Introduction
While Chile presents a fast-growing research environment for Phase 1 clinical trials, the complexities involved can pose significant challenges for MedTech and Biopharma companies. With advantages such as:
- A diverse patient population
- Streamlined regulatory processes
- Impressive retention rates
Chile stands out as a strategic hub for early-stage studies. Despite the promising landscape, sponsors often find themselves grappling with regulatory complexities and the challenge of engaging patients effectively.
How can sponsors effectively overcome these obstacles while maximizing the benefits of conducting trials in this promising market? Addressing these challenges is crucial for sponsors to fully capitalize on the advantages that Chile’s clinical trial landscape offers.
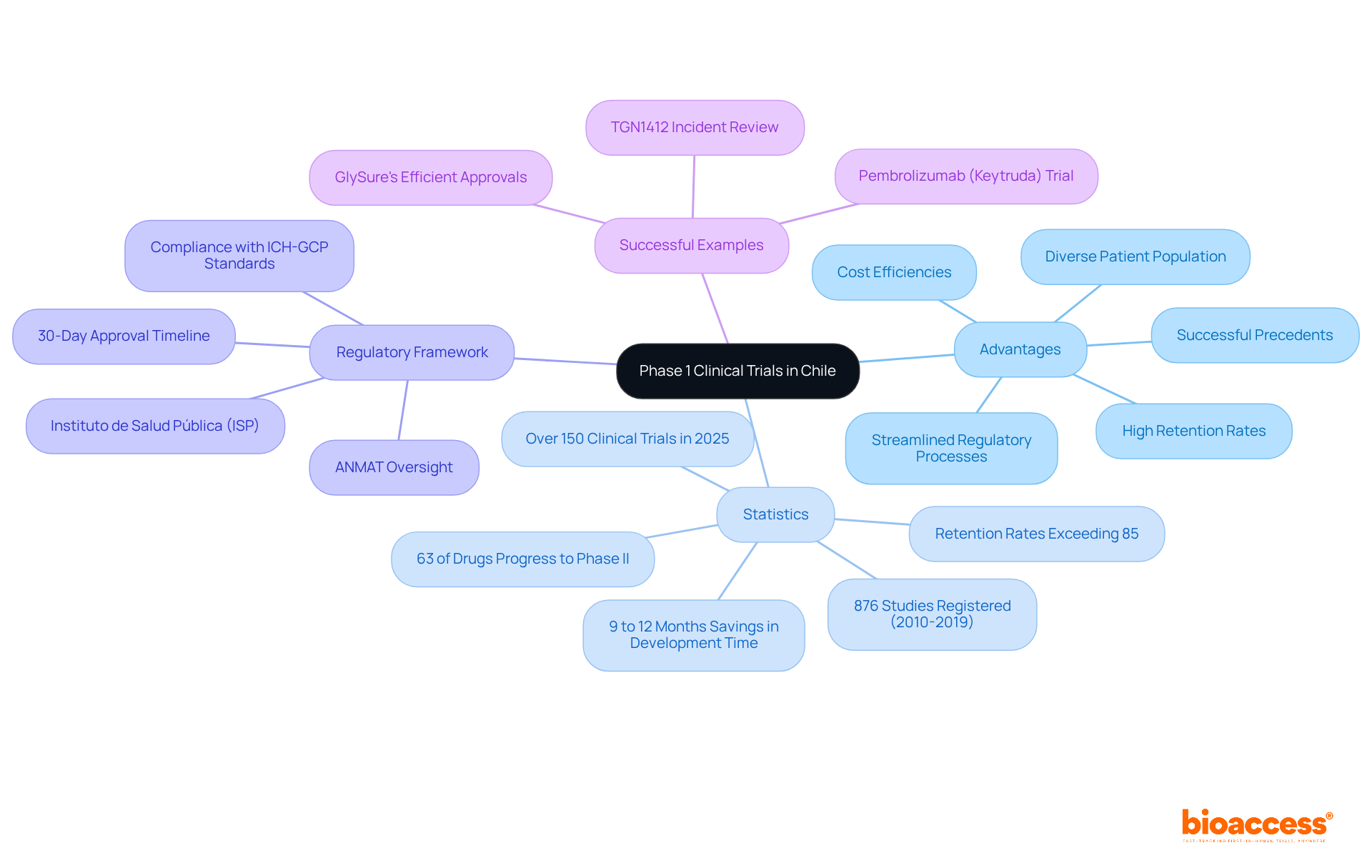
Understand Phase 1 Clinical Trials in Chile
Phase 1 clinical evaluations are crucial for determining the safety and efficacy of new medications and medical devices, yet many face challenges in navigating the complexities of clinical research. These studies typically involve a small group of 20 to 80 healthy participants and primarily evaluate safety, tolerability, and pharmacokinetics. The goal is to determine the maximum tolerated dose (MTD) and identify potential side effects. Conducting a phase 1 clinical trial in Chile presents several advantages. These include a diverse patient base, streamlined approval processes, and significant cost reductions. The expertise of bioaccess® further amplifies these benefits.
