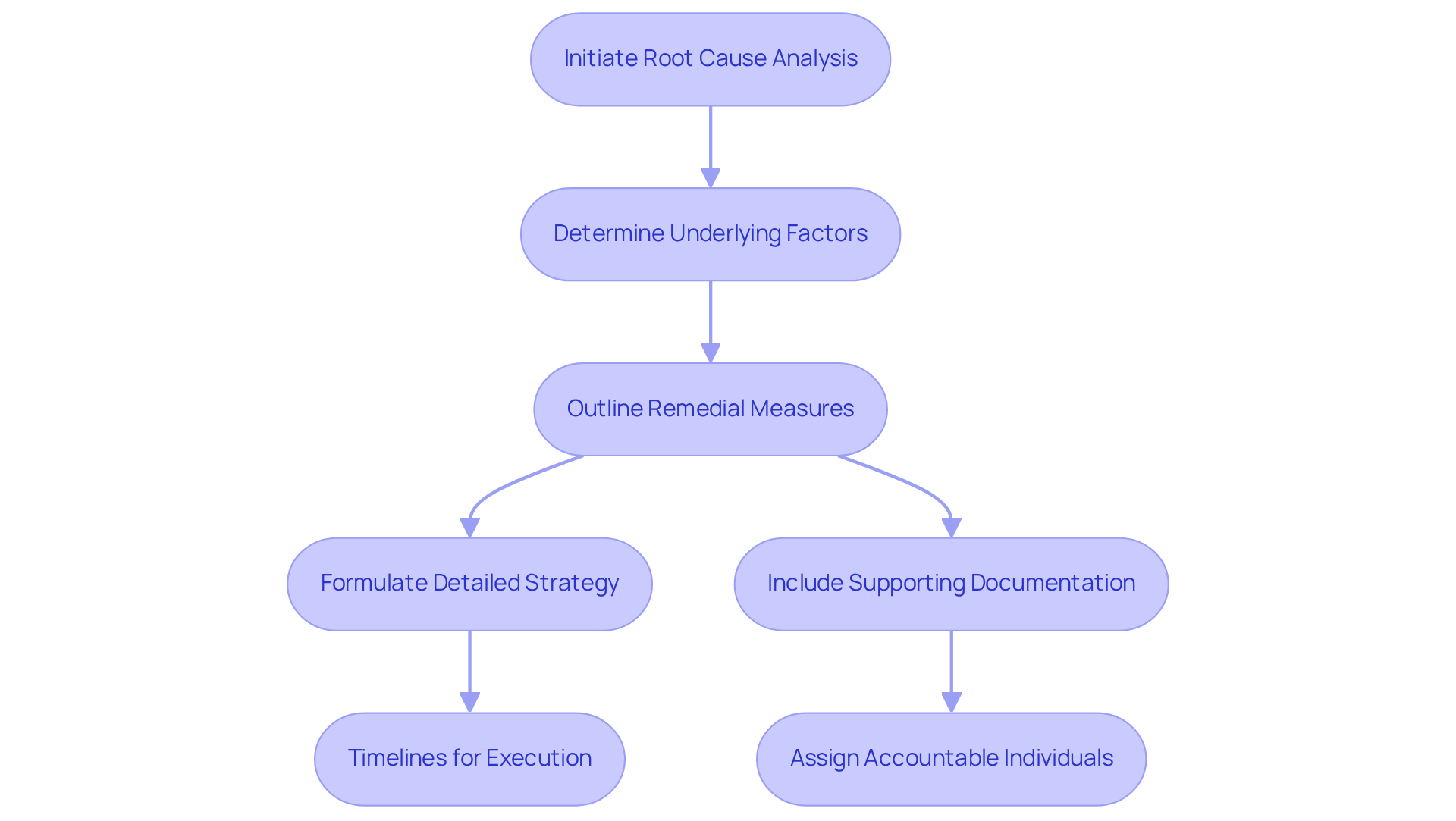
Develop a Comprehensive Response Strategy
To develop a comprehensive , initiate a of the issues identified in the warning letter. It is crucial to determine the underlying factors that led to the violations and outline specific for each issue.
