Introduction
The aseptic fill-finish manufacturing landscape is rapidly evolving, driven by the increasing demand for biologics and the necessity for stringent quality control measures. As the industry faces mounting challenges—ranging from regulatory compliance to contamination risks—understanding the key principles and innovations in this field becomes crucial for success.
What strategies can organizations implement to navigate these complexities and ensure the safety and efficacy of their products? This article delves into ten essential insights that illuminate the path to achieving excellence in aseptic fill-finish manufacturing.
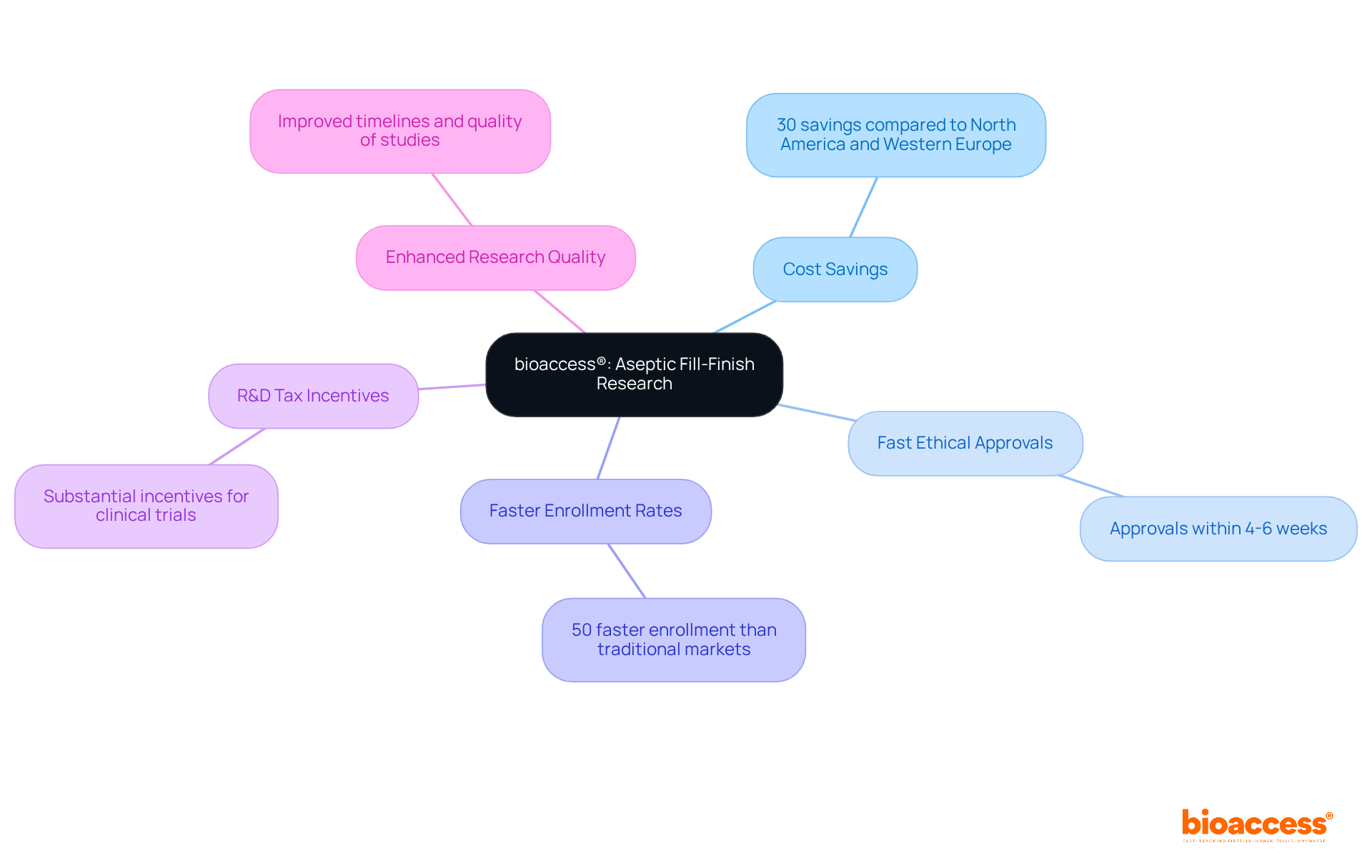
bioaccess®: Accelerating Aseptic Fill-Finish Research in Latin America
bioaccess® is at the forefront of in Latin America, leveraging Colombia’s competitive advantages to facilitate rapid .
With compared to North America and Western Europe, bioaccess® accelerates ethical approvals within just 4-6 weeks, significantly enhancing .
The company boasts than those in traditional markets, supported by a population of over 50 million, with 95% of individuals covered by universal healthcare.
Furthermore, Colombia presents , amplifying its attractiveness for .
This strategic positioning not only expedites research timelines but also elevates the overall quality of , empowering to achieve for their solutions and bring them to market with greater efficiency.
