Introduction
Navigating the intricate landscape of medical device registration presents significant challenges, particularly with the looming deadlines for GUDID compliance. As manufacturers endeavor to meet stringent regulatory standards, grasping the essential steps for compliance is paramount for success in a competitive market.
This article provides a comprehensive checklist that delineates the crucial actions manufacturers must undertake to achieve GUDID compliance. It explores the benefits of adherence, not only for securing regulatory approval but also for enhancing patient safety and operational efficiency.
How can manufacturers effectively streamline their compliance processes while remaining ahead of the evolving regulatory landscape?
bioaccess®: Accelerate GUDID Compliance for Medical Devices
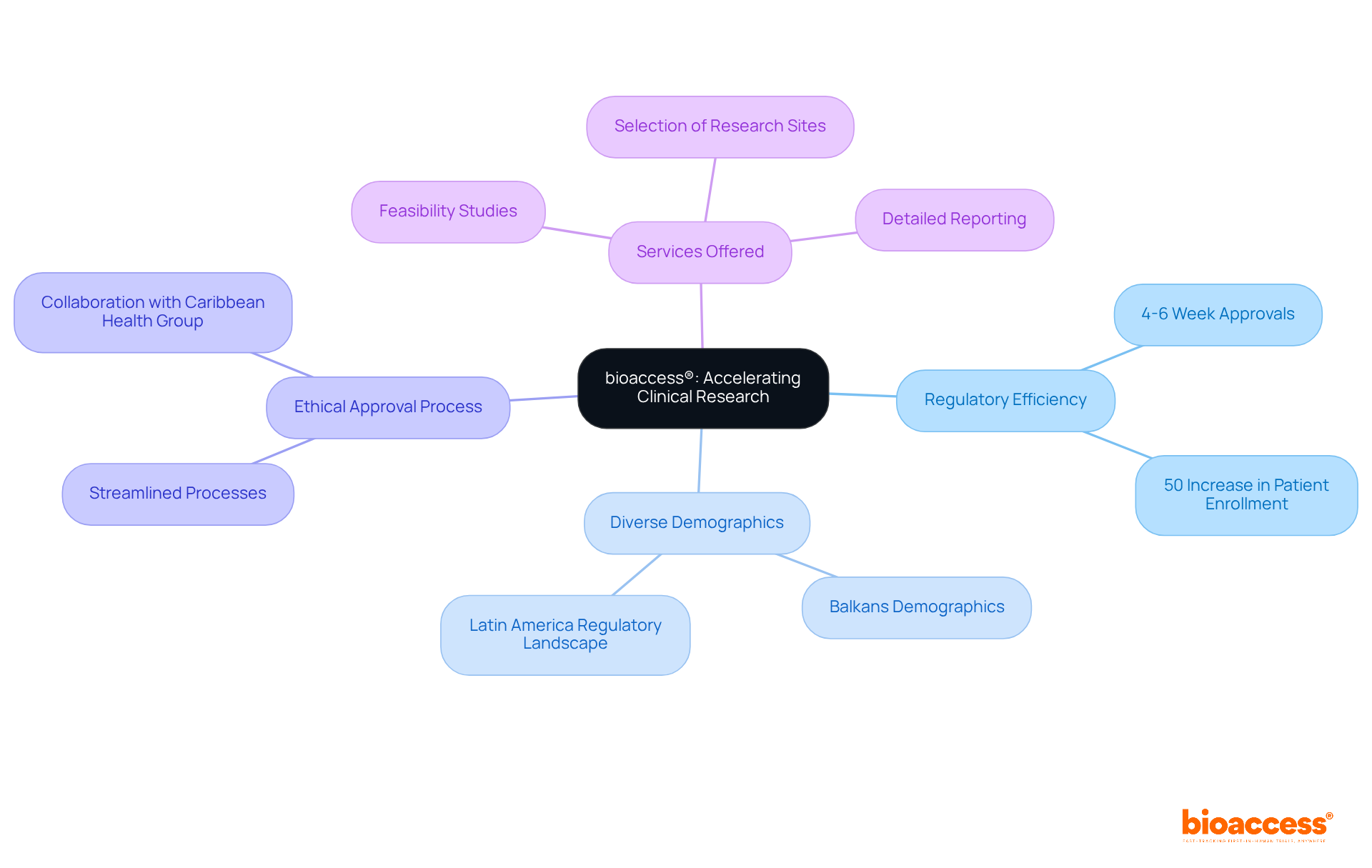
bioaccess® excels in accelerating adherence for medical products by leveraging its extensive knowledge of regulatory frameworks across Latin America, the Balkans, and Australia. With an impressive capability to secure in just 4-6 weeks and enhance enrollment rates by 50%, bioaccess® offers a streamlined approach for manufacturers to navigate the complexities of registration. This tailored strategy not only ensures prompt adherence but also mitigates delays in the .
Industry leaders underscore the critical importance of , emphasizing its role in ensuring product safety and effectiveness in real-world settings. Successful registrations, such as those achieved by NuView for its liquid biopsy diagnostic test, exemplify the efficacy of bioaccess®’s strategies in facilitating .
As continue to evolve, the speed at which companies can achieve compliance will significantly influence their ability to capitalize on in Latin America, where over 10,000 medical products are already available globally.
Colombia emerges as a prime destination for due to its , offering compared to North America and Western Europe, alongside a that spans just 90-120 days. Furthermore, the Colombian government provides R&D tax incentives, including a 100% tax deduction for investments in science and technology, enhancing the appeal of conducting trials in the country.
The collaboration between bioaccess™ and Caribbean Health Group aims to position Barranquilla as a leading hub for in Latin America, supported by Colombia’s Minister of Health. Additionally, the partnership with Welwaze Medical Inc. for the Celbrea® launch highlights bioaccess®’s commitment to facilitating market access and reinforces the necessity of partnering with organizations like bioaccess® to navigate regulatory challenges and expedite the commercialization of life-saving technologies.
Understand UDI Format: Key to GUDID Entry
The format comprises two essential elements: the and the . The DI serves as a required, fixed element that identifies both the labeler and the specific version or model of the apparatus. In contrast, the PI provides supplementary details, including the lot or batch number and expiration date.
Mastering this format is vital for manufacturers, as it ensures precise input of equipment information into the GUDID. This accuracy is crucial for and compliance, especially with the set to commence in Q4 2024, culminating in a by Q2 2026.
Regulatory specialists emphasize that adherence to not only enhances traceability but also protects patients and clinicians by preventing counterfeit products from infiltrating the market. As producers prepare for these changes, understanding the becomes a critical component of successful and overall market readiness.
