Introduction
Mastering clinical trial management in Argentina is essential for researchers aiming to navigate its complex regulatory landscape effectively. This unique environment demands not only a nuanced understanding of regulations but also the implementation of effective project management strategies. By exploring best practices, researchers can uncover invaluable insights that streamline processes and significantly enhance the success rates of their studies. Yet, challenges such as recruitment hurdles and compliance complexities persist.
So, how can clinical trial teams adeptly maneuver through this intricate landscape to achieve their objectives?
Understand Regulatory Frameworks for Clinical Trials in Argentina
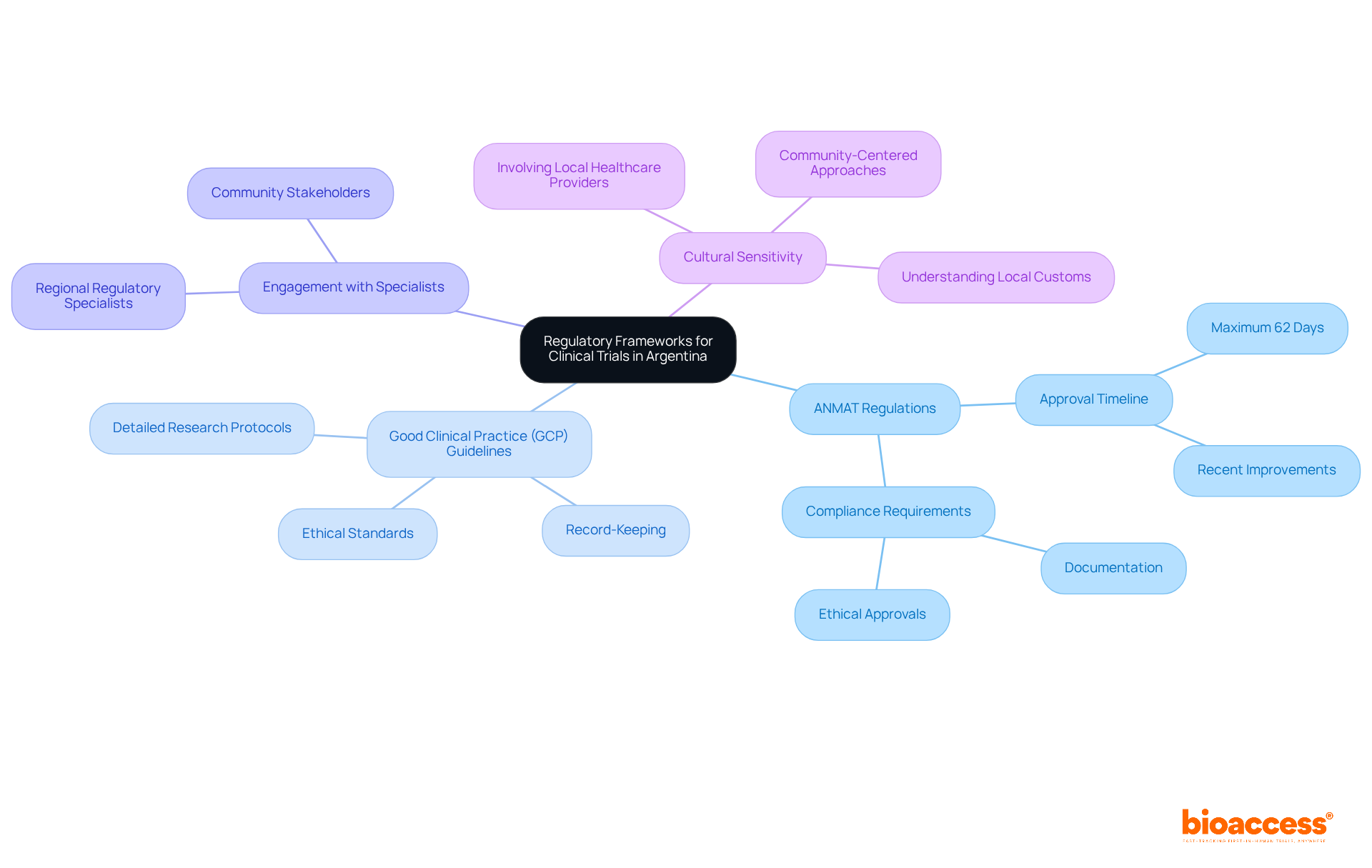
Navigating the regulatory environment in Argentina is crucial for the success of research studies. The National Administration of Drugs, Food and Medical Technology (ANMAT) has implemented significant reforms, establishing a legally binding maximum approval timeline of 62 days for clinical studies. Understanding ANMAT regulations, especially the Good Clinical Practice (GCP) guidelines, is vital. These guidelines mandate the preparation of detailed research protocols, meticulous record-keeping, and strict adherence to ethical standards.