In 2025, the nation conducted over 150 research studies, reflecting a robust research environment supported by a well-established healthcare system. The regulatory framework, overseen by the Instituto de Salud Pública (ISP), ANMAT, and other agencies, ensures compliance with ICH-GCP standards, facilitating FDA-bridgeable data acceptance for future submissions. The evaluation period for clinical study approvals in the region typically spans around 30 days, facilitating faster timelines compared to many other areas.
In fact, Chile’s retention rates exceed 85%, which is impressive and enhances the reliability of trial outcomes. The streamlined regulatory procedures have enabled companies like GlySure to realize savings of 9 to 12 months in development time due to quicker approvals. This efficiency is vital for startups seeking to secure funding and progress their research programs.
Successful instances of Phase 1 studies in the region include the pembrolizumab (Keytruda) investigation, which highlighted the significance of thorough safety monitoring and adaptive dose escalation, ultimately resulting in market approval. Such occurrences highlight the potential for this nation to act as a strategic center for early-stage studies, merging speed, cost-effectiveness, and a nurturing compliance environment, especially through the capabilities of bioaccess®.
In summary, the advantages of conducting Phase 1 trials in Chile include:
- A diverse and accessible patient population
- Streamlined regulatory processes with quick approval timelines facilitated by bioaccess®
- High retention rates that enhance data reliability
- Significant cost efficiencies that benefit startups
- Successful precedents that demonstrate the effectiveness of the local clinical trial environment.
Navigate Chilean Regulatory Requirements
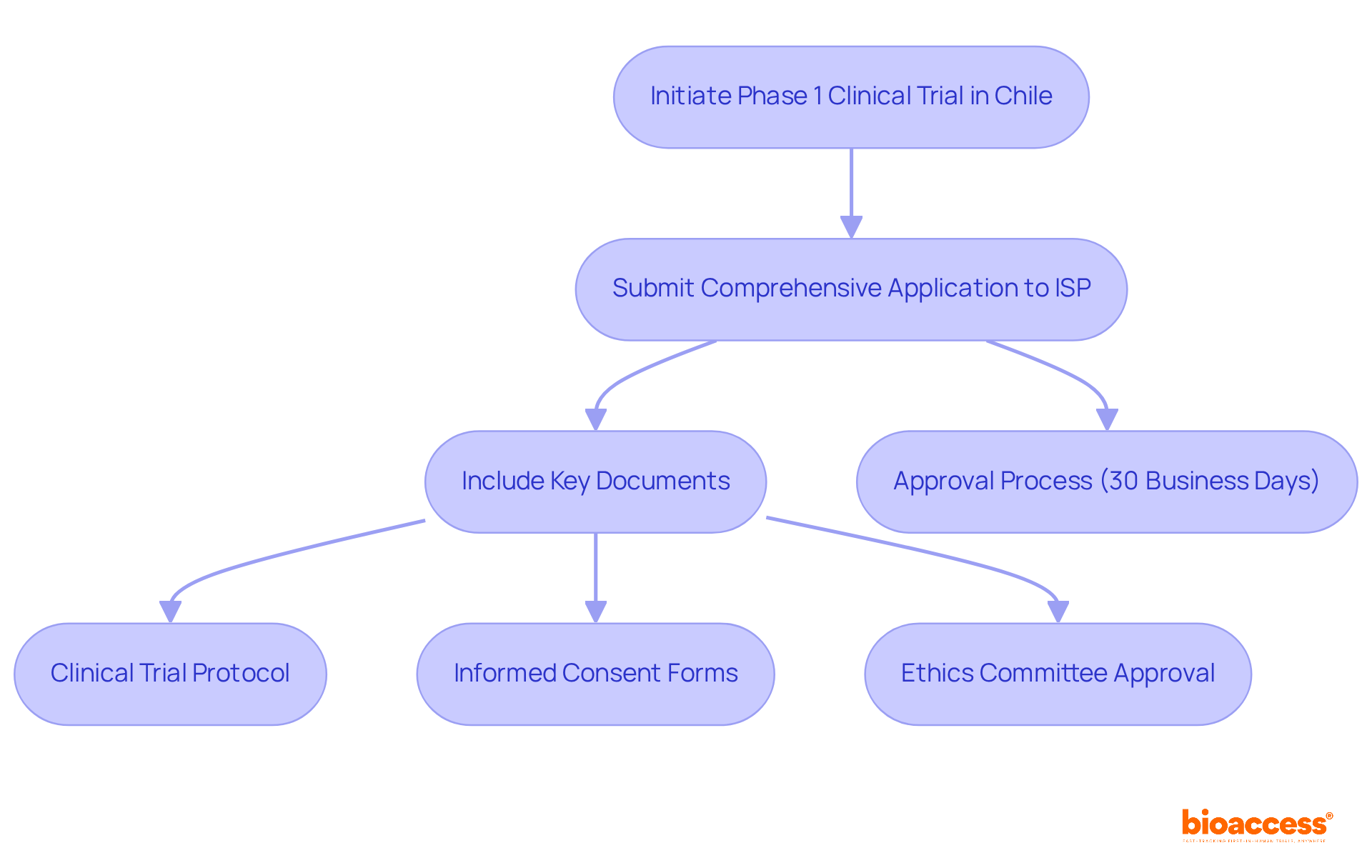
Navigating the complexities of initiating a phase 1 clinical trial in Chile can be daunting for sponsors. To commence this process, they must present a comprehensive application to the Instituto de Salud Pública (ISP). This application should include the clinical study protocol, informed consent forms, and investigator brochures. The approval process typically takes about 30 business days, during which the ISP reviews the submission for compliance with local regulations. Key documents required for submission include:
