Introduction
Navigating the complex landscape of medical device labeling requirements is crucial for manufacturers aiming to achieve FDA compliance. With approximately 30% of medical devices failing to meet these standards, understanding the essential guidelines is not just beneficial—it’s imperative for patient safety and market success. As the stakes rise, manufacturers must ensure their labels convey accurate information while adhering to the stringent regulations set forth by the FDA. This article delves into the ten key labeling requirements that every Medtech firm must master to thrive in the competitive healthcare landscape.
bioaccess®: Accelerate Compliance with FDA Medical Device Labeling Requirements
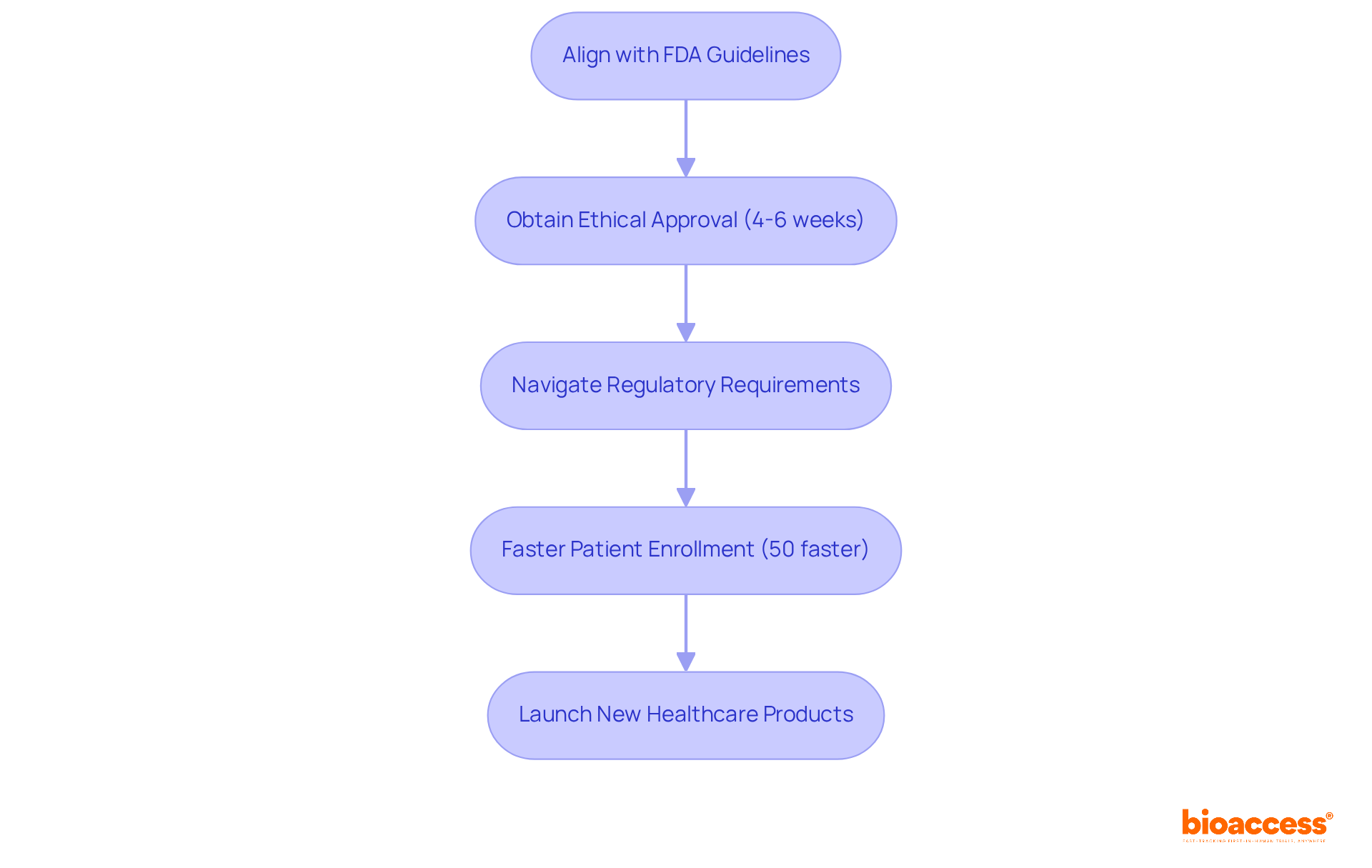
bioaccess® is dedicated to empowering Medtech firms as they navigate the intricate FDA for healthcare product identification. By emphasizing the acceleration of compliance, bioaccess® leverages its profound understanding of regulatory frameworks, particularly the fda, to ensure that clients efficiently meet all necessary guidelines.
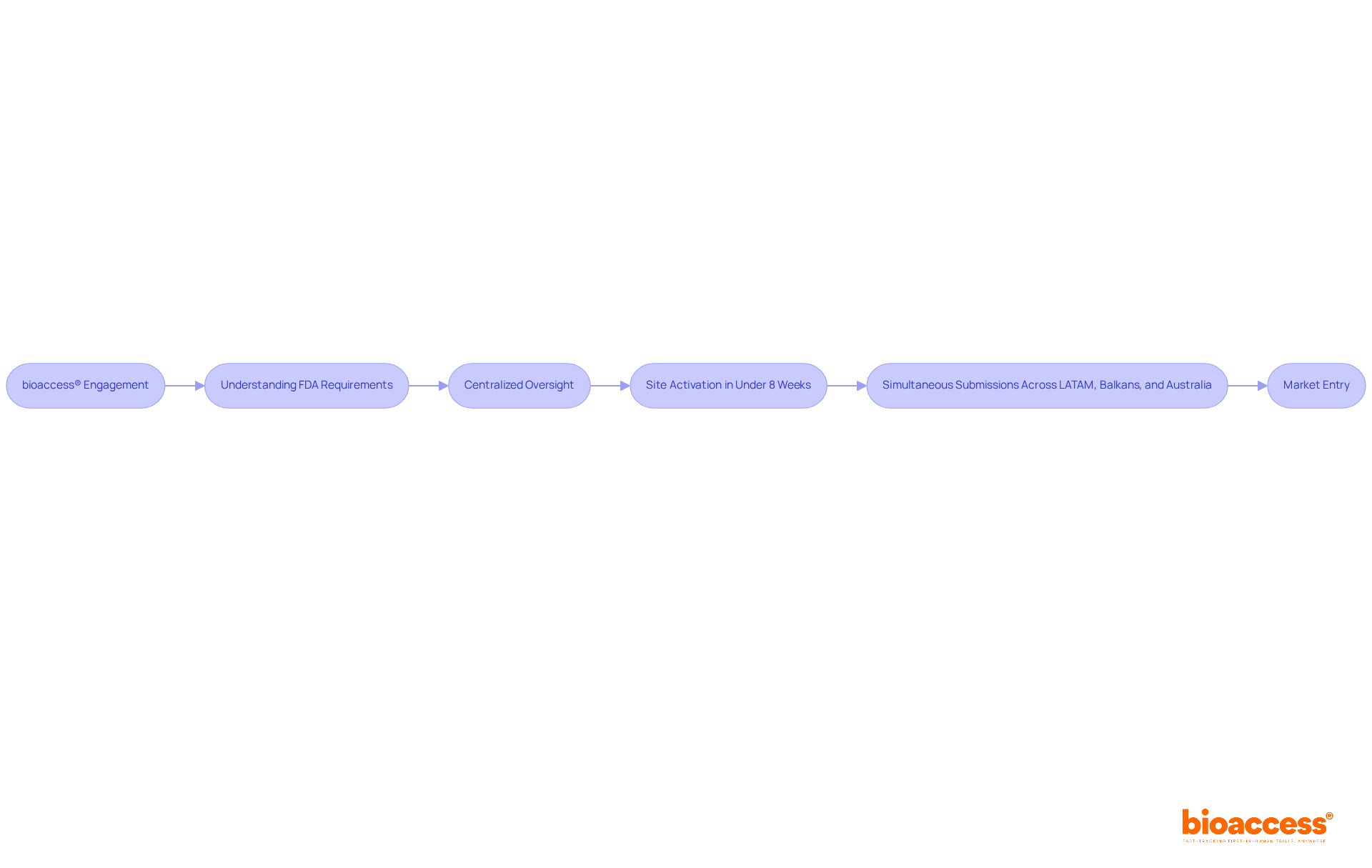
With its global-first clinical flexibility and provision of under centralized oversight, bioaccess® enables faster market entry for groundbreaking healthcare products. This includes:
- Site activation in under 8 weeks
- The capability for simultaneous submissions across LATAM, the Balkans, and Australia