![]()
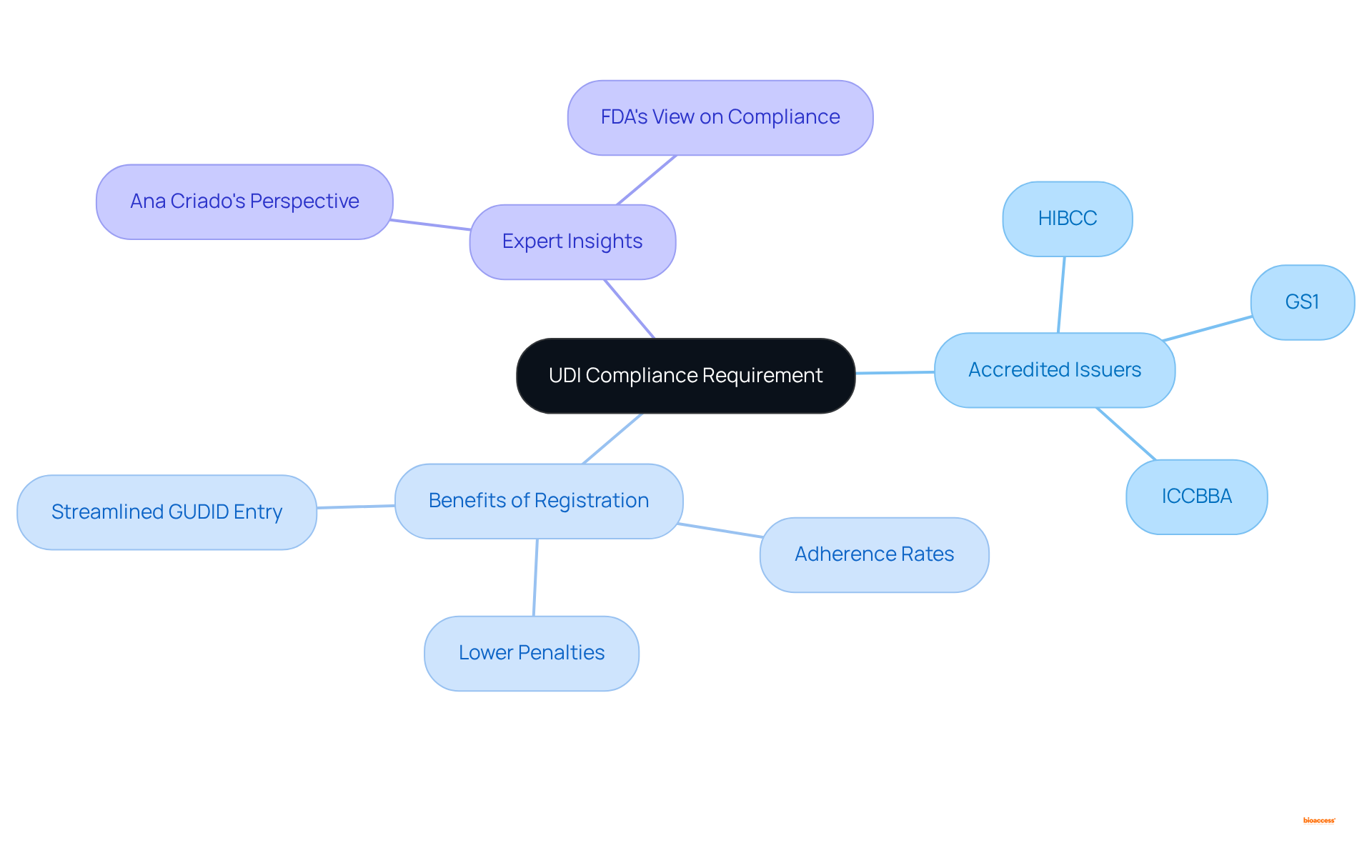
Register with an FDA-Accredited UDI Issuer: A Compliance Requirement
To satisfy , producers must enroll with an -, a crucial step for acquiring a . This UDI must be prominently displayed on the device label and packaging. The recognizes several , including:
- GS1
- HIBCC
- ICCBBA
Collaborating with an accredited issuer not only ensures that UDIs adhere to but also streamlines the process of . Statistics indicate that manufacturers listed with UDI issuers achieve greater adherence rates, significantly lowering the risk of penalties and delays in market access. This proactive strategy is essential for producers aiming to navigate the complexities of effectively. Experts like Ana Criado, Director of Regulatory Affairs and a consultant with extensive experience in biomedical engineering and health economics, emphasize the necessity of following these regulations to ensure successful market entry for .
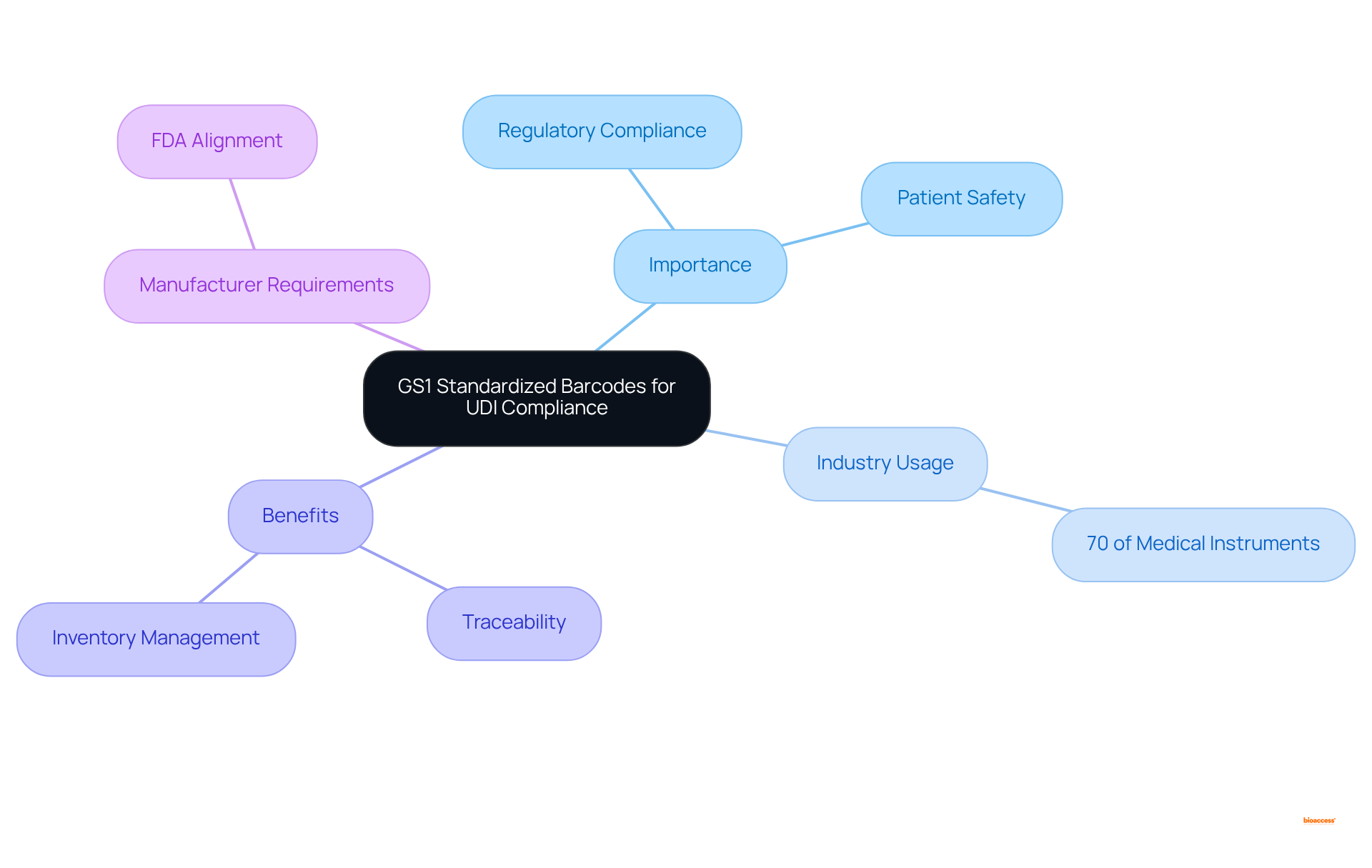
Obtain GS1 Standardized Barcodes: Essential for UDI Compliance
Obtaining is essential for achieving . These barcodes must be displayed on labels and packaging in both human-readable and . Currently, approximately 70% of for , highlighting the importance of gudid in the industry.