Engaging with regional regulatory specialists can provide essential insights, facilitating smoother interactions with ANMAT. This approach not only decreases delays but also enhances study efficiency. Recent updates indicate that the average timeline for medical research approvals in Argentina has improved, showcasing the positive impact of these regulatory changes on the research landscape.
Furthermore, bioaccess’s Global Trial Accelerators™ offer critical insights into regulatory updates and market entry strategies. This includes addressing cultural sensitivity in study design, which is essential for effectively engaging the Argentine population. Ensuring that research resonates with local customs and health practices is key to successful outcomes.
Implement Effective Project Management Strategies
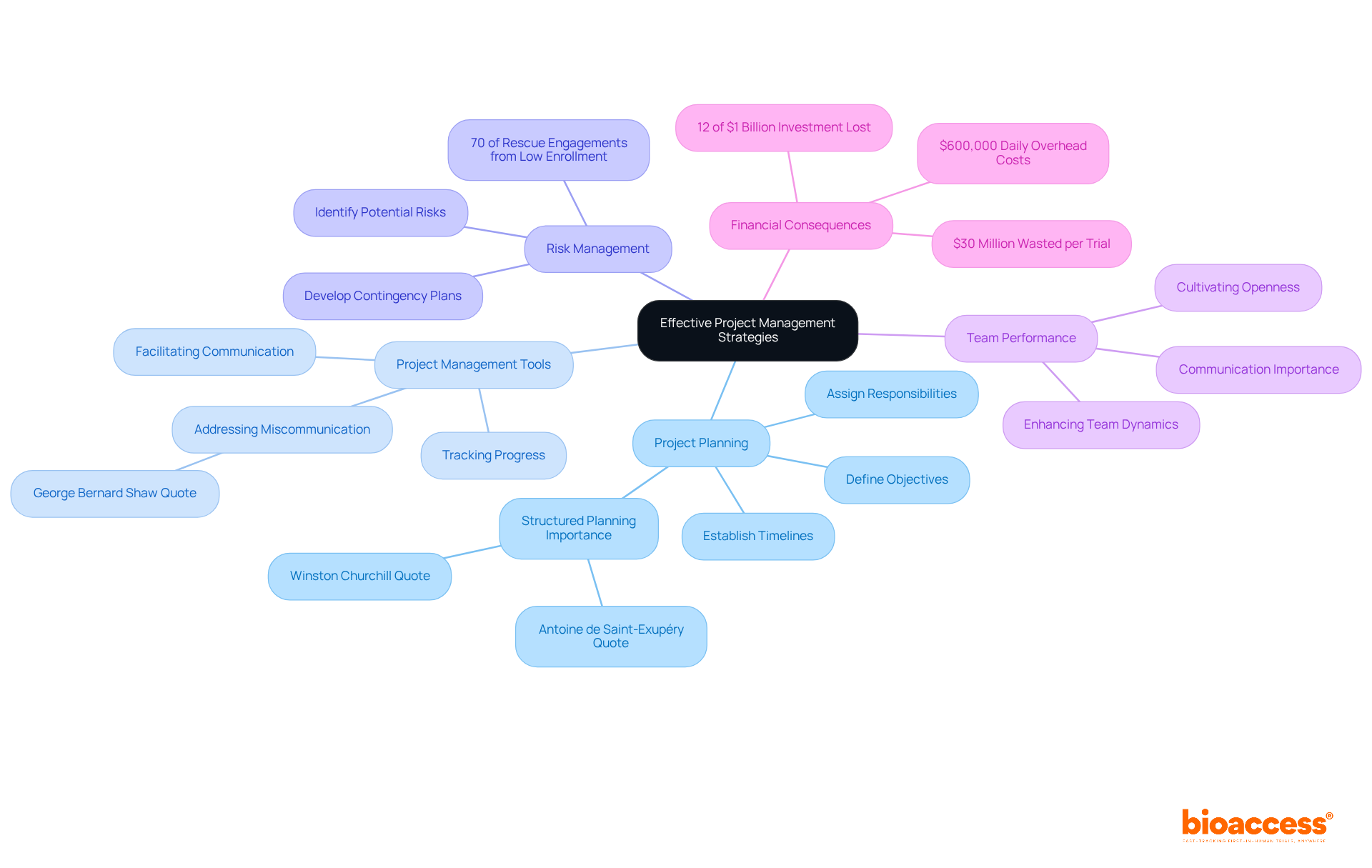
Implementing strong project management strategies is essential for the success of research studies. A clear project plan that defines objectives, timelines, and responsibilities is the foundation of effective management. As Winston Churchill wisely stated, “Let our advance worrying become advance thinking and planning,” underscoring the necessity of structured planning in achieving research goals.