This ensures that documentation requirements are met promptly while delivering tailored solutions and expert support for in diverse regions.
General Labeling Provisions: Key FDA Guidelines for Medical Devices
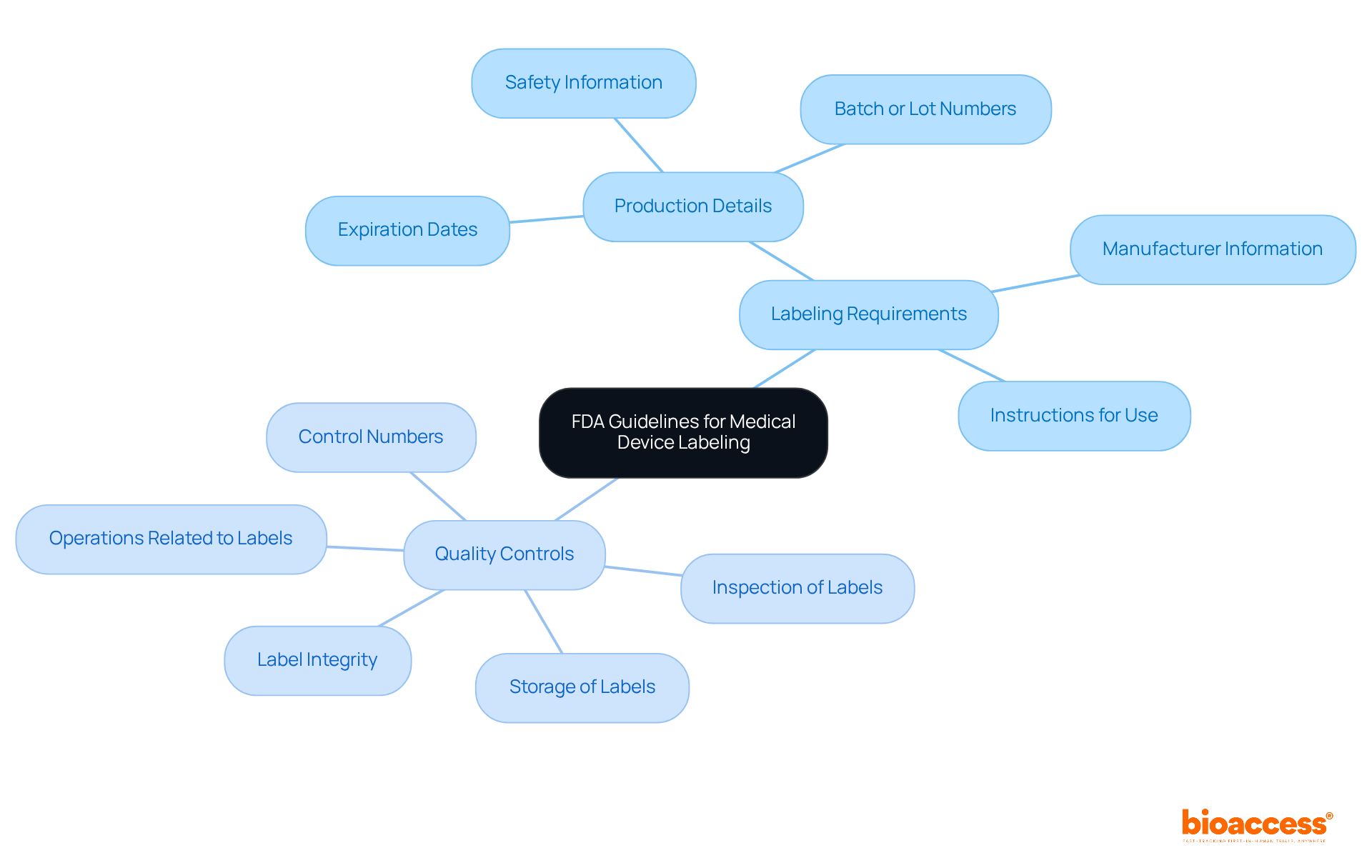
The that the ensure that medical instrument labels are accurate and non-deceptive, a fundamental principle that underpins patient well-being and regulatory compliance. Central to these regulations are general marking provisions, which require that the label includes the name and location of the manufacturer, packer, or distributor, along with sufficient instructions for use. These guidelines are essential, ensuring users possess the and effectively, which is critical for and adherence to regulations.
Notably, research indicates that approximately 30% of medical devices fail to meet FDA marking standards, leading to compliance issues and potential delays in market entry. Manufacturers must concentrate on :
- Label integrity
- Inspection of labels
- Storage of labels
- Operations related to labels
- Control numbers
to guarantee compliance with these guidelines. Moreover, the set forth by the FDA’s highlight the necessity of including such as expiration dates, safety information, and batch or lot numbers. Accurate labeling not only , underscoring the imperative for manufacturers to prioritize .