The GS1 system offers a globally recognized standard that enhances traceability, ensuring items are accurately identified throughout the supply chain. Notably, leading manufacturers have embraced GS1 barcodes to streamline their registration processes and improve inventory management.
Experts in assert that effective barcode implementation not only facilitates but also enhances by enabling the gudid for precise tracking of . Manufacturers must ensure their barcodes align with to mitigate potential issues during audits and inspections, thereby safeguarding their market position and reputation.
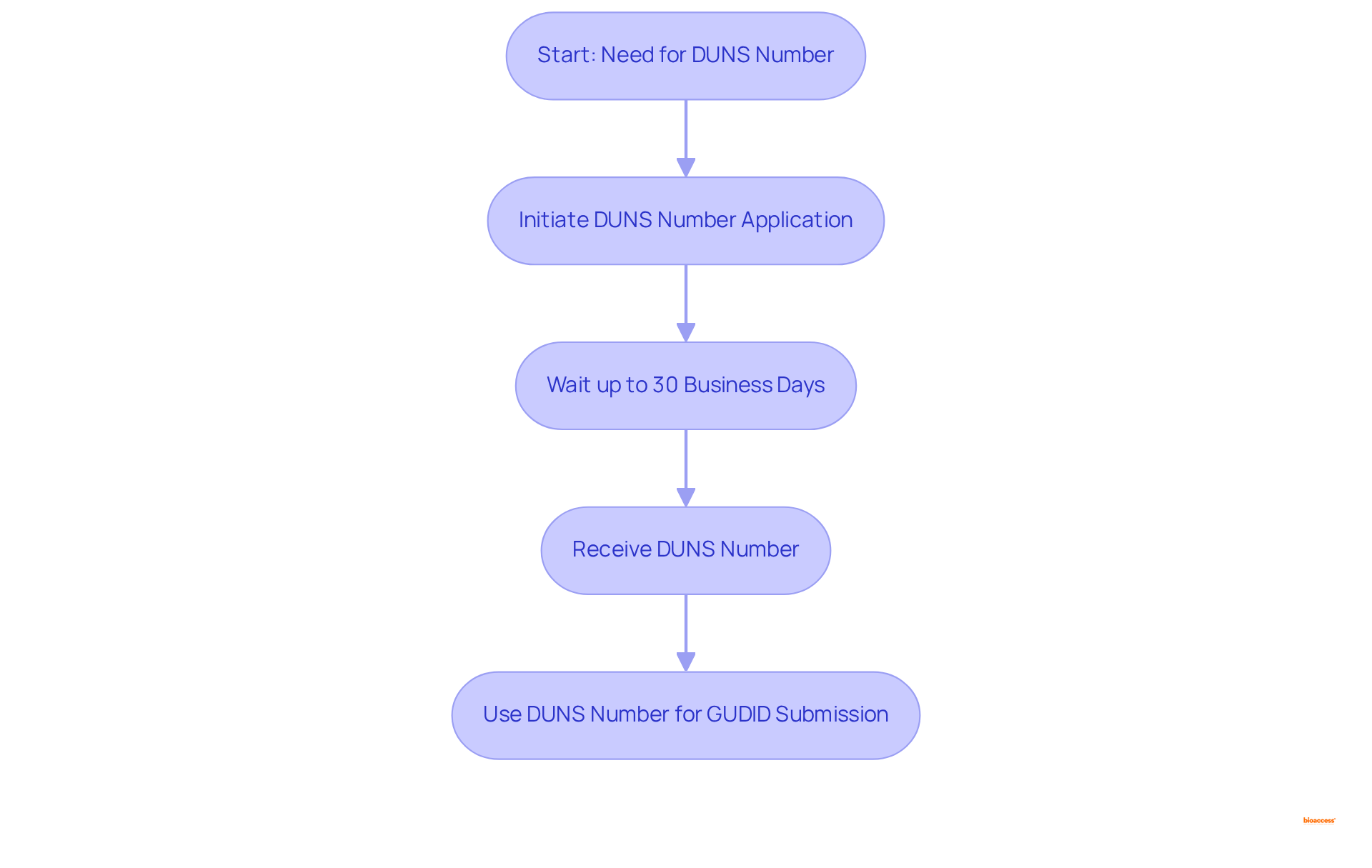
Request a DUNS Number: Necessary for GUDID Identification
To sign up for an account, a (DUNS) number, a distinct identifier issued by Dun & Bradstreet that confirms the legal identity of the labeler organization. This is crucial for guaranteeing the precision and traceability of the information provided to the database.
The average time to acquire a can take up to 30 business days, making it essential for manufacturers to initiate this process early in their to avoid potential delays. Industry leaders emphasize that efficient preparation, including obtaining a , is vital for a . Adam Newman, Head of Marketing, asserts, “Effective preparation is essential for a .”
Furthermore, producers who have effectively managed the underscore the significance of the and its relation to the and enhancing compliance. By prioritizing the acquisition of a , which is free of charge, manufacturers can ensure they meet relevant requirements efficiently, thereby minimizing and facilitating smoother market access.
