Introduction
The landscape of Class II medical devices is rapidly evolving, driven by technological advancements and stringent regulatory requirements. As these devices account for a significant portion of healthcare innovations, navigating the complex approval processes necessitates a deep understanding of the critical factors for clinical success.
What insights can empower manufacturers not only to comply with regulations but also to accelerate their market entry and enhance patient safety? This article delves into ten essential insights that illuminate the opportunities and challenges faced by stakeholders in the Class II medical device sector, offering a comprehensive roadmap for achieving clinical success in an increasingly competitive environment.
bioaccess®: Accelerating Clinical Research for Class II Medical Devices
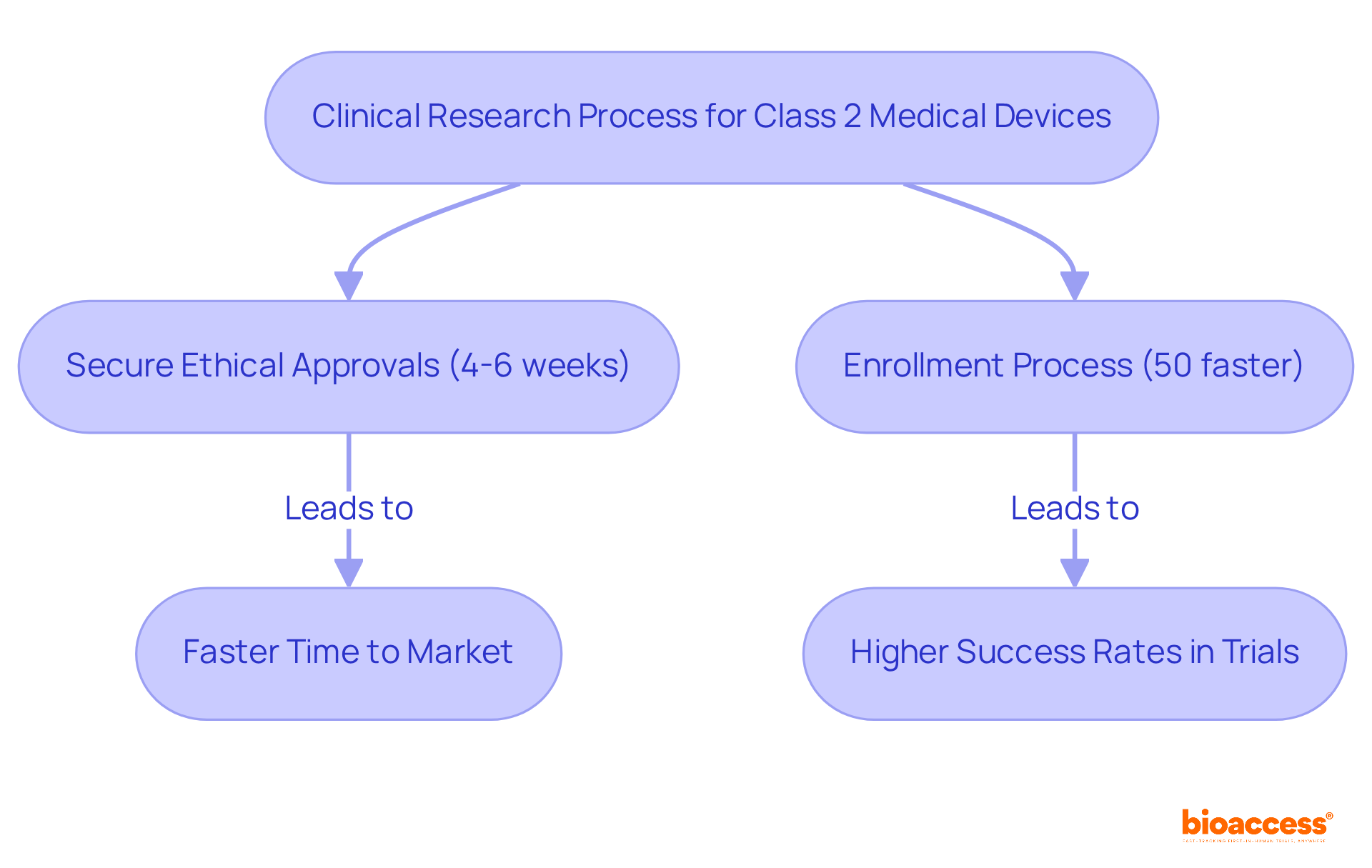
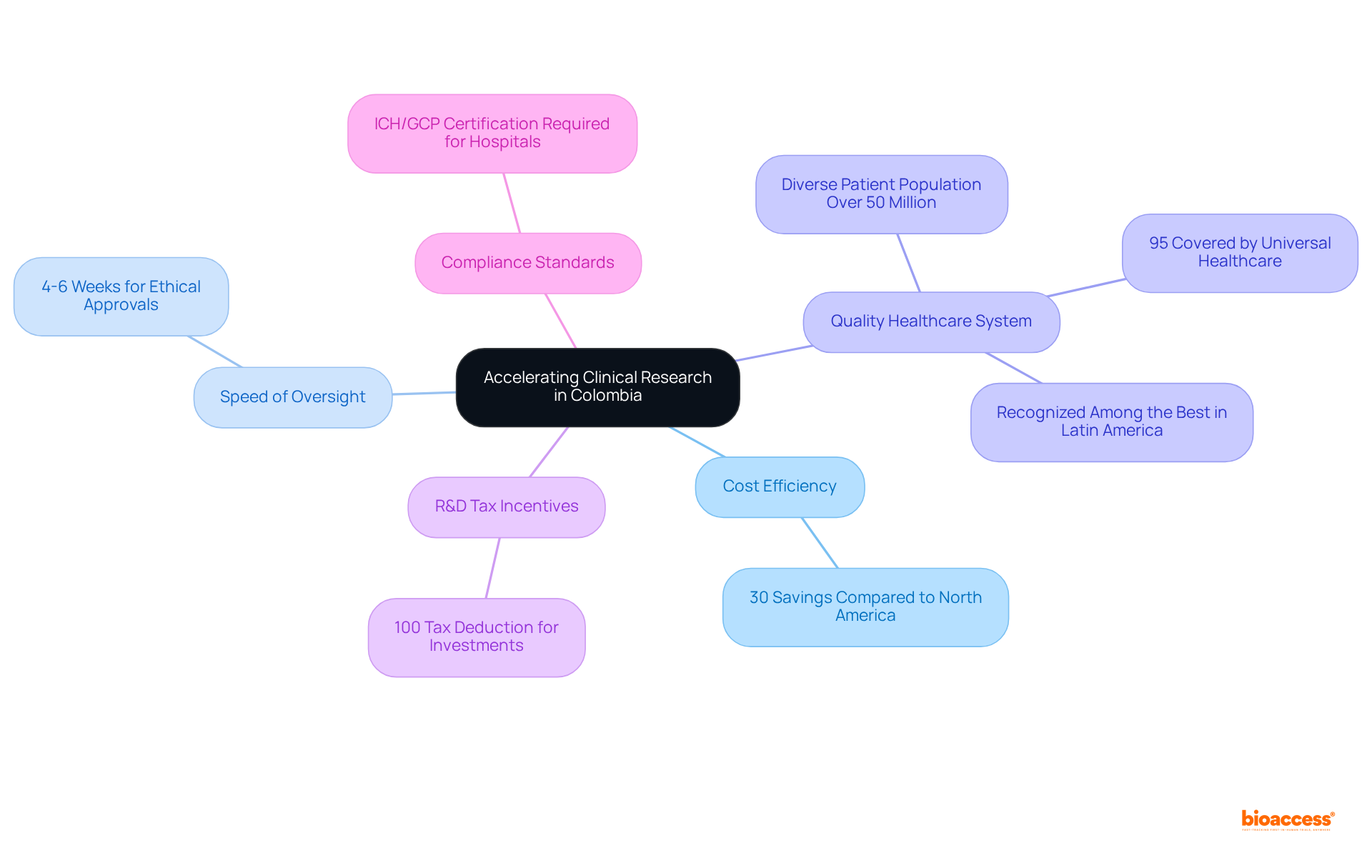
bioaccess® leverages its extensive expertise and regional advantages to specifically for . emerges as a premier destination, distinguished by its cost efficiency, offering savings exceeding 30% compared to trials conducted in North America or Western Europe. The speed of oversight is exceptional, with , facilitated by the streamlined processes of INVIMA, ‘s National Food and Drug Surveillance Institute. Moreover, the country is home to a , and a diverse patient population exceeding 50 million, with 95% covered by universal healthcare.
In addition, provides , further enhancing its appeal for . Hospitals in must undergo a stringent prior to conducting , ensuring adherence to high standards of quality and compliance. These factors significantly reduce the , underscoring the importance of agility for Medtech innovators aiming to swiftly and efficiently launch their products.