Required Elements on FDA-Compliant Medical Device Labels
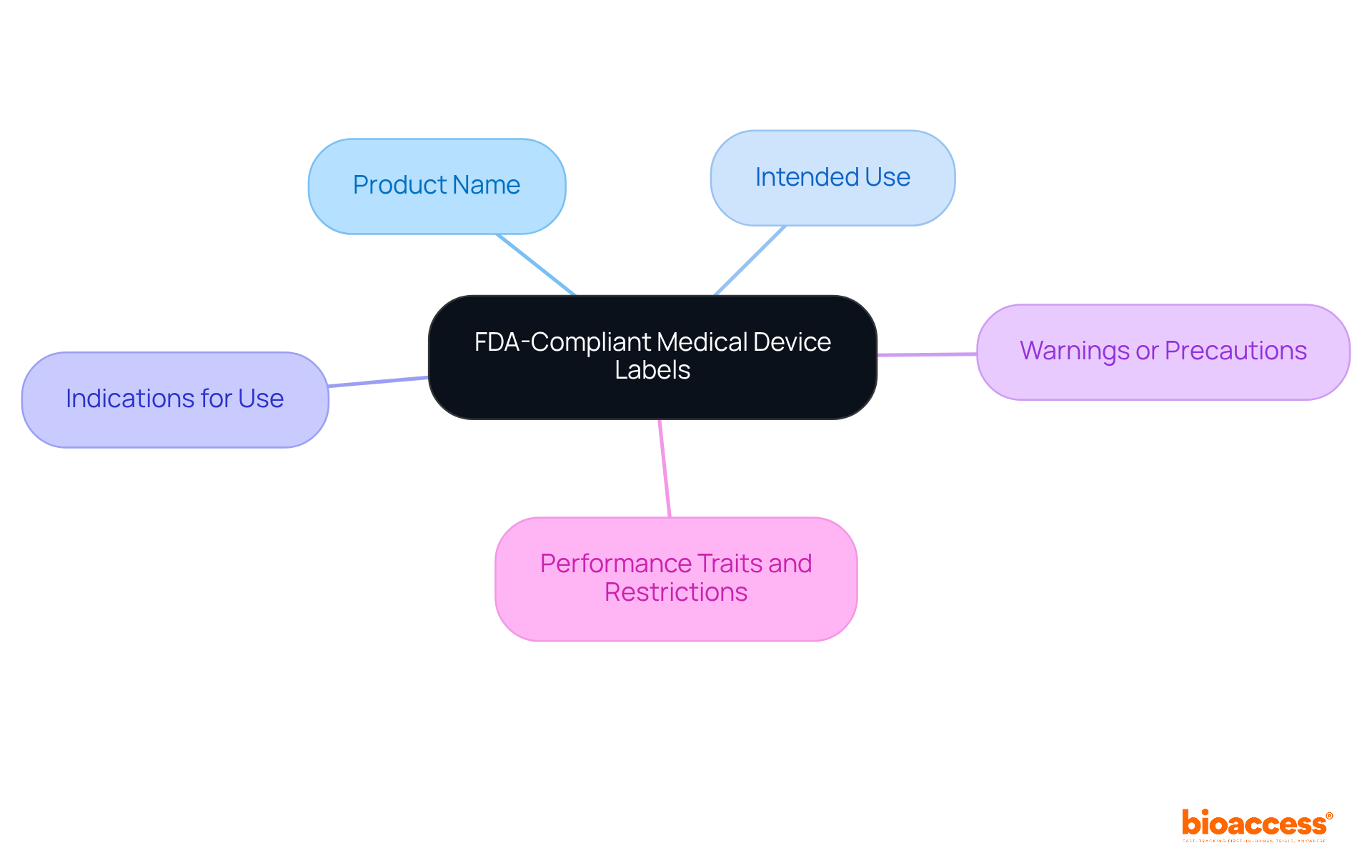
The mandate that must include several , such as:
- Product name
- Any warnings or precautions
Additionally, labels are required to provide details on the equipment’s and any applicable restrictions. The inclusion of these elements is crucial; it not only helps manufacturers comply with but also enhances user comprehension of the product.
Unique Device Identification (UDI): Essential for FDA Labeling Compliance
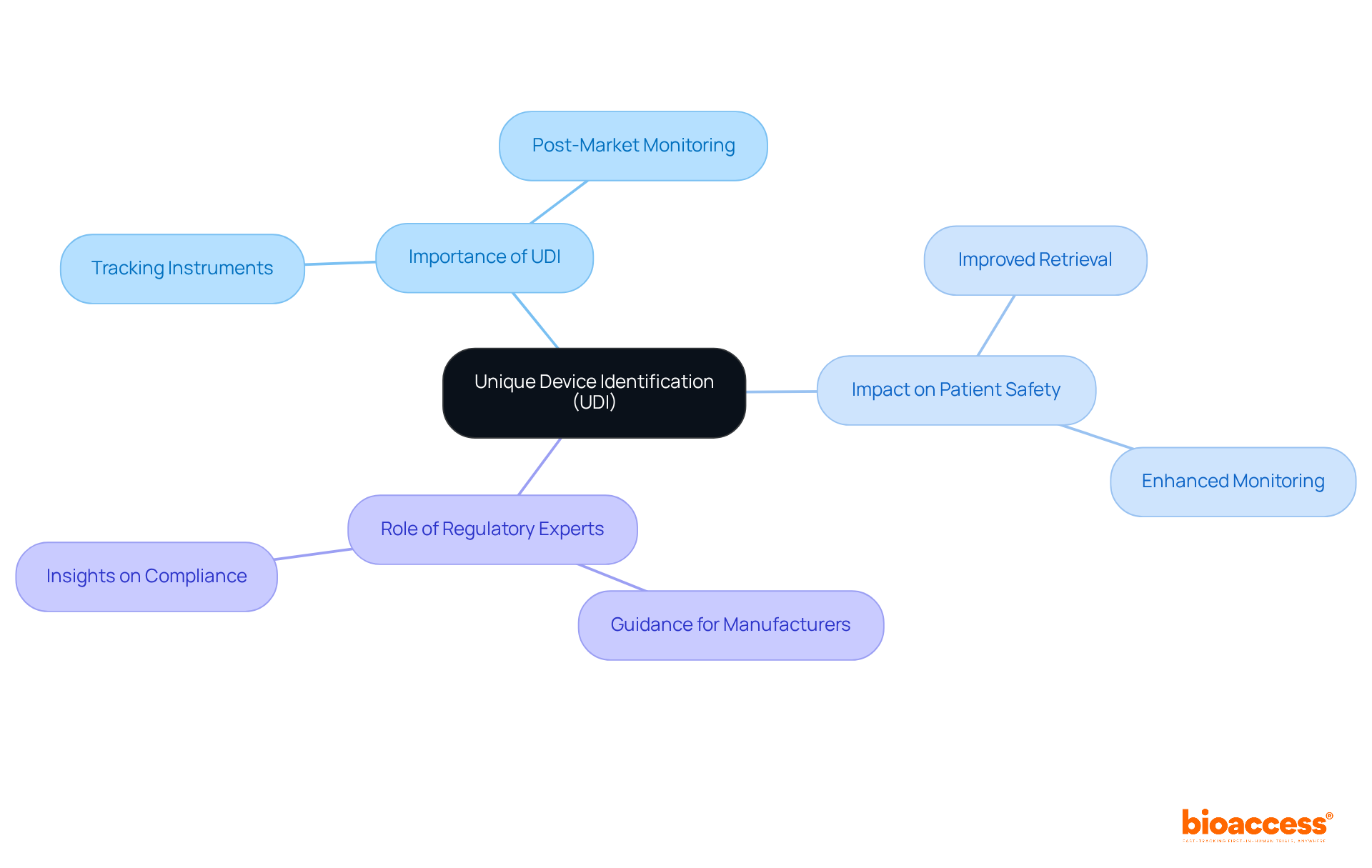
The mandates that most healthcare instruments carry a unique identifier, facilitating easier tracking and recognition. This system significantly enhances by aiding in the retrieval of instruments and improving . Compliance with the FDA and UDI requirements is essential for manufacturers to meet , ensuring that their products can be effectively monitored throughout their lifecycle.
