Introduction
Understanding the various types of FDA meetings is essential for anyone involved in clinical research. These gatherings serve as critical touchpoints between sponsors and the regulatory agency. By mastering the nuances of each meeting type – from high-priority discussions to informal INTERACT sessions – stakeholders can significantly enhance their engagement strategies and align their objectives with FDA expectations. However, navigating the complexities of these meetings poses a challenge: how can sponsors ensure that their requests are not only well-structured but also resonate with FDA priorities?
Understand FDA Meeting Types
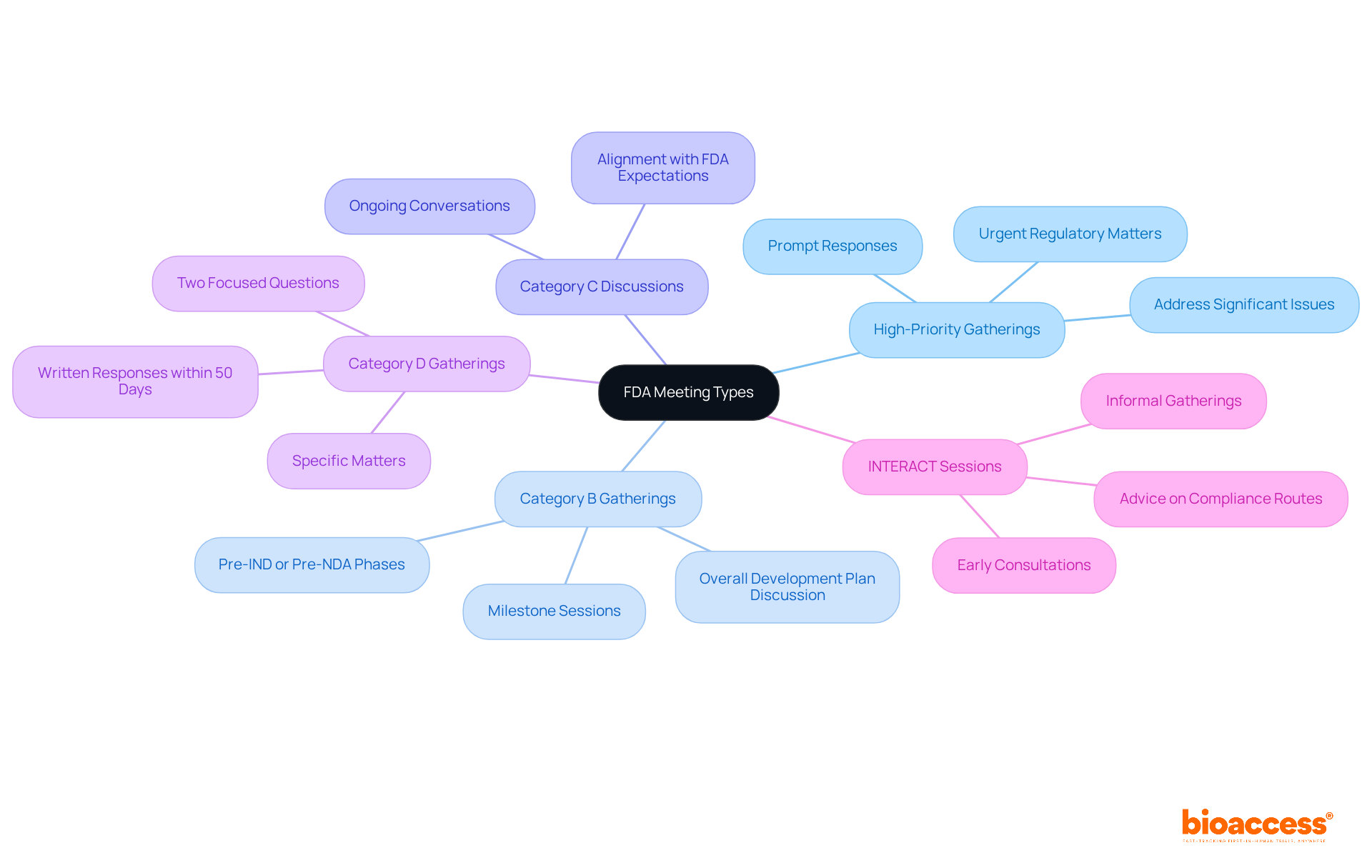
The FDA conducts various formal gatherings to enhance communication between sponsors and the agency, which is crucial for effective engagement in . Understanding these meeting types is essential:
- : These sessions address significant issues that could stall a product’s development. They are often requested when urgent regulatory matters arise, ensuring prompt responses to critical challenges.