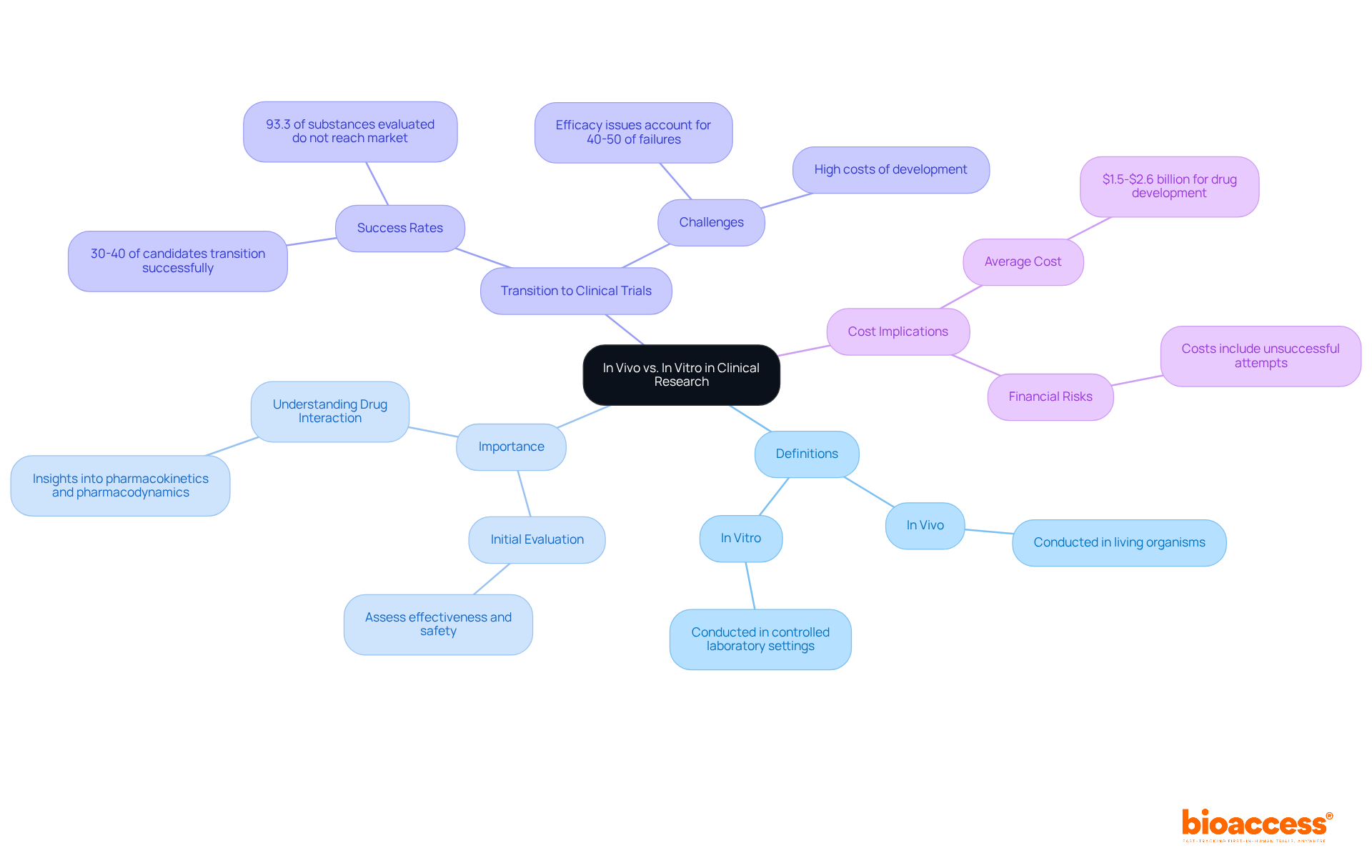
Understanding Class II Medical Devices: Definition and Classification
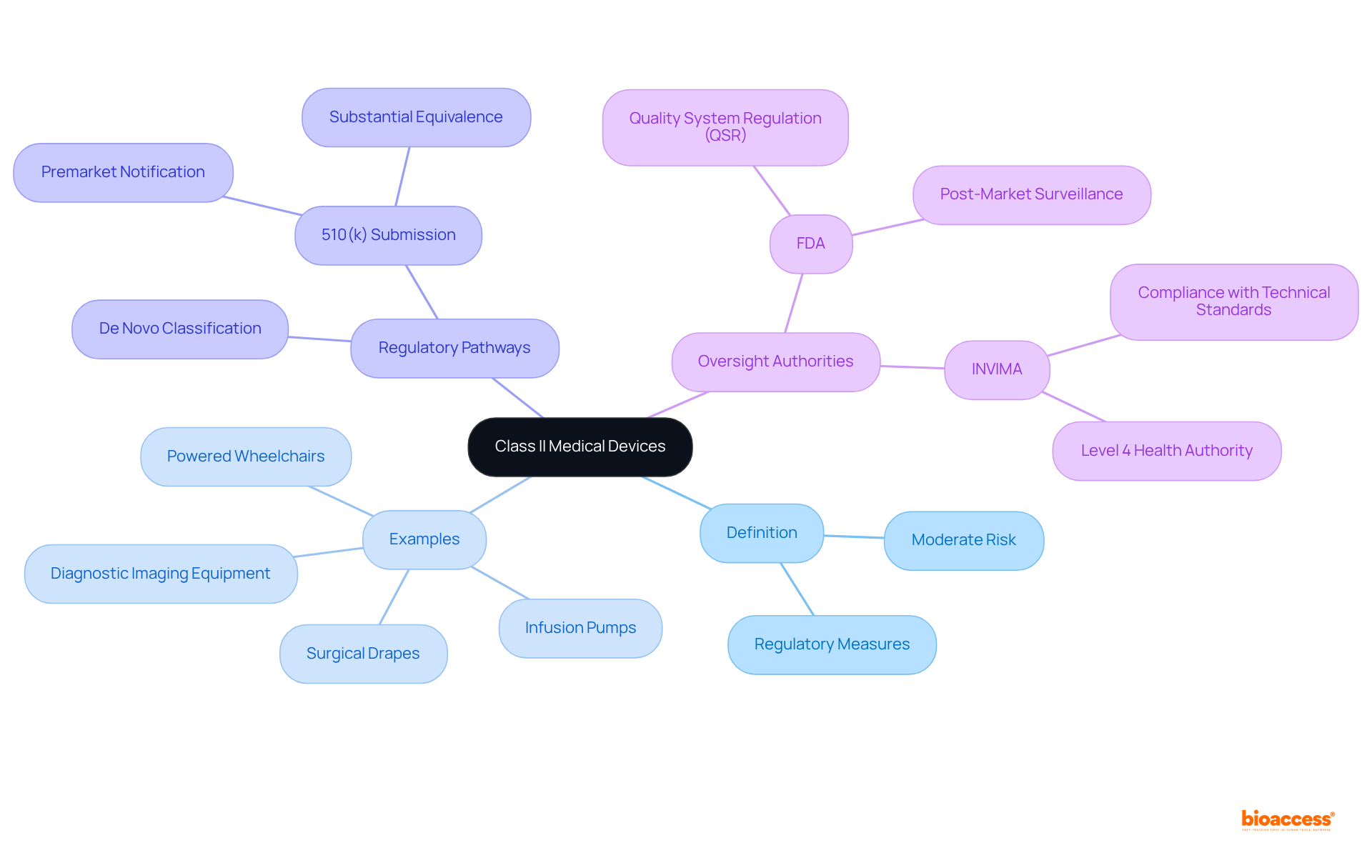
are classified as healthcare items that pose a moderate risk to patients, necessitating specific regulatory measures to ensure their safety and efficacy. This category encompasses a diverse array of products, including:
- infusion pumps
- powered wheelchairs
- surgical drapes
- diagnostic imaging equipment
Notably, recent data indicates that Category II instruments account for , underscoring their significance in the healthcare market. Most require a , commonly referred to as a , which shows that the new item is substantially equivalent to an existing, legally marketed product. This process is crucial for verifying that these products fulfill before they are introduced to the market.
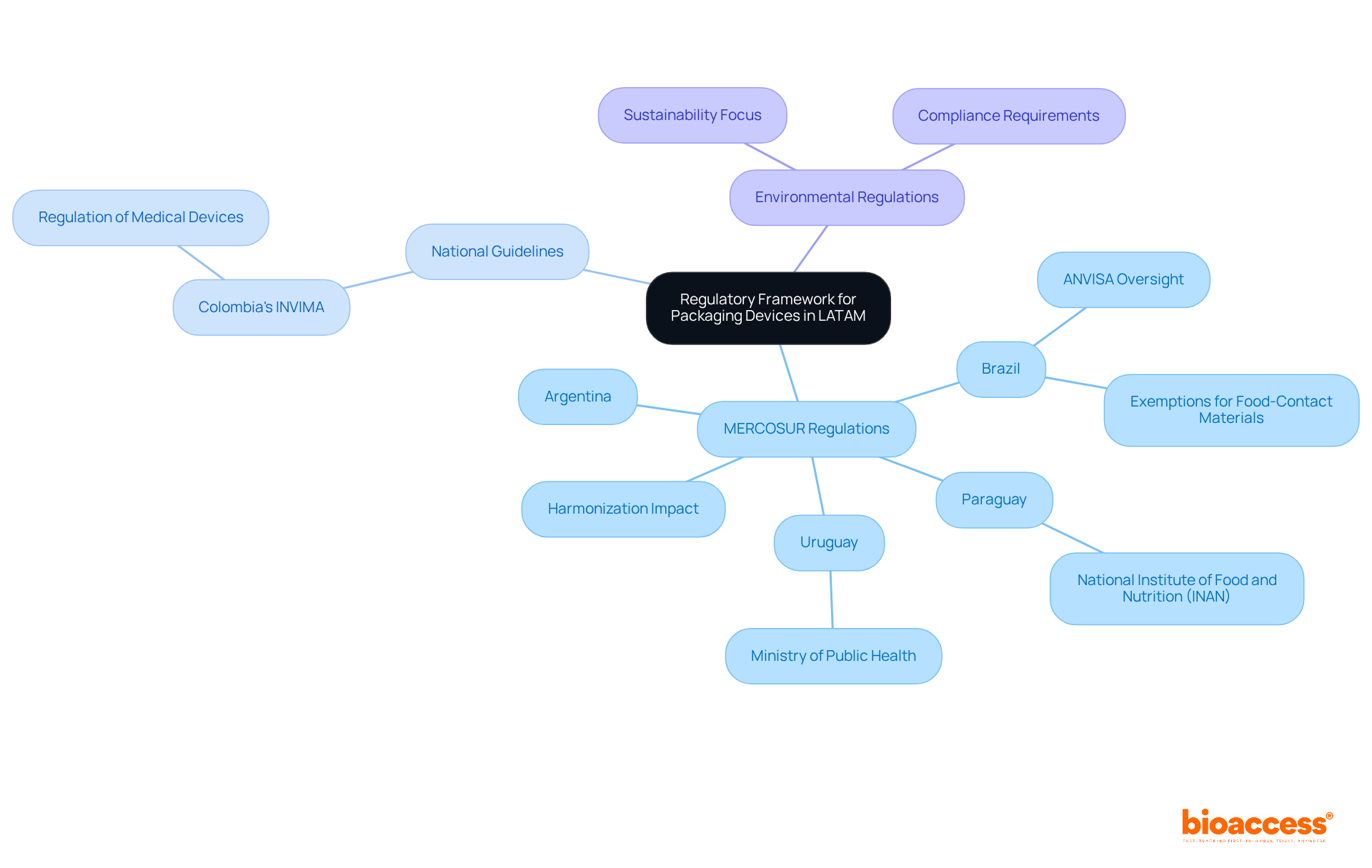
In Colombia, the (Instituto Nacional de Vigilancia de Medicamentos y Alimentos), which plays a vital role in the inspection and oversight of the marketing and production of health products, including . INVIMA’s Directorate for and other Technologies ensures compliance with technical standards and guarantees that products meet essential safety and quality criteria. Furthermore, INVIMA is recognized as a Level 4 health authority by PAHO/WHO, highlighting its expertise in health regulation.
Regulatory specialists emphasize that while certain Category II items may be exempt from the 510(k) requirement, they are still obligated to adhere to general controls and maintain compliance with . Understanding the intricacies of , including their classification and , is essential for successful market entry and patient safety. Additionally, is a critical component of compliance for Category II products, ensuring ongoing oversight of their performance and safety.