- Clinical Trial Protocol: This document outlines the study design, objectives, and methodology, serving as the foundation for the trial.
- Informed Consent Forms: These documents ensure that participant rights and safety are prioritized, providing essential information about the study.
- Ethics Committee Approval: Securing consent from an accredited ethics committee is essential before commencing the study, ensuring compliance with ethical standards.
Sponsors must also ensure ongoing compliance with ICH-GCP guidelines during the trial. This is crucial for maintaining integrity and trust in the research process. Clear communication with the ISP is vital to address any queries or concerns during the review process, facilitating a smoother approval pathway. Have you considered how compliance landscapes in neighboring countries like Brazil and Colombia could impact your trial? Proactive engagement with oversight bodies can significantly streamline operations and enhance the likelihood of timely approvals.
Understanding the compliance landscapes in neighboring countries is essential. For example, Brazil’s ANVISA and Colombia’s INVIMA have their own specific requirements and timelines that can influence trial planning in the region. Significantly, the enactment of Law 20.850 has resulted in a 20% reduction in the number of medical studies registered in the region, emphasizing the necessity of effectively navigating the compliance framework. As Julio G. Martinez-Clark, CEO of bioaccess®, mentions, “In the dynamic landscape of research, navigating the regulatory framework in this region is crucial for success.” This underscores why sponsors need to be well-prepared and informed as they embark on their research journey in this growing market. Without thorough preparation, sponsors risk delays and complications that could jeopardize their research objectives in this competitive market.