- : These milestone sessions occur at crucial stages in the development process, such as or pre-New Drug Application (NDA) phases. They provide an opportunity to discuss the overall development plan and receive .
- : These address additional subjects not covered in Categories A or B. Generally utilized for ongoing conversations about a product’s development, they enable sponsors to maintain alignment with .
- Category D Gatherings: Focused discussions on specific matters that don’t require the immediacy of a Category A session but still need prompt input. These gatherings are particularly beneficial for addressing specific compliance inquiries, using a structured approach that limits questions to two targeted issues. provide written responses within 50 days, making them a quicker option compared to Type C discussions.
- : Informal gatherings that allow sponsors to discuss their development programs with FDA personnel and receive advice on compliance routes. These sessions are designed for early consultations, helping to identify potential issues before significant investments are made.
In 2023, the FDA reported that Type A and B discussions achieved over 90% success rates, underscoring their importance in the regulatory process. By familiarizing yourself with these , you can better prepare for your interactions with the FDA and ensure that your objectives align with the appropriate format.
Request an FDA Meeting
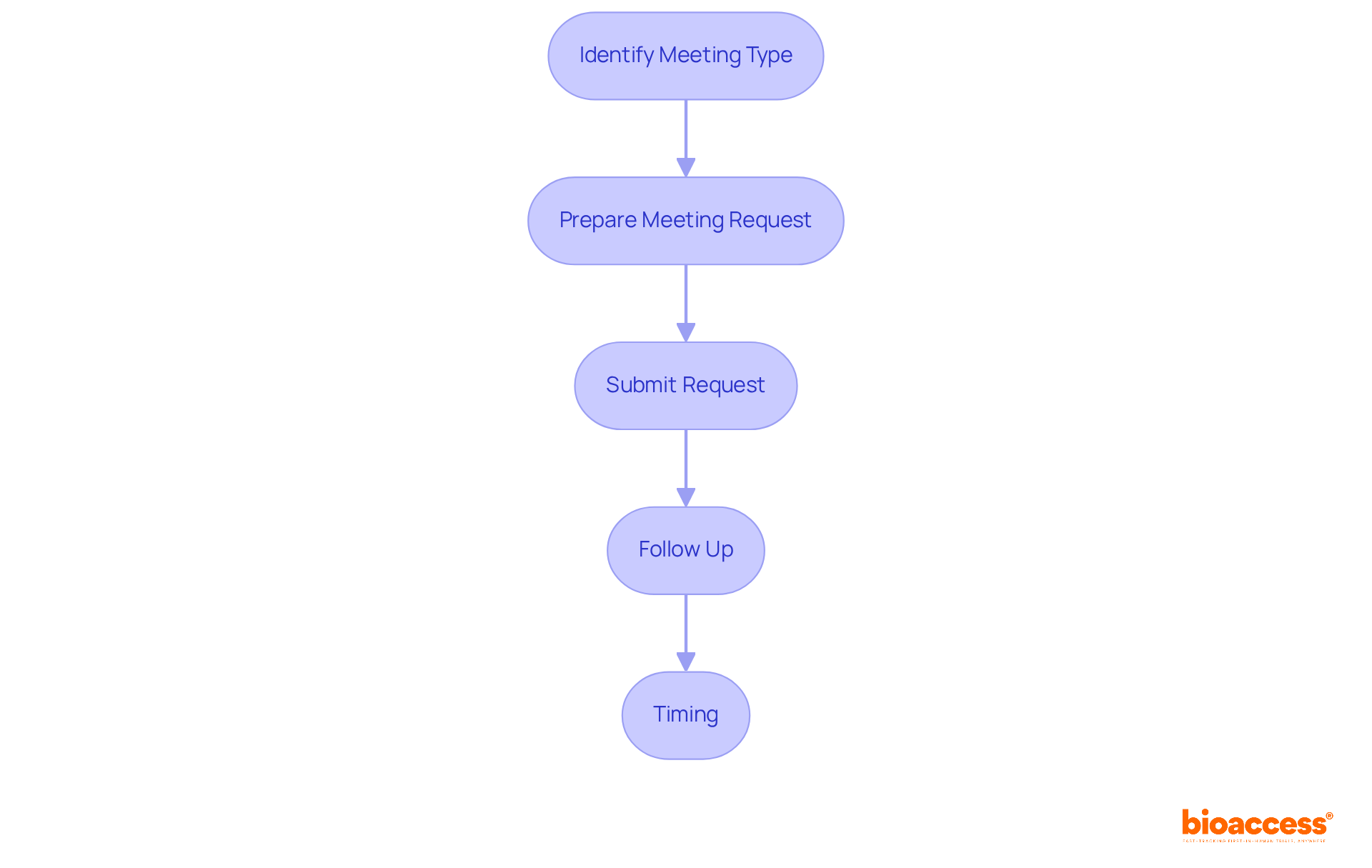
To effectively request a meeting with the FDA, it’s crucial to follow these essential steps:
-
Identify the : Determine the appropriate based on your objectives, such as Type A, B, C, D, or INTERACT. Each serves distinct purposes and has specific requirements that you must understand.