Utilizing effective project management tools significantly enhances tracking progress and facilitates communication among team members. Miscommunication is a common issue in research, as highlighted by George Bernard Shaw. Regularly reviewing project milestones allows for timely adjustments to address emerging challenges. It’s alarming that nearly 90% of drug candidates fail in clinical development due to inefficiencies, making proactive management crucial.
Effective risk management is equally vital. Identifying potential risks early and developing contingency plans can mitigate issues before they escalate. With 70% of rescue engagements stemming from low enrollment, proactive planning becomes essential. Cultivating an environment of openness and teamwork can improve team performance, ensuring that the project stays on schedule and achieves its goals.
The financial consequences of inadequate project performance are substantial. An estimated $30 million is squandered per attempt due to inefficiencies, highlighting the essential need for effective project management. By embracing these strategies, research teams can enhance their chances of success and drive impactful outcomes.
Recruit and Retain Qualified Research Personnel
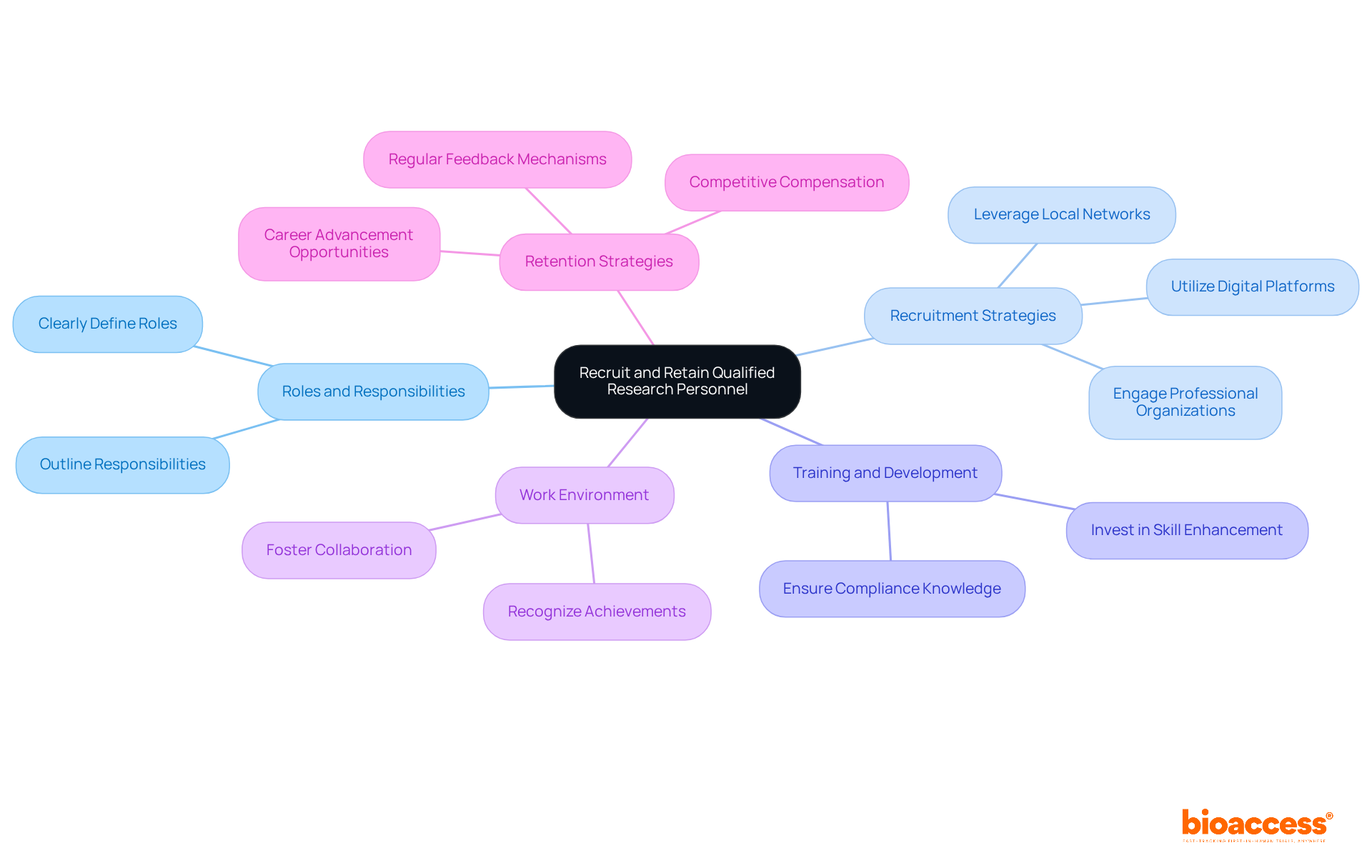
Recruiting and retaining qualified research personnel is essential for the success of clinical studies. Clearly outlining the roles and responsibilities required for your assessment is the first step. Actively seek candidates with relevant experience through local networks and professional organizations. Once you have the right team in place, invest in their training and development to enhance their skills, ensuring they are well-versed in legal procedures and compliance requirements.