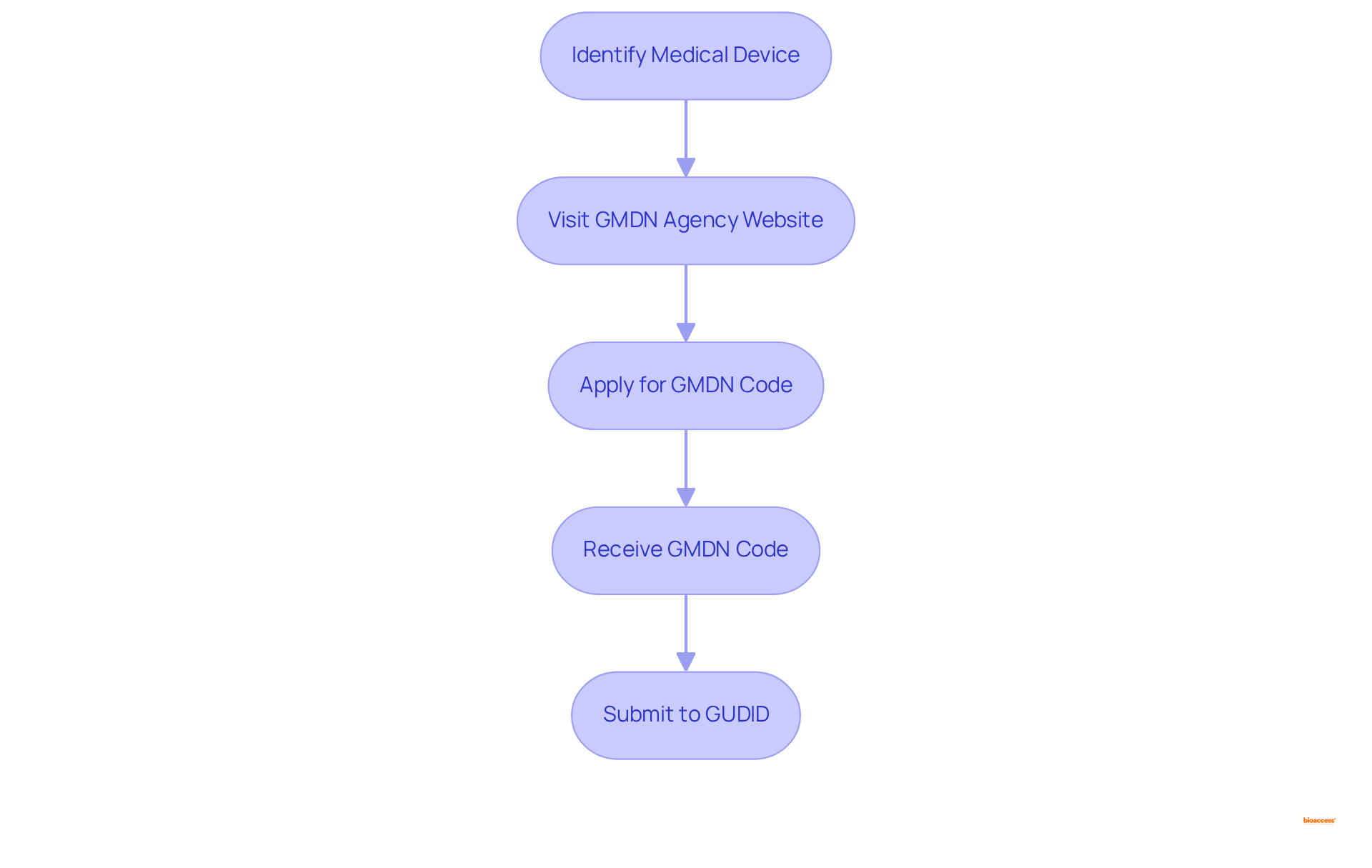
Apply for GMDN: Classify Your Medical Device for GUDID
To ensure accurate categorization for submission to the GUDID, manufacturers must acquire a for their medical products. The that involved. Each is required to have at least one , which aids regulatory bodies and in accurately identifying and categorizing devices. This classification is essential, as adherence to s is increasingly mandated by , including the GUDID, facilitating smoother and alignment with international standards. As emphasized by specialists such as Ana Criado, Director of Regulatory Affairs, . Manufacturers can easily obtain s through the GMDN Agency’s website, ensuring they meet the necessary criteria for successful submissions.
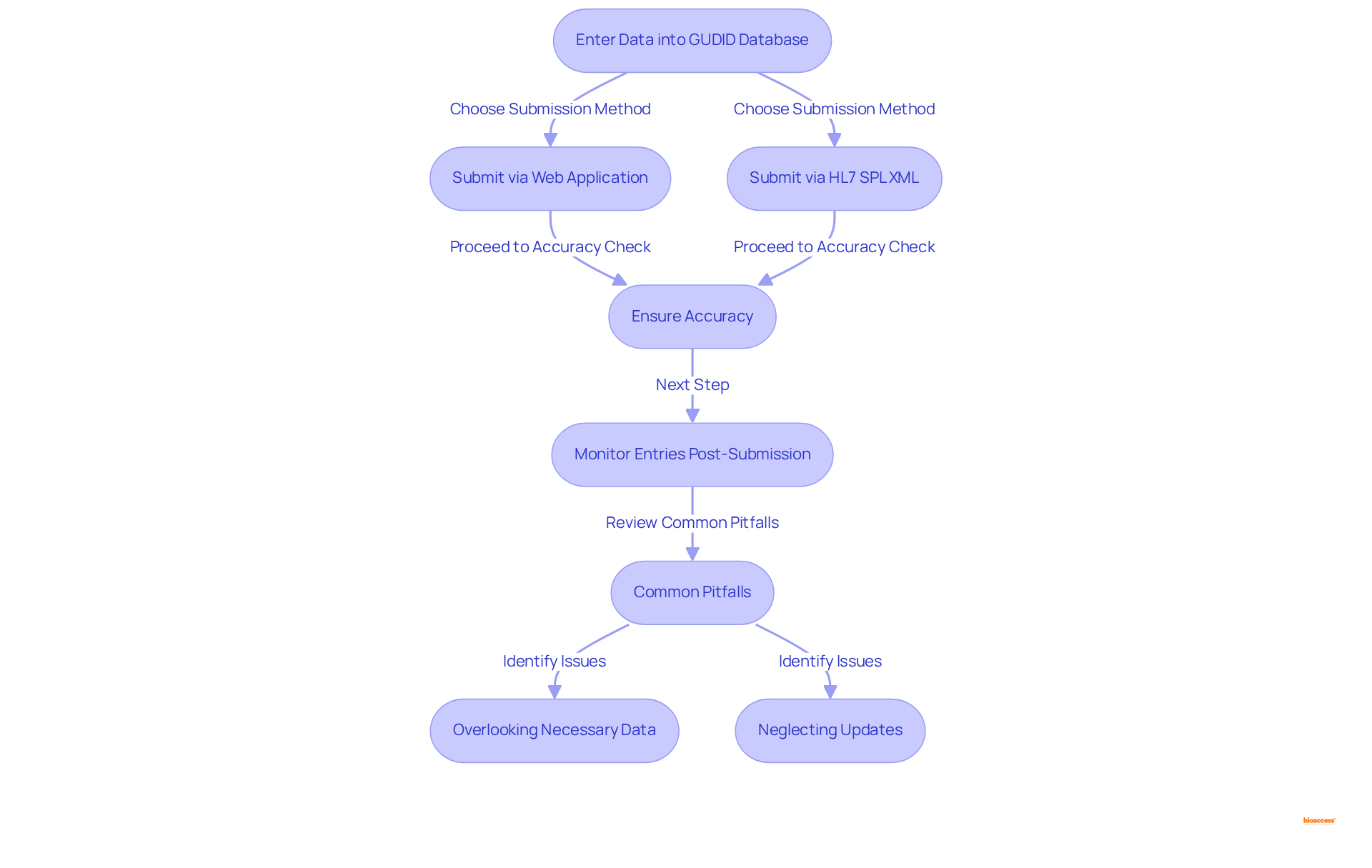
Enter Data into the GUDID Database: Final Compliance Step
The final phase in meeting the necessary standards necessitates the meticulous input of all required information into the database. Producers can choose to submit their data through the web application or via HL7 SPL XML file submissions, with the decision primarily influenced by their submission volume. For those submitting higher volumes, the presents significant advantages, enabling automated submissions that greatly streamline the process.