-
Prepare a Meeting Request: Draft a formal request that includes:
- A .
- Specific topics for discussion, ensuring they align with .
- Relevant background information, including a product overview and clinical synopsis, to provide necessary context.
- Limit your inquiries during gatherings to a maximum of 10 to ensure clarity and focus.
-
Submit the Request: Send your appointment request to the relevant FDA division, typically through the FDA’s Electronic Submission Gateway (ESG). Ensure that your submission adheres to the for compliance.
-
Follow Up: After submission, promptly follow up with the FDA to confirm receipt and inquire about the scheduling process. Early notification to your Regulatory Project Manager (RPM) can expedite responses for urgent requests.
-
Timing: well in advance, ideally 2-3 months before your desired appointment date. This allows sufficient time for scheduling and increases the likelihood of securing your preferred date.
By following these steps, you can enhance the completeness of your request and improve the chances of receiving a . Notably, the FDA has granted over 663 out of 858 Type D inquiries from 2023 to June 2024, indicating a favorable response rate when requests are well-structured and clear. early in the process can also enhance your approach, aid in formulating questions, and help evade typical obstacles, ensuring a fruitful interaction with the FDA.
Prepare for FDA Meetings
Effective preparation for an is crucial and involves several key steps that can significantly enhance the likelihood of a productive outcome:
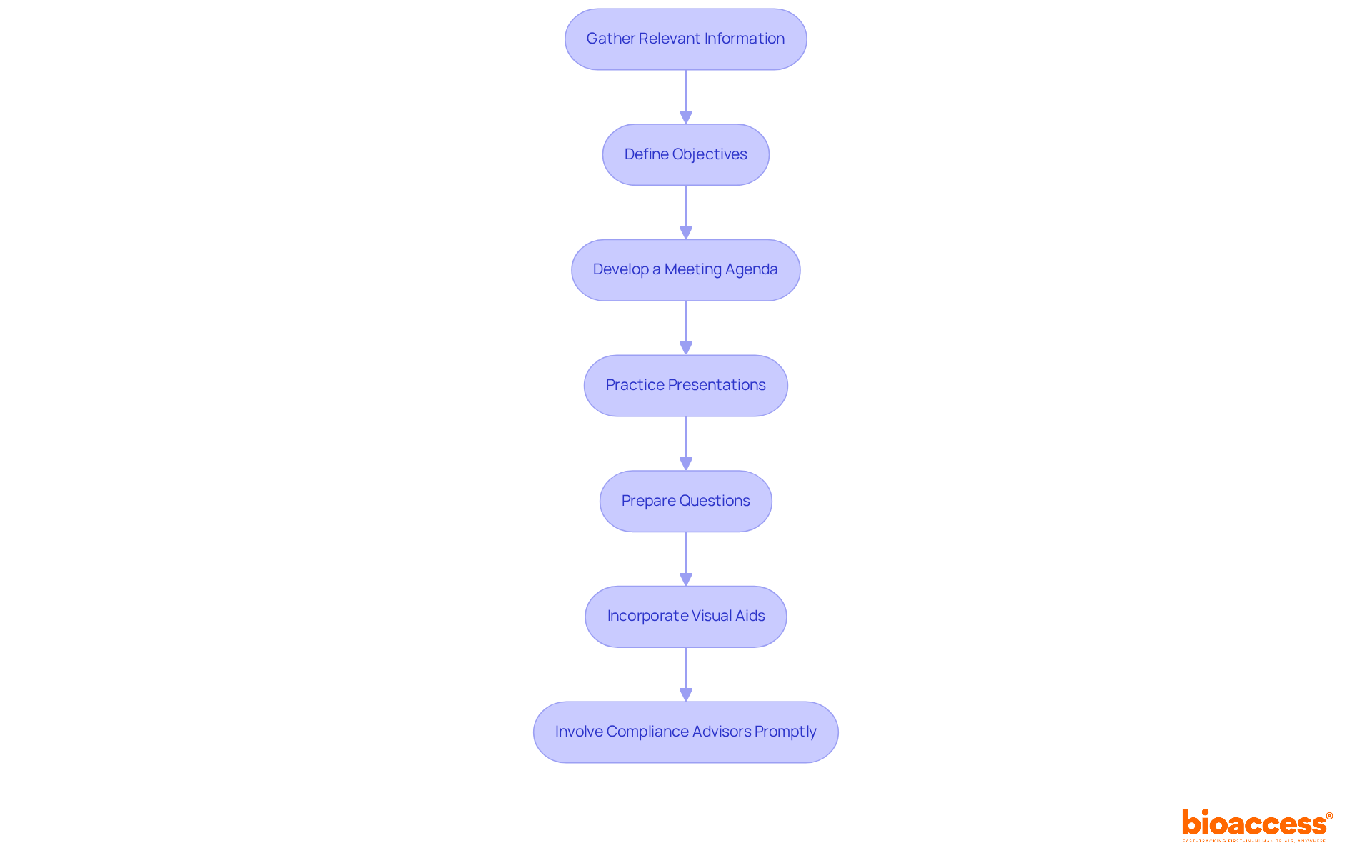
- Gather Relevant Information: Compile all necessary documents, including past session minutes, data summaries, and any pertinent compliance guidance. This foundational step ensures that all participants are .
- Define Objectives: Clearly outline the goals of the discussion. Research indicates that common objectives include seeking feedback on pivotal trial designs, clarifying compliance requirements, and optimizing development plans. Establishing specific objectives helps focus discussions and aligns expectations with the type.