Execute Phase 1 Trials: Key Steps and Strategies
Executing a phase 1 clinical trial in Chile involves unique challenges that necessitate strategic planning and execution. Here are several key steps to consider:
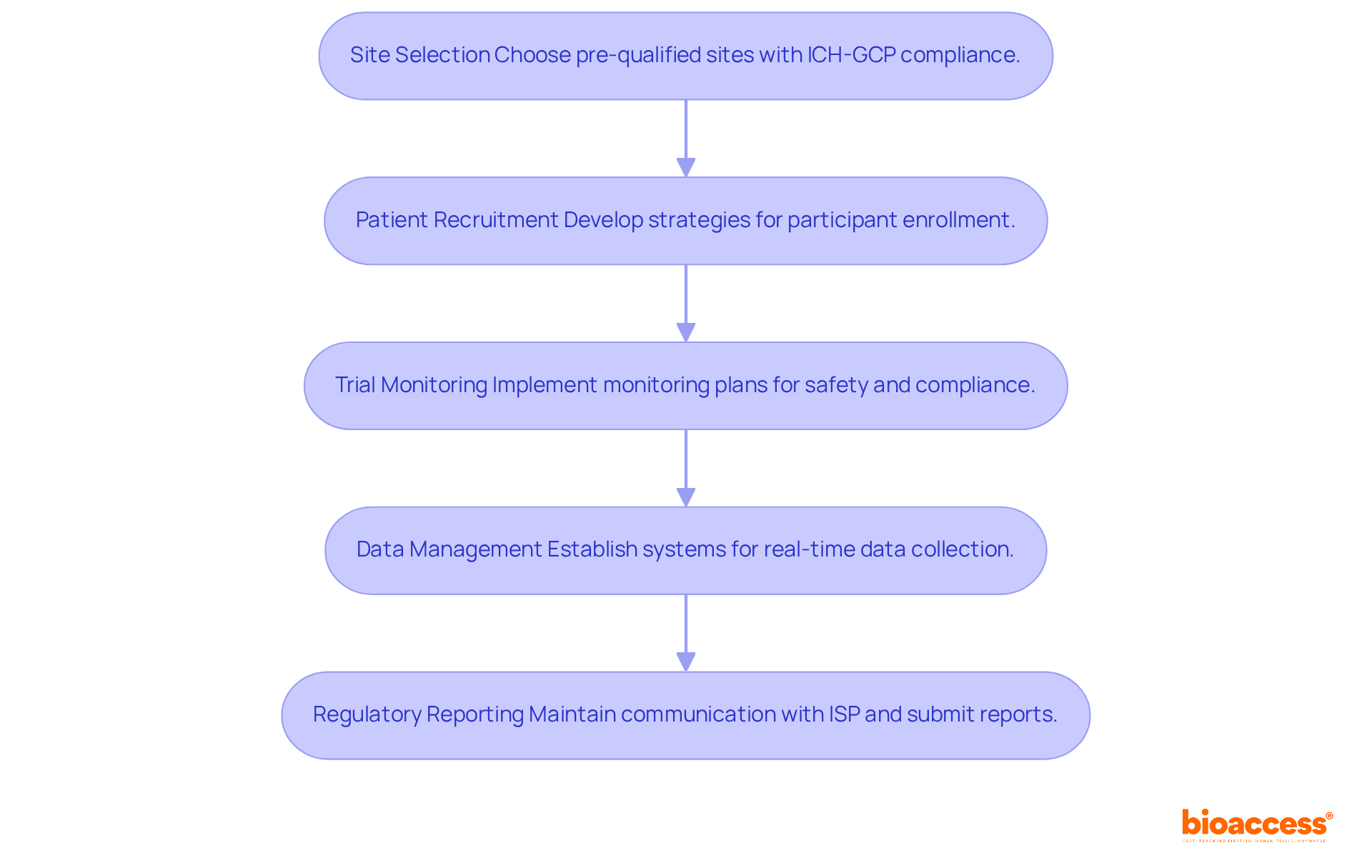
- Site Selection: Choose from a network of pre-qualified clinical research locations that comply with ICH-GCP standards and have demonstrated experience in conducting Phase 1 studies. Ensure that the site has the necessary infrastructure and qualified personnel to support the study effectively.