Precision and thoroughness are paramount; any discrepancies can result in and potential penalties. Industry statistics indicate that the typical adherence rate for producers entering information into the database hovers around 75%, highlighting the critical need for .
Post-submission, it is essential for manufacturers to consistently to maintain standards. This proactive approach not only mitigates risks but also ensures that all records correspond with , thereby enhancing the overall integrity of the submission process.
As emphasize, common pitfalls in include:
- Overlooking
- Neglecting to update records swiftly, which can jeopardize adherence efforts
Navigate Global UDI Challenges: Insights for Manufacturers
Manufacturers face considerable challenges when navigating , which differ significantly across countries. These challenges include diverse regulations, adherence timelines, and data submission procedures. For instance, the U.S. implemented UDI regulations in 2013, mandating adherence for Class III products by September 24, 2014, and Class I products by December 8, 2022. In contrast, Saudi Arabia has set deadlines for high-risk items by September 1, 2023, and medium to low-risk items by September 1, 2024. The European Union’s UDI system is anticipated to be fully operational by the end of Q2 2025, with adherence beginning on January 1, 2026.
To effectively manage these complexities, manufacturers must prioritize staying informed about the latest and engage with local experts who possess a deep understanding of each market’s nuances. Developing a that accommodates these variations is essential. This strategy should incorporate that capture critical attributes such as device identifiers and production identifiers, ensuring compliance with regulations like the .
Moreover, leveraging platforms such as RegDesk, which provides across more than 120 markets, can facilitate the navigation of UDI regulations. This platform aids manufacturers in preparing and publishing international submissions, thereby and minimizing the risk of penalties, which could lead to loss of business and reputation. By adopting these strategies and consistently reviewing their UDI approaches in response to evolving regulations, manufacturers can enhance their operational efficiency in the global marketplace.
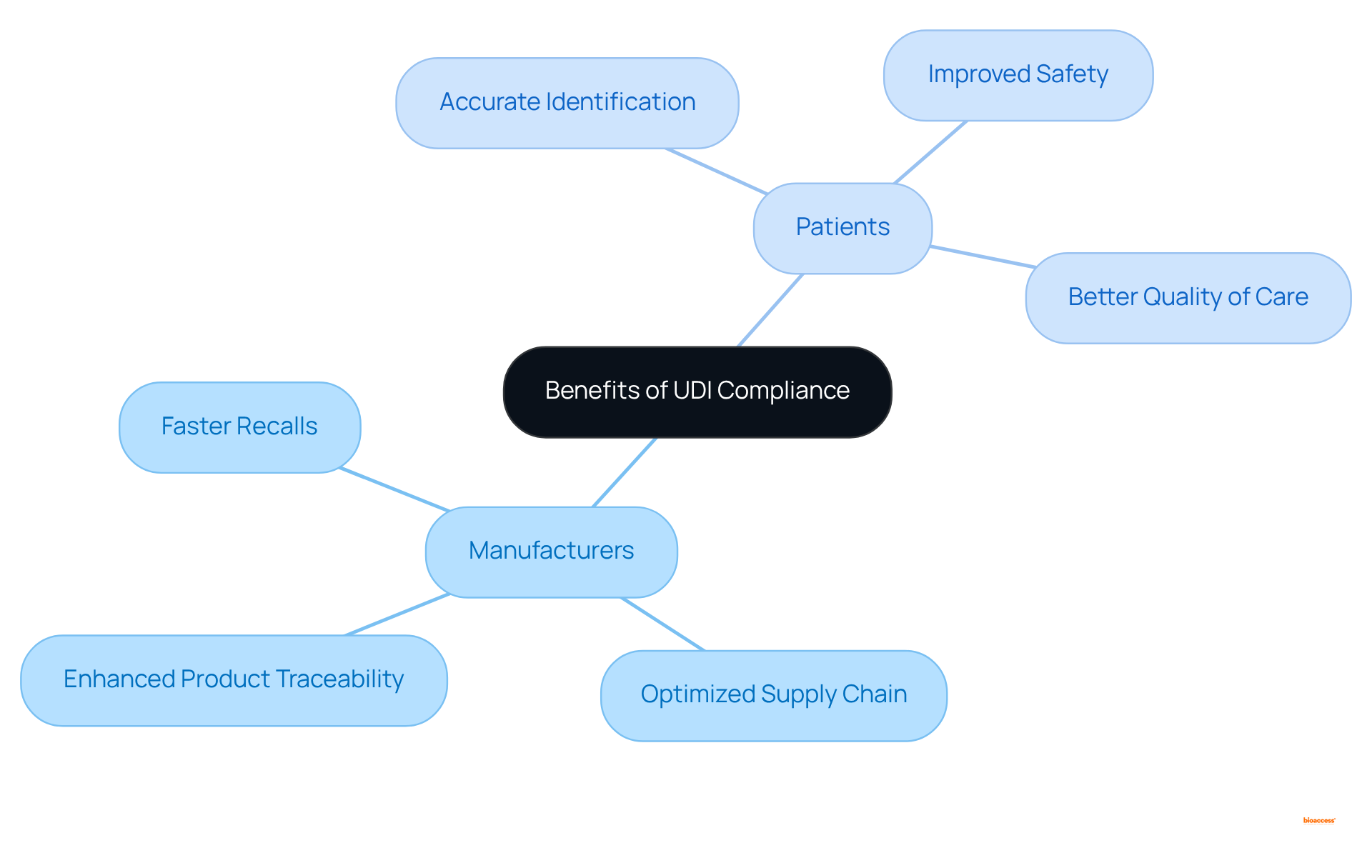
Leverage UDI Compliance: Benefits for Manufacturers and Patients
Achieving UDI conformity presents significant advantages for both manufacturers and patients, particularly through the implementation of the gudid. For manufacturers, the , optimizes supply chain processes, and facilitates quicker recalls in the event of adverse incidents, which is essential for mitigating risks associated with equipment failures.
Experts, including Ana Criado, Director of Regulatory Affairs and a consultant with extensive experience in biomedical engineering and health economics, emphasize that is critical for navigating the complex and beyond.
For patients, guarantees accurate identification of , which is vital for ensuring safety and improving the quality of care. Healthcare professionals assert that , as facilitated by gudid, is directly linked to heightened patient safety, enabling timely alerts regarding recalls and compatibility issues.