Creating a positive work environment is crucial. Foster collaboration and recognize achievements to keep morale high. Implement retention strategies such as competitive compensation, career advancement opportunities, and regular feedback to maintain motivation and engagement. Research indicates that 80% of medical studies fail to meet initial enrollment expectations, highlighting the critical importance of effective recruitment methods. Additionally, 19% of studies are closed due to insufficient enrollment, underscoring the need for proactive measures.
As Tiffany Ashton, Director of Operations and Clinical Trial Manager, aptly states, “Recruitment and retention are a team effort-and when we work together toward a common goal, we are more likely to achieve success.” By focusing on these strategies, you can enhance your team’s capabilities and contribute significantly to the success of your research studies.
Leverage Local Patient Populations for Faster Recruitment
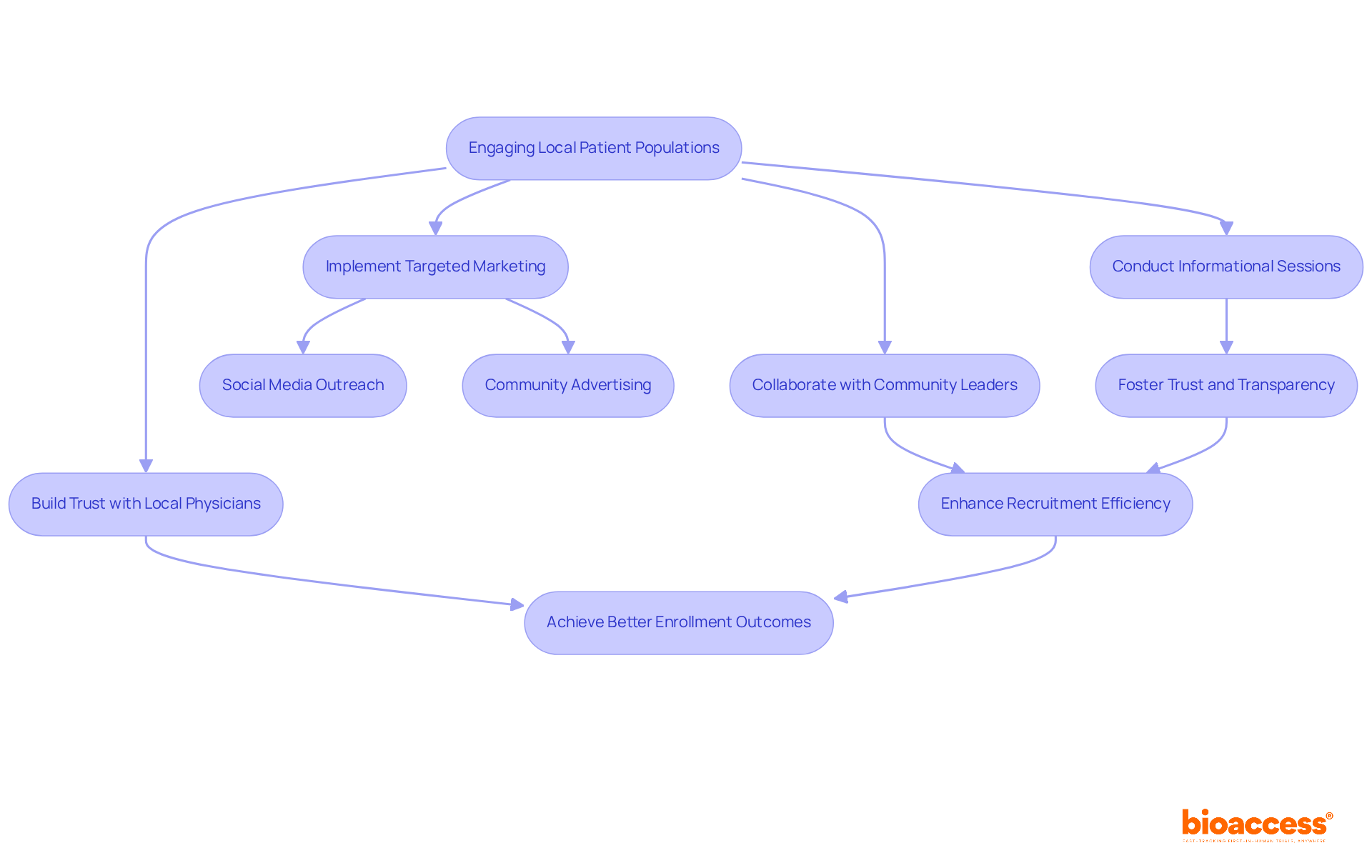
Engaging local patient populations is essential for enhancing recruitment in clinical studies. By collaborating with community leaders and healthcare providers, awareness about the study and its potential benefits can significantly increase. Informational sessions allow potential participants to grasp the process and voice any concerns, fostering trust and transparency.
Targeted marketing strategies, such as social media outreach and community advertising, are crucial for reaching diverse patient populations. Research shows that nearly half of potential participants discover clinical studies through media and the internet, underscoring the necessity for a robust online presence.
Building trust within the community is vital; involving local physicians as advocates can bolster credibility and encourage participation. Effective community engagement strategies not only accelerate recruitment timelines but also enhance the overall quality of study data. By cultivating strong relationships with local communities, studies can achieve better enrollment outcomes and ultimately support more successful research initiatives.