Experts in , such as Ana Criado, who possesses extensive experience at Colombia’s regulatory agency INVIMA and serves as a consultant for global companies, play a crucial role in guiding manufacturers through these requirements. Her insights are instrumental in ensuring that medical equipment adheres to UDI standards, ultimately contributing to enhanced and efficient market supervision.
Adequate Directions for Use: A Must for Medical Device Labels
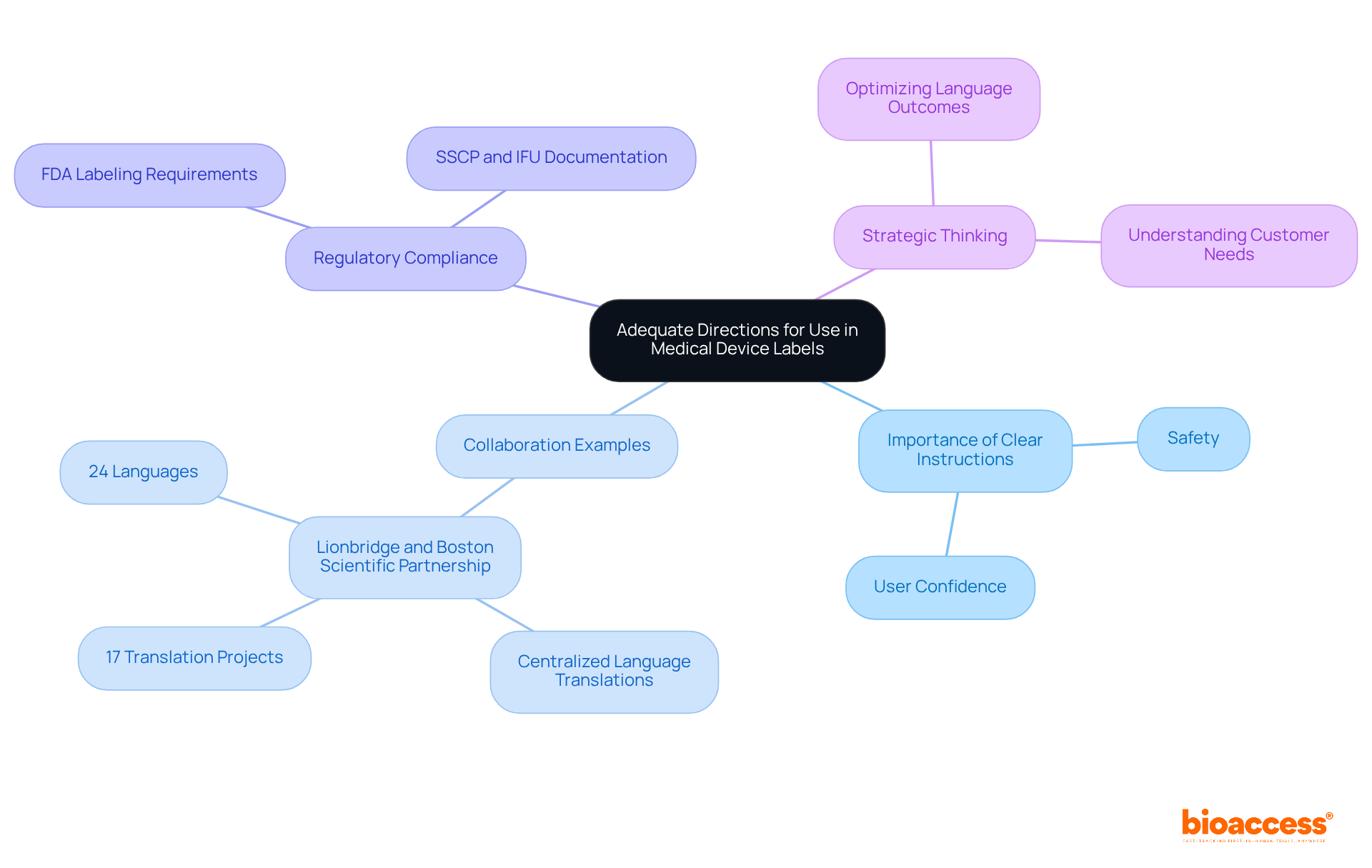
Medical equipment labels must provide , which are crucial for ensuring security and compliance with the . These instructions should encompass all necessary steps for safe operation, including preparation, usage, and maintenance. Comprehensive and understandable directions not only facilitate adherence to but also bolster user confidence and safety.
A notable example is the , which demonstrated that centralizing language translations for and Instructions for Use (IFUs) across 17 translation projects in 24 languages resulted in significant efficiencies and . This situation underscores the significance of , especially for high-risk items, where precise instructions can mitigate hazards and improve user experience.
Regulatory specialists emphasize that well-organized guidelines are essential for complying with the , thereby enhancing clarity and ensuring that users can operate equipment safely and effectively.
As Pia Windelov, VP of Life Sciences Strategy and Product Marketing, stated, ‘We’re eager to understand your needs and share how our innovative capabilities can empower you to break barriers and expand your global reach.’ This statement highlights the importance of in the realm of medical equipment identification.