Furthermore, a majority of moderate- and high-risk products are now labeled with a gudid, which underscores the pervasive compliance within the sector. By prioritizing , manufacturers not only meet regulatory requirements but also play a crucial role in and building trust in their products.
This commitment to safety and quality ultimately cultivates a more reliable healthcare environment, where patients can confidently rely on the tools that support their health. To fully leverage these benefits, manufacturers should and stay informed about regulatory developments.
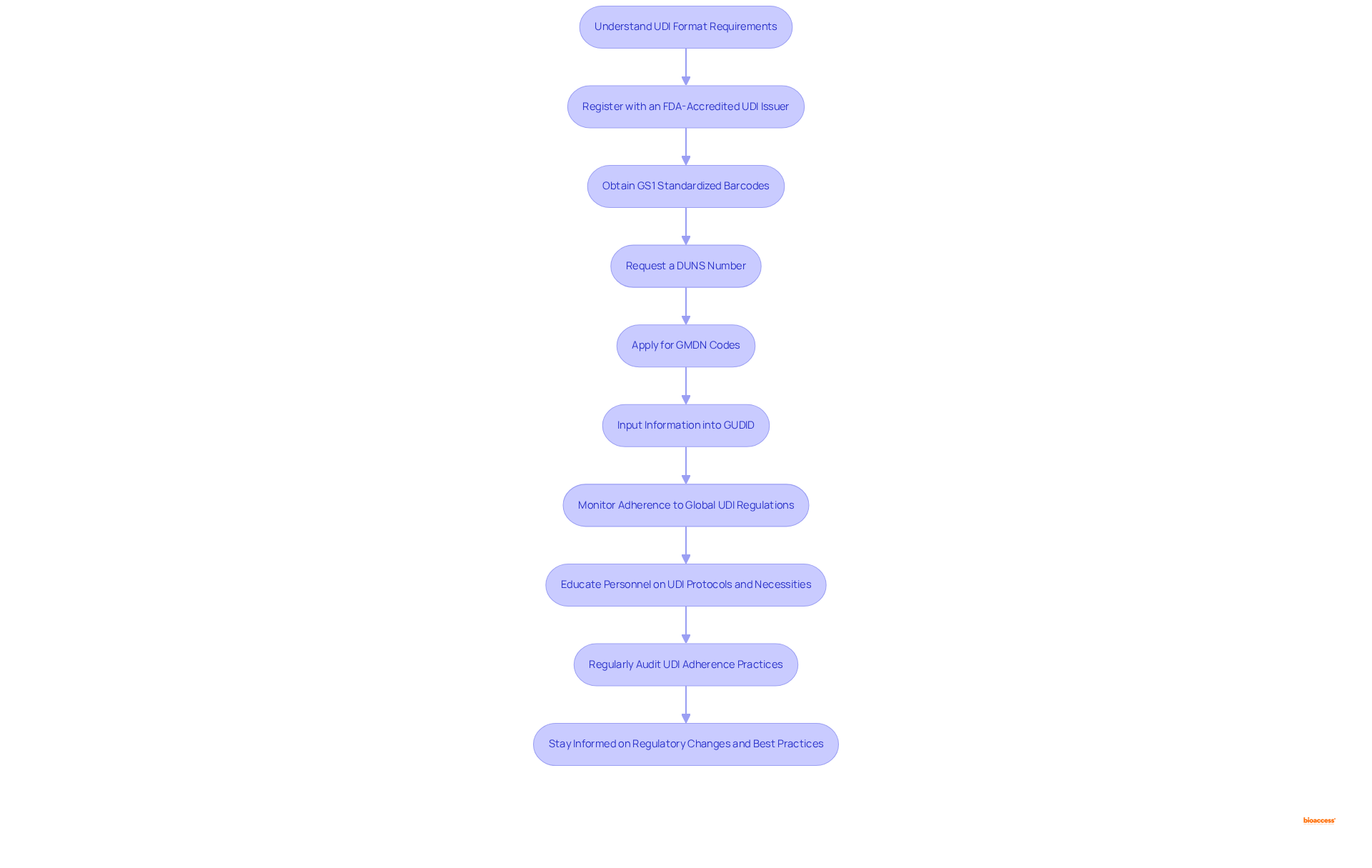
Checklist for UDI Compliance: Essential Considerations for Manufacturers
To achieve effectively, manufacturers must follow these essential steps:
- : It is crucial to familiarize yourself with the specific UDI format mandated by the FDA, which includes the Device Identifier (DI) and Production Identifier (PI).
- : Ensure registration with a recognized UDI issuer to obtain the necessary identifiers for your products.
- : Acquire GS1 barcodes that comply with UDI standards for accurate tracking and identification.
- Request a DUNS Number: Obtain a Data Universal Numbering System (DUNS) number, essential for identifying your business in the global marketplace.
- : Secure Global Medical Device Nomenclature (GMDN) codes to classify your appropriately.
- Input Information into GUDID: Accurately input all required information into the GUDID database to ensure adherence.
- : Stay vigilant about changes in UDI regulations across different markets to maintain conformity.
- Educate Personnel on UDI Protocols and Necessities: Establish thorough training initiatives for your team to guarantee comprehension of UDI procedures, significantly improving regulatory effectiveness. , with 85% of respondents reporting improved preparedness for adherence deadlines.
- Regularly Audit UDI Adherence Practices: Conduct frequent audits of your UDI adherence practices to identify and rectify any gaps, ensuring alignment with regulatory standards. A case study reveals that companies saved an average of $2.86 million by performing regular audits, underscoring the significance of this step.
- Stay Informed on and Best Practices: Continuously monitor updates in UDI regulations and industry best practices to adjust your adherence strategies accordingly.
Regulatory experts emphasize that understanding the intent behind UDI—primarily traceability—is crucial for successful implementation. As Jon Speer notes, “This should not be a foreign concept to anybody. This is a concept of traceability – that’s all a UDI is – traceability.” By following these steps, manufacturers can not only meet compliance requirements but also enhance their operational efficiency and product safety.
Conclusion
Achieving GUDID compliance is essential for medical device manufacturers aiming to navigate the complexities of regulatory requirements and ensure timely market access. This process involves a series of critical steps, from understanding the UDI format to accurately entering data into the GUDID database. Each stage plays a pivotal role in not only meeting compliance standards but also enhancing the overall safety and effectiveness of medical devices in the market.
Key arguments outlined in the article highlight the importance of:
- Understanding UDI format requirements
- Registering with FDA-accredited UDI issuers
- Acquiring GS1 standardized barcodes
- Obtaining a DUNS number
- Acquiring GMDN codes for proper identification and classification
The article emphasizes the necessity of meticulous data entry into the GUDID database, as well as the importance of continuous monitoring and training to maintain compliance with evolving regulations.