- Patient Recruitment: Develop a robust recruitment strategy that includes outreach to local healthcare providers, leveraging patient registries, and utilizing targeted digital marketing campaigns. Engaging with community organizations can significantly raise awareness about the study and its benefits. However, with only 60 oncologists in Chile, recruiting participants remains a challenge. Innovative recruitment strategies are essential to ensure adequate participant enrollment. With bioaccess®, sponsors can achieve 50% faster enrollment through pre-qualified networks, enhancing recruitment efficiency.
- Trial Monitoring: Implement a comprehensive monitoring plan to ensure adherence to the protocol and regulatory requirements. Regular evaluations of participant safety and data integrity are essential throughout the study, particularly considering the rigorous safety assessments necessary in Phase 1 research.
- Data Management: Establish a data management system that facilitates real-time data collection and analysis. This system should ensure that any adverse events are swiftly reported and addressed, preserving the integrity of the study.
- Regulatory Reporting: Maintain ongoing communication with the Instituto de Salud Pública (ISP) and submit necessary reports or updates as required by regulatory guidelines. This includes timely safety reports and protocol modifications, which can cut bureaucratic delays in the approval process by over 30%. Moreover, the ISP’s initiatives to optimize these processes can greatly improve the overall timeline for executing the phase 1 clinical trial in Chile. Leveraging bioaccess®’s pre-negotiated site contracts can lead to $25K savings per patient, allowing sponsors to invest these savings into R&D or future funding milestones.
By concentrating on these strategic actions, sponsors can utilize the local advantages of the region, including expedited ethics approvals and adherence to local standards, to improve patient recruitment success rates in phase 1 clinical trial in Chile. Ultimately, by leveraging local advantages and innovative strategies, sponsors can significantly enhance the success of their phase 1 clinical trial in Chile, paving the way for future advancements in clinical research.
Overcome Challenges in Phase 1 Trials