Furthermore, strategic thinking regarding the SSCP and its interdependent documents is vital for ensuring that supports .
Avoiding False or Misleading Statements: Compliance in Medical Device Labeling
Labels must avoid any inaccurate or deceptive claims regarding the product’s capabilities or performance. This requirement is not only essential for but also vital for upholding the integrity of the healthcare product industry. In Colombia, the plays a crucial role in this oversight, ensuring that all health products, including , adhere to rigorous quality and effectiveness standards. Manufacturers must substantiate all claims made on labels with evidence, as misleading information can result in serious consequences for and legal repercussions. The classification of INVIMA as a Level 4 health authority by the Pan American Health Organization/World Health Organization underscores its capacity to enforce these regulations, further emphasizing the significance of precise .
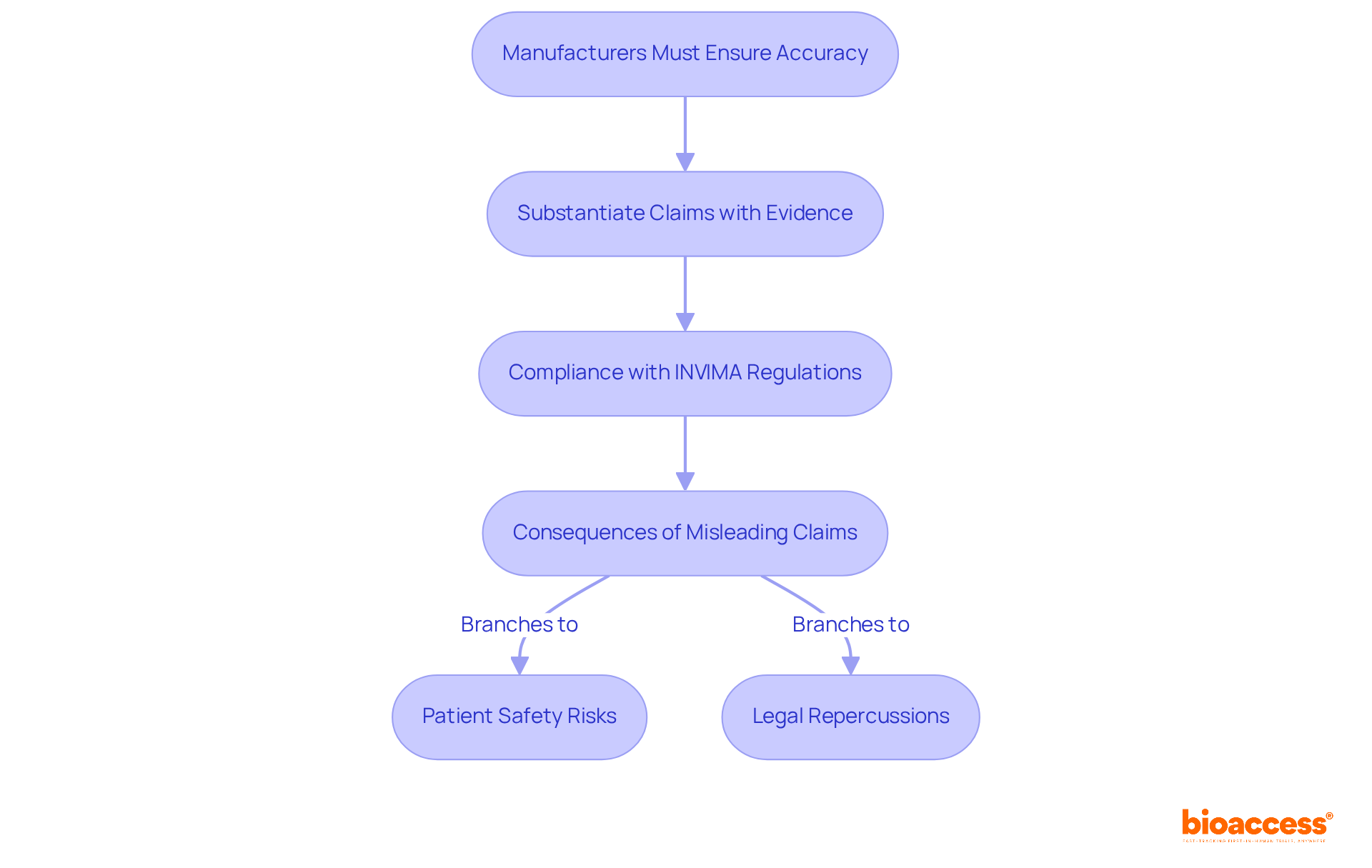
Prominence of Statements: Ensuring Clarity in Medical Device Labels
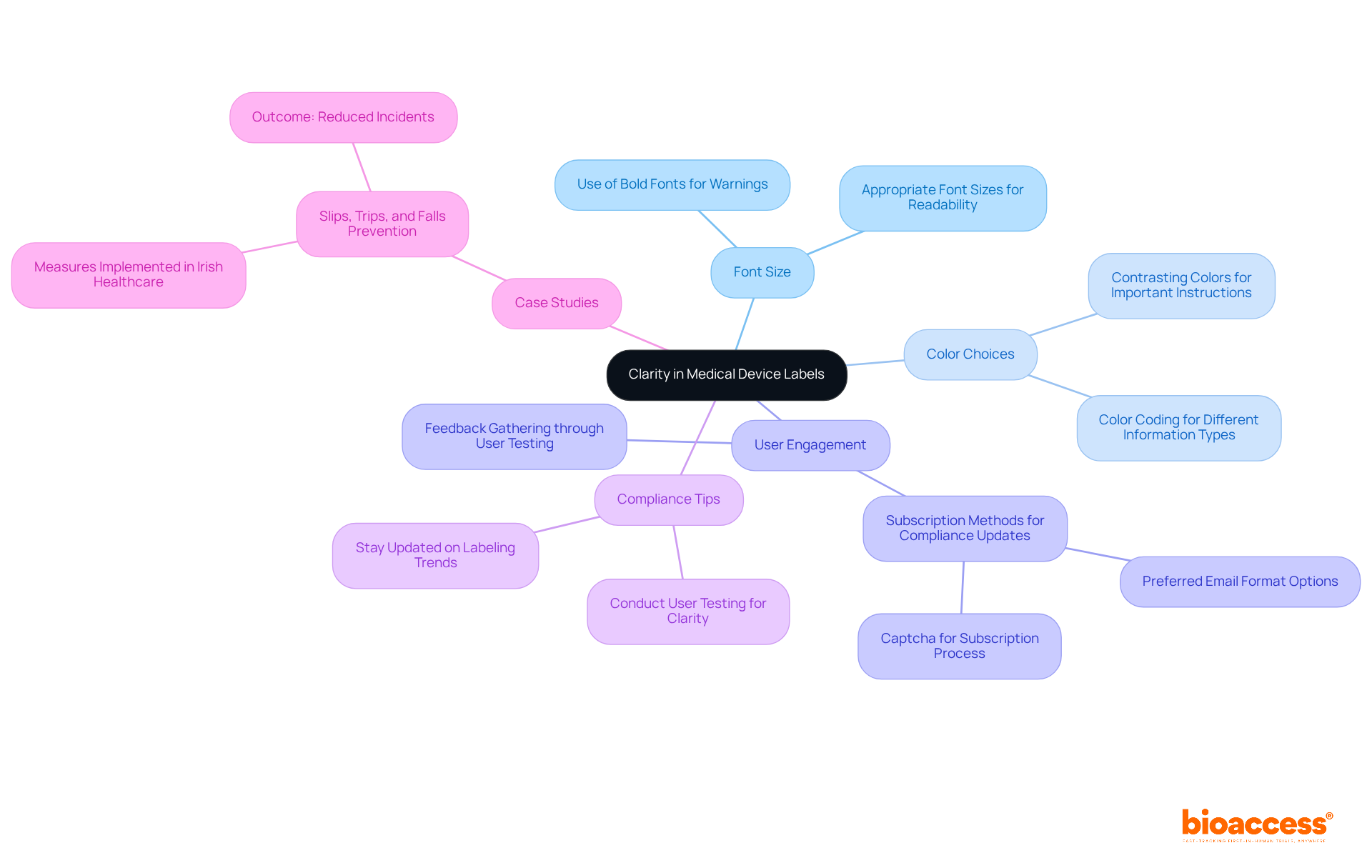
The FDA mandates that ensure critical statements on labels are presented in a manner that maximizes . This involves selecting appropriate font sizes, colors, and placements to ensure that is easily discernible. For instance, employing bold fonts for warnings and contrasting colors for important instructions can significantly enhance user awareness. By emphasizing the importance of these statements, manufacturers enable faster recognition of usage guidelines, ultimately and enhancing user protection.