Ultimately, the benefits of adhering to GUDID compliance extend beyond regulatory requirements; they significantly enhance patient safety and trust in medical devices. By prioritizing UDI compliance, manufacturers not only streamline their operational processes but also contribute to a more reliable healthcare environment. Engaging with experts and utilizing resources like bioaccess® can further ease this journey, ensuring that manufacturers are well-equipped to meet compliance deadlines and improve patient outcomes. Taking proactive steps now will pave the way for successful market entry and foster a culture of safety and accountability in the medical device industry.
Frequently Asked Questions
What is bioaccess® and how does it assist in GUDID compliance for medical devices?
bioaccess® accelerates adherence for medical products by utilizing its extensive knowledge of regulatory frameworks across Latin America, the Balkans, and Australia. It helps manufacturers navigate the complexities of registration, securing ethical approvals in 4-6 weeks and enhancing enrollment rates by 50%.
Why is regulatory adherence important for medical devices?
Regulatory adherence is critical for ensuring product safety and effectiveness in real-world settings. It influences the successful registration of medical devices, which is essential for timely market access.
What are the benefits of conducting clinical trials in Colombia?
Colombia offers cost efficiency for clinical trials, with savings of over 30% compared to North America and Western Europe. The regulatory review process typically spans 90-120 days, and the Colombian government provides R&D tax incentives, including a 100% tax deduction for investments in science and technology.
What is the Unique Device Identifier (UDI) format and its components?
The UDI format consists of two elements: the Device Identifier (DI), which identifies the labeler and specific version or model of the device, and the Production Identifier (PI), which provides additional details like the lot number and expiration date.
Why is mastering the UDI format important for manufacturers?
Mastering the UDI format is essential for accurately inputting equipment information into the GUDID, ensuring effective tracking and compliance, especially with the upcoming UDI/Device Registration period starting in Q4 2024 and culminating in a compliance deadline by Q2 2026.
What must producers do to meet UDI compliance standards?
Producers must enroll with an FDA-accredited UDI issuer to acquire a unique UDI for each medical item. This UDI must be displayed on the device label and packaging.
Which organizations are recognized as FDA-accredited UDI issuers?
The FDA recognizes several accredited issuing agencies, including GS1, HIBCC, and ICCBBA.
How does collaborating with an accredited UDI issuer benefit manufacturers?
Collaborating with an accredited issuer ensures compliance with FDA regulations and streamlines the process of entering the GUDID, leading to greater adherence rates and reduced risks of penalties and delays in market access.
List of Sources
- bioaccess®: Accelerate GUDID Compliance for Medical Devices
- bioaccessla.com (https://bioaccessla.com/blog/how-to-navigate-the-costs-of-fda-approval-for-medical-devices-a-step-by-step-guide)
- emergobyul.com (https://emergobyul.com/news/us-fda-looking-medical-device-registrants-udi-gudid-compliance)
- Is Mexico A Prime Location For Medical Device Clinical Trials? (https://meddeviceonline.com/doc/is-mexico-a-prime-location-for-medical-device-clinical-trials-0001)
- Argentina’s Thriving Medical Device Market Amid An Economic Plunge (https://lifescienceleader.com/doc/argentina-s-thriving-medical-device-market-amid-an-economic-plunge-0001)
- Understand UDI Format: Key to GUDID Entry
- regdesk.co (https://regdesk.co/fda-guidance-on-gudid-device-identifier-di-record)
- prweb.com (https://prweb.com/releases/rfid_and_enhanced_udi_tracking_updates_announced_by_matrix_it_medical_tracking_systems/prweb13144214.htm)
- UDI for Medical Devices: Codes & Examples [Ultimate Guide] (https://greenlight.guru/blog/udi-medical-devices)
- qad.com (https://qad.com/blog/2024/07/global-udi-compliance-good-for-patients-and-medical-device-manufacturers)
- Unique Device Identification (UDI) – Healthcare | GS1 (https://gs1.org/industries/healthcare/udi)
- Register with an FDA-Accredited UDI Issuer: A Compliance Requirement
- emergobyul.com (https://emergobyul.com/news/us-fda-looking-medical-device-registrants-udi-gudid-compliance)
- bioworld.com (https://bioworld.com/articles/703341-fda-tells-industry-to-get-serious-about-udi-compliance)
- Understanding FDA Requirements for UDI Compliance in Medical Devices | Key Steps & Benefits (https://ddismart.com/blog/understanding-fda-requirements-for-unique-device-identification-udi-compliance)
- 130+ Compliance Statistics & Trends to Know for 2026 (https://secureframe.com/blog/compliance-statistics)
- 115 Compliance Statistics You Need To Know in 2023 – Drata (https://drata.com/blog/compliance-statistics)
- Obtain GS1 Standardized Barcodes: Essential for UDI Compliance
- Best Supply Chain Quotes (https://linkedin.com/pulse/best-supply-chain-quotes-daniel-stanton)