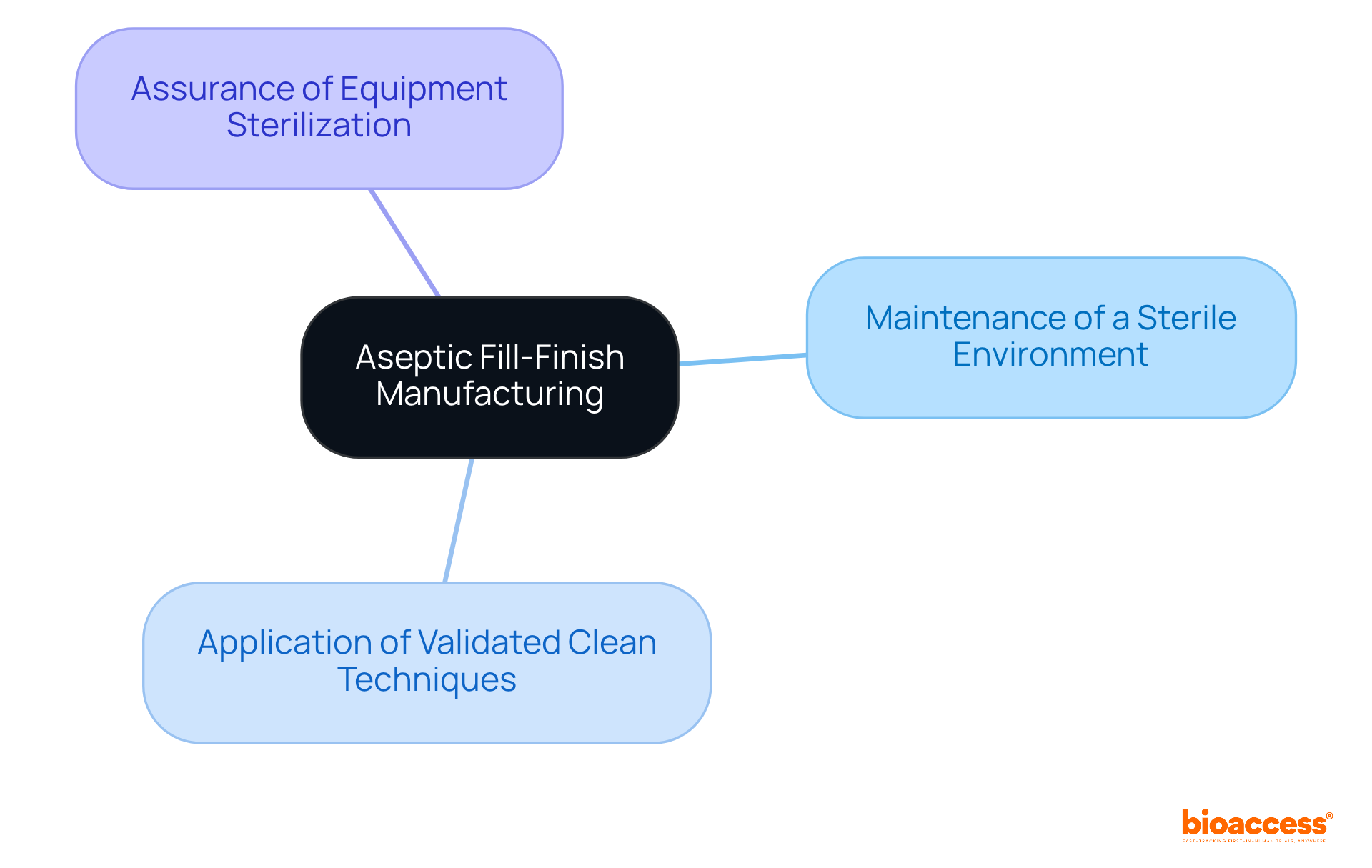
Understanding Aseptic Fill-Finish Manufacturing: Key Principles
serves as a cornerstone operation within the , focusing on the into containers under meticulously regulated conditions. This process is underpinned by key principles, including:
- The maintenance of a
- The application of
- The assurance that all equipment is thoroughly sterilized
The primary objective of this procedure is to , which is essential for of the final product—an aspect that is paramount for .
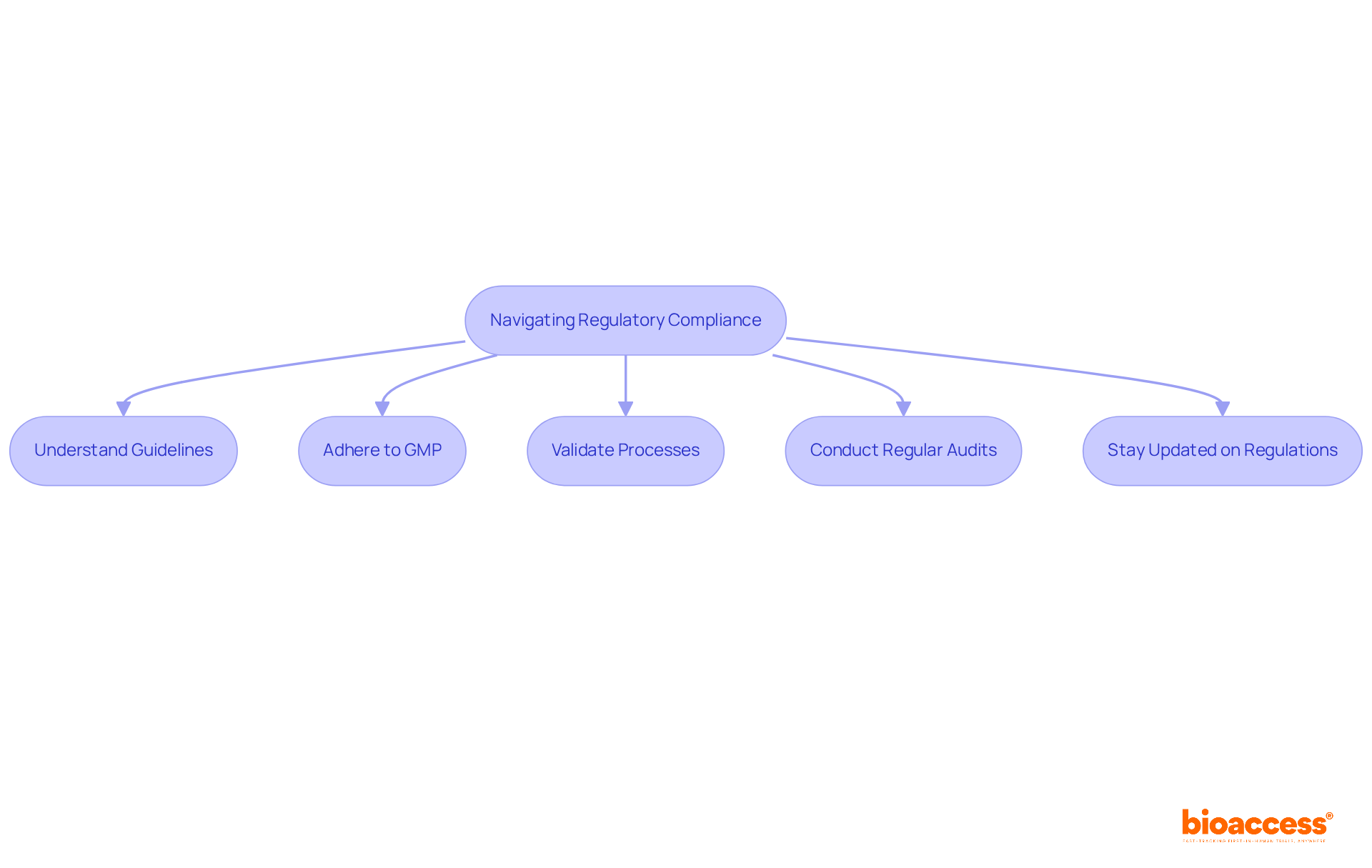
Navigating Regulatory Compliance in Aseptic Fill-Finish Operations
Navigating in requires a comprehensive understanding of the guidelines established by . Adherence to (GMP) is essential, necessitating companies to validate all processes meticulously. Regular audits and inspections are critical in maintaining compliance, helping to avert costly delays in product approvals. Furthermore, is vital for upholding operational integrity and ensuring patient safety.