- Develop a Meeting Agenda: Create a structured agenda that prioritizes discussion topics and allocates time for each item. Sharing this agenda with all participants in advance fosters preparedness and encourages meaningful contributions.
- Practice Presentations: Conduct internal rehearsals to ensure that all team members are familiar with their roles and the content they will present. This practice can help identify potential challenges and refine the delivery of key messages.
- Prepare Questions: Formulate specific questions to guide the discussion and ensure that all critical points are addressed during the gathering. Grouping questions by discipline-clinical, non-clinical, and CMC-can enhance the productivity of discussions by bringing appropriate FDA experts to the table.
- Incorporate Visual Aids: Utilize visual aids such as flowcharts and tables in your briefing documents to simplify complex data. This can expedite the FDA’s review process and enhance understanding.
- Involve Compliance Advisors Promptly: Engage compliance advisors early in the process to for the gathering. Their insights can help clarify objectives and identify pressing questions.
By following these , particularly leveraging , including and , you can significantly improve the effectiveness of your interactions in the context of the type. This approach not only facilitates but also advances your clinical development goals, especially for startups navigating the complexities of FDA submissions in Latin America.
Conduct Effective FDA Meetings
To conduct an effective , consider the following strategies:
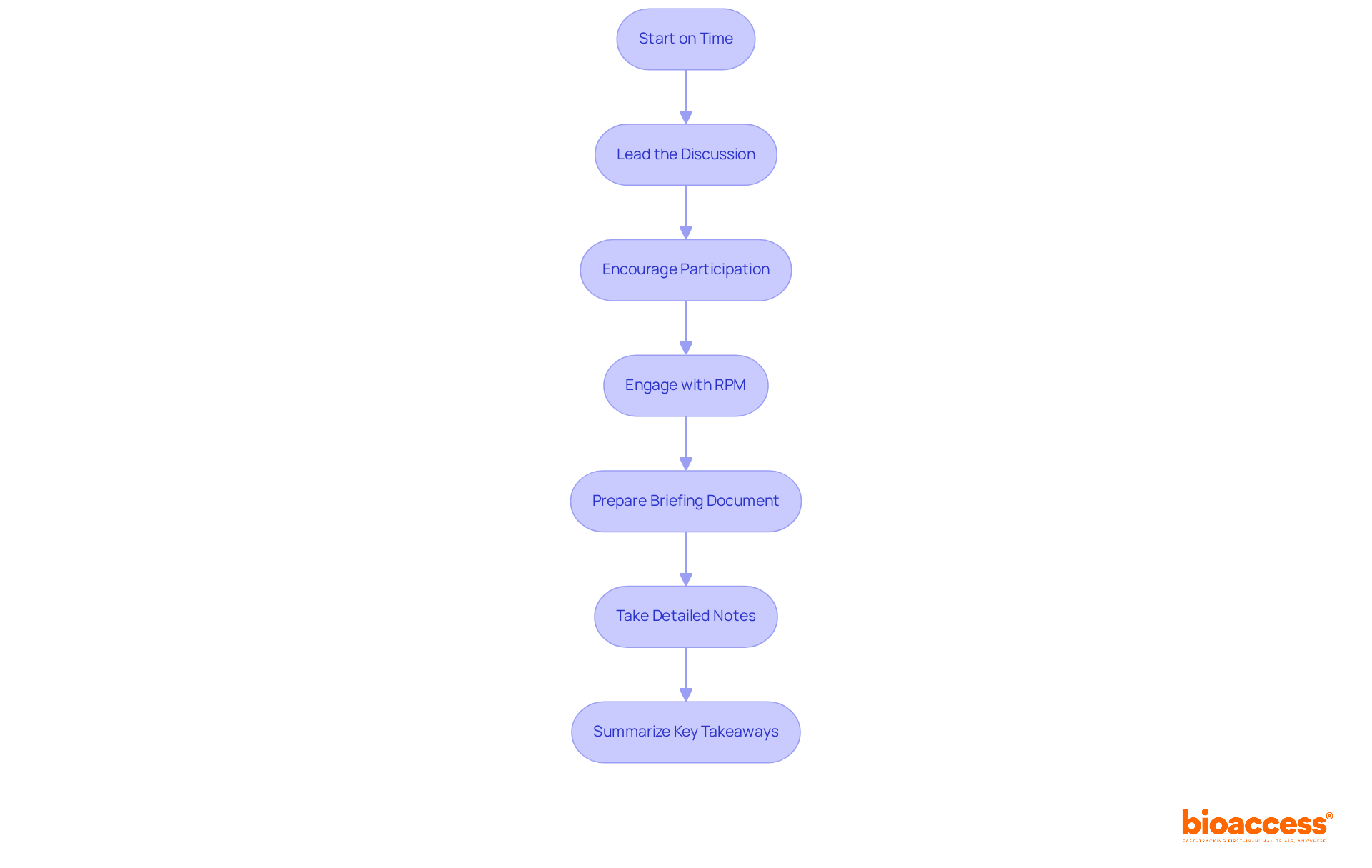
- Start on Time: Begin the session promptly to respect the FDA’s schedule and demonstrate professionalism. Timeliness is crucial, as it sets a positive tone and reflects your commitment to the process.
- Lead the Discussion: Take charge of the gathering by guiding the conversation according to the agenda. Ensure that all topics are covered and that participants remain focused. Clear leadership helps maintain momentum and direction throughout the gathering.
- : Foster an environment where all team members feel comfortable contributing. Encourage questions and discussions to clarify points, as diverse input can lead to more comprehensive insights and solutions. Remember, the FDA advises sponsors to avoid asking more than 10 questions during discussions to maintain clarity and focus.