Integrating user engagement tactics, such as straightforward subscription methods for updates on compliance requirements, can further improve awareness. The in illustrates how effective communication of safety information can lead to improved outcomes. Furthermore, staying updated on current trends and news regarding assists manufacturers in effectively adjusting their packaging strategies.
To ensure compliance, manufacturers should also consider actionable tips, such as conducting to gather feedback on the clarity and visibility of product information. This approach can lead to continuous improvement in practices regarding labels.
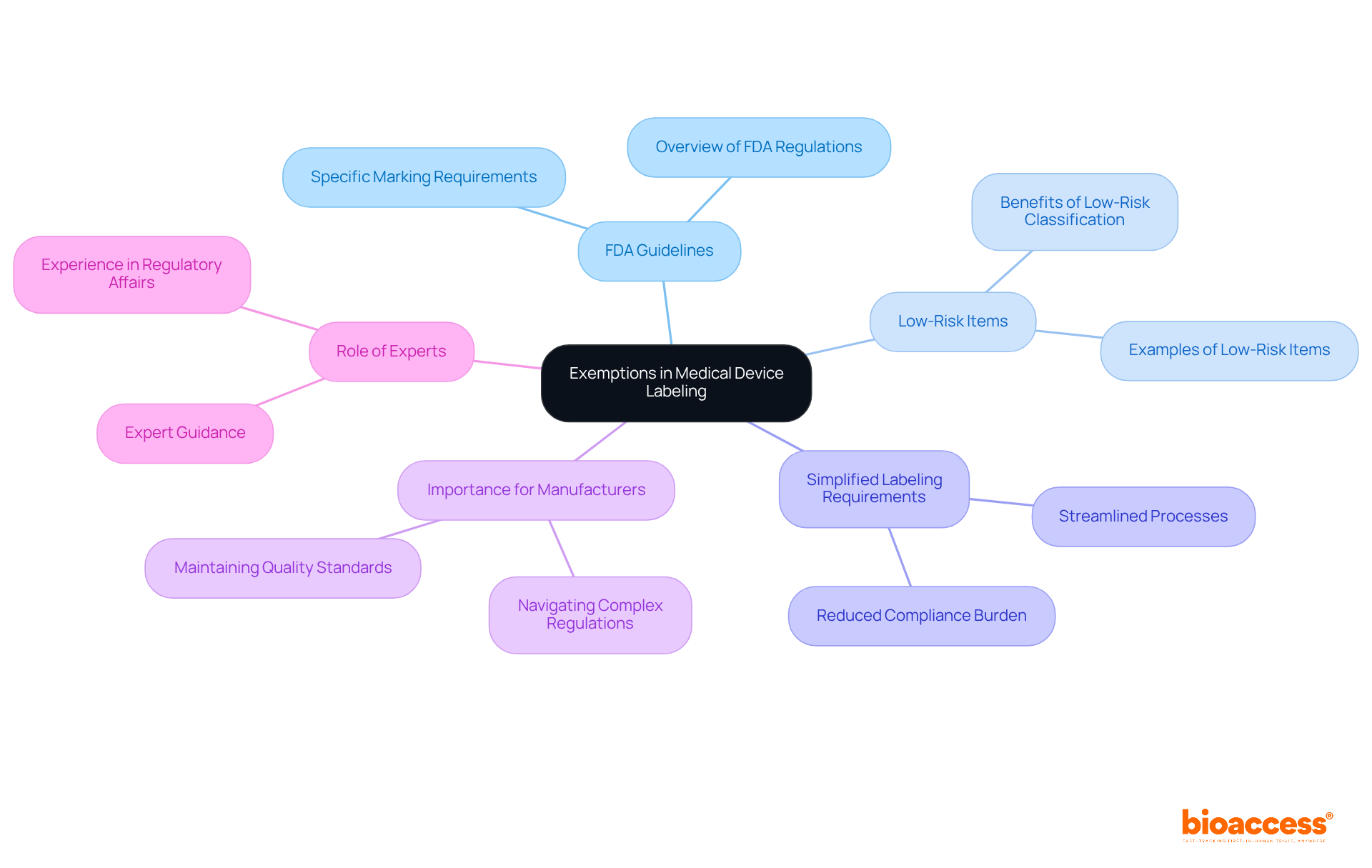
Exemptions in Medical Device Labeling: Understanding FDA Guidelines
Certain medical instruments may be exempt from specific under . For instance, items classified as low-risk often benefit from . Understanding these exemptions is crucial for manufacturers, as it helps them navigate the FDA while avoiding being overwhelmed by unnecessary regulations and still adhering to . With experts like Ana Criado, Director of at bioaccess, who possesses extensive experience in and biomedical engineering, manufacturers can effectively navigate these complexities.