Navigating the Approval Process for Class II Medical Devices
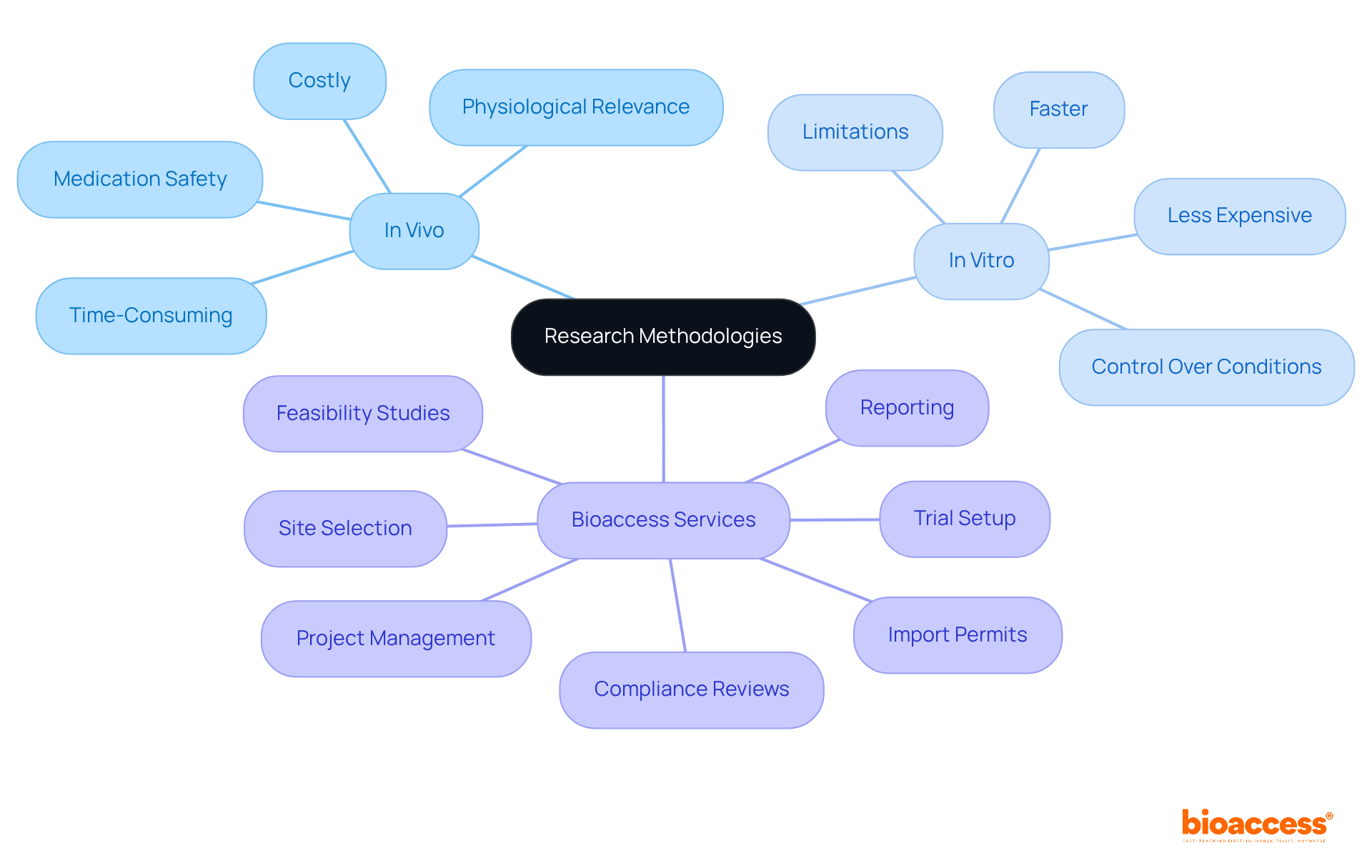
The approval process for requires the submission of a to the FDA, which must include . This submission is not merely a formality; it requires and, in certain instances, to substantiate the claims presented. Engaging compliance consultants early in the process can significantly streamline submissions and enhance the likelihood of a favorable outcome. Recent statistics reveal that approximately 67% of 510(k) submissions prompted , highlighting the critical nature of thorough preparation. Successful manufacturers have effectively leveraged compliance consultants to navigate these complexities, ensuring adherence to and accelerating the approval timeline.
At bioaccess, our extensive encompass:
- Feasibility studies
- Site selection
- Compliance reviews
- Trial setup
- Import permits
- Project management
- Reporting
These elements are crucial for maintaining and can substantially influence the success of the approval process. The 510(k) review process typically aims for a decision within 90 days; however, delays may arise due to incomplete submissions or the necessity for further clarification. By understanding these steps and challenges—particularly —manufacturers can better equip themselves for success in the competitive landscape of .
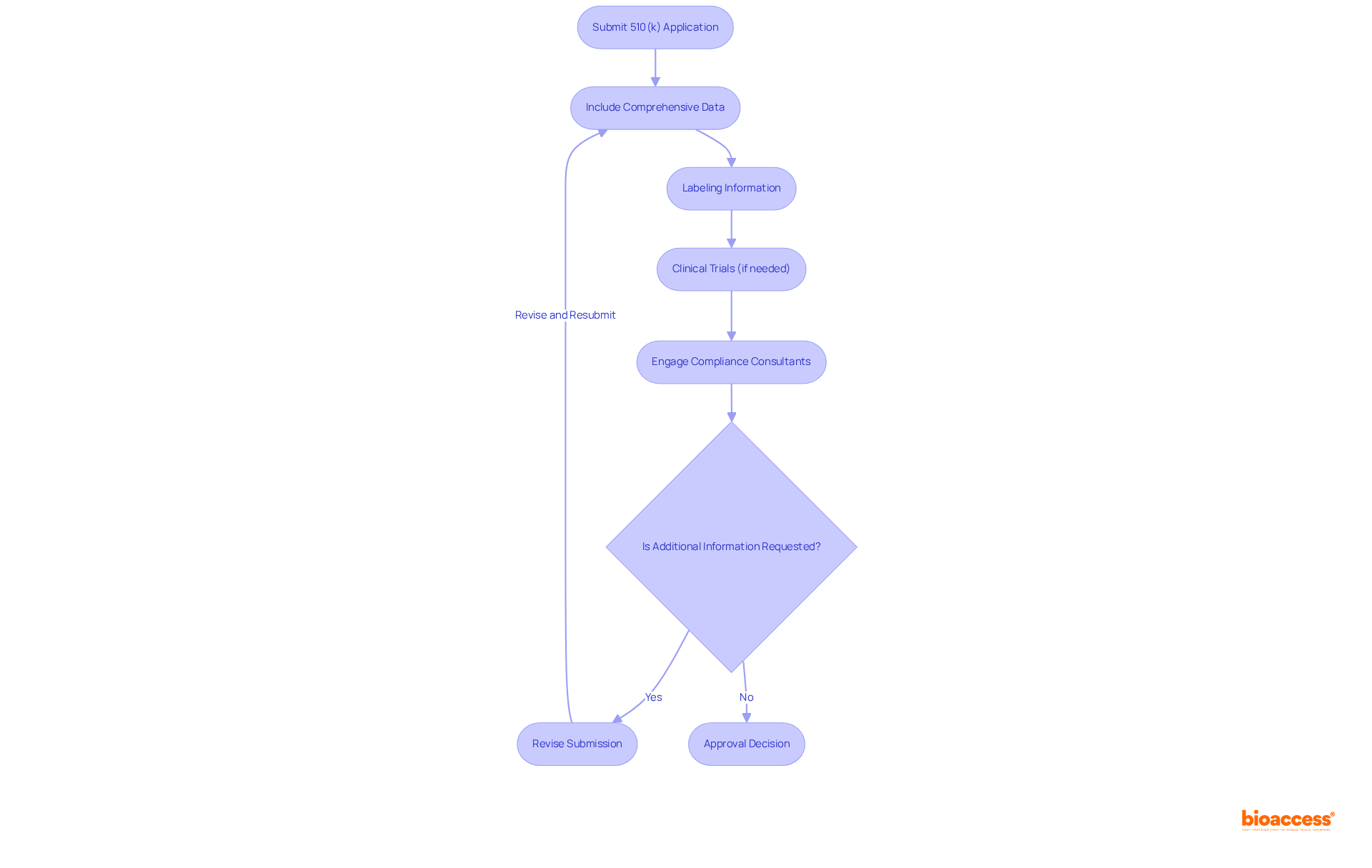
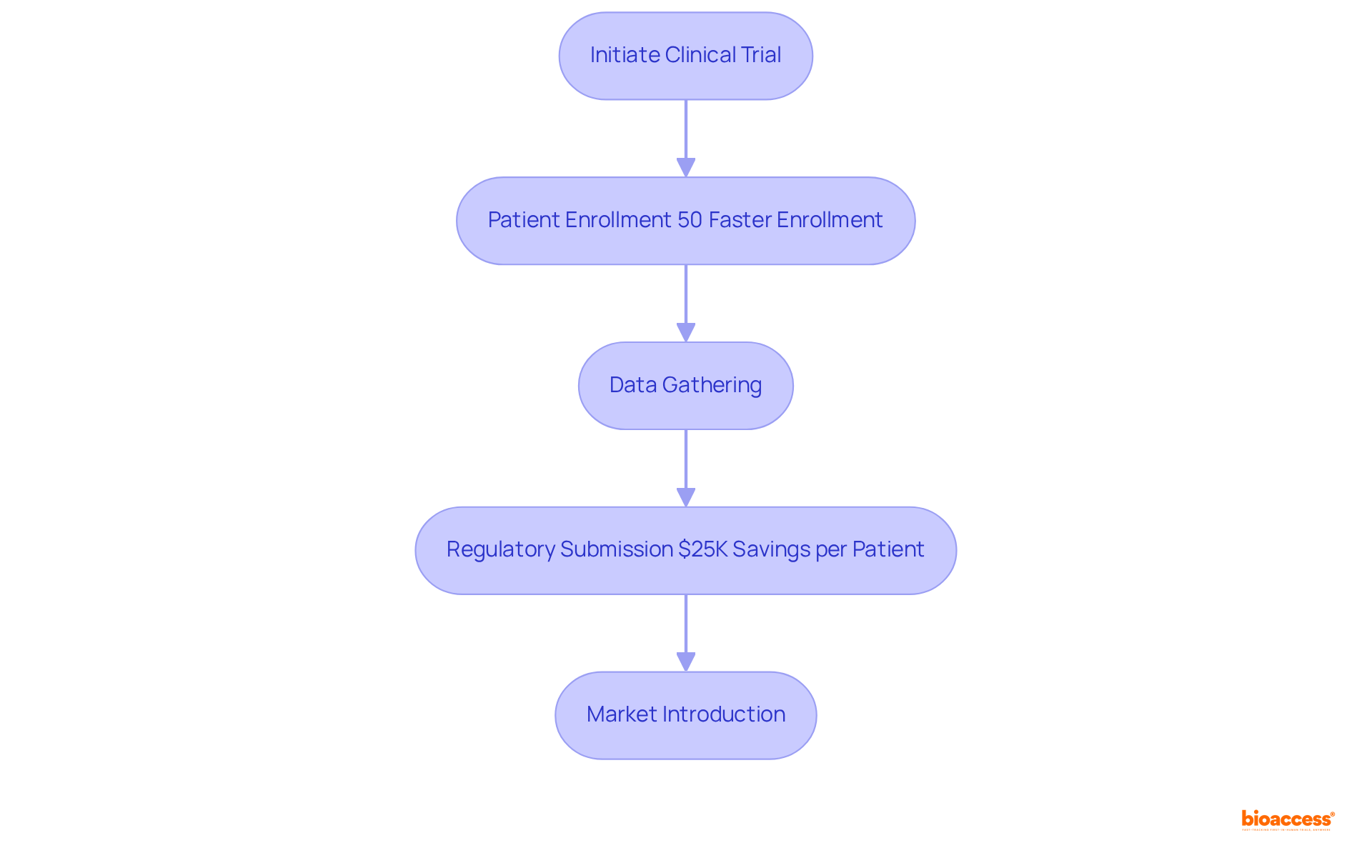
The Role of Clinical Trials in Class II Medical Device Development
are essential for gathering data on their performance in . These trials vary in size and scope, influenced by the system’s complexity and compliance requirements. Notably, with bioaccess®, than at traditional Western sites, significantly enhancing . This acceleration supports and results in an with , eliminating rework and delays. Well-structured trials not only increase the trustworthiness of the product in the marketplace but also lead to .