- gov.uk (https://gov.uk/government/consultations/consultation-on-the-future-regulation-of-medical-devices-in-the-united-kingdom/outcome/chapter-4-registration-and-udi)
- digitaldefynd.com (https://digitaldefynd.com/IQ/motivational-supply-chain-management-quotes)
- How GS1 Standards Improve Medical Device Safety & Efficiency (https://mavenprofserv.com/blog/the-gs1-advantage-for-medical-device)
- Supply Chain Quotes: Take it to the next level. – (https://supplychaintoday.com/supply-chain-quotes)
- Request a DUNS Number: Necessary for GUDID Identification
- reedtech.com (https://reedtech.com/knowledge-center/gudid-account-creation-five-steps-to-follow)
- registrarcorp.com (https://registrarcorp.com/blog/medical-devices/medical-device-registration/gudid)
- reedtech.com (https://reedtech.com/knowledge-center/one-step-in-fda-udi-compliance-the-dun-bradstreet-duns-number)
- matrixone.health (https://matrixone.health/blog/gudid-must-knows-the-fda-global-unique-device-identification-database)
- insider.thefdagroup.com (https://insider.thefdagroup.com/p/fda-gudid-guidance)
- Apply for GMDN: Classify Your Medical Device for GUDID
- vistaar.ai (https://vistaar.ai/blog/2024-recap-medical-device-key-regulation-guidance-in-usa-eu-asia-and-row)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC3704061)
- emmainternational.com (https://emmainternational.com/gmdn-codes)
- mpo-mag.com (https://mpo-mag.com/breaking-news/gmdn-codes-explainedaccording-to-the-gmdn-age)
- obelis.net (https://obelis.net/news/the-importance-of-gmdn-codes)
- Enter Data into the GUDID Database: Final Compliance Step
- regulatorydoctor.us (https://regulatorydoctor.us/f/understanding-the-fda’s-new-udi-guidance)
- tga.gov.au (https://tga.gov.au/resources/event/webinars/udi-global-manufacturer-learnings-and-project-update)
- insider.thefdagroup.com (https://insider.thefdagroup.com/p/fda-gudid-guidance)
- Navigate Global UDI Challenges: Insights for Manufacturers
- qad.com (https://qad.com/blog/2024/07/global-udi-compliance-good-for-patients-and-medical-device-manufacturers)
- regdesk.co (https://regdesk.co/global-medical-device-udi-requirements-a-quick-reference-guide-for-united-states-europe-canada-japan-australia)
- Quick reference guide – global medical device UDI requirements and timelines (https://rimsys.io/blog/quick-reference-guide-global-udi-requirements-and-timelines)
- innovit.com (https://innovit.com/udi-compliance)
- Leverage UDI Compliance: Benefits for Manufacturers and Patients
- tandfonline.com (https://tandfonline.com/doi/full/10.2147/MDER.S364539)
- needle.tube (https://needle.tube/resources-7/The-Impact-of-UDI-Regulations-on-Hospital-Supply-and-Equipment-Management)
- Benefits of a UDI System (https://fda.gov/medical-devices/unique-device-identification-system-udi-system/benefits-udi-system)
- pew.org (https://pew.org/en/research-and-analysis/fact-sheets/2016/07/unique-device-identifiers-improve-safety-and-quality)
- Advancing Patient Safety Surrounding Medical Devices: A Health System Roadmap to Implement Unique Device Identification at the Point of Care – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC8645947)
- Checklist for UDI Compliance: Essential Considerations for Manufacturers
- 115 Compliance Statistics You Need To Know in 2023 – Drata (https://drata.com/blog/compliance-statistics)
- greenlight.guru (https://greenlight.guru/blog/focusing-on-intent-of-udi-requirements-fda-final-guidance)
- 130+ Compliance Statistics & Trends to Know for 2026 (https://secureframe.com/blog/compliance-statistics)
- 100+ Compliance Statistics You Should Know in 2025 (https://sprinto.com/blog/compliance-statistics)
- insights.citeline.com (https://insights.citeline.com/MT103707/Manufacturers-Scramble-To-Meet-Looming-Unique-Device-Identification-Deadline–But-Too-Many-Are-Unprepared)



 — the larger the slice, the greater the savings from that category. Each slice of the pie shows how much each cost factor contributes to the total savings in virtual clinical trials — the larger the slice, the greater the savings from that category.](https://images.tely.ai/telyai/vsfswxcc-each-slice-of-the-pie-shows-how-much-each-cost-factor-contributes-to-the-total-savings-in-virtual-clinical-trials-the-larger-the-slice-the-greater-the-savings-from-that-category.webp)


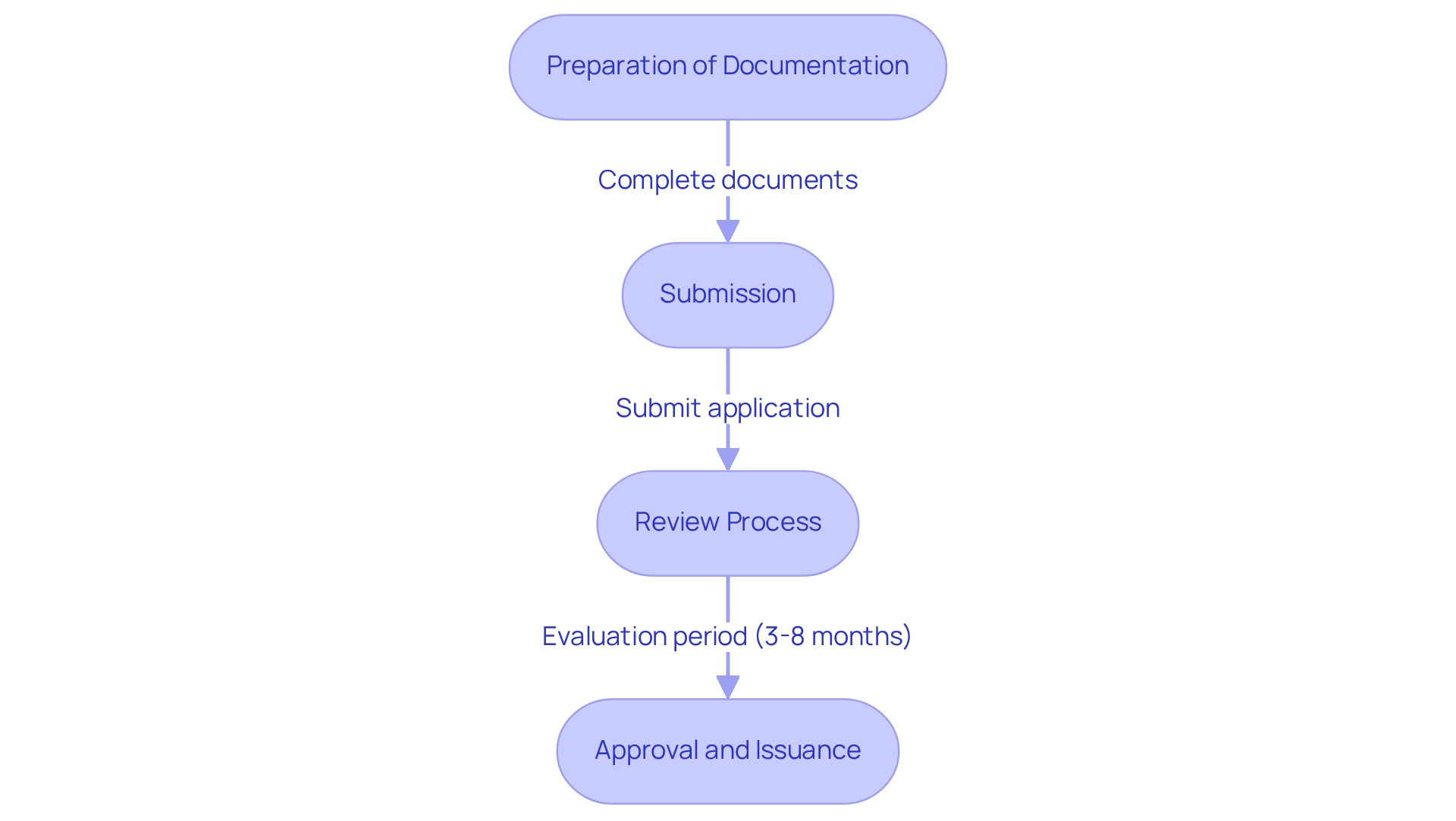
 and study efficiency. Each box shows a step in the clinical study management process. Follow the arrows to see how each step leads into the next, ultimately enhancing patient recruitment and study efficiency.](https://images.tely.ai/telyai/cmiyxnfq-each-box-shows-a-step-in-the-clinical-study-management-process-follow-the-arrows-to-see-how-each-step-leads-into-the-next-ultimately-enhancing-patient-recruitment-and-study-efficiency.webp)



 and see how each branch connects to important details like their role, advancements, effectiveness statistics, risks, and surgeons' views. Explore the central idea of surgical staplers and see how each branch connects to important details like their role, advancements, effectiveness statistics, risks, and surgeons' views.](https://images.tely.ai/telyai/gdmadftk-explore-the-central-idea-of-surgical-staplers-and-see-how-each-branch-connects-to-important-details-like-their-role-advancements-effectiveness-statistics-risks-and-surgeons-views.webp)