Formulate a that encompasses:
- Assigning accountable individuals
Your response must be clear, concise, and directly address each point raised by the . Additionally, include any supporting documentation that illustrates your .
Implement Corrective Actions and Monitor Compliance
Once have been identified, it is imperative to implement them effectively. Assign responsibilities to team members while ensuring they possess the necessary resources to execute their tasks. Establish a to evaluate the effectiveness of the , making adjustments as required. It is essential to consistently examine adherence to and internal policies to avoid receiving a and prevent future violations.
The should be utilized in to uncover root causes and effectively prevent issues. Document all actions taken and maintain records using for future reference, as this will be crucial for any follow-up communications related to the .
Moreover, leveraging , such as feasibility studies and site selection, can significantly enhance adherence efforts. This proactive approach not only safeguards against potential issues but also reinforces the organization’s commitment to , which is vital for Directors of Clinical Research in ensuring compliance and operational excellence.
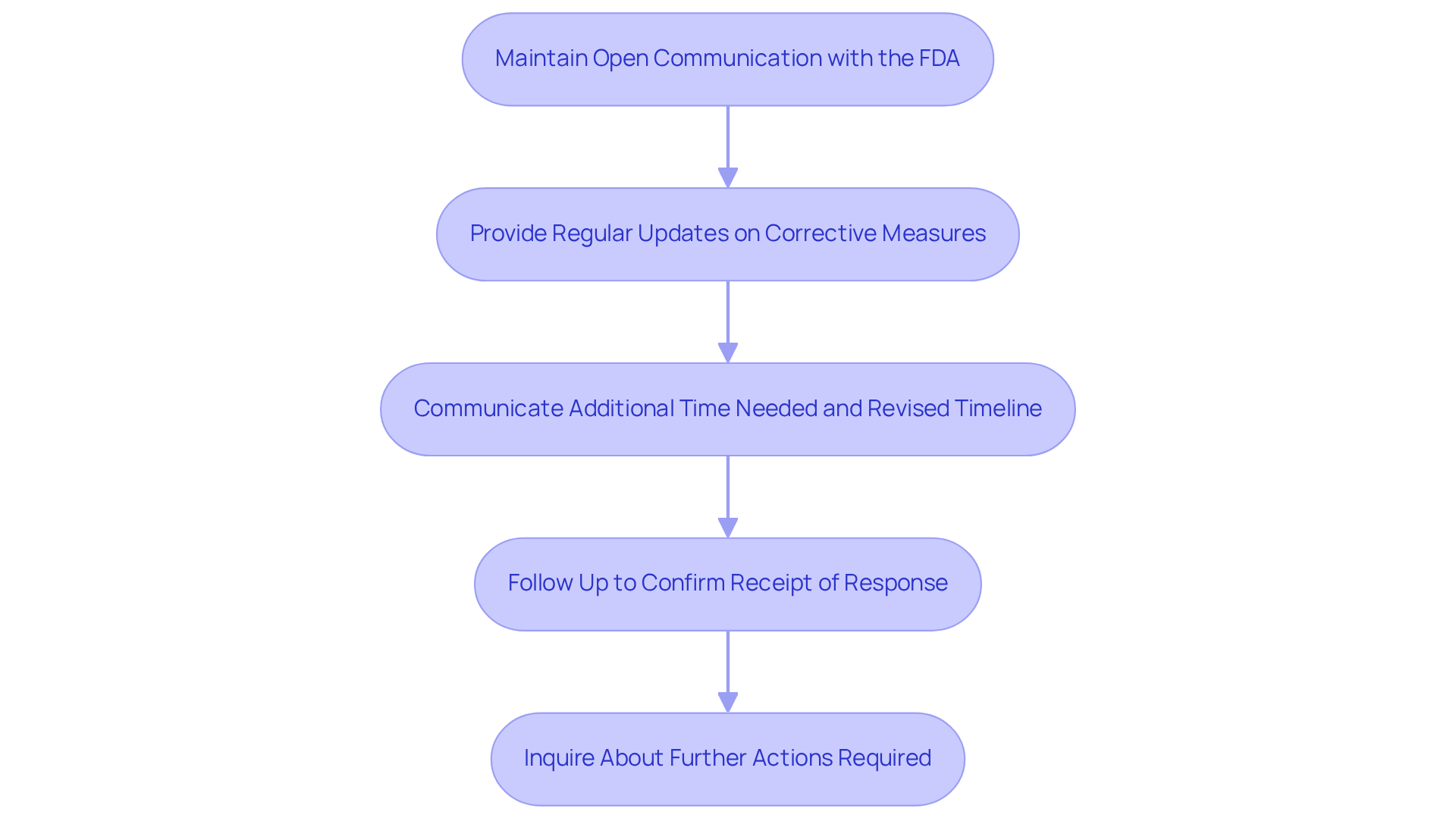
Maintain Open Communication with the FDA
Maintaining with the is crucial throughout the response process, especially when dealing with from us. Regular updates on the status of your and any challenges faced should be provided. If additional time is required to implement changes, it is essential to communicate this to the along with a revised timeline. Following the submission of your response, a follow-up is necessary to confirm receipt and to inquire about any further actions required. Building a not only facilitates smoother interactions in the future but also demonstrates your , which can help prevent a .
Conclusion
Receiving a warning letter from the USFDA marks a pivotal moment for any organization, underscoring regulatory violations while simultaneously presenting an opportunity to affirm a commitment to compliance. Addressing such a letter effectively transcends merely mitigating immediate concerns; it serves as a chance to bolster operational integrity and avert future infractions.
This article delineates five essential steps to adeptly navigate this intricate process:
- Grasping the significance of the warning letter
- Initiating prompt response actions
- Formulating a comprehensive response strategy
- Executing corrective measures
- Sustaining transparent communication with the FDA