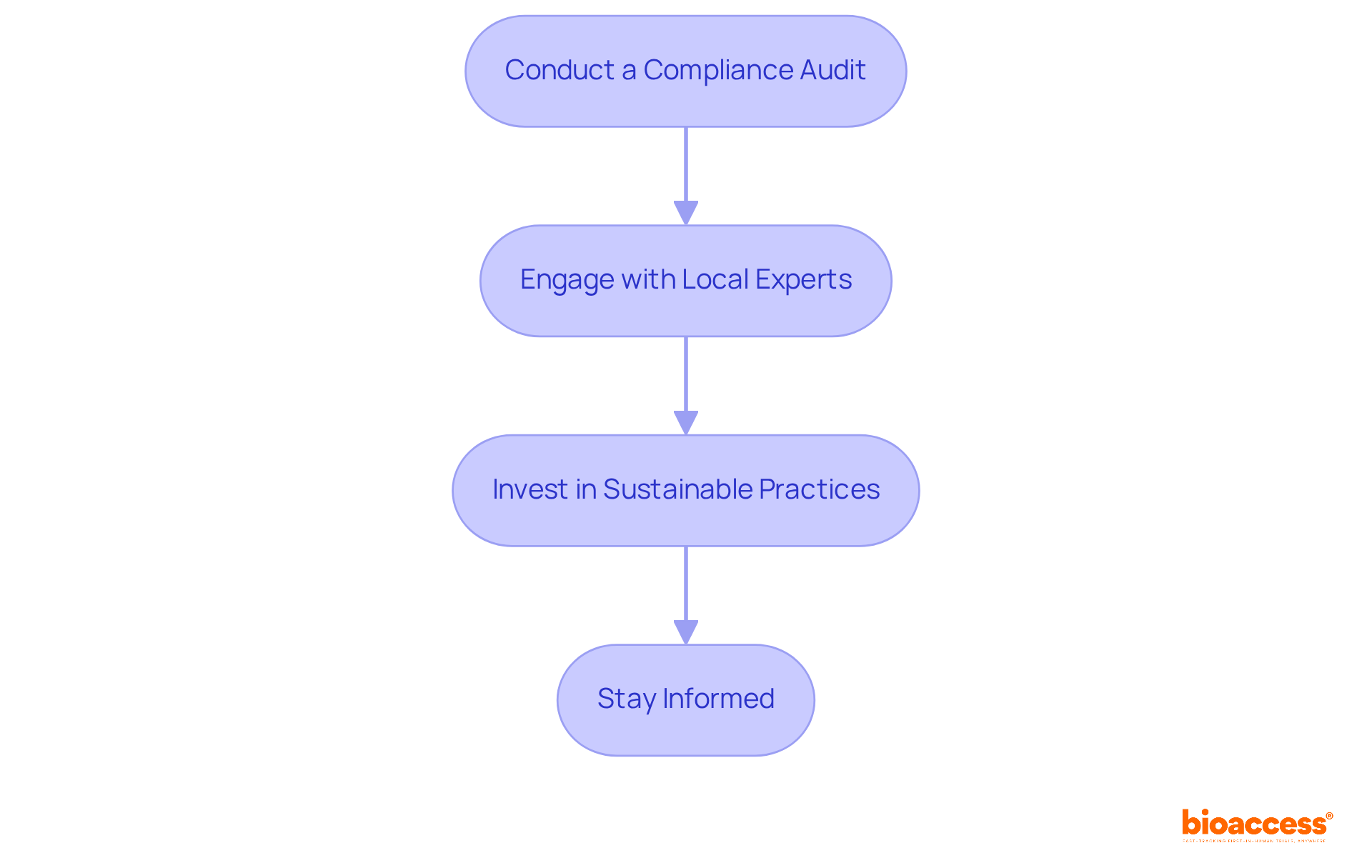
Post-Market Surveillance: Ensuring Safety for Class II Medical Devices
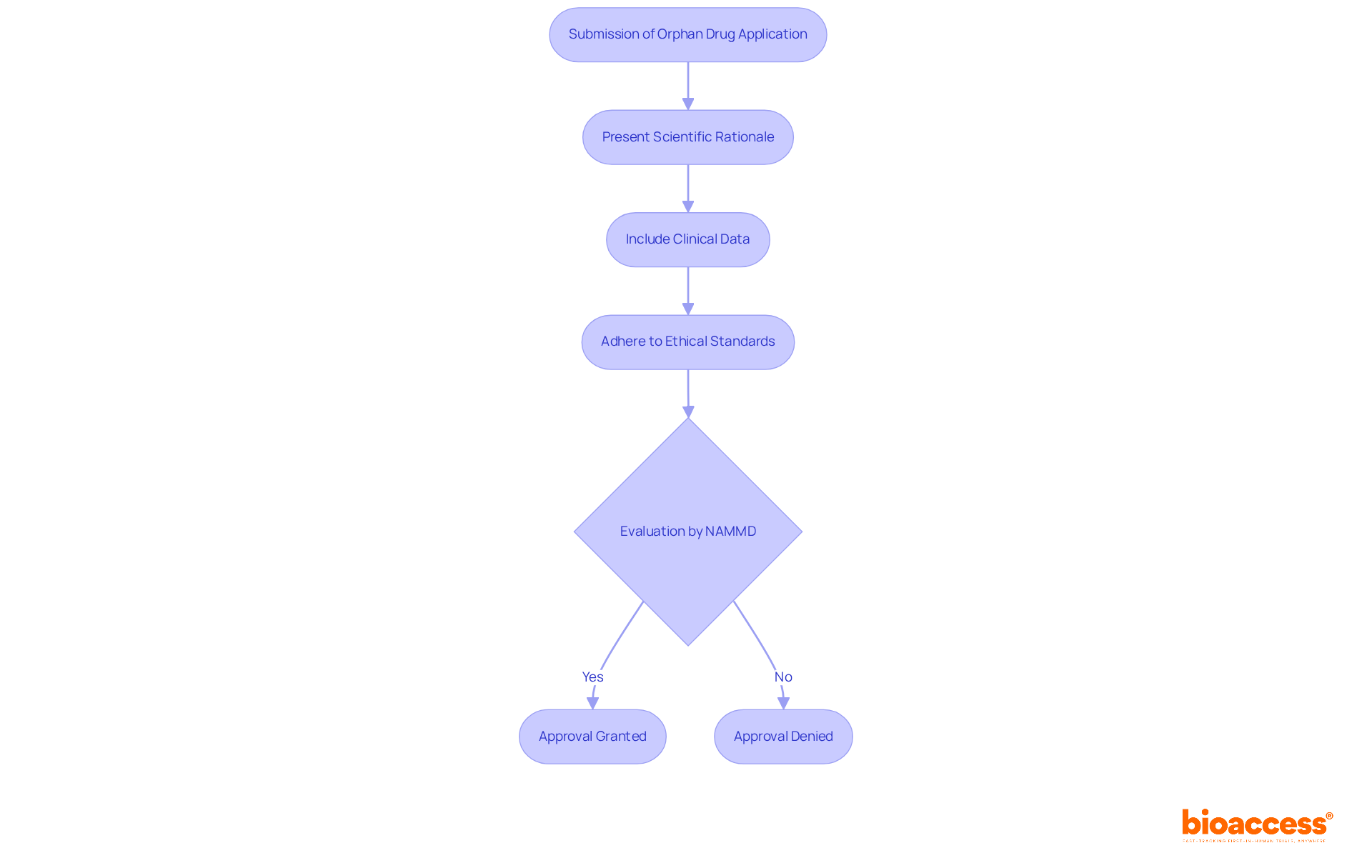
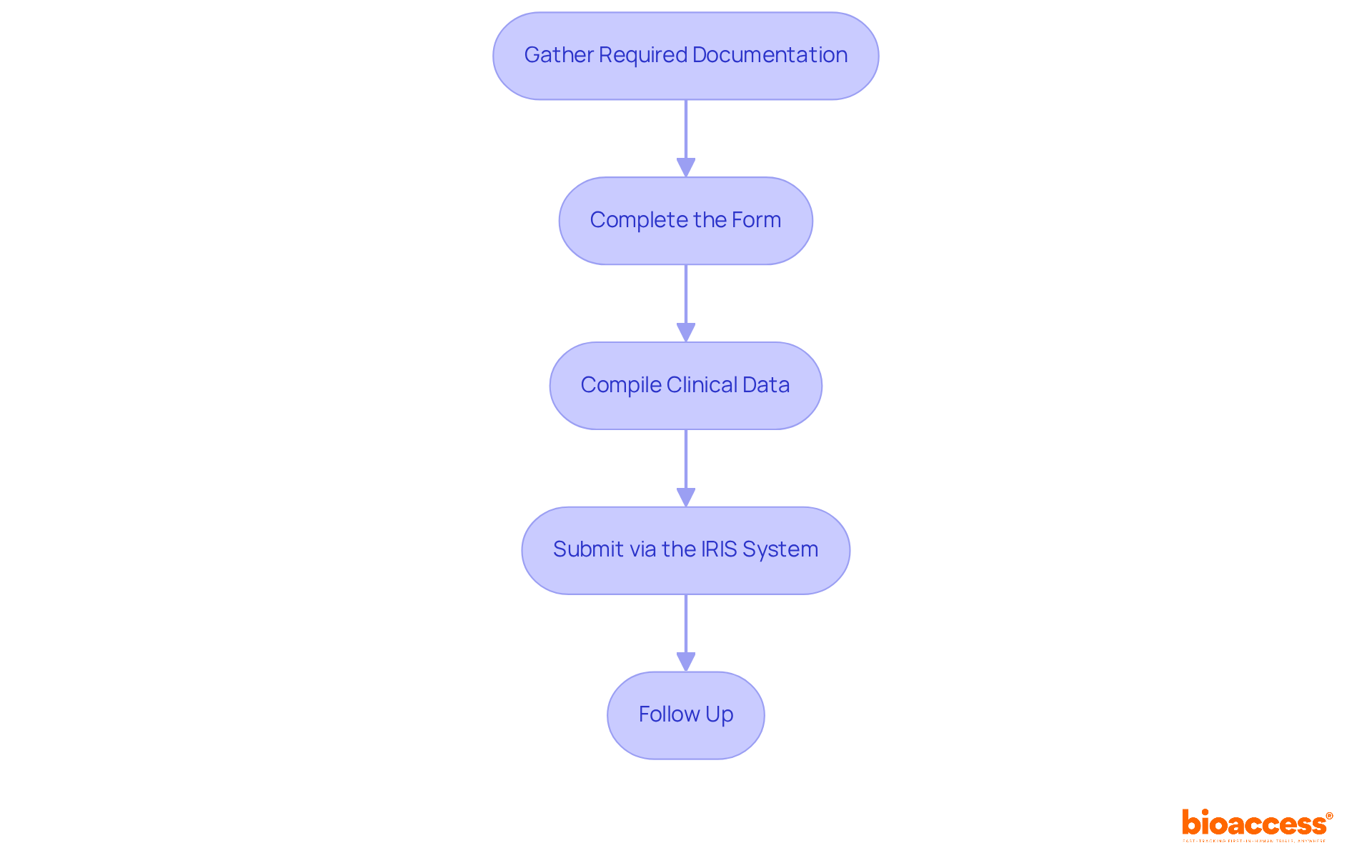
is essential for the continuous monitoring of following their approval for use. Producers are required to report negative incidents swiftly and conduct regular to ensure compliance with established standards. This proactive vigilance is crucial, as it enables the early identification of potential issues, allowing for timely corrective actions that safeguard patient health. Notably, approximately 70% of medical devices in Brazil are under continuous supervision, underscoring the significance of adhering to .