Specialists like Ana Criado, Director of Regulatory Affairs and CEO of Mahu Pharma, underscore the significance of of the sterile production process, particularly concerning biologics and pharmaceuticals. As the , businesses must prioritize regulatory adherence to foster trust and reliability in their manufacturing processes.
With projections indicating an increase in the sterile packaging market from USD 7.5 billion in 2025 to USD 13.3 billion by 2035, the becomes increasingly evident. Successful case studies further illustrate the in achieving operational success.
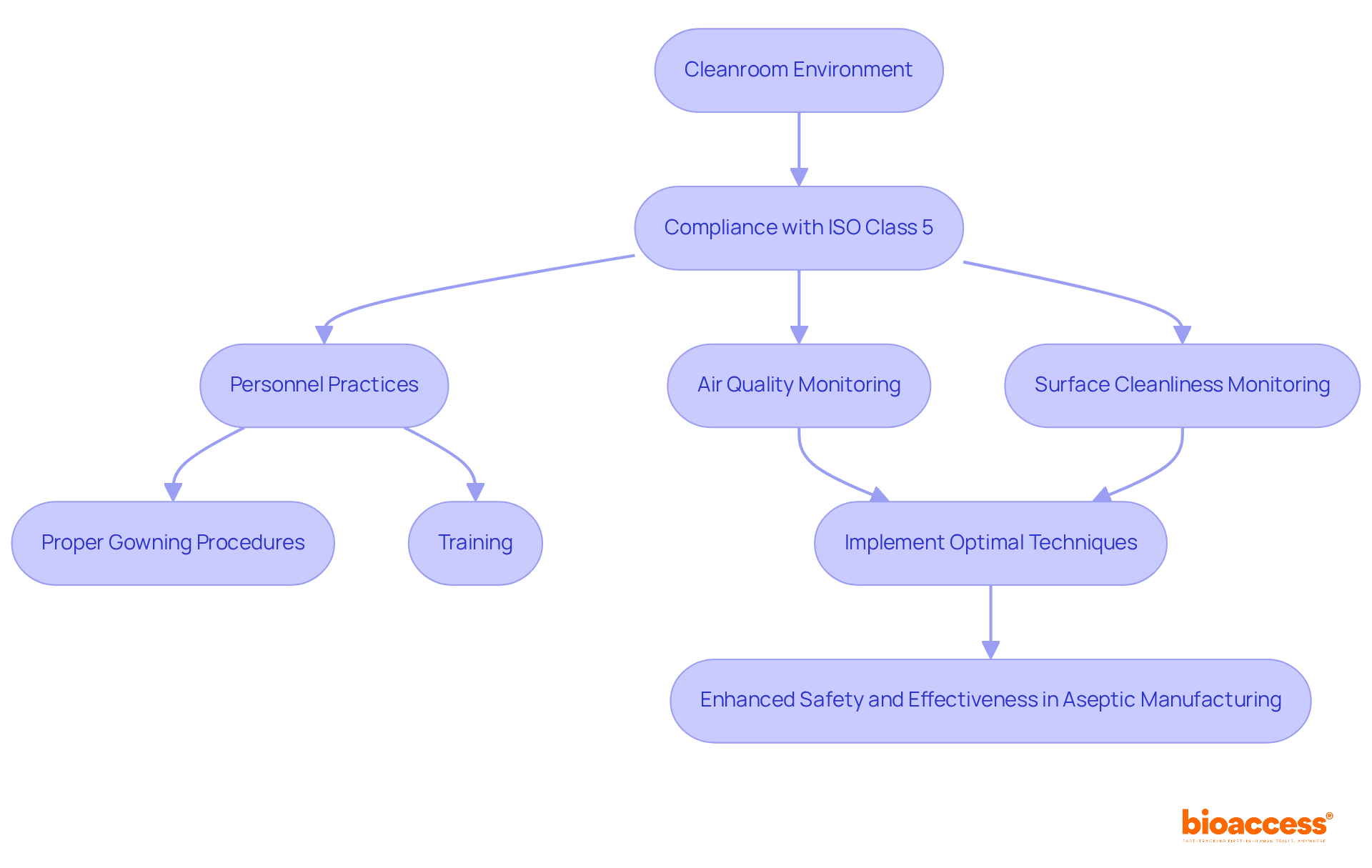
The Role of Cleanrooms in Ensuring Aseptic Conditions
Cleanrooms play a pivotal role in sterile packaging production, providing a meticulously regulated environment that significantly reduces contamination risks. To meet stringent standards such as , these facilities must limit airborne particle levels to a maximum of 3,520 particles (0.5 microns or larger) per cubic meter of air.
Consistent monitoring of air quality, surface cleanliness, and personnel practices is essential for maintaining the integrity of the . This includes adhering to , crucial for ensuring optimal air quality.
Proper gowning procedures and comprehensive training for personnel are imperative, as they directly impact . Research indicates that the number of operators in a cleanroom can greatly influence particle loads, highlighting the necessity for effective training and operational protocols.
By implementing these optimal techniques, organizations can ensure , thereby enhancing the safety and effectiveness of .