Furthermore, with bioaccess’s approach, clinical studies can realize $25K savings per patient, achieve 50% faster enrollment through pre-qualified networks, and enjoy a 30% reduction in overall study costs. Given that over 60% of sites fail to meet their original patient enrollment goals and nearly 80% of patient recruitment timelines are unmet, these strategies are increasingly critical.
Conclusion
Mastering clinical trial management in Argentina is essential for advancing medical research and improving patient care. It demands a thorough understanding of the regulatory landscape, effective project management strategies, and a commitment to recruiting and retaining qualified personnel. By integrating these critical elements, research teams can adeptly navigate the complexities of clinical studies, ensuring compliance and operational efficiency.
Key insights throughout this discussion underscore the necessity of familiarizing oneself with ANMAT regulations, implementing structured project management practices, and leveraging local patient populations for recruitment. Engaging regulatory specialists, utilizing effective management tools, and fostering a positive work environment are pivotal steps that collectively enhance the likelihood of successful outcomes in clinical trials.
In summary, the path to successful clinical trial management in Argentina transcends mere regulatory adherence; it involves building robust teams and actively engaging with local communities. By adopting these best practices, research teams can significantly bolster their chances of success, ultimately contributing to advancements in medical science and patient care. Embracing these strategies today can pave the way for a more efficient and impactful clinical research landscape in Argentina.
Frequently Asked Questions
What is the role of ANMAT in clinical trials in Argentina?
The National Administration of Drugs, Food and Medical Technology (ANMAT) is responsible for regulating clinical trials in Argentina, ensuring compliance with established guidelines and standards.