Phase 1 trials in Chile face unique challenges that demand strategic solutions for successful outcomes:
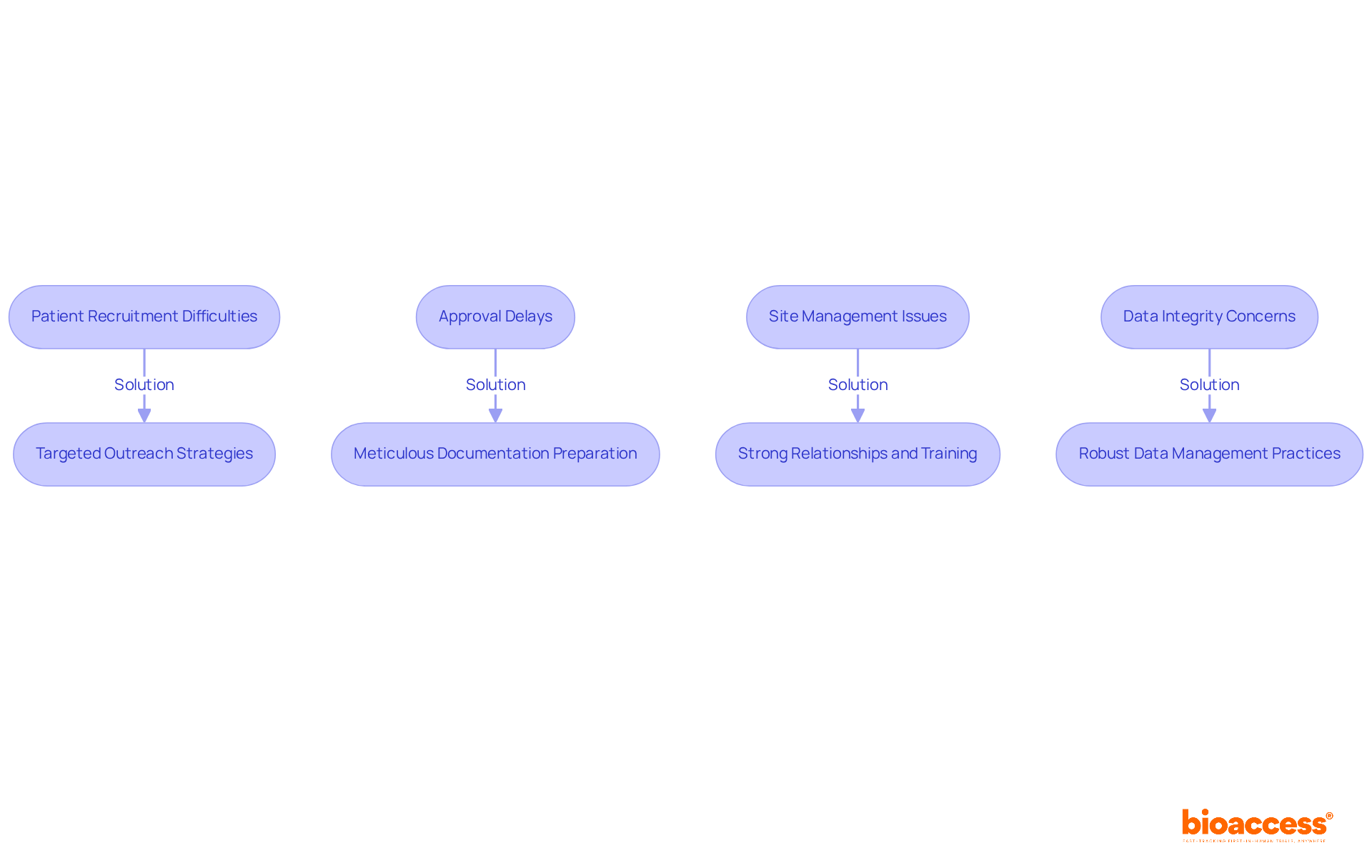
- Patient Recruitment Difficulties: Recruitment efforts often stall due to a lack of eligible participants, creating a significant barrier to success. To tackle this issue, consider targeted outreach strategies that connect with local healthcare providers and community organizations. Utilizing mobile clinics and health fairs can enhance visibility and trust, making it easier to identify potential participants.
- Approval Delays: Navigating the compliance landscape in Chile can be complex, often leading to setbacks in study initiation. To mitigate this, ensure that all documentation is meticulously prepared and compliant with the Instituto de Salud Pública (ISP) requirements before submission. Engaging consultants with expertise in local regulations can further streamline the process and help anticipate potential obstacles.
- Site Management Issues: Ensuring that clinical research locations are adequately staffed and equipped is crucial. Fostering strong relationships with site staff and providing comprehensive training on the study protocol can enhance site readiness and compliance.
- Data Integrity Concerns: Maintaining high data quality and integrity is essential for regulatory submissions. Implement robust data management practices, including regular audits and real-time monitoring, to identify and rectify discrepancies promptly.
By proactively addressing these challenges, sponsors can significantly enhance the likelihood of successful phase 1 clinical trial Chile outcomes, leveraging the region’s strategic advantages in speed and cost efficiency for early-stage clinical trials. Addressing these challenges not only improves trial outcomes but also positions sponsors to capitalize on Chile’s advantages in clinical research.
Conclusion
Phase 1 clinical trials in Chile present a compelling opportunity for MedTech and Biopharma companies to harness unique advantages. With a diverse patient population, streamlined regulatory processes, and significant cost efficiencies, these factors facilitate faster trial execution. They also enhance the quality and reliability of the data collected, positioning Chile as an attractive destination for early-stage clinical research.