- (RPM): Leverage the expertise of the FDA’s RPM for support in organizing your discussion. can accelerate responses and enhance the overall gathering experience.
- Prepare a : Ensure that your briefing document summarizes and includes specific follow-up questions. A well-prepared document is essential for a productive FDA gathering and helps clarify the objectives of your discussion.
- : Document key points, decisions made, and action items during the discussion. This practice ensures accountability and provides a reference for follow-up, enhancing the .
- : At the conclusion of the gathering, summarize the main points discussed and any agreed-upon next steps. This reinforces understanding and sets clear expectations for follow-up actions, ensuring that all participants leave with a shared understanding of the outcomes.
By implementing these strategies, you can enhance the effectiveness of your type and improve the chances of achieving your . Engaging in well-structured discussions and adhering to best practices will not only facilitate smoother interactions but also increase the likelihood of favorable outcomes.
Conclusion
Understanding the various types of FDA meetings is crucial for anyone involved in clinical research and regulatory affairs. These meetings, from high-priority gatherings to informal INTERACT sessions, offer essential opportunities for sponsors to engage with the FDA, clarify expectations, and navigate the complexities of drug development. Mastering the nuances of each meeting type can significantly enhance the effectiveness of these interactions and align objectives with regulatory requirements.
The article outlined key categories of FDA meetings, highlighting the critical importance of Type A and Type B discussions, which have demonstrated impressive success rates. It detailed the necessary steps to request a meeting, emphasizing clarity, structure, and timely communication. Furthermore, effective preparation and conduct during these meetings are vital components for achieving favorable outcomes, supported by practical strategies to ensure productive discussions.