What is the maximum approval timeline for clinical studies set by ANMAT?
ANMAT has established a legally binding maximum approval timeline of 62 days for clinical studies.
Why is understanding Good Clinical Practice (GCP) guidelines important?
Understanding GCP guidelines is vital as they mandate the preparation of detailed research protocols, meticulous record-keeping, and strict adherence to ethical standards in clinical trials.
How can engaging with regional regulatory specialists benefit clinical trials?
Engaging with regional regulatory specialists can provide essential insights, facilitate smoother interactions with ANMAT, decrease delays, and enhance study efficiency.
What recent trends have been observed in the approval timelines for medical research in Argentina?
Recent updates indicate that the average timeline for medical research approvals in Argentina has improved, reflecting the positive impact of regulatory changes on the research landscape.
What resources does bioaccess provide for navigating regulatory frameworks?
Bioaccess’s Global Trial Accelerators™ offer critical insights into regulatory updates and market entry strategies, aiding in the navigation of the regulatory environment for clinical trials.
Why is cultural sensitivity important in study design for clinical trials in Argentina?
Cultural sensitivity is essential for effectively engaging the Argentine population, ensuring that research resonates with local customs and health practices, which is key to successful outcomes.
List of Sources
- Understand Regulatory Frameworks for Clinical Trials in Argentina
- ANMAT updates clinical research regulation in Argentina | Intrials Clinical Research posted on the topic | LinkedIn (https://linkedin.com/posts/intrials-clinical-research_intrials-activity-7386780565036765185-0lEy)
- Challenges In Designing Trials For Argentina K… | bioaccess® (https://bioaccessla.com/blog/challenges-in-designing-trials-for-argentina-key-considerations)
- Executive Forecast – Conversation: Argentina’s Clinical Trial Renaissance: Interview with Mariel Peitiado – President, Cámara Argentina de Organizaciones de Investigación Clínica (CAOIC), Argentina (https://executiveforecast.com/conversation/argentinas-clinical-trial-renaissance-interview-with-mariel-peitiado—president-camara-argentina-de-organizaciones-de-investigacion-clinica-caoic-argentina)
- Argentina is entering a new regulatory era — and ANMAT already has the blueprint (https://linkedin.com/pulse/argentina-entering-new-regulatory-era-anmat-already-schiavone-md-n5izf)
- Implement Effective Project Management Strategies
- The Role of Project Management in Clinical Research Success (https://xceneresearch.com/project-management-in-clinical-research)
- 35 Best Project Management Quotes (https://projectmanager.com/blog/10-best-project-management-quotes)
- toggl.com (https://toggl.com/blog/project-management-quotes)
- The 62 Most Inspiring Project Management Quotes (https://inloox.com/company/blog/articles/the-62-most-inspiring-project-management-quotes)
- Recruit and Retain Qualified Research Personnel
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- 2025 Trends In Patient Recruitment: From Disruption To Precision (https://clinicalleader.com/doc/trends-in-patient-recruitment-from-disruption-to-precision-0001)
- A Primer on the Importance of Recruitment and Retention in Clinical Trials – ACRP (https://acrpnet.org/2023/04/18/a-primer-on-the-importance-of-recruitment-and-retention-in-clinical-trials)
- Recruitment and retention of participants in randomised controlled trials: a review of trials funded and published by the United Kingdom Health Technology Assessment Programme (https://bmjopen.bmj.com/content/7/3/e015276)
- autocruitment.com (https://autocruitment.com/blogs/current-challenges-in-clinical-trial-patient-recruitment-and-enrollment)
- Leverage Local Patient Populations for Faster Recruitment
- appliedclinicaltrialsonline.com (https://appliedclinicaltrialsonline.com/view/enrollment-performance-weighing-facts)
- Patient Recruitment for Clinical Trials | Veristat (https://veristat.com/patient-recruitment-for-clinical-trials)
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- Evaluation of factors associated with recruitment rates in early phase clinical trials based on the European Clinical Trials Register data (https://ascpt.onlinelibrary.wiley.com/doi/10.1111/cts.13659)
- 2025 Trends In Patient Recruitment: From Disruption To Precision (https://clinicalleader.com/doc/trends-in-patient-recruitment-from-disruption-to-precision-0001)

Leave a Reply