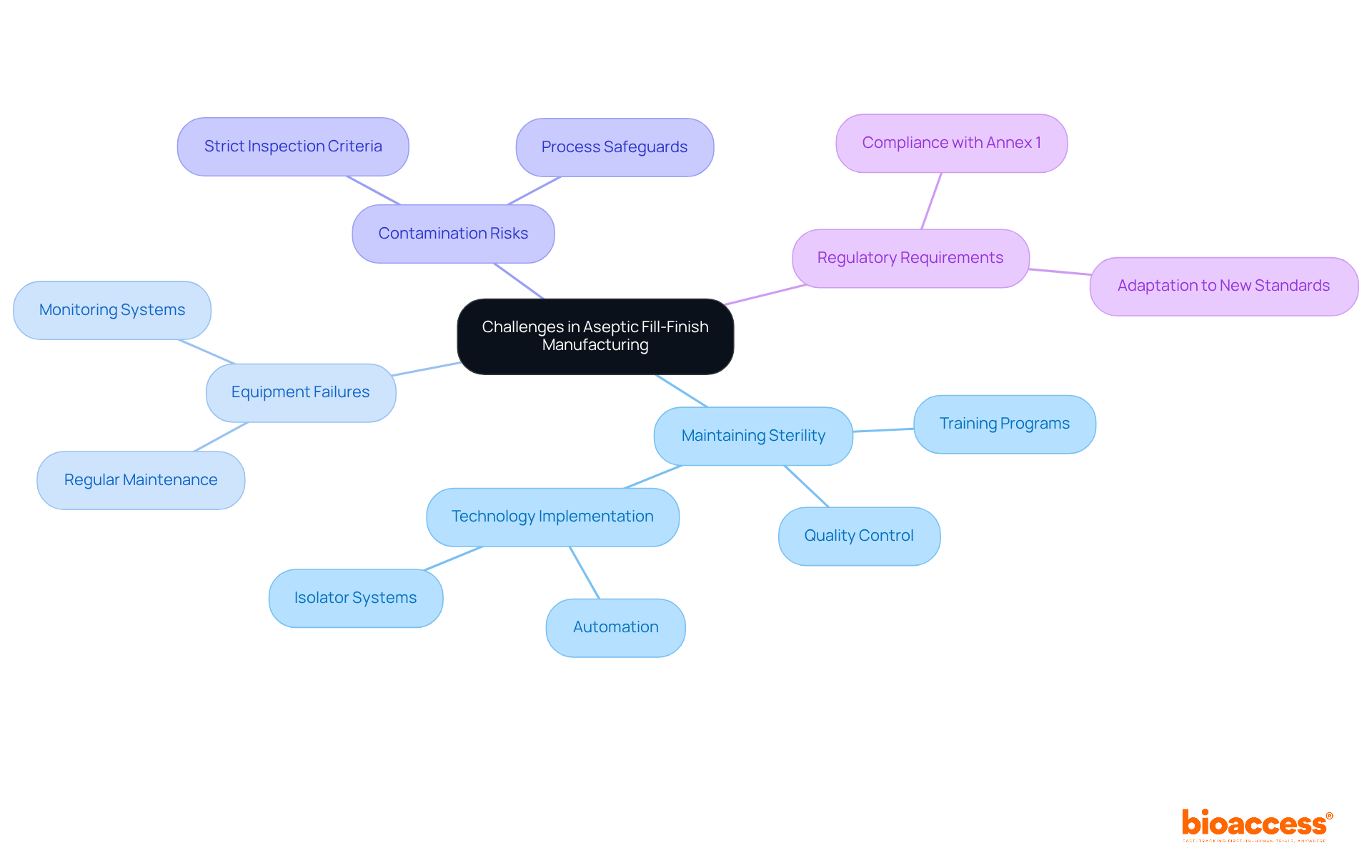
Identifying Challenges in Aseptic Fill-Finish Manufacturing
presents significant challenges that can critically affect product quality and patient safety. Maintaining sterility, managing complex formulations, and navigating are among the key issues. Notably, equipment failures pose a considerable risk; nearly 60% of surveyed organizations reported , with biopharmaceutical facilities experiencing an average batch failure once every 40.6 weeks. This underscores the urgent need for . Contamination risks remain a primary concern, as even minor breaches can result in severe consequences.
To address these challenges, implementing a multifaceted approach that incorporates is essential. Establishing robust training programs enhances staff competency, while regular equipment maintenance is crucial for reducing failures. Moreover, a stringent emphasis on is vital. The implementation of , such as automation and isolator systems, is increasingly relevant. There is a notable trend toward enhanced automation in sterile procedures, including the use of robots for transfers between processing stages. This not only ensures compliance with evolving regulatory standards but also aligns with the revised EU GMP Annex 1 guidance, which emphasizes contamination control and the strategic use of technology to minimize human intervention.