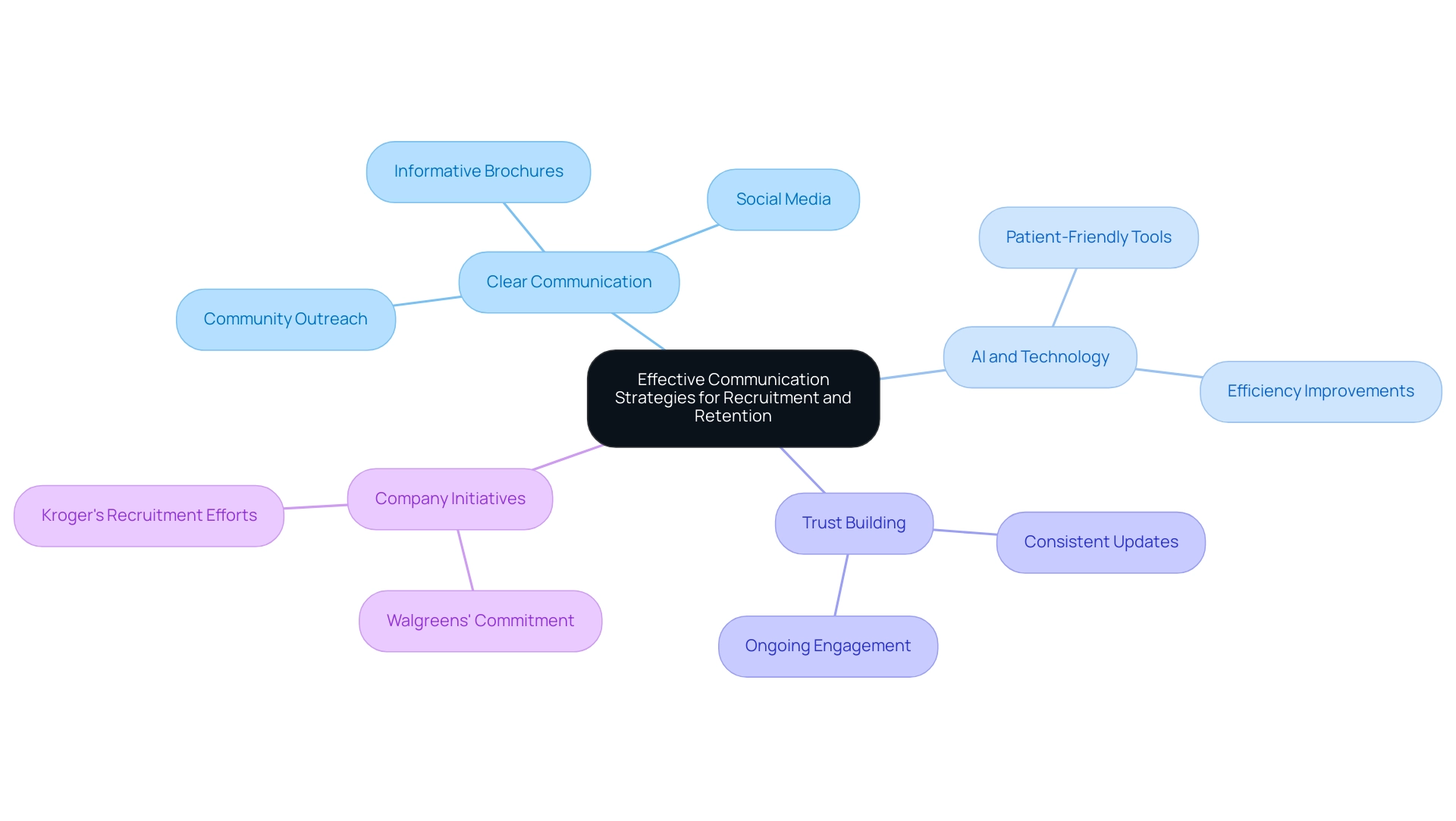
Effective communication with governing organizations, such as ANVISA, is vital for ensuring compliance and fostering collaboration. Organizations that implement structured training programs and robust reporting systems experience significantly higher compliance rates, thereby enhancing their ability to manage adverse events effectively.
The Periodic Safety Update Report (PSUR) serves as a critical tool for systematic risk management for . As the oversight environment evolves, remaining knowledgeable and adaptable in compliance approaches is essential for manufacturers to navigate the complexities of post-market monitoring effectively.
With experts like Ana Criado and Katherine Ruiz, who possess extensive experience in regulatory affairs and biomedical engineering, organizations can gain a deeper understanding of these requirements and .
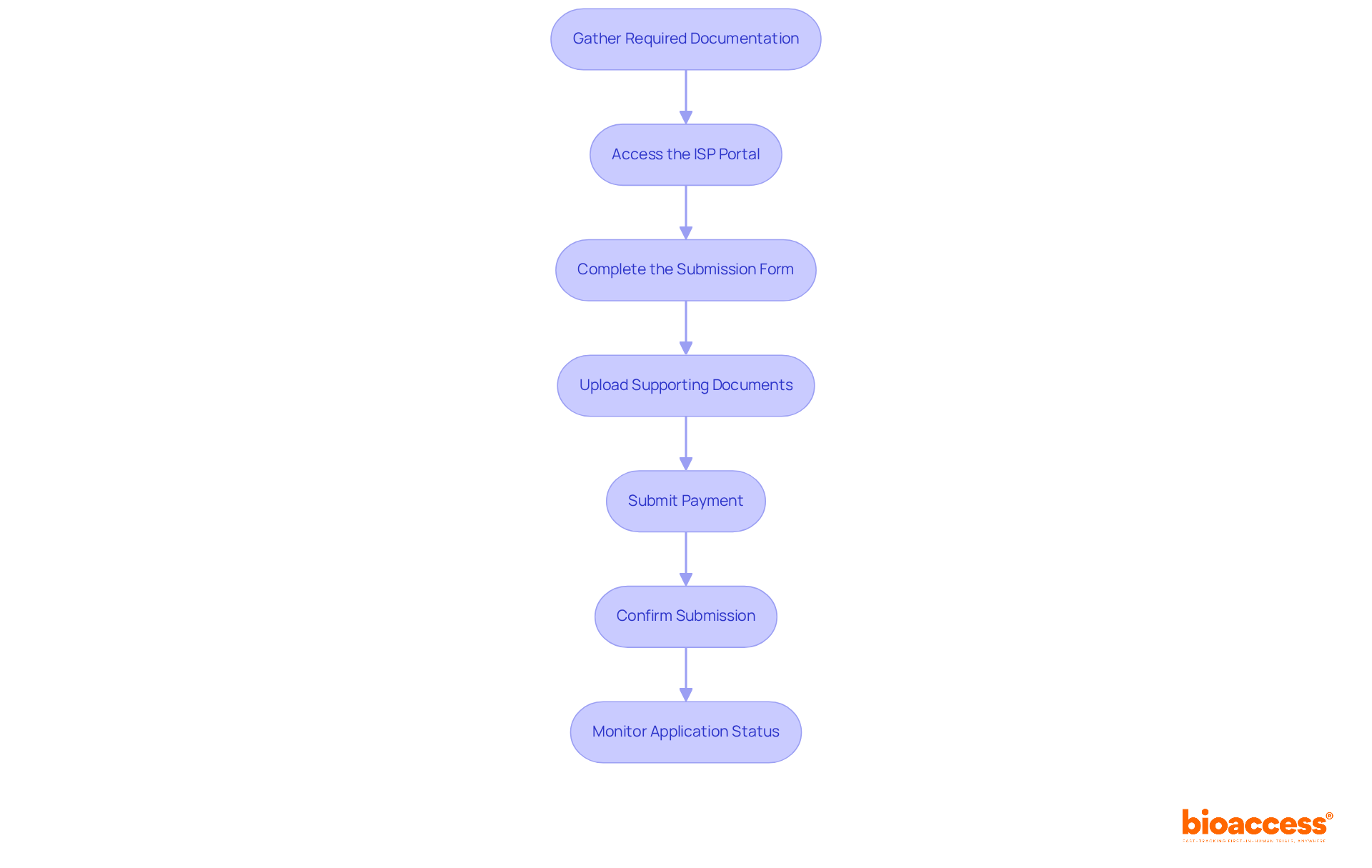
 process. Follow the arrows to see how monitoring and communication lead to safety and compliance. Each box represents a critical step in the post-market surveillance process. Follow the arrows to see how monitoring and communication lead to safety and compliance.](https://images.tely.ai/telyai/jfnquvoo-each-box-represents-a-critical-step-in-the-post-market-surveillance-process-follow-the-arrows-to-see-how-monitoring-and-communication-lead-to-safety-and-compliance.webp)
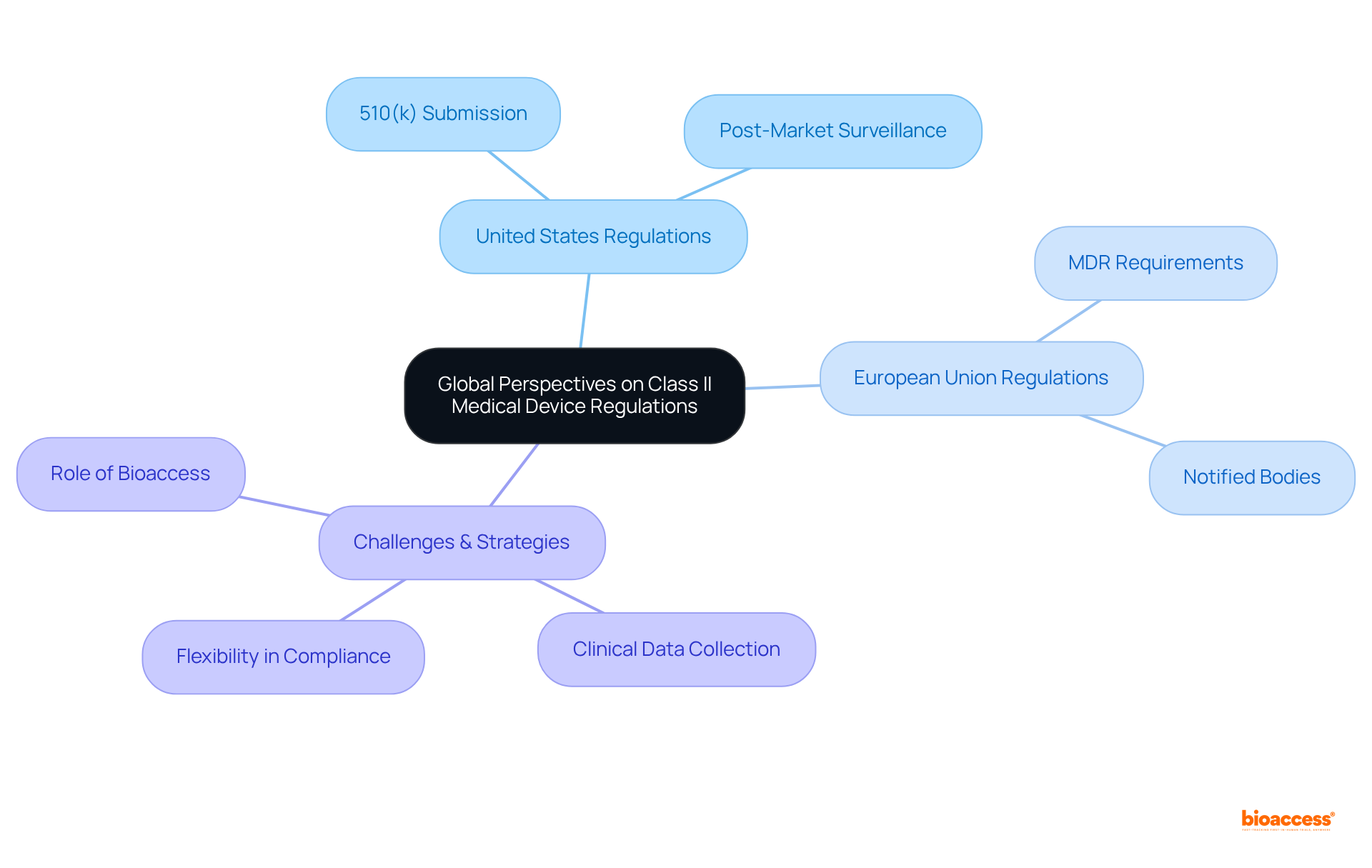
Global Perspectives: Class II Medical Device Regulations Around the World
Regulations for reveal significant differences among nations, necessitating a sophisticated comprehension from producers. In the United States, the to demonstrate substantial equivalence to a legally marketed product, thereby ensuring safety and effectiveness. Conversely, the European Union operates under the , which imposes stricter requirements for pre-market assessments and ongoing . This governing framework, effective since May 2021, has elevated the standards for and compliance, compelling manufacturers to adapt their strategies for successful market entry.