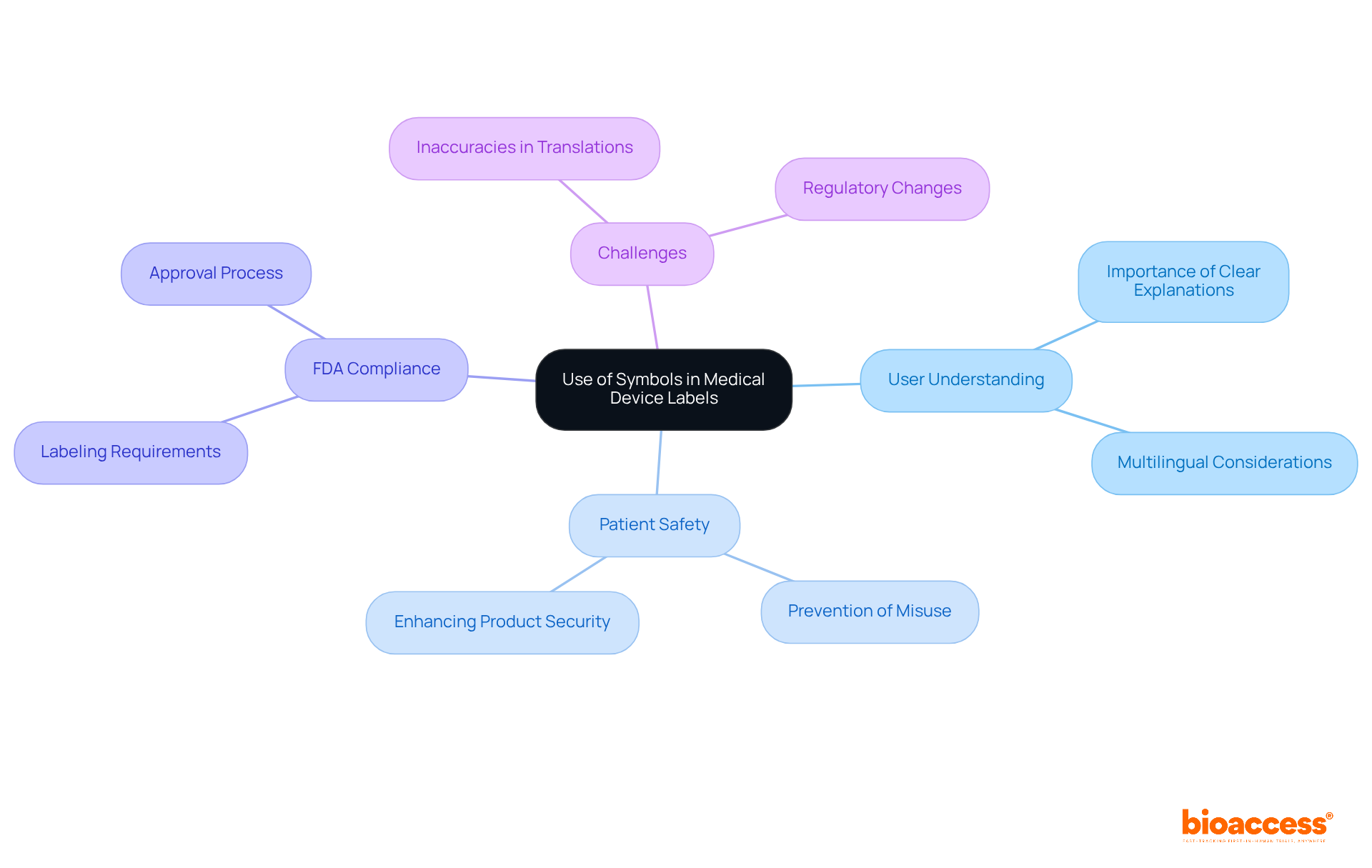
Use of Symbols: Enhancing FDA-Compliant Medical Device Labels
serve as a crucial tool for conveying essential information quickly and efficiently. The FDA advocates for their use, particularly in multilingual environments, to enhance user understanding. However, it is imperative that these symbols are accompanied by by all users.
Efficient application of symbols can greatly enhance the usability of , ultimately aiding in better patient safety and care. For instance, the integration of symbols has been shown to and caregivers, thereby preventing serious health consequences.
As the worldwide healthcare apparatus sector, valued at around 577 billion and consisting of about 2 million various types of healthcare instruments, continues to grow, the significance of becomes increasingly vital. By adopting these symbols, manufacturers can not only comply with the but also promote better understanding among users, ensuring that healthcare instruments are utilized safely and effectively.
As noted by Sarah Moore, “the primary benefit of medical device markings is that it .” Furthermore, challenges such as the requirement for labels in national languages may lead to inaccuracies in translations, posing safety risks. Therefore, is crucial for effective categorization.