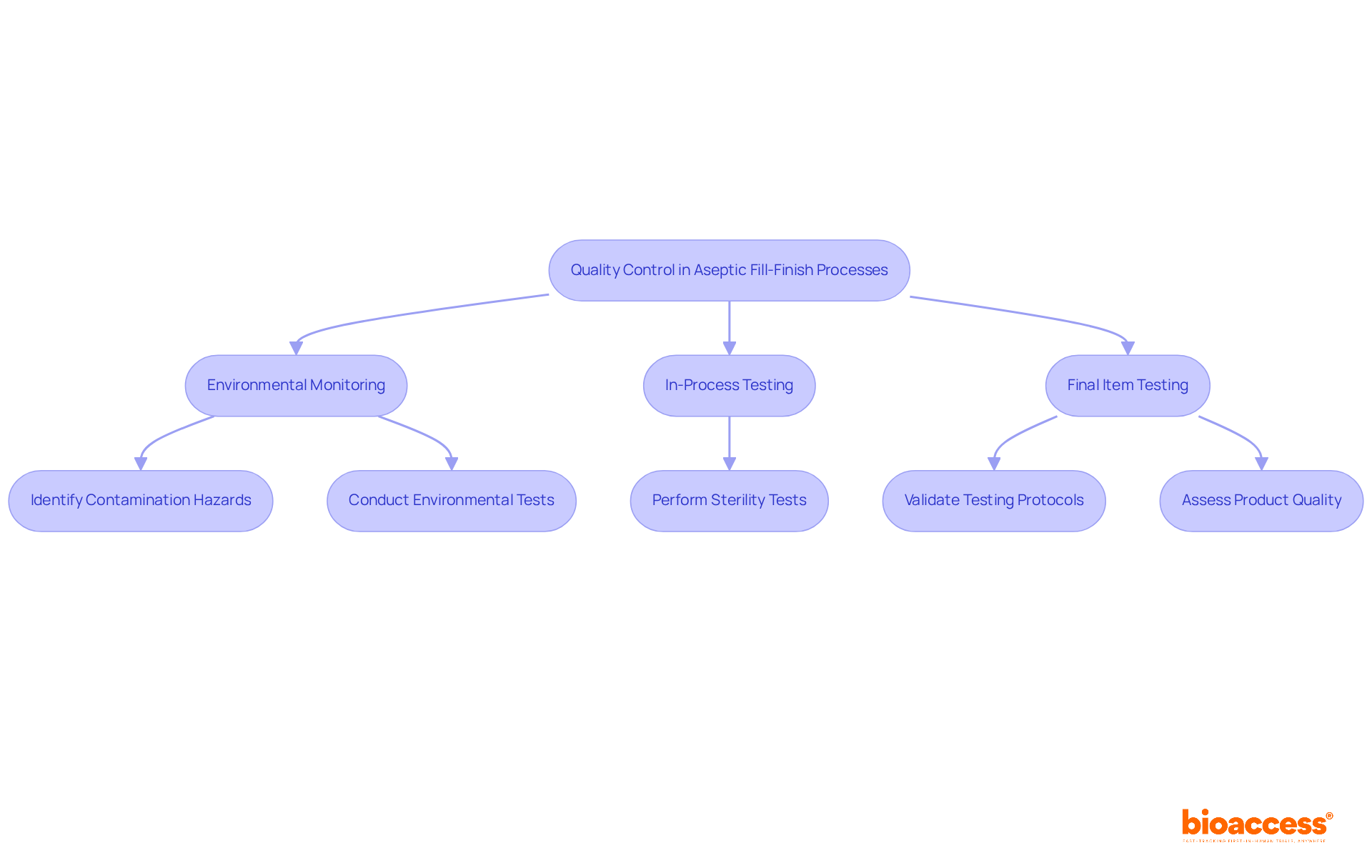
Implementing Quality Control in Aseptic Fill-Finish Processes
Implementing quality control in es is crucial for ensuring the safety and efficacy of the item. This entails thorough , in-process testing, and final item testing at every production stage. is especially critical, as it aids in identifying potential contamination hazards that could jeopardize quality. Statistics suggest that the from USD 6.04 billion in 2024 to USD 17.17 billion by 2034, highlighting the in tackling public health issues, particularly as noncommunicable diseases (NCDs) represent 71% of all global fatalities.
must be well-trained and equipped with advanced tools to swiftly identify and rectify any deviations from established protocols. of quality assurance methods are essential for ensuring adherence to strict regulatory requirements and improving overall quality. , including automation and digitalization, have further enhanced the precision and effectiveness of these processes, ensuring that sterile conditions are maintained throughout production.
The impact of robust on cannot be overstated; it is a fundamental component in preventing contamination and ensuring that products meet the highest safety standards. As Colleen Dixon, CEO of Selkirk Pharma, stated, “Time is of the essence in . Delays in the might result in missed trial milestones and major financial setbacks.” As the sterile packaging market continues to expand, fueled by , the focus on efficient will be crucial in protecting public health.
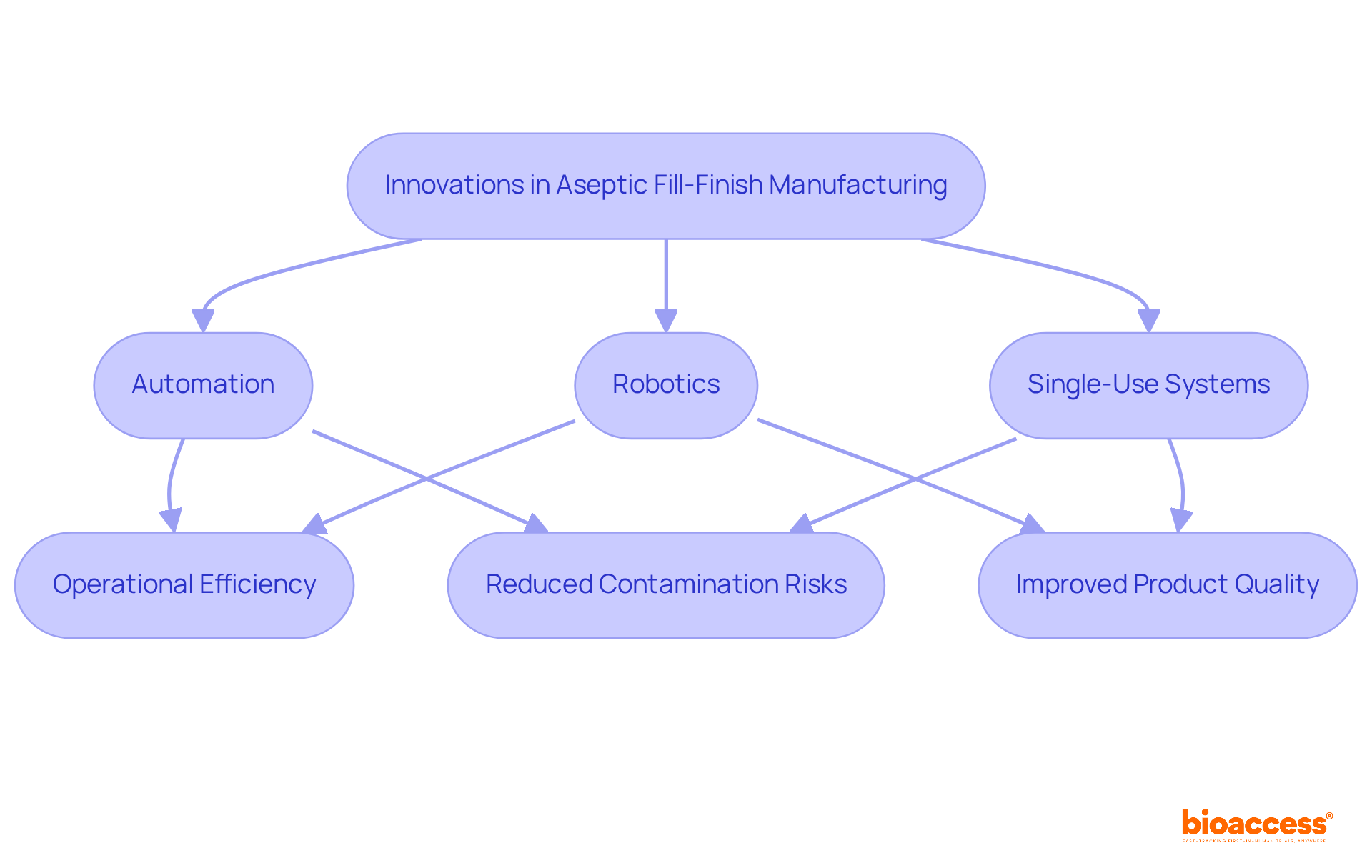
Innovations Transforming Aseptic Fill-Finish Manufacturing
The manufacturing environment for is undergoing a significant transformation driven by advancements in automation, robotics, and single-use systems. These innovations not only enhance operational efficiency but also reduce contamination risks associated with the , ultimately improving product quality.