Key insights from this guide highlight the importance of understanding the regulatory landscape and effective patient recruitment strategies. Navigating the complexities of regulatory approvals can be daunting for sponsors. By addressing challenges such as approval delays and site management issues, companies can significantly enhance their chances of success. Compliance with ICH-GCP standards and maintaining clear communication with regulatory bodies like the Instituto de Salud Pública (ISP) is crucial for ensuring trial integrity and timely approvals.
In conclusion, the potential for successful Phase 1 clinical trials in Chile is immense, driven by the region’s advantages in speed, cost-effectiveness, and a supportive regulatory environment. To truly capitalize on these benefits, companies need to prioritize thorough preparation and strategic planning to overcome challenges and maximize their research outcomes. By embracing these strategies, companies not only enhance their research outcomes but also solidify Latin America’s position in the global clinical research arena.
Frequently Asked Questions
What are Phase 1 clinical trials?
Phase 1 clinical trials are initial studies conducted to evaluate the safety, tolerability, and pharmacokinetics of new medications and medical devices, typically involving 20 to 80 healthy participants.
What is the main goal of Phase 1 clinical trials?
The main goal is to determine the maximum tolerated dose (MTD) and identify potential side effects of the new treatment.
What advantages does Chile offer for conducting Phase 1 clinical trials?
Chile offers a diverse patient base, streamlined approval processes, significant cost reductions, and a robust healthcare system which supports clinical research.
How many research studies were conducted in Chile in 2025?
In 2025, Chile conducted over 150 research studies, reflecting a strong research environment.
What regulatory bodies oversee clinical trials in Chile?
The regulatory framework is overseen by the Instituto de Salud Pública (ISP), ANMAT, and other agencies that ensure compliance with ICH-GCP standards.
What is the typical evaluation period for clinical study approvals in Chile?
The evaluation period for clinical study approvals in Chile typically spans around 30 days.
What is the retention rate for participants in clinical trials in Chile?
Chile boasts a retention rate exceeding 85%, which enhances the reliability of trial outcomes.
How do streamlined regulatory procedures benefit companies conducting trials in Chile?
Streamlined regulatory procedures have enabled companies to save 9 to 12 months in development time due to quicker approvals, which is vital for startups seeking funding.
Can you provide an example of a successful Phase 1 study conducted in Chile?
An example is the investigation of pembrolizumab (Keytruda), which demonstrated the importance of thorough safety monitoring and adaptive dose escalation, ultimately leading to market approval.
What are the key benefits of conducting Phase 1 trials in Chile?
Key benefits include a diverse and accessible patient population, quick approval timelines, high retention rates, significant cost efficiencies, and successful precedents that demonstrate the effectiveness of the local clinical trial environment.
List of Sources
- Understand Phase 1 Clinical Trials in Chile
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Phase I Clinical Trials Explained: Objectives, Risks & Process (https://ccrps.org/clinical-research-blog/phase-i-clinical-trials-explained-objectives-risks-amp-process)
- A Comprehensive Guide to Phase 1 Clinical Trials | ICON news and blogs (https://careers.iconplc.com/blogs/2023-10/a-guide-to-phase-1-clinical-trials)
- Navigate Chilean Regulatory Requirements
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Regulatory Guide for Latin America | bioaccess® (https://bioaccessla.com/regulatory-guide)
- Execute Phase 1 Trials: Key Steps and Strategies
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- A Comprehensive Guide to Phase 1 Clinical Trials | ICON news and blogs (https://careers.iconplc.com/blogs/2023-10/a-guide-to-phase-1-clinical-trials)
- Optimizing Site Selection and Management for Clinical Trial Success (https://studypages.com/blog/optimizing-site-selection-and-management-for-clinical-trial-success)
- Keys to Maximize Patient Recruitment (https://linical.com/articles-research/keys-to-maximize-patient-recruitment)
- Overcome Challenges in Phase 1 Trials
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- Patient Recruitment Strategies for Clinical Trials (https://freyrsolutions.com/blog/patient-recruitment-strategies-for-clinical-trials)
- Clinical Trials Patient Recruitment in Latin America | H Clinical (https://hclinical.com/patient-recruitment)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)

Leave a Reply