Statistics indicate that account for 43% of all medical devices in the U.S., underscoring their market significance. However, presents challenges; manufacturers must continuously collect and submit reports to Notified Bodies in the EU, while also adhering to the FDA’s requirements. Industry leaders emphasize the necessity of understanding these international regulatory differences to mitigate risks and enhance market access. For instance, the FDA’s Unique Identification (UDI) system enhances traceability, whereas the EU’s EUDAMED system aims to improve transparency in the regulation of products.
As manufacturers strive to comply with these diverse regulations, they often implement tailored strategies to meet the unique requirements of each market. This flexibility is essential for ensuring that innovative can reach patients safely and contribute to improved healthcare outcomes worldwide. Comprehensive , such as those offered by bioaccess, are invaluable in this process. From feasibility studies and site selection to , trial setup, import permits, and nationalization of investigational equipment, bioaccess provides essential assistance in navigating the complexities of regulatory requirements. Their expertise in project management and reporting ensures that these tools can effectively reach patients, ultimately enhancing healthcare outcomes. To learn more about how bioaccess can assist with your , consider booking a meeting with our experts.
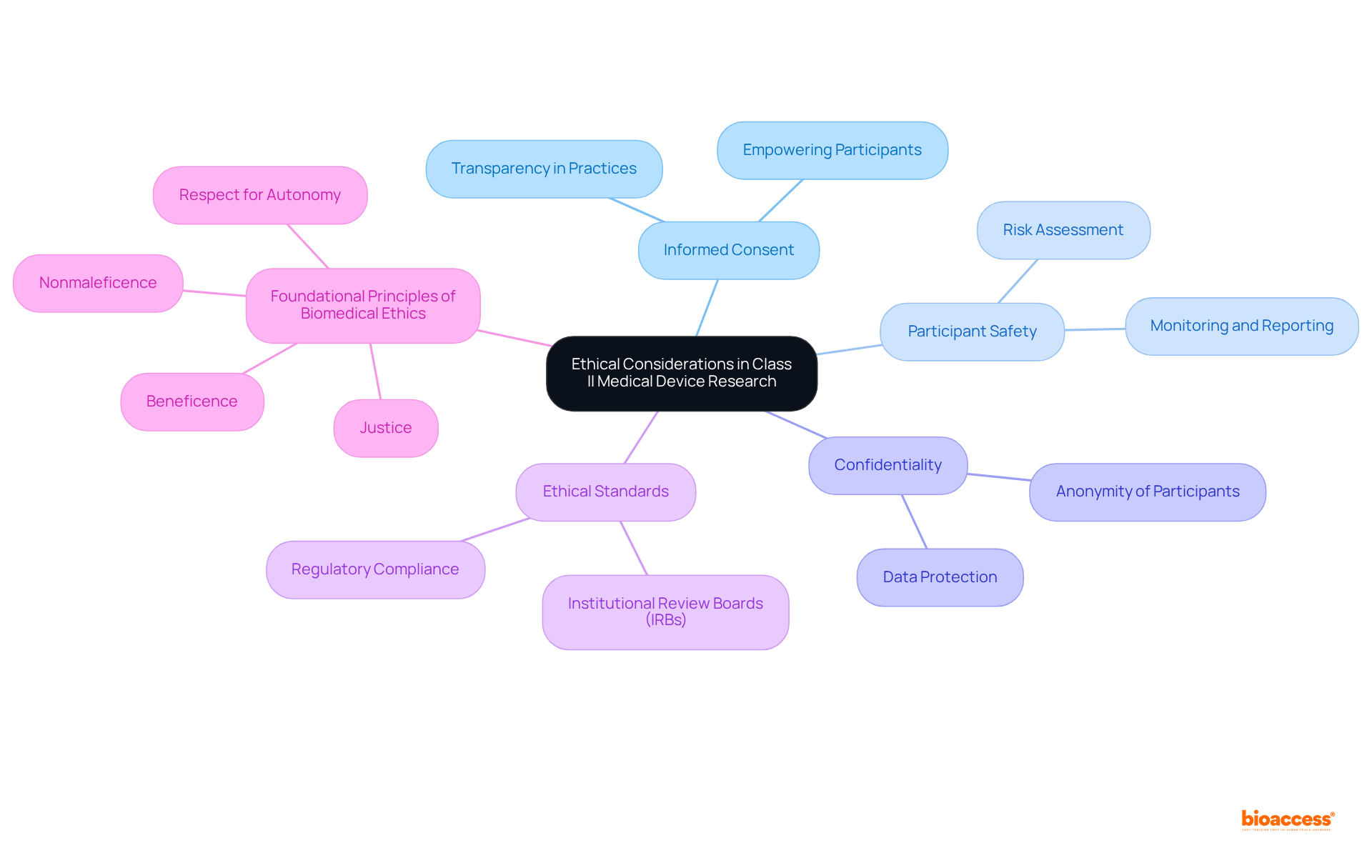
Ethical Considerations in Class II Medical Device Research and Approval
are paramount, focusing on the acquisition of , ensuring participant safety, and maintaining confidentiality. Researchers are required to adhere strictly to the established by regulatory authorities and institutional review boards (IRBs), which are essential in safeguarding clinical equipoise during equipment testing.
Adhering to these is vital for cultivating trust between researchers and participants, thereby enhancing the integrity of the research process. practices must be transparent and comprehensive, empowering participants to make informed decisions regarding their involvement. This approach not only protects participant rights but also fortifies the ethical foundation of .
As bioethicists emphasize, prioritizing and participant safety is crucial for the credibility of , especially regarding , where balancing innovation with ethical responsibility is imperative. Moreover, the —beneficence, nonmaleficence, respect for autonomy, and justice—should guide researchers in fulfilling their ethical obligations.
The World Medical Association Declaration of Helsinki also provides essential ethical principles for research involving human subjects, underscoring the . By integrating these principles and guidelines, researchers can ensure that their studies not only advance healthcare technology but also uphold the highest .
Additionally, bioaccess offers comprehensive that encompass feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting. These services are designed to aid researchers in navigating the complex compliance landscape, ensuring that are met throughout the research process.