For instance, adeptly manage various operational needs, significantly decreasing human involvement and the potential for errors. The market for services related to is projected to grow from USD 6.0 billion in 2024 to USD 8.3 billion by 2033, reaching USD 16.9 billion by 2034. This growth underscores the rising demand for .
Furthermore, the integration of digital solutions for monitoring and data analysis enables real-time adjustments to manufacturing processes, ensuring compliance with stringent . This capability is crucial as regulatory bodies like the US FDA and EMA mandate rigorous validation and documentation for sterile filling automation.
Companies that embrace these innovations are strategically positioned to meet the escalating demand for , which require es that are vital in various therapeutic areas, including oncology and immunology.
As the sector evolves, it is imperative for firms to stay informed about technological advancements such as to maintain a competitive edge in the sterile packaging market. The adoption rates of automation in sterile manufacturing are steadily rising, driven by the necessity for higher sterility levels and operational efficiency, particularly in the . This trend is expected to continue, with and single-use technologies, further revolutionizing the sterile packaging processes.
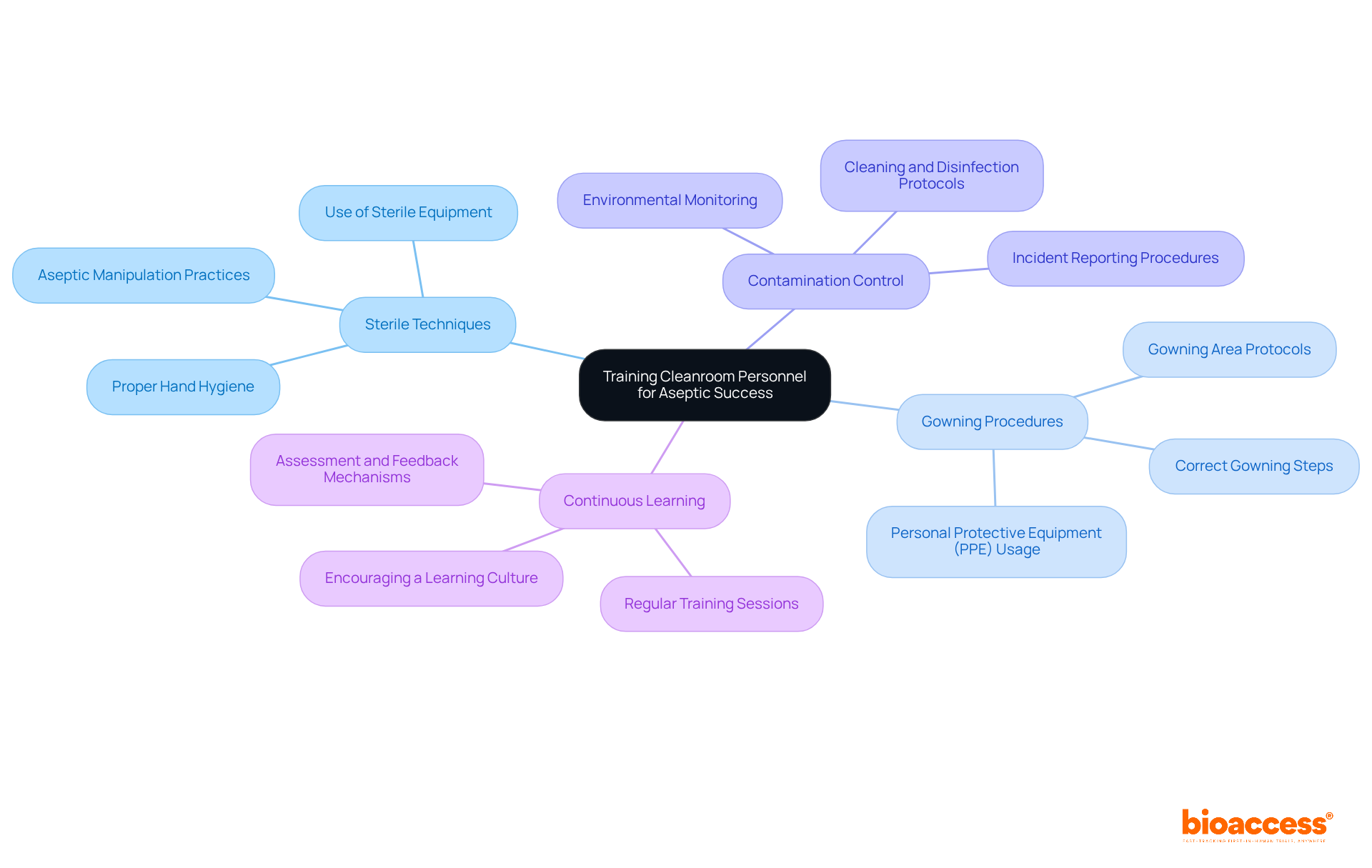
Training Cleanroom Personnel for Aseptic Success
is essential in the realm of aseptic fill finish production. It is imperative that personnel are well-versed in , , and . Regular and assessments are crucial in ensuring compliance with industry standards and best practices. Furthermore, fostering a culture of not only enhances team performance but also contributes significantly to the success of clean operations.