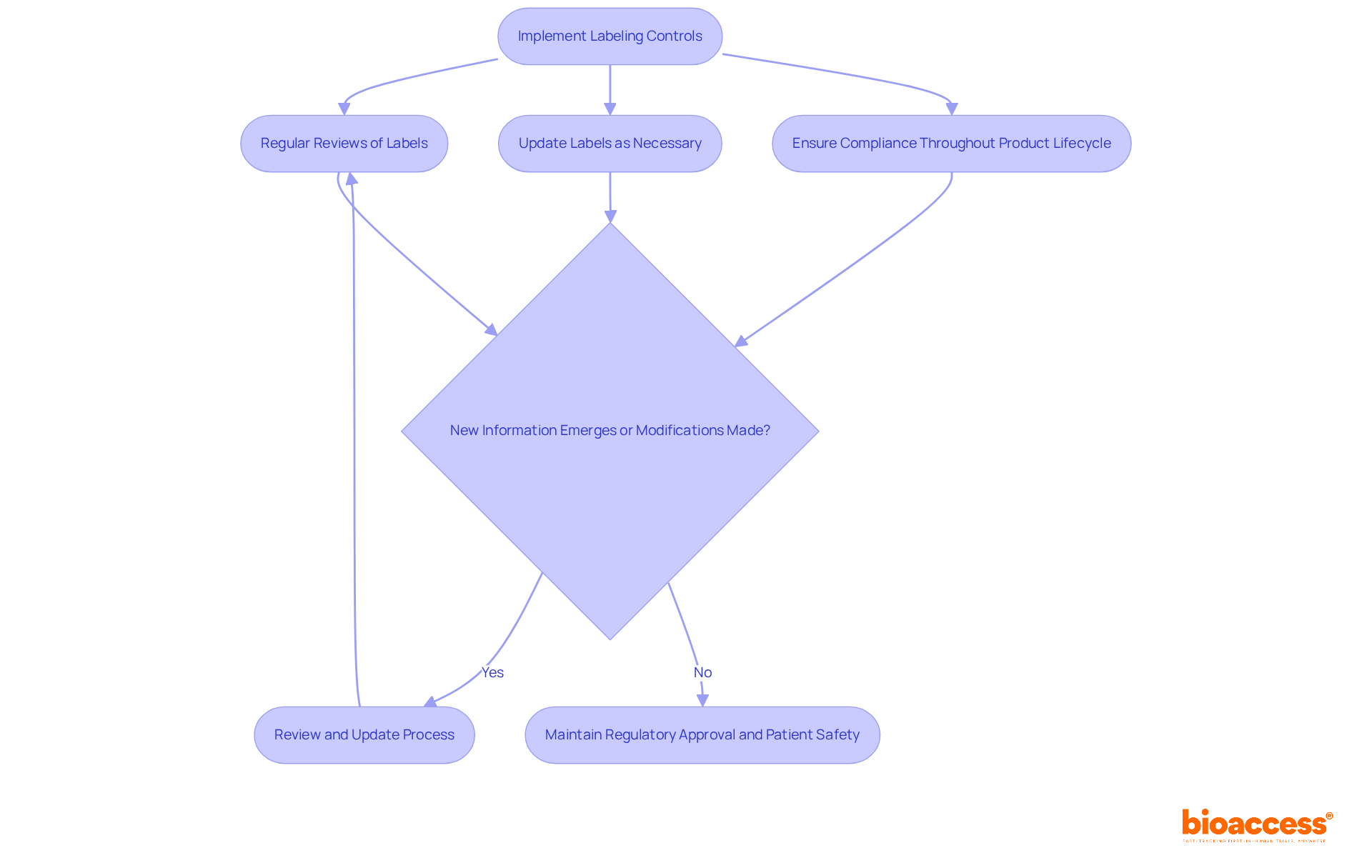
Labeling Controls Under FDA QSR: Ensuring Ongoing Compliance
Under the FDA’s , it is imperative for manufacturers to implement stringent FDA. These controls ensure that all labeling remains accurate and compliant with the throughout the . Regular reviews and updates to labels are not merely best practices; they are essential, particularly when new information emerges or modifications are made to the device. Ongoing compliance is crucial—not only for maintaining but also for safeguarding .
Conclusion
Navigating the intricate landscape of FDA medical device labeling requirements is essential for manufacturers striving to ensure compliance and promote patient safety. This article has delineated the critical elements and guidelines that must be adhered to, underscoring the necessity of precise and clear labeling practices. By grasping these requirements, manufacturers can effectively convey vital information regarding their products, thereby enhancing user comprehension and safeguarding health outcomes.
Key insights from the article emphasize the imperative of including fundamental elements such as:
- Product names
- Intended uses
- Warnings on labels
- Adherence to unique device identification standards
Furthermore, the discussion highlights the significance of providing adequate directions for use and avoiding false or misleading statements, all of which contribute to a transparent and trustworthy healthcare environment. Manufacturers are urged to implement continuous labeling controls to maintain compliance throughout the product lifecycle, ensuring that updates and changes are promptly reflected.
The importance of these labeling requirements cannot be overstated, as they not only fulfill regulatory obligations but also play a crucial role in protecting patients and healthcare providers alike. As the medical device landscape continues to evolve, remaining informed about FDA guidelines and best practices is vital. Manufacturers should prioritize compliance, leverage expert insights, and adopt innovative labeling strategies to enhance clarity and effectiveness, ultimately fostering a safer healthcare ecosystem for all.
Frequently Asked Questions
What is bioaccess® and how does it assist Medtech firms?
bioaccess® helps Medtech firms navigate FDA medical device labeling requirements by accelerating compliance and providing a deep understanding of regulatory frameworks, ensuring that clients meet necessary guidelines efficiently.
What are the benefits of using bioaccess® for medical device labeling?
bioaccess® offers global-first clinical flexibility, FDA/EMA/MDR-ready datasets, and enables faster market entry for healthcare products, including site activation in under 8 weeks and simultaneous submissions across multiple regions.
What are the key FDA guidelines for medical device labeling?
The FDA mandates that medical device labels must be accurate and non-deceptive, including the manufacturer’s name and location, as well as sufficient instructions for use, which are essential for patient safety and regulatory compliance.
What are the common compliance issues with medical device labeling?
Approximately 30% of medical devices fail to meet FDA marking standards, which can lead to compliance issues and delays in market entry.
What quality controls should manufacturers focus on to ensure compliance?
Manufacturers should concentrate on five key quality controls: label integrity, inspection of labels, storage of labels, operations related to labels, and control numbers.
What specific information must be included on FDA-compliant medical device labels?
FDA-compliant medical device labels must include the product name, intended use, indications for use, warnings or precautions, performance traits, and any applicable restrictions.
Why is accurate labeling important for medical devices?
Accurate labeling is crucial for safeguarding patients, sustaining the integrity of the healthcare system, and ensuring clear communication about the product, which enhances user comprehension and compliance with regulations.
List of Sources
- General Labeling Provisions: Key FDA Guidelines for Medical Devices
- FDA Medical Device Labeling: The Complete Guide To Compliance| Registrar Corp (https://registrarcorp.com/blog/medical-devices/medical-device-registration/medical-device-labeling)
- greenlight.guru (https://greenlight.guru/blog/medical-device-labeling)
- rqmis.com (https://rqmis.com/blog/the-critical-importance-of-accurate-labeling-on-medical-products)
- highpowervtls.com (https://highpowervtls.com/2025/04/why-medical-device-labeling-is-important)
- loftware.com (https://loftware.com/resources/blog/2024/the-importance-of-accurate-labeling-in-medical-devices)
- Adequate Directions for Use: A Must for Medical Device Labels
- lionbridge.com (https://lionbridge.com/case-study/boston-scientific-case-study)
- Prominence of Statements: Ensuring Clarity in Medical Device Labels
- hsa.ie (https://hsa.ie/eng/your_industry/health_and_social_care_sector/healthcare_case_studies)
- Use of Symbols: Enhancing FDA-Compliant Medical Device Labels
- news-medical.net (https://news-medical.net/health/Beyond-the-surface-Unraveling-the-hidden-power-of-medical-device-labeling.aspx)







. Each box represents a step in the quality control process. Follow the arrows to see how each measure contributes to ensuring the quality of the Development Safety Update Report.](https://images.tely.ai/telyai/owyncqiq-each-box-represents-a-step-in-the-quality-control-process-follow-the-arrows-to-see-how-each-measure-contributes-to-ensuring-the-quality-of-the-development-safety-update-report.webp)