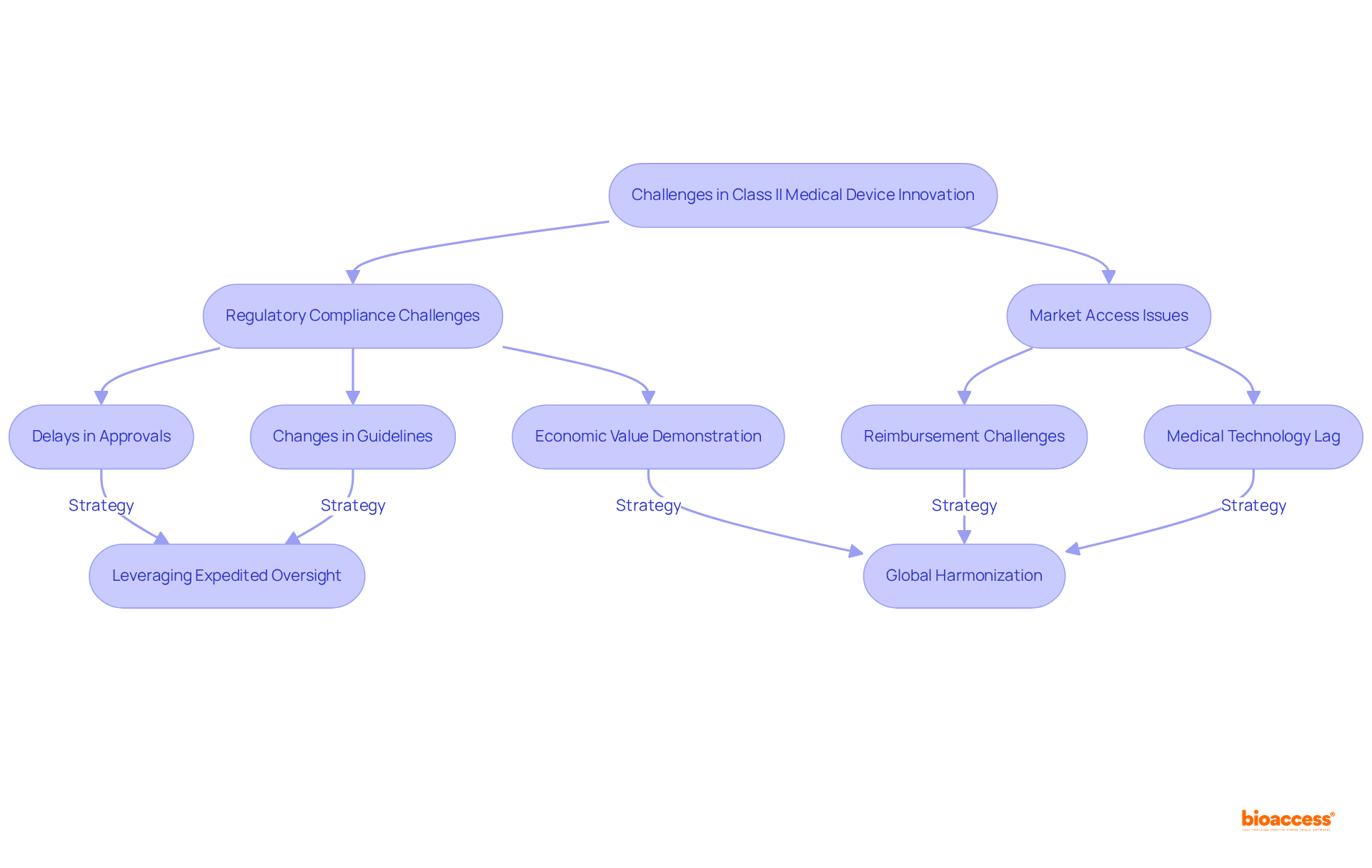
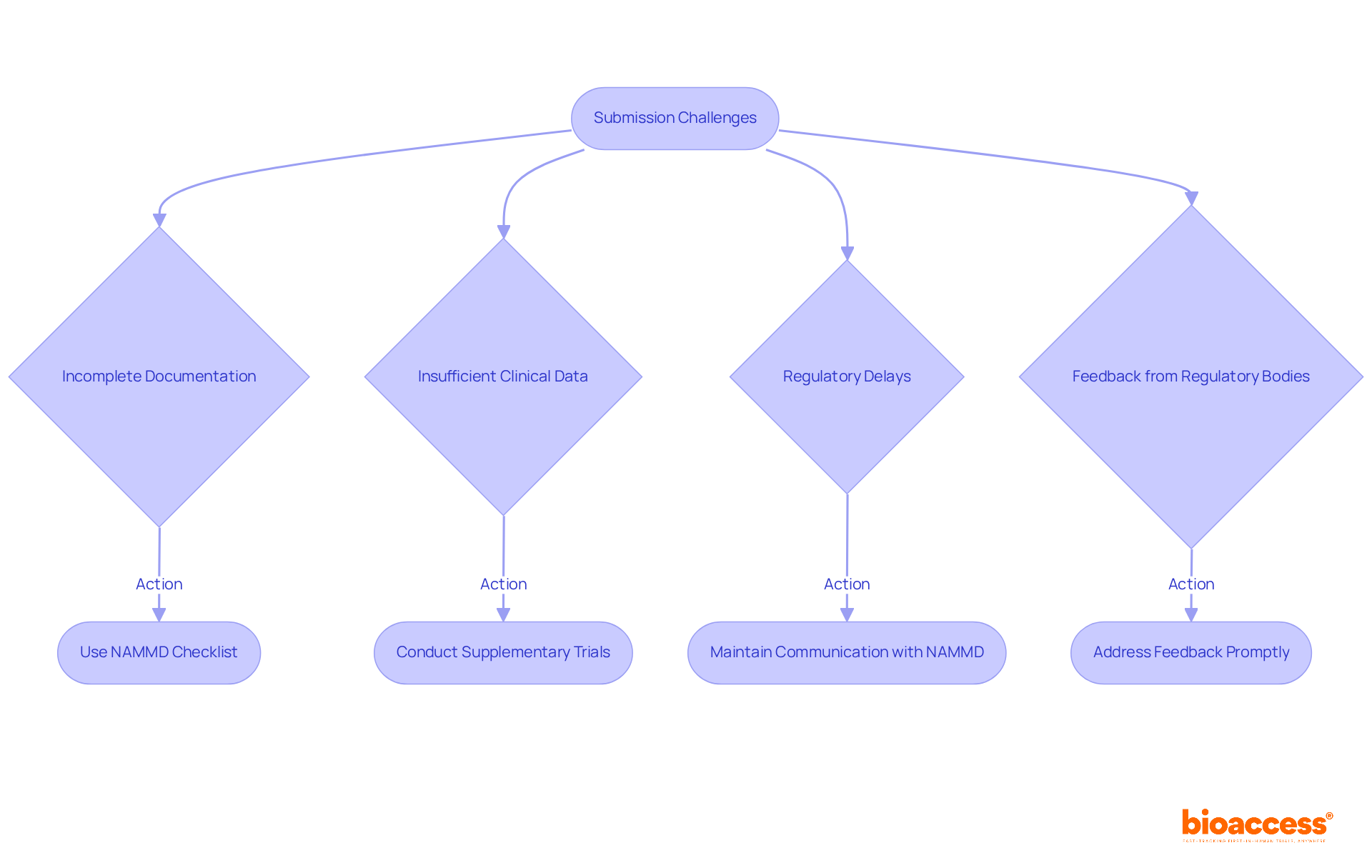
Challenges in Innovating Class II Medical Devices: Regulatory Hurdles and Market Access
Creating requires and market entry hurdles. Manufacturers frequently face considerable delays in securing approvals due to rigorous regulatory demands and unforeseen guideline changes. For instance, the , which allows market access by demonstrating substantial equivalence to existing products, can be a lengthy endeavor, particularly when additional information or modifications are necessary. Furthermore, poses another significant challenge, as many manufacturers struggle to in a competitive landscape.
Statistics indicate that compliance challenges can lead to a ‘,’ delaying access to innovative advancements. This delay is particularly pronounced in , which are categorized as moderate risk and requiring more oversight compared to Class I items. Industry experts underscore the necessity of comprehending these challenges and formulating proactive strategies to tackle them. For example, leveraging the expedited oversight in regions like can facilitate quicker approvals and market entry, as evidenced by , which achieves enrollment 50% faster than traditional markets.
Recent insights from industry leaders emphasize the critical need for to streamline approval processes and minimize discrepancies that impede market entry. As the healthcare equipment landscape continues to evolve, manufacturers must remain adaptable and informed to effectively navigate the regulatory environment and seize market opportunities.
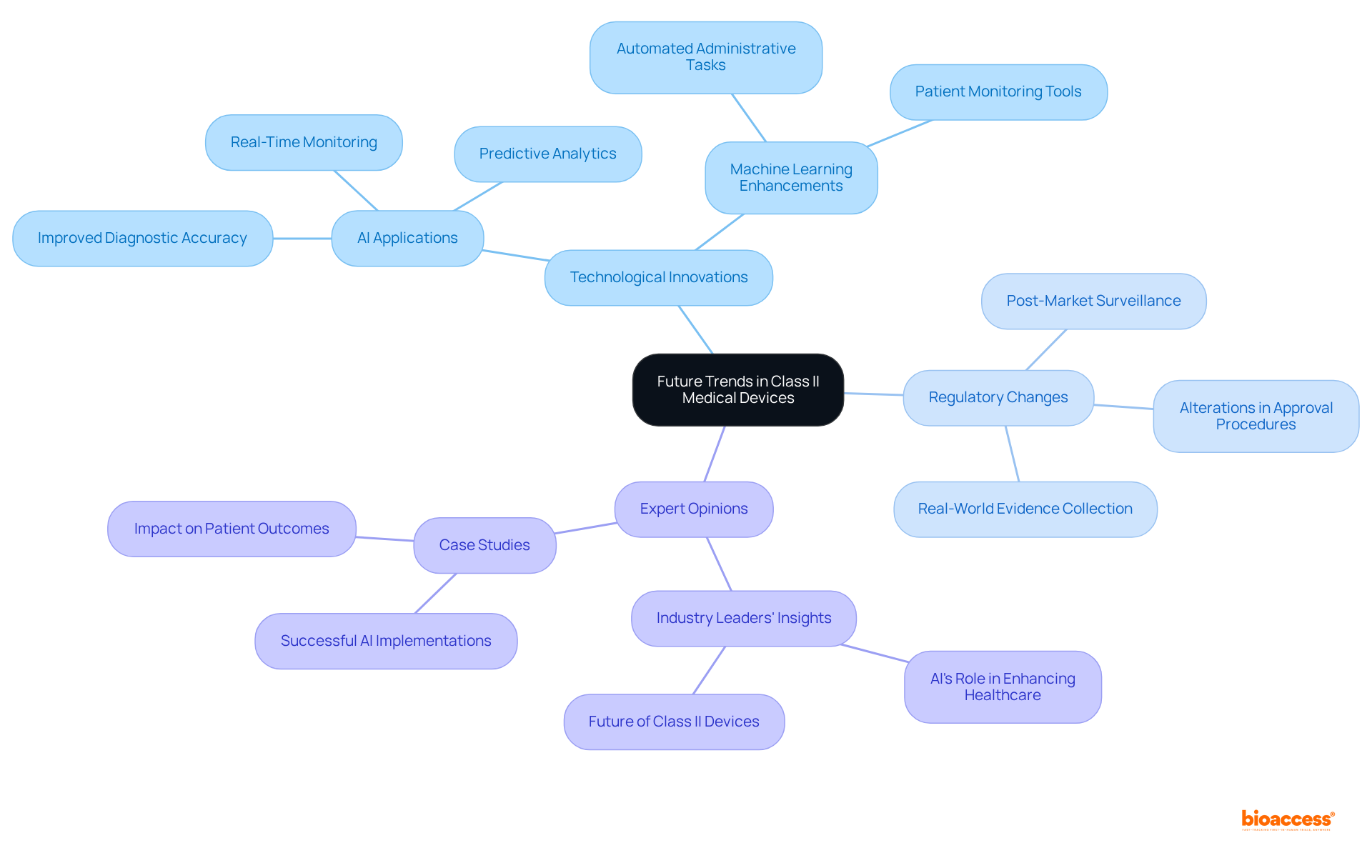
Future Trends in Class II Medical Devices: Innovations and Regulatory Changes
The future of is set for transformation through the integration of , particularly artificial intelligence (AI) and machine learning (ML). These innovations not only enhance design and functionality but also streamline processes such as and real-time monitoring. For instance, AI algorithms are increasingly utilized to improve diagnostic accuracy and automate administrative tasks, significantly reducing the burden on healthcare providers.
Regulatory bodies are adapting to these technological advancements by placing greater emphasis on and the collection of . This shift may lead to alterations in approval procedures, ensuring that products remain effective and safe throughout their lifecycle. Consequently, manufacturers must stay vigilant and responsive to these evolving regulatory landscapes to maintain compliance and foster innovation.
Statistics indicate that the integration of AI in medical technology development is accelerating, with a notable increase in since 2018. This trend underscores the growing reliance on AI technologies in healthcare, particularly in the segment, where instruments often undergo the , emphasizing substantial equivalence to existing products.
Case studies highlight the successful application of AI in various , demonstrating how these technologies can enhance and operational efficiency. For example, AI-driven tools are being developed to assist in real-time patient monitoring and , which are crucial for timely interventions.
Expert opinions suggest that the future of will increasingly depend on the capacity to leverage AI and ML for innovation. As noted by industry leaders, the potential for AI to enhance healthcare services and is significant, with many anticipating that these technologies will become essential to the development and operation of future health instruments.
In conclusion, staying informed about these trends and actively engaging in the evolving will be vital for producers aiming to thrive in the competitive arena of .