Future Trends in Aseptic Fill-Finish Manufacturing
The future of manufacturing is increasingly characterized by a , automation, and the incorporation of like artificial intelligence (AI) and machine learning. These innovations enhance while significantly reducing waste and improving product quality.
As the demand for and biologics escalates, manufacturers are compelled to adopt more . Companies that proactively embrace these trends will be strategically positioned to meet , ensuring their competitiveness in a rapidly changing landscape.
The emphasis on sustainability is particularly crucial, as regulatory agencies tighten standards, urging organizations to innovate in their sterile processes, especially in , while .
Moreover, the successful adoption of AI in sterile manufacturing is revolutionizing workflows, with statistics indicating that organizations leveraging these technologies can achieve .
As the industry evolves, staying ahead of these trends will be essential for success.
Key Takeaways on Aseptic Fill-Finish Manufacturing
Essential insights on sterile production highlight the necessity of maintaining a and adhering to . Notably, approximately 80% of product recalls are associated with packaging-related issues, emphasizing the .
The market, which is valued at USD 16.00 billion in 2024, is projected to grow at a CAGR of 8.90% through 2034, with expectations to reach USD 37.53 billion by 2034, driven by the . Furthermore, —such as single-use systems and AI-driven methods—are essential for enhancing efficiency and sustaining sterility throughout production.
As Vishakha Agrawal notes, ‘The process involves filling and sealing in containers like vials, syringes, and ampules.’ Successful strategies for encompass thorough training and the implementation of advanced monitoring systems, which are vital for mitigating risks associated with maintaining sterility.
As the industry continues to evolve, it is imperative for organizations to stay informed about to excel in the competitive landscape of .
Conclusion
The landscape of aseptic fill-finish manufacturing is shaped by critical insights that emphasize the importance of maintaining sterile environments and adhering to rigorous regulatory standards. This process not only safeguards the efficacy and safety of pharmaceutical products but also plays a vital role in the broader healthcare ecosystem. As the demand for biologics and biopharmaceuticals continues to surge, the need for efficient and compliant aseptic fill-finish operations has never been more pressing.
Key arguments outlined in this article highlight the multifaceted challenges faced by manufacturers, including:
- The necessity for stringent quality control
- The pivotal role of cleanrooms
- The impact of emerging technologies
Innovations such as automation and AI-driven solutions are transforming operations, driving efficiency, and reducing contamination risks. Furthermore, the importance of thorough training for personnel cannot be overstated, as it directly influences the success of aseptic processes.
Looking ahead, organizations must remain vigilant and adaptable in the face of evolving market demands and regulatory landscapes. Embracing advancements in technology and fostering a culture of continuous improvement will be essential for maintaining a competitive edge in aseptic fill-finish manufacturing. The commitment to excellence in this field not only enhances product safety but also contributes significantly to public health outcomes, underscoring the critical nature of aseptic manufacturing in today’s healthcare environment.
Frequently Asked Questions
What is bioaccess® and what role does it play in aseptic fill-finish research in Latin America?
bioaccess® is a leader in aseptic fill-finish packaging research in Latin America, utilizing Colombia’s advantages to expedite clinical trials and enhance regulatory efficiency.
How does bioaccess® achieve cost savings in clinical trials?
bioaccess® offers cost savings exceeding 30% compared to North America and Western Europe, which contributes to its competitive edge in conducting clinical trials.
What is the timeline for ethical approvals in Colombia as facilitated by bioaccess®?
Ethical approvals in Colombia can be achieved in just 4-6 weeks, significantly improving the regulatory process.
How do enrollment rates for clinical trials in Colombia compare to traditional markets?
Enrollment rates in Colombia are 50% faster than those in traditional markets, aided by a population of over 50 million and a high percentage of universal healthcare coverage.
What are the R&D tax incentives available in Colombia?
Colombia offers substantial R&D tax incentives, making it an attractive location for conducting clinical trials.
What is the primary objective of aseptic fill-finish manufacturing?
The primary objective of aseptic fill-finish manufacturing is to prevent contamination during the sterile filling of medications, which is crucial for ensuring the safety and efficacy of the final product.
What are the key principles of aseptic fill-finish manufacturing?
Key principles include maintaining a sterile environment, applying validated clean techniques, and ensuring all equipment is thoroughly sterilized.
Why is regulatory compliance important in aseptic fill-finish operations?
Regulatory compliance is vital to adhere to guidelines set by regulatory bodies like the FDA and EMA, ensuring Good Manufacturing Practices (GMP) are followed to maintain product safety and efficacy.
What role do audits and inspections play in regulatory compliance?
Regular audits and inspections are critical for maintaining compliance, helping to prevent costly delays in product approvals.
What is the projected growth of the sterile packaging market?
The sterile packaging market is projected to grow from USD 7.5 billion in 2025 to USD 13.3 billion by 2035, highlighting the increasing importance of compliance in the industry.