Ultimately, successful navigation of FDA meetings can greatly influence the trajectory of a product’s development. By leveraging the insights and best practices discussed, stakeholders can foster more productive engagements with the FDA, ensuring that their objectives are met while advancing public health goals. Engaging with compliance advisors early and adhering to structured meeting protocols will streamline the process and enhance the potential for successful regulatory interactions.
Frequently Asked Questions
What is the purpose of FDA meetings?
FDA meetings enhance communication between sponsors and the agency, which is crucial for effective engagement in clinical research.
What are High-Priority Gatherings?
High-Priority Gatherings address significant issues that could stall a product’s development and are requested when urgent regulatory matters arise to ensure prompt responses to critical challenges.
What are Category B Gatherings?
Category B Gatherings are milestone sessions that occur at crucial stages in the development process, such as pre-Investigational New Drug (IND) or pre-New Drug Application (NDA) phases, allowing sponsors to discuss the overall development plan and receive essential feedback from the FDA.
What topics are covered in Category C Discussions?
Category C Discussions address additional subjects not covered in Categories A or B, enabling ongoing conversations about a product’s development and helping sponsors maintain alignment with FDA expectations.
What is the focus of Category D Gatherings?
Category D Gatherings focus on specific matters that don’t require the immediacy of a Category A session but still need prompt input, particularly for addressing specific compliance inquiries with a structured approach.
What are INTERACT Sessions?
INTERACT Sessions are informal gatherings that allow sponsors to discuss their development programs with FDA personnel and receive advice on compliance routes, designed for early consultations to identify potential issues before significant investments are made.
What success rates were reported for Type A and B discussions in 2023?
In 2023, the FDA reported that Type A and B discussions achieved over 90% success rates, highlighting their importance in the regulatory process.
How can understanding FDA meeting types benefit sponsors?
Familiarizing yourself with these FDA meeting types helps sponsors better prepare for interactions with the FDA and ensure that their objectives align with the appropriate meeting format.
List of Sources
- Understand FDA Meeting Types
- Formal FDA meeting types (https://bgtcplaybook.document360.io/docs/formal-fda-meeting-types)
- FDA Meeting Preparation Consulting: A Founder’s Guide – J&J Compliance Consulting Inc. (https://jjccgroup.org/type-b-meeting-fda-guide)
- MMS | Data CRO | Global Clinical Research & Health Data Analytics Company (https://mmsholdings.com/perspectives/pdufa-vii-fda-meeting-updates)
- Leveraging FDA Type A Meetings to Overcome Regulatory Hurdles (https://synergbiopharma.com/blog/fda-type-a-meeting)
- Navigating the Type D FDA Meeting: What Sponsors Need to Know (https://facetlifesciences.com/2025/03/10/fda-type-d-meetings)
- Request an FDA Meeting
- Pre-IND Meetings with the FDA: Everything You Need to Know (https://thefdagroup.com/blog/pre-ind-meeting)
- Leveraging FDA Type A Meetings to Overcome Regulatory Hurdles (https://synergbiopharma.com/blog/fda-type-a-meeting)
- MMS | Data CRO | Global Clinical Research & Health Data Analytics Company (https://mmsholdings.com/perspectives/pdufa-vii-fda-meeting-updates)
- Prepare for FDA Meetings
- Assessment of FDA Premarket Approval Process and Suggestions for Improvement (https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2824671)
- Pre-IND Meetings with the FDA: Everything You Need to Know (https://thefdagroup.com/blog/pre-ind-meeting)
- Navigating FDA Meetings: Drug Development & Approval Path (https://allucent.com/resources/blog/inroads-approval-navigating-formal-fda-meetings-optimize-your-drug-development-program)
- The Value of FDA Pre-Submission Meetings & Enhancements under PDUFA VI – Food and Drug Law Institute (FDLI) (https://fdli.org/2017/12/value-fda-pre-submission-meetings-enhancements-pdufa-vi)
- Conduct Effective FDA Meetings
- Leveraging FDA Type A Meetings to Overcome Regulatory Hurdles (https://synergbiopharma.com/blog/fda-type-a-meeting)
- MMS | Data CRO | Global Clinical Research & Health Data Analytics Company (https://mmsholdings.com/perspectives/pdufa-vii-fda-meeting-updates)
- Seven Steps for Success at FDA Meetings (https://3dcommunications.us/latest-thinking/posts/seven-steps-for-success-at-fda-meetings)

Leave a Reply