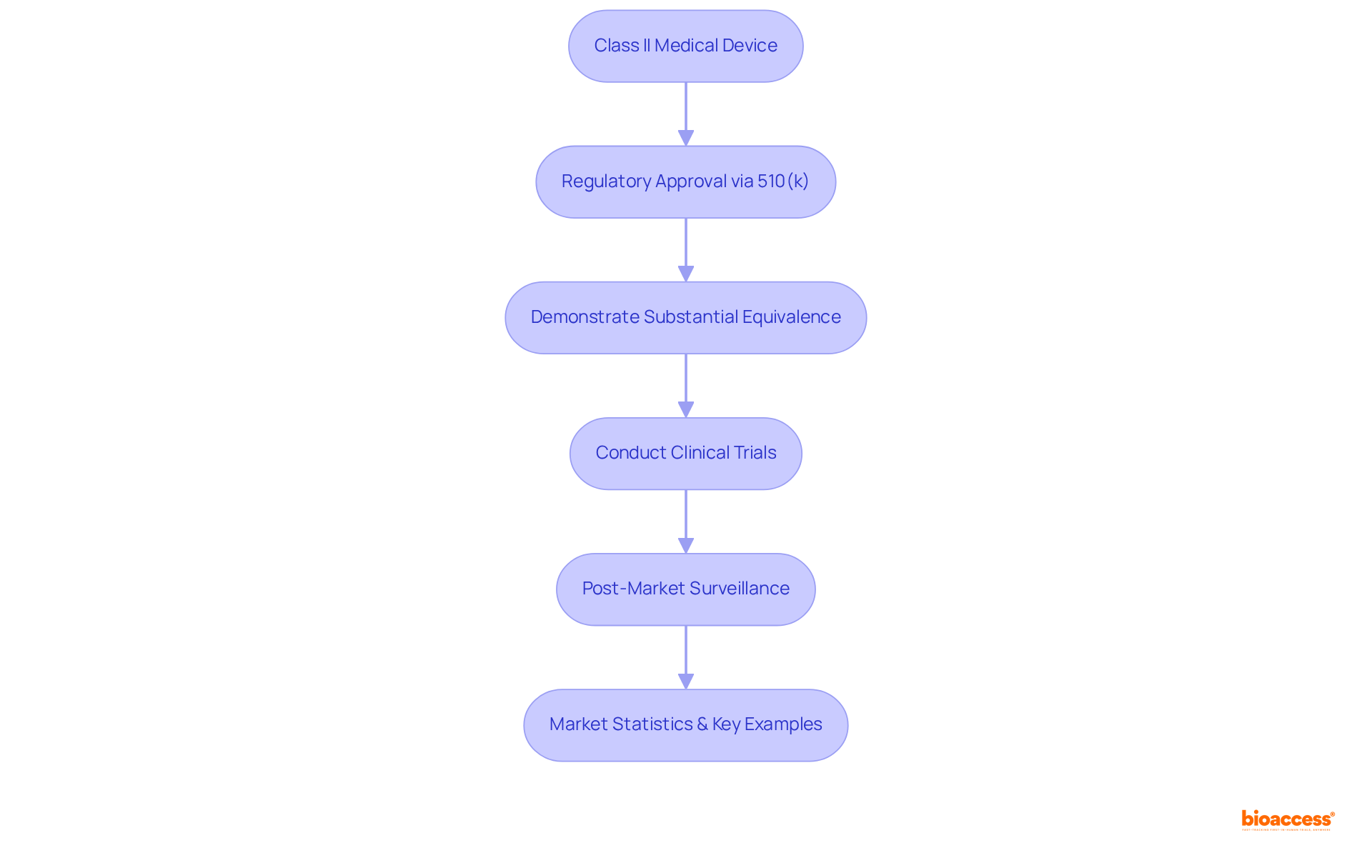
Key Takeaways: Essential Facts About Class II Medical Devices
, categorized as moderate-risk products, require primarily through the . This pathway mandates that manufacturers demonstrate to existing products, thereby ensuring safety and efficacy. play a vital role in this process, providing the essential data needed to validate product performance prior to market entry. Furthermore, is crucial for ongoing compliance, allowing manufacturers to monitor product performance and address any that may arise after commercialization.
The global market for Category II medical instruments is robust, with these instruments accounting for approximately 43% of all applications. Noteworthy examples include blood pressure cuffs, pregnancy tests, and powered wheelchairs, illustrating the diverse range of products within this category. The market is anticipated to experience significant growth, propelled by advancements in technology and an increasing demand for innovative healthcare solutions.
Successful navigation of the is exemplified by companies that have adeptly leveraged existing predicate products to accelerate their market entry. For instance, the FDA’s streamlined review process has enabled numerous Class II products to reach the market more swiftly than their Class III counterparts, which require more extensive clinical data.
Experts emphasize the importance of understanding , as the FDA’s centralized approval system contrasts with the EU’s decentralized approach, where approvals can differ substantially among member states. This complexity necessitates that manufacturers remain vigilant to changes in regulations and market dynamics to ensure compliance and maintain a competitive advantage.
In conclusion, the landscape for is marked by a mix of compliance challenges and opportunities. Staying informed about market trends, regulatory updates, and the significance of and is essential for manufacturers striving for success in this evolving sector. To enhance your and improve your chances of successful market entry, consider leveraging bioaccess®’s expertise in .
Conclusion
The journey of class II medical devices from conception to market is a complex yet vital process that necessitates a thorough understanding of regulatory frameworks, clinical trials, and ongoing compliance. This article has underscored the critical aspects contributing to the success of these devices, emphasizing the necessity of navigating the 510(k) approval process, leveraging clinical trials for robust data collection, and implementing effective post-market surveillance to ensure patient safety and device efficacy.
Key insights reveal significant cost efficiencies and expedited timelines available through conducting clinical trials in regions like Colombia, the imperative to understand international regulatory differences, and the ethical considerations that must guide research practices. Furthermore, the growing integration of advanced technologies such as AI and machine learning is poised to transform the landscape of class II medical devices, paving the way for innovative solutions that enhance patient outcomes and streamline regulatory compliance.
As the market for class II medical devices continues to evolve, it is essential for manufacturers to stay informed about these trends and actively engage with compliance requirements. Embracing the opportunities presented by accelerated clinical research and comprehending the regulatory landscape can lead to successful market entry and improved healthcare solutions. The future of class II medical devices is promising, and those who adapt to these changes will undoubtedly play a crucial role in advancing medical technology for the benefit of patients worldwide.
Frequently Asked Questions
What is bioaccess® and what does it offer for clinical research?
bioaccess® is an organization that accelerates clinical research specifically for class II medical devices, leveraging its expertise and regional advantages in Colombia, which is known for its cost efficiency and fast oversight processes.
Why is Colombia considered a prime location for clinical trials?
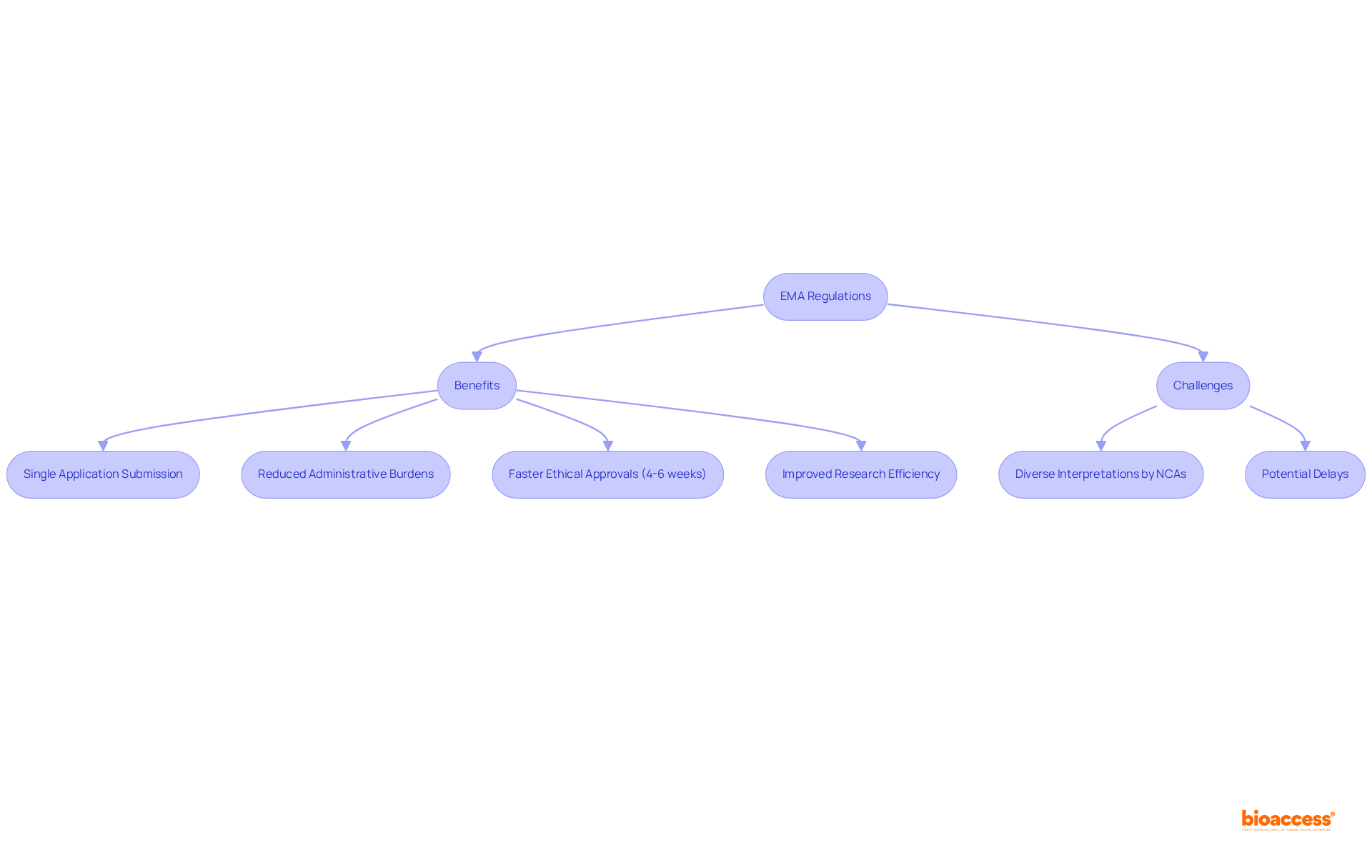
Colombia offers significant cost savings exceeding 30% compared to North America or Western Europe, exceptional speed in ethical approvals (4-6 weeks), a high-quality healthcare system, and a diverse patient population of over 50 million, with 95% covered by universal healthcare.
What are the tax incentives available for R&D in Colombia?
Colombia provides substantial R&D tax incentives, including a 100% tax deduction for investments in science, technology, and innovation projects, enhancing its attractiveness for conducting clinical trials.
What is the classification of Class II medical devices?
Class II medical devices are healthcare items that pose a moderate risk to patients and require specific regulatory measures to ensure safety and efficacy. Examples include infusion pumps, powered wheelchairs, surgical drapes, and diagnostic imaging equipment.
What is the significance of the 510(k) submission for Class II medical devices?
The 510(k) submission is crucial as it demonstrates that the new device is substantially equivalent to an existing, legally marketed product, ensuring it meets safety and efficacy standards before market introduction.
What role does INVIMA play in Colombia’s regulatory landscape for medical devices?
INVIMA (Instituto Nacional de Vigilancia de Medicamentos y Alimentos) oversees the marketing and production of health products, ensuring compliance with technical standards and safety criteria. It is recognized as a Level 4 health authority by PAHO/WHO.