List of Sources
- bioaccess®: Accelerating Aseptic Fill-Finish Research in Latin America
- linkedin.com (https://linkedin.com/pulse/latin-america-biologics-fill-finish-service-vthyc)
- dataintelo.com (https://dataintelo.com/report/aseptic-fill-finish-service-market)
- insightaceanalytic.com (https://insightaceanalytic.com/report/aseptic-fill-finish-market-/1880)
- expertmarketresearch.com (https://expertmarketresearch.com/reports/aseptic-fill-finish-manufacturing-market?srsltid=AfmBOooBlg6dYYJhekrNus7dZEgqouZTHwLwL-mMi7daMtOZlAt0cyIC)
- fairfieldmarketresearch.com (https://fairfieldmarketresearch.com/report/aseptic-fill-finish-market)
- Understanding Aseptic Fill-Finish Manufacturing: Key Principles
- ast-inc.com (https://ast-inc.com/blogs/ast-featured-in-the-medicine-maker-expert-roundtable-on-aseptic-fill-finish-manufacturing)
- rootsanalysis.com (https://rootsanalysis.com/press-releases/aseptic-fill-finish-manufacturing-market.html)
- expertmarketresearch.com (https://expertmarketresearch.com/reports/aseptic-fill-finish-manufacturing-market?srsltid=AfmBOopyqg0akwobTWEYXCueWC3SZKFPHZETBDrx0TMFyN4F4fSYy3D5)
- insightaceanalytic.com (https://insightaceanalytic.com/report/aseptic-fill-finish-market-/1880)
- Navigating Regulatory Compliance in Aseptic Fill-Finish Operations
- outsourcedpharma.com (https://outsourcedpharma.com/doc/quality-and-regulatory-compliance-in-fill-finish-the-foundation-for-excellence-in-every-injection-0001)
- insightaceanalytic.com (https://insightaceanalytic.com/report/aseptic-fill-finish-market-/1880)
- rootsanalysis.com (https://rootsanalysis.com/reports/aseptic-fill-finish-market.html)
- towardshealthcare.com (https://towardshealthcare.com/insights/aseptic-fill-finish-market-sizing)
- The Role of Cleanrooms in Ensuring Aseptic Conditions
- blog.bccresearch.com (https://blog.bccresearch.com/whats-behind-the-growth-of-the-cleanroom-market)
- datamintelligence.com (https://datamintelligence.com/research-report/cleanroom-technologies-market)
- ISO 5 Cleanroom Standards for 14644-1 Certification (FS209E Class 100) (https://terrauniversal.com/blog/iso-class-5-cleanroom-standards-for-ISO-14644-1-clean-room-classes-particulate)
- designtekconsulting.com (https://designtekconsulting.com/post/from-particles-to-patients-the-critical-role-of-contaminant-cleanroom-control-in-pharma)
- tandfonline.com (https://tandfonline.com/doi/full/10.1080/03639045.2022.2043352)
- Identifying Challenges in Aseptic Fill-Finish Manufacturing
- pharmtech.com (https://pharmtech.com/view/facing-the-future-of-aseptic-manufacturing)
- pharmaceuticalmanufacturer.media (https://pharmaceuticalmanufacturer.media/pharmaceutical-industry-insights/pharmaceutical-manufacturing-insights/a-game-of-risk-the-challenges-faced-in-aseptic-processing)
- bioprocessintl.com (https://bioprocessintl.com/bioanalytical-methods/biotech-facilities-average-a-batch-failure-every-40-6-weeks)
- innovatrix.eu (https://innovatrix.eu/what-challenges-are-associated-with-fill-finish-manufacturing)
- biospectrumasia.com (https://biospectrumasia.com/news/95/20156/the-challenges-faced-by-the-aseptic-and-sterile-fill-and-finish-industry.html)
- Implementing Quality Control in Aseptic Fill-Finish Processes
- insightaceanalytic.com (https://insightaceanalytic.com/report/aseptic-fill-finish-market-/1880)
- dataintelo.com (https://dataintelo.com/report/global-pharmaceutical-aseptic-fill-finish-cmo-market)
- towardshealthcare.com (https://towardshealthcare.com/insights/aseptic-fill-finish-market-sizing)
- verifiedmarketreports.com (https://verifiedmarketreports.com/product/pharmaceutical-aseptic-fill-finish-cmo-market-size-and-forecast)
- Innovations Transforming Aseptic Fill-Finish Manufacturing
- insightaceanalytic.com (https://insightaceanalytic.com/report/aseptic-fill-finish-market-/1880)
- expertmarketresearch.com (https://expertmarketresearch.com/reports/aseptic-fill-finish-manufacturing-market?srsltid=AfmBOoplHK1U8la89eImYAA-hg7KcCC3ch8brACcxgQ8wAs0SmIxfXEh)
- marketintelo.com (https://marketintelo.com/report/aseptic-filling-automation-market)
- towardshealthcare.com (https://towardshealthcare.com/insights/aseptic-fill-finish-manufacturing-market-sizing)
- Training Cleanroom Personnel for Aseptic Success
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC10937679)
- asepticenclosures.com (https://asepticenclosures.com/personnel-aseptic-practices-training)
- researchgate.net (https://researchgate.net/publication/363320798_The_role_of_simulation-based_training_in_healthcare-associated_infection_HAI_prevention)
- aricjournal.biomedcentral.com (https://aricjournal.biomedcentral.com/articles/10.1186/s13756-025-01587-6)
- fabtechnologies.com (https://fabtechnologies.com/a-quick-guide-to-training-to-ensure-cleanroom-best-practice)
- Future Trends in Aseptic Fill-Finish Manufacturing
- reportsanddata.com (https://reportsanddata.com/report-detail/aseptic-fill-finish-manufacturing-market)
- expertmarketresearch.com (https://expertmarketresearch.com/reports/aseptic-fill-finish-manufacturing-market?srsltid=AfmBOoqyM5nmt3E-1uLLJ1eePp_FIMUgIjb5jceGJK_b9Y0IxrvrnP5g)
- towardshealthcare.com (https://towardshealthcare.com/insights/aseptic-fill-finish-market-sizing)
- Automation Statistics 2026: Comprehensive Industry Data and Market Insights (https://thunderbit.com/blog/automation-statistics-industry-data-insights)
- Key Takeaways on Aseptic Fill-Finish Manufacturing
- expertmarketresearch.com (https://expertmarketresearch.com/reports/aseptic-fill-finish-manufacturing-market?srsltid=AfmBOorlcUpf5p5eH0QhBajo5e1TWGchkCnLrQbijConCBLTf8vcURd4)
- finance.yahoo.com (https://finance.yahoo.com/news/emerging-trends-aseptic-fill-finish-160000230.html)
- towardshealthcare.com (https://towardshealthcare.com/insights/aseptic-fill-finish-manufacturing-market-sizing)
- marketsandmarkets.com (https://marketsandmarkets.com/PressReleases/fill-finish-manufacturing.asp)

Leave a Reply