Each of these steps is crucial not only for resolving current issues but also for cultivating a culture of compliance and continuous improvement within the organization.
Ultimately, the importance of addressing a warning letter extends far beyond mere regulatory compliance; it embodies a commitment to quality and safety standards that can protect a company’s reputation and operational success. By adopting proactive measures and engaging constructively with the FDA, organizations can transform potential crises into invaluable learning experiences, ensuring they are better prepared to meet regulatory expectations moving forward.
Frequently Asked Questions
What is the significance of an FDA warning letter?
An FDA warning letter signifies that the agency has identified significant violations of regulations during inspections. It highlights specific concerns and provides the recipient with an opportunity to address these issues.
What are the potential consequences of ignoring an FDA warning letter?
Ignoring an FDA warning letter can lead to severe consequences, including enforcement actions, product recalls, and damage to the company’s reputation.
What should be the immediate response upon receiving an FDA warning letter?
The immediate response should be to formally acknowledge the receipt of the letter and conduct an internal review of the cited issues.
Who should be involved in the internal review process after receiving a warning letter?
A dedicated team should be assembled, including members from quality assurance, regulatory affairs, and relevant department heads, to thoroughly assess the findings.
Why is it important to document the response actions taken after receiving a warning letter?
Documenting the response actions is crucial to record all steps undertaken and to create a clear timeline for addressing the issues mentioned in the letter.
How quickly must a company communicate with the FDA after receiving a warning letter?
Typically, a company has 15 days to inform the FDA of its intent to respond and outline its initial actions.
What is the benefit of a proactive approach when responding to an FDA warning letter?
A proactive approach demonstrates the company’s commitment to compliance and sets the stage for a constructive dialogue with the FDA.
List of Sources
- Initiate Immediate Response Actions
- Case Study Report | PDF | Pharmaceutical Drug | Pharmaceutical Industry (https://scribd.com/document/139268588/Case-Study-Report)
- Implement Corrective Actions and Monitor Compliance
- qualityze.com (https://qualityze.com/blogs/fda-form-483-and-warning-letters)

Leave a Reply