Introduction
Investigational Device Exemptions (IDEs) are a cornerstone in the realm of clinical trials, enabling sponsors to legally ship investigational devices for clinical studies. This mechanism is crucial for generating the data needed to demonstrate the safety and effectiveness of medical devices, aligning with the FDA’s mission to protect public health. IDEs ensure that investigational devices undergo thorough regulatory scrutiny before market entry, fostering the development of innovative medical products.
Clinical research, underpinned by well-structured studies, is vital for advancing public health. The FDA’s commitment to harmonizing human subject protection regulations with the Department of Health and Human Services’ Common Rule exemplifies efforts to streamline clinical research while safeguarding participants’ rights. This regulatory alignment is essential for investigators and sponsors managing IDEs amidst the complexities of global medical device development.
The significant number of injuries and deaths linked to medical devices underscores the necessity of robust regulatory oversight. Adhering to IDE requirements not only enhances the development of safer medical devices but also improves patient outcomes and advances medical knowledge.
Understanding Investigational Device Exemptions (IDEs)
play a crucial role in the environment of trials, permitting sponsors to legally transport investigational devices for use in experiments. This mechanism is indispensable as it enables the rigorous collection of data necessary to substantiate the of . As part of the FDA’s broader mandate to protect , Ideas facilitate the development of innovative medical products by ensuring that investigational devices are subjected to stringent before reaching the market.
Effective, well-structured medical research is essential for enhancing , and the FDA actively encourages the creation of trustworthy evidence through these investigations. As one FDA report indicates, the agency is committed to harmonizing human subject protection regulations with the Department of Health and Human Services’ Common Rule, aiming to streamline while safeguarding participant rights. This harmonization effort underscores the importance of compliance with FDA regulations, particularly for investigators and sponsors navigating the complexities of IDEs.
The global nature of the medical device industry presents several challenges, including ensuring compliance with varied regulatory standards and addressing safety concerns during product development. Over a ten-year period, more than 1.7 million injuries and 83,000 deaths in the United States were potentially linked to , highlighting the . By adhering to IDE requirements, sponsors can contribute to the development of safer and more effective , ultimately enhancing patient outcomes and advancing medical knowledge.

Types of Device Studies Under 21 CFR Part 812
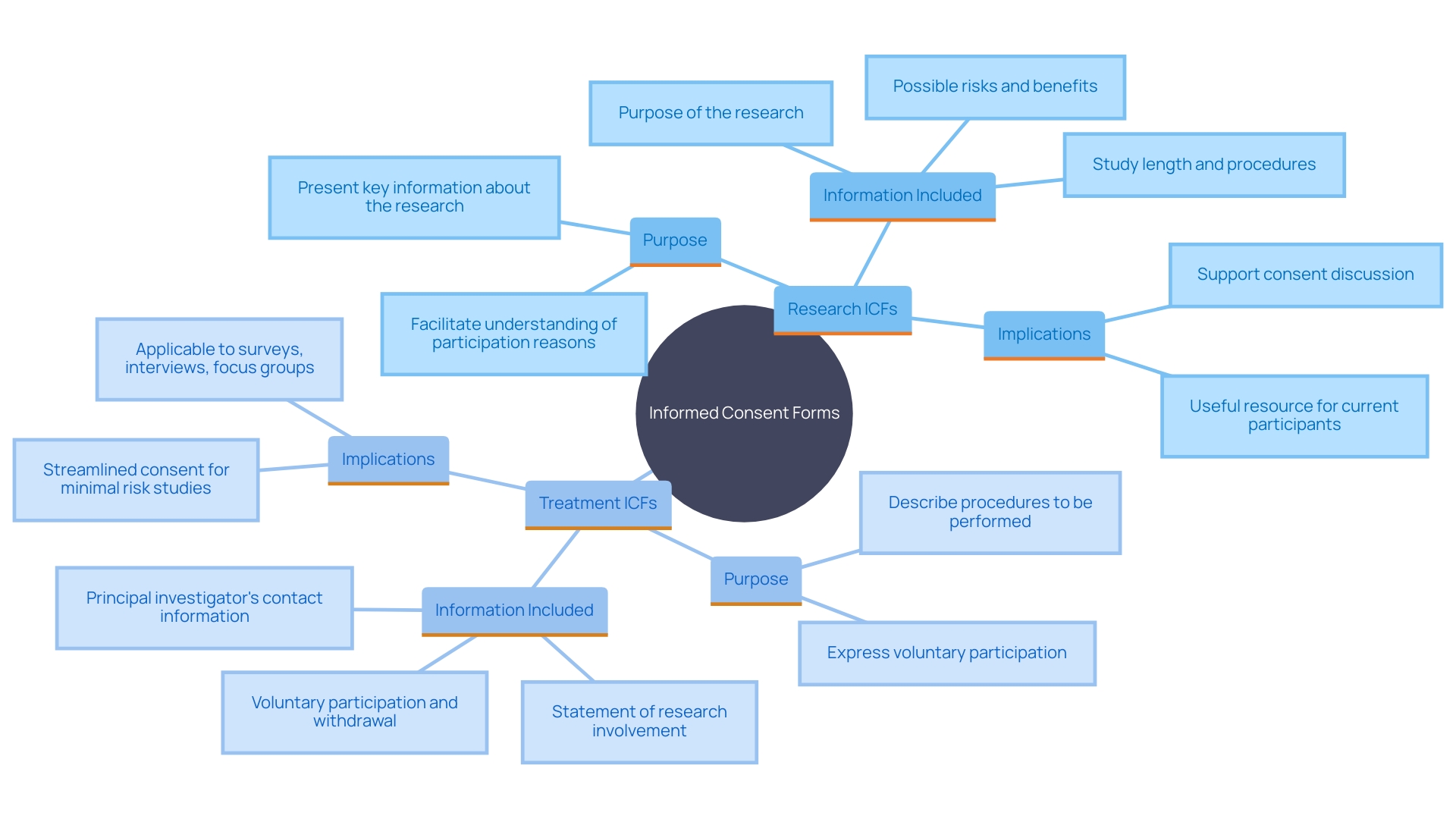
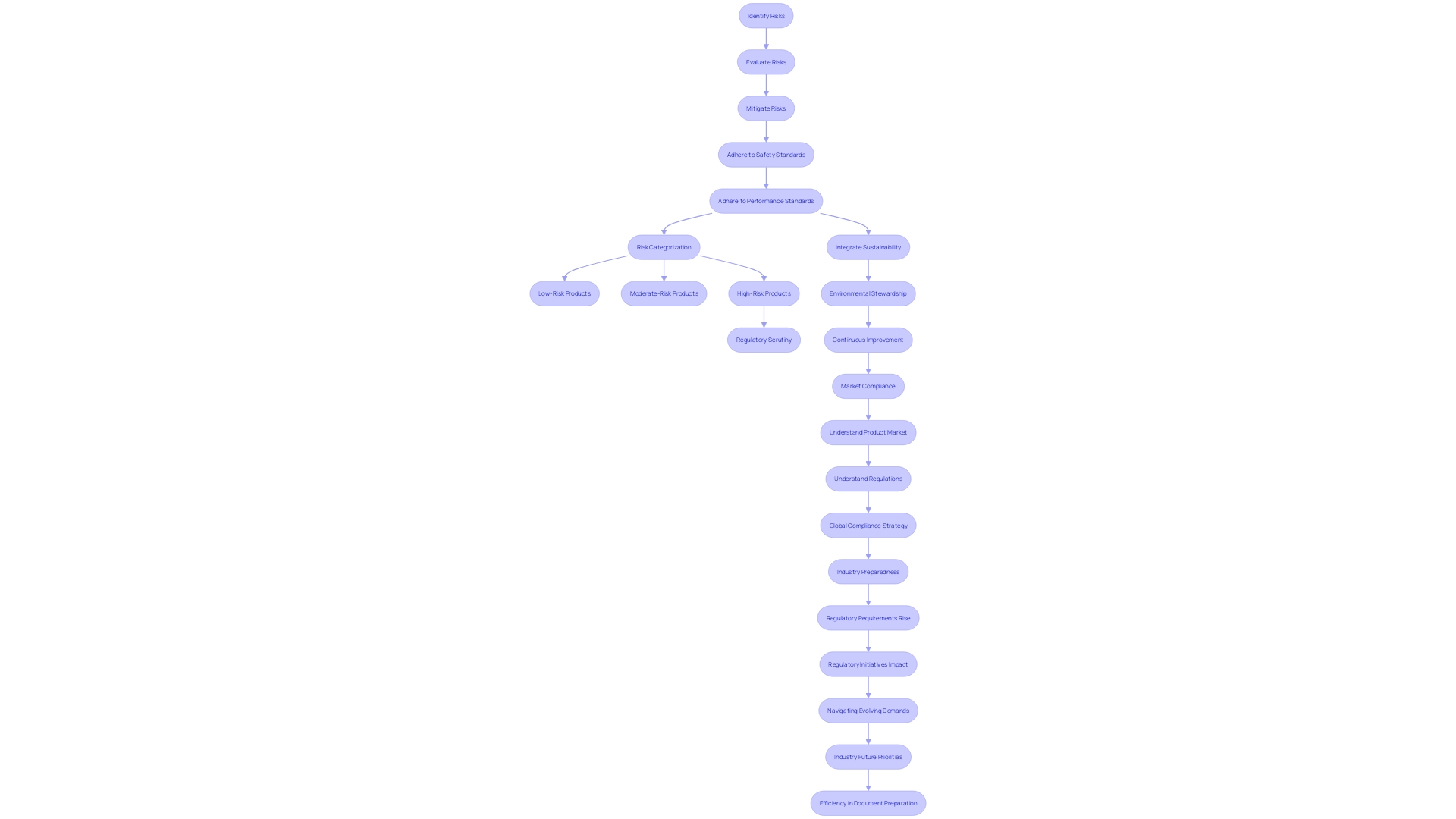
21 CFR Part 812 outlines various categories for , specifically exempt evaluations, , and . Each classification has distinct and oversight. SR examinations are subject to more rigorous scrutiny due to their potential to present a serious risk to the health, safety, or welfare of a subject. In contrast, NSR studies, while still regulated, are considered to pose less risk. Understanding these differences is essential for ensuring and procedures, thus protecting the integrity and .
IDE Application and Approval Process
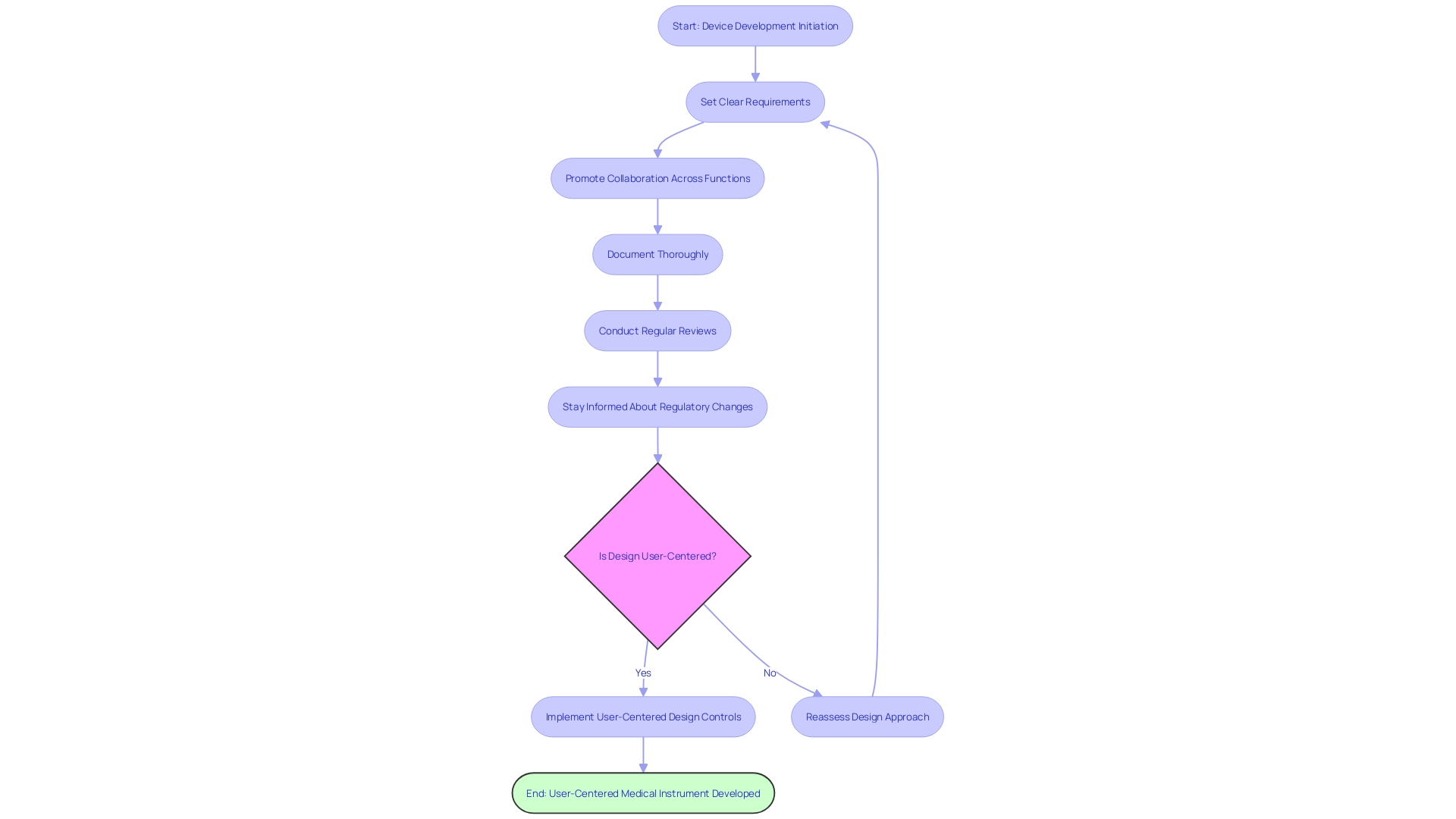
The is a critical step in initiating . This procedure necessitates submitting a comprehensive plan that details the aims, methodology, and adherence to regulatory standards. The FDA meticulously reviews this application to ensure that the proposed research aligns with the required . Familiarity with the is paramount, particularly concerning the . An IDE allows the utilization of non-cleared devices in trials to collect crucial safety and effectiveness information. Overcoming challenges in the IDE submission, such as , is vital for securing . This process not only facilitates pre-market research but also contributes significantly to the development and evaluation of medical devices’ post-market performance.
Institutional Review Board (IRB) Approval and Oversight
are crucial ethics committees tasked with the protection of participants’ rights and welfare in . These boards meticulously review , documents, and ongoing adherence to ethical standards. The significance of IRBs can’t be overstated as they ensure that are treated with respect and care throughout the research process.
IRBs were officially established under the , a response to unethical research practices, such as the infamous Tuskegee Syphilis Study. Today, approximately 2,300 IRBs exist in the U.S., overseeing both academic and commercial research. Their primary role is to evaluate the purpose of the research, procedures, risks and benefits, and consent forms, ensuring compliance with federal regulations and institutional criteria.
is a fundamental step in conducting ethical medical research. ‘Without it, research cannot legally continue, underscoring the board’s critical function in safeguarding public health and maintaining trust in medical research.’.

Participant Safety and Informed Consent
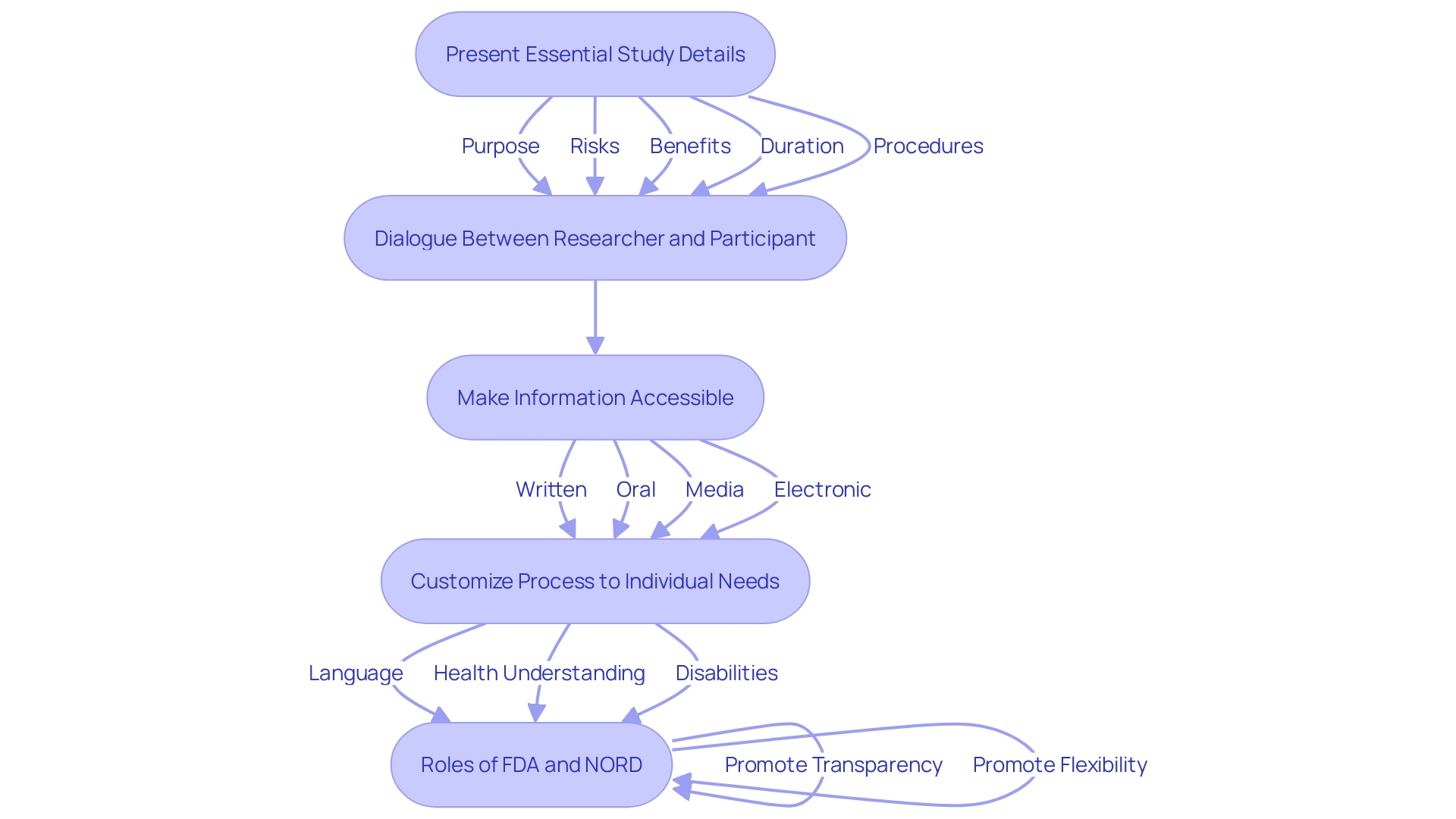
Ensuring is a cornerstone of . The is designed to provide potential participants with comprehensive information about the study, including risks, benefits, and the . Recent guidance emphasizes the importance of presenting key information in a clear and concise manner at the beginning of the consent document. This includes the purpose of the research, expected duration, procedures, and compensation for research-related injuries.
The National Organization for Rare Disorders (NORD) has praised efforts to make informed consent more accessible, highlighting the need to address language barriers, sensory impairments, and health literacy levels. Innovative approaches, such as videos, have been recommended to tailor the process to individual needs, ensuring participants fully understand the risks and benefits.
often involve participants who may not directly benefit from the research but contribute to future advancements. For example, elderly patients with transthyretin-mediated amyloidosis engage in studies hoping to assist future generations. Despite the personal toll, including invasive tests and new side effects, their motivation often stems from a desire to improve outcomes for others.
Addressing the complexity of is crucial, as they have become increasingly burdensome. Originally intended to help potential participants in making educated choices, these papers now frequently surpass twenty pages and are composed at an advanced reading level, creating barriers to enrollment, particularly among marginalized groups.
The draft guidance encourages the use of key information as a guide to support discussions between investigators and potential participants. This approach aims to facilitate comprehension and ensure , building trust between researchers and participants.
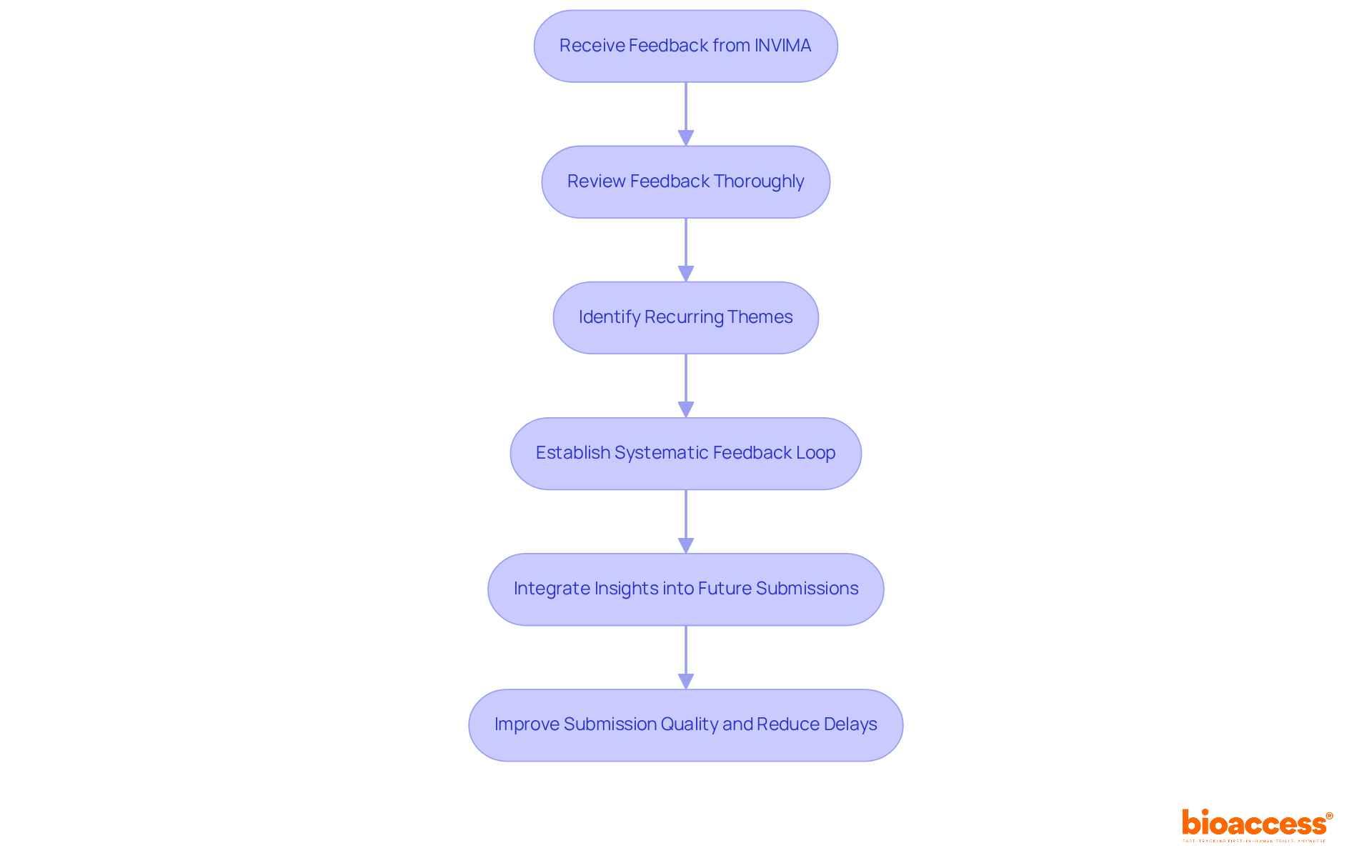
Record Keeping and Reporting Requirements
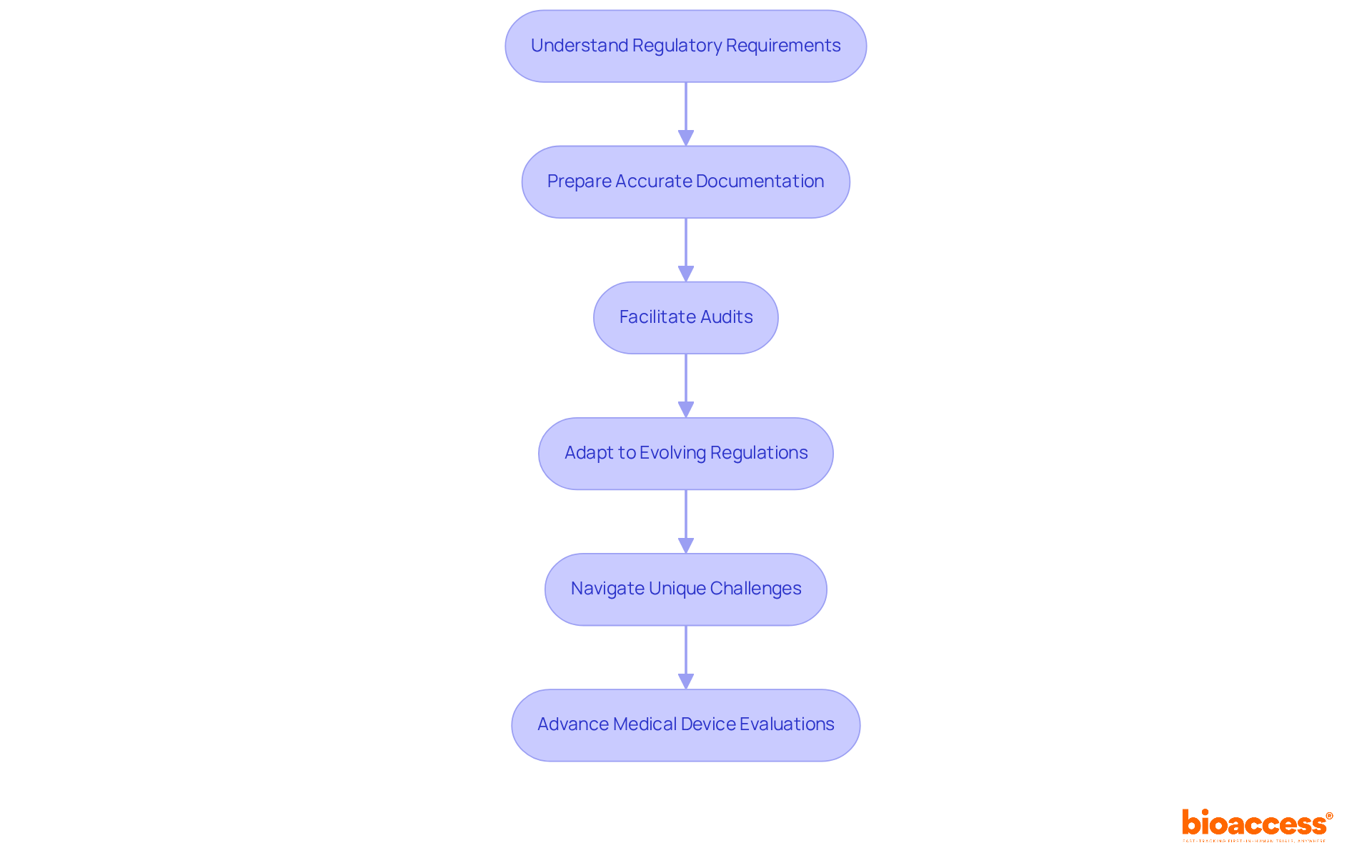
Maintaining accurate records and adhering to are critical components of compliance with . Sponsors must keep detailed documentation of all study-related activities, including and . This guarantees the integrity of by offering a thorough account of the research’s execution and any problems faced. Regular reporting to the is essential for transparency and accountability. During the last 25 years, the criteria for information components in research documentation have developed, mirroring alterations in study reporting regulations and guidelines. Consequently, older records on platforms like ClinicalTrials. Gov may lack information that has since become mandatory. Modernized reporting systems now require extensive information submissions to ensure thorough oversight. To further enhance public health protection, regulatory agencies like the FDA focus their compliance and risk management processes on high-impact areas. This approach is designed to maintain flexibility, enabling rapid and effective responses to emerging public health threats.
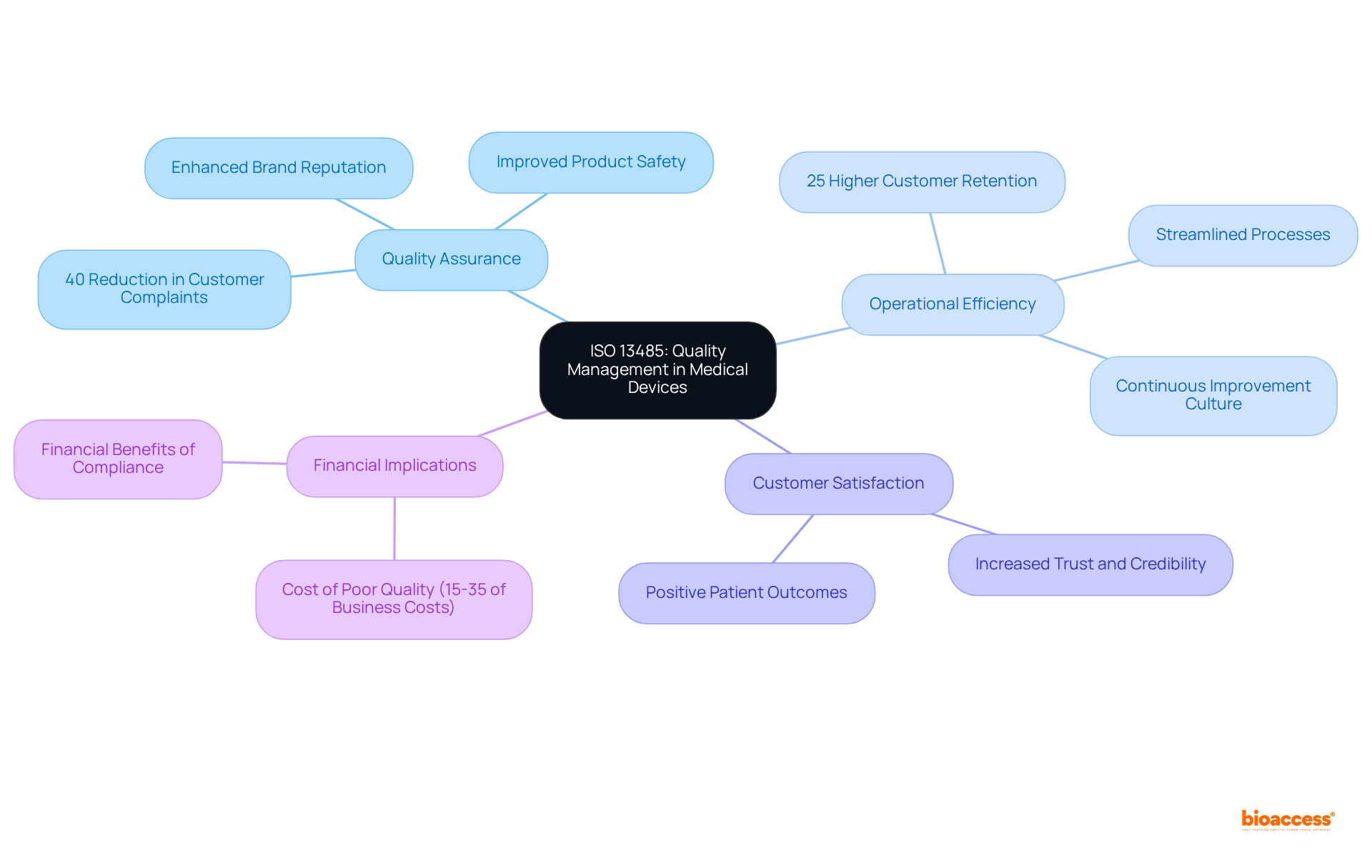
Quality Assurance and Quality Control Measures
Implementing measures is indispensable for maintaining the integrity of . These measures ensure that studies comply with and that the information gathered is both reliable and valid. The World Health Organization (WHO) estimates there are two million different kinds of globally, each potentially impacting millions of lives. is crucial in this context since it involves human subjects and devices that may be widely used. Article 62 of the European Union Medical Device Regulation (EU MDR) emphasizes that must prioritize the rights, safety, dignity, and well-being of participants while ensuring that the information generated is scientifically valid, reliable, and robust.
often utilize time- and labor-intensive methods for information collection, creating burdens on clinicians and patients. This is further complicated by the absence of simultaneous information from other sources, which could provide significant insights into a participant’s health. For instance, information from daily activities or thorough patient-reported details is often absent, impacting the validity and generalizability of the findings.
Public and private investments over the past two decades have significantly advanced electronic health record (EHR) adoption, health information interoperability, and information standards. These advancements offer a strong basis for enhancing healthcare information management practices. However, there is still an urgent need to build on this infrastructure to support reusable research trial capabilities. The FDA also encourages the use of automated processes for information validation, steering away from manual methods that are prone to errors.
Outstanding management of medical information is essential for business to, establishing the foundation of submissions to regulatory authorities. In the US, approximately 10-15% of successful 510(k) submissions for Class II devices depend on study data, and all Class III devices necessitate extensive evaluations to establish safety and effectiveness. Thus, comprehending and implementing strong QA and QC practices are essential for the overall success of research and the following approval and marketability of .
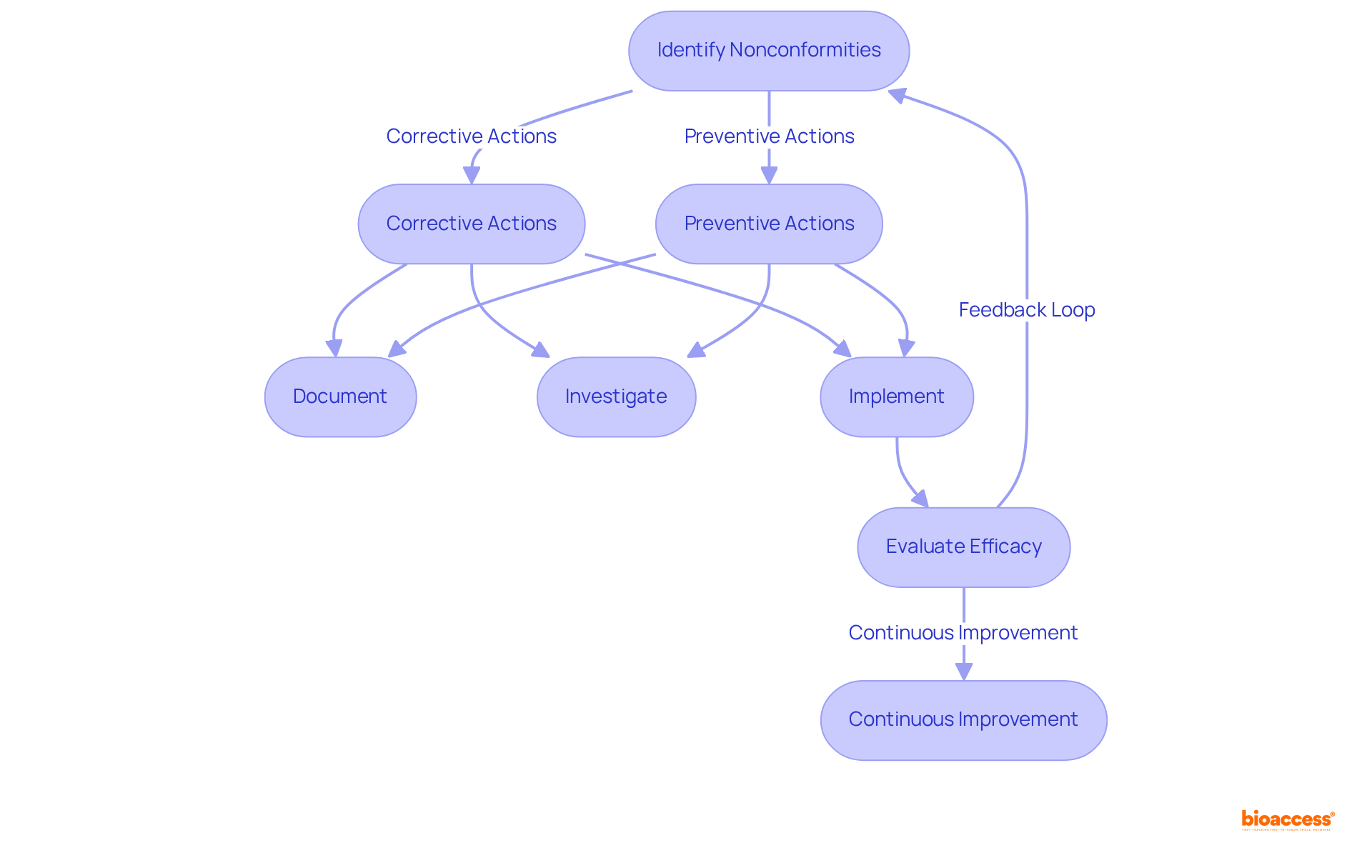
 in clinical trials, highlighting their importance in ensuring reliable and valid research outcomes. This mind map illustrates the interconnected concepts surrounding quality assurance (QA) and quality control (QC) in clinical trials, highlighting their importance in ensuring reliable and valid research outcomes.](https://tely.blob.core.windows.net/telyai/this-mind-map-illustrates-the-interconnected-concepts-surrounding-quality-assurance-qa-and-quality-control-qc-in-clinical-trials-highlighting-their-importance-in-ensuring-reliable-and-valid-research-outcomes.jpg)
Monitoring and Compliance with 21 CFR Part 812
Ensuring adherence to requires ongoing supervision of research studies. This involves a multitude of activities such as regular audits, inspections, and reviews of study processes and data. Effective are crucial in identifying potential issues early, thereby allowing for timely interventions and maintaining the integrity of the clinical study. ‘ are pivotal in testing and establishing the safety and efficacy of new treatments or interventions, making stringent monitoring indispensable.’.
ClinicalTrials.gov functions as an essential archive for summarizing research protocols and results information, which must be regularly updated by sponsors and investigators. Throughout the last 25 years, the criteria for information components on this platform have developed, mirroring alterations in research reporting regulations and guidelines. Given the importance of accurate and comprehensive data, ongoing monitoring ensures that study records are consistent with current standards.
Furthermore, the in protecting public health by assuring the safety, effectiveness, and security of medical products. This encompasses supervising research studies and making certain they comply with set standards. For example, the FDA’s recent release of the “Direct-to-Consumer Prescription Drug Advertisements” final rule highlights the agency’s dedication to clear and transparent communication of drug information, which is essential to the public’s trust in medical research.
Monitoring also involves the use of , which provide an additional layer of oversight. These committees are essential in assessing the safety and efficacy data during a study, making recommendations on whether to continue, modify, or terminate the research based on interim findings. This guidance is part of the FDA’s broader efforts to enhance regulatory processes, foster innovation, and advance public health protection.
Ultimately, the role of effective monitoring cannot be overstated. It ensures that clinical trials are conducted to the highest standards, and the well-being of participants.
Conclusion
The exploration of Investigational Device Exemptions (IDEs) highlights their critical function in the landscape of clinical trials. These exemptions not only allow sponsors to legally ship investigational devices but also ensure that thorough data collection occurs to validate the safety and effectiveness of medical devices. The FDA’s commitment to regulatory oversight is paramount, particularly in light of significant concerns regarding medical device-related injuries and fatalities.
By adhering to IDE requirements, sponsors can enhance patient outcomes and contribute to medical knowledge, fostering the development of safer medical products.
Understanding the different categories of device studies under 21 CFR Part 812, as well as the IDE application and approval process, is essential for compliance and the integrity of clinical trials. The role of Institutional Review Boards (IRBs) in safeguarding participant rights cannot be overstated, as they ensure ethical standards are upheld throughout research. Furthermore, the informed consent process must be transparent and accessible, addressing the diverse needs of potential participants to facilitate comprehension and trust.
Record keeping, reporting requirements, and the implementation of quality assurance and quality control measures are vital for maintaining the integrity of clinical trials. Continuous monitoring and compliance with regulatory standards ensure that clinical research adheres to established guidelines, ultimately safeguarding public health. By focusing on these aspects, the medical device industry can not only navigate the complexities of clinical trials but also contribute to the advancement of innovative and effective medical solutions.
Frequently Asked Questions
What are Investigational Device Exemptions (IDEs)?
IDEs are regulatory mechanisms that allow sponsors to legally transport investigational medical devices for use in clinical trials. They are essential for gathering data to demonstrate the safety and effectiveness of these devices.
Why are IDEs important for public health?
IDEs enable the collection of rigorous data necessary for evaluating medical devices, ensuring they undergo strict regulatory scrutiny before market release. This process helps protect public health.
How does the FDA support IDEs?
The FDA encourages the development of credible evidence through clinical investigations and works to harmonize regulations that protect human subjects, aiming to streamline the research process while ensuring participant rights.
What are the risks associated with medical devices?
Over the past decade, there have been significant reports of injuries and deaths linked to medical devices in the U.S., highlighting the necessity for robust regulatory oversight to enhance safety.
What categories of device evaluations are outlined in 21 CFR Part 812?
The categories include exempt evaluations, significant risk (SR) evaluations, and non-significant risk (NSR) evaluations. SR evaluations face more stringent regulatory requirements due to higher potential risks.
What is the IDE application process?
This process involves submitting a detailed plan to the FDA that outlines the study’s objectives, methodology, and compliance with regulations. The FDA reviews the application to ensure it meets safety and efficacy standards.
What role do Institutional Review Boards (IRBs) play in clinical trials?
IRBs are ethics committees that protect participants’ rights and welfare by reviewing research protocols and informed consent documents, ensuring compliance with ethical standards.
Why is informed consent important in clinical trials?
Informed consent provides potential participants with essential information about the study, including risks and benefits, allowing them to make educated decisions about their participation.
What are the responsibilities of sponsors regarding record-keeping and reporting?
Sponsors must maintain detailed records of all study-related activities and report any adverse events or protocol deviations to ensure transparency and uphold the integrity of the research.
How do quality assurance (QA) and quality control (QC) contribute to clinical trials?
QA and QC measures ensure compliance with regulatory standards, making the collected data reliable and valid. They are crucial for the overall success of research and the approval of medical devices.
What monitoring practices are recommended under 21 CFR Part 812?
Ongoing supervision through regular audits, inspections, and reviews is vital for identifying potential issues early and maintaining the integrity of clinical studies.
How does the FDA ensure the safety and effectiveness of medical products?
The FDA oversees research studies to ensure compliance with regulations and standards, enhancing public health through rigorous monitoring and transparent communication of drug information.
What is the role of Data Monitoring Committees (DMCs)?
DMCs assess safety and efficacy data during studies and provide recommendations on whether to continue, modify, or terminate research based on interim findings.
List of Sources
- Understanding Investigational Device Exemptions (IDEs)
- fdli.org (https://fdli.org/2023/10/the-accelerated-approval-program-desiderata-for-a-proper-solution-to-the-untimely-completion-of-confirmatory-trials)
- fda.gov (https://fda.gov/about-fda/reports/reports-agency-policies-and-initiatives)
- gao.gov (https://gao.gov/products/gao-24-106699?utm_medium=social&utm_source=twitter&utm_campaign=usgao)
- elsevier.com (https://elsevier.com/industry/medical-device-regulation?dgcid=RN_CM_Sourced_400006591#2-mdr-webinars)
- fda.gov (https://fda.gov/drugs/news-events-human-drugs/fda-clinical-investigator-training-course-citc-2023-12062023)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-march-15-2024)
- infomeddnews.com (https://infomeddnews.com/tdb_templates/41424-2)
- fda.gov (https://fda.gov/news-events/fda-voices/increasing-options-clinical-research-facilitate-medical-product-development)
- fda.gov (https://fda.gov/regulatory-information/search-fda-guidance-documents/real-world-evidence-considerations-regarding-non-interventional-studies-drug-and-biological-products)
- nam.edu (https://nam.edu/regenerative-medicine-case-study-for-understanding-and-anticipating-emerging-science-and-technology)
- gao.gov (https://gao.gov/products/gao-24-106699?utm_medium=social&utm_source=twitter&utm_campaign=usgao)
- wonder.cdc.gov (https://wonder.cdc.gov)
- Types of Device Studies Under 21 CFR Part 812
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-november-21-2023)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-september-6-2024)
- ecfr.gov (https://ecfr.gov/current/title-21/chapter-I/subchapter-H/part-807/subpart-A)
- nam.edu (https://nam.edu/regenerative-medicine-case-study-for-understanding-and-anticipating-emerging-science-and-technology)
- ecfr.gov (https://ecfr.gov/current/title-21/chapter-I/subchapter-H/part-807/subpart-A)
- IDE Application and Approval Process
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-march-15-2024)
- infomeddnews.com (https://infomeddnews.com/tdb_templates/41424-2)
- greenlight.guru (https://greenlight.guru/blog/how-to-set-up-clinical-studies-to-comply-with-us-fda-regulations)
- medidata.com (https://medidata.com/en/company/eclinical-solutions?utm_source=twitter&utm_medium=sm&utm_content=e-clinical-15-years&utm_campaign=gl-q422-partner)
- greenlight.guru (https://greenlight.guru/blog/planning-an-fda-submission)
- elsevier.com (https://elsevier.com/industry/medical-device-regulation?dgcid=RN_CM_Sourced_400006591#2-mdr-webinars)
- fda.gov (https://fda.gov/about-fda/cdrh-innovation/medical-device-coverage-initiatives-connecting-payors-payor-communication-task-force)
- fda.gov (https://fda.gov/regulatory-information/search-fda-guidance-documents/platform-technology-designation-program-drug-development)
- fdli.org (https://fdli.org/2023/10/the-accelerated-approval-program-desiderata-for-a-proper-solution-to-the-untimely-completion-of-confirmatory-trials)
- fda.gov (https://fda.gov/about-fda/reports/reports-agency-policies-and-initiatives)
- dev.to (https://dev.to/rajatp/the-journey-from-idea-to-launch-a-step-by-step-guide-to-software-development-515m)
- venngage.com (https://venngage.com/blog/how-to-write-a-research-proposal)
- Institutional Review Board (IRB) Approval and Oversight
- catalyst.harvard.edu (https://catalyst.harvard.edu/regulatory/video-series-participant-data?utm_source=Twitter&utm_medium=Social%20&utm_campaign=Participant%20Data%20Videos%20)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- undark.org (https://undark.org/2024/02/26/institutional-review-board-slow)
- statnews.com (https://statnews.com/2024/07/18/institutional-review-boards-must-assess-trials-scientific-merit?utm_campaign=rss)
- mailchi.mp (https://mailchi.mp/jhu/bioethicsbulletin-2519368-8a3ajiivxd-2520380)
- content.govdelivery.com (https://content.govdelivery.com/accounts/UKRI/bulletins/376c384?utm_source=Twitter&utm_medium=social&utm_campaign=Orlo)
- jonesday.com (https://jonesday.com/en/insights/2024/01/fda-final-rule-irb-waiver-of-informed-consent-for-minimalrisk-investigations)
- fda.gov (https://fda.gov/news-events/fda-voices/increasing-options-clinical-research-facilitate-medical-product-development)
- fda.gov (https://fda.gov/about-fda/economic-impact-analyses-fda-regulations/institutional-review-board-waiver-or-alteration-informed-consent-minimal-risk-clinical-0)
- fda.gov (https://fda.gov/about-fda/reports/reports-agency-policies-and-initiatives)
- venngage.com (https://venngage.com/blog/how-to-write-a-research-proposal)
- Participant Safety and Informed Consent
- scientia.global (https://scientia.global/mr-anthony-keyes-understanding-and-improving-clinical-trial-compliance)
- researchamerica.org (https://researchamerica.org/press-releases-statements/large-majority-of-americans-say-consideration-of-clinical-trial-participation-should-be-a-part-of-regular-health-care)
- rethinkingclinicaltrials.org (https://rethinkingclinicaltrials.org/news/grand-rounds-february-2-2024-strategies-for-improving-public-understanding-of-fda-and-the-products-it-regulateswhy-should-we-care-and-what-might-we-do-susan-c-winckler-rph-esq)
- bioethicstoday.org (https://bioethicstoday.org/blog/academic-and-private-partnership-to-improve-informed-consent-forms-using-a-data-driven-approach)
- jamanetwork.com (https://jamanetwork.com/journals/jama/article-abstract/2820280)
- medpagetoday.com (https://medpagetoday.com/opinion/second-opinions/107038)
- nucats.northwestern.edu (https://nucats.northwestern.edu/about/news/2024/tnn-success.html)
- raps.org (https://raps.org/news-and-articles/news-articles/2024/5/stakeholders-welcome-fda-s-informed-consent-‘key-i?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- nature.com (https://nature.com/articles/s41591-023-02665-1)
- rethinkingclinicaltrials.org (https://rethinkingclinicaltrials.org/news/grand-rounds-february-23-2024-virtual-vigilance-monitoring-of-decentralized-clinical-trials-adrian-hernandez-md-christopher-j-lindsell-phd)
- Record Keeping and Reporting Requirements
- fda.gov (https://fda.gov/regulatory-information/search-fda-guidance-documents/use-data-monitoring-committees-clinical-trials?utm_content=bufferf9e54&utm_medium=social&utm_source=twitter.com&utm_campaign=buffer)
- fda.gov (https://fda.gov/about-fda/reports/reports-agency-policies-and-initiatives)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-march-15-2024)
- histalk2.com (https://histalk2.com/2024/10/06/monday-morning-update-10-7-24)
- raps.org (https://raps.org/news-and-articles/news-articles/2023/12/fda-compliance-chiefs-detail-fy2023-trends,-plans?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- medpagetoday.com (https://medpagetoday.com/opinion/second-opinions/107038)
- scientia.global (https://scientia.global/mr-anthony-keyes-understanding-and-improving-clinical-trial-compliance)
- jamanetwork.com (https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2811814)
- fda.gov (https://fda.gov/drugs/news-events-human-drugs/fda-clinical-investigator-training-course-citc-2023-12062023)
- nlm.nih.gov (https://nlm.nih.gov/pubs/techbull/mj24/mj24_Clinical_Trials_Study_Record_Modernization.html)
- fda.gov (https://fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/anish-s-shah-mdsiyan-clinical-research-674073-12122023)
- Quality Assurance and Quality Control Measures
- medtechintelligence.com (https://medtechintelligence.com/news_article/fda-adopts-iso-13485-with-qmsr-final-rule)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-march-15-2024)
- med-technews.com (https://med-technews.com/medtech-insights/medtech-regulatory-insights/simplifying-samd-regulatory-compliance-with-ai-driven-expert)
- jamanetwork.com (https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2811814)
- nlm.nih.gov (https://nlm.nih.gov/pubs/techbull/mj24/mj24_Clinical_Trials_Study_Record_Modernization.html)
- fda.gov (https://fda.gov/regulatory-information/search-fda-guidance-documents/use-data-monitoring-committees-clinical-trials?utm_content=bufferf9e54&utm_medium=social&utm_source=twitter.com&utm_campaign=buffer)
- greenlight.guru (https://greenlight.guru/blog/guide-clinical-data-management-medtech)
- Navigating Clinical Evaluations and Investigations in MedTech (https://greenlight.guru/blog/navigating-clinical-evaluations-and-investigations-in-medtech)
- jamanetwork.com (https://jamanetwork.com/journals/jama/fullarticle/2822037?utm_source=twitter&utm_campaign=content-shareicons&utm_content=article_engagement&utm_medium=social&utm_term=080624)
- greenlight.guru (https://greenlight.guru/blog/data-management-and-reporting-in-fda-regulated-clinical-trials)
- Monitoring and Compliance with 21 CFR Part 812
- rethinkingclinicaltrials.org (https://rethinkingclinicaltrials.org/news/grand-rounds-february-2-2024-strategies-for-improving-public-understanding-of-fda-and-the-products-it-regulateswhy-should-we-care-and-what-might-we-do-susan-c-winckler-rph-esq)
- fda.gov (https://fda.gov/regulatory-information/search-fda-guidance-documents/use-data-monitoring-committees-clinical-trials?utm_content=bufferf9e54&utm_medium=social&utm_source=twitter.com&utm_campaign=buffer)
- fda.gov (https://fda.gov/about-fda/reports/reports-agency-policies-and-initiatives)
- fda.gov (https://fda.gov/drugs/news-events-human-drugs/fda-clinical-investigator-training-course-citc-2023-12062023)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-november-21-2023)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-december-19-2023)
- scientia.global (https://scientia.global/mr-anthony-keyes-understanding-and-improving-clinical-trial-compliance)
- nlm.nih.gov (https://nlm.nih.gov/pubs/techbull/mj24/mj24_Clinical_Trials_Study_Record_Modernization.html)







 (CDI) Process Flowchart: Clinical Documentation Improvement (CDI) Process](https://tely.blob.core.windows.net/telyai/flowchart-clinical-documentation-improvement-cdi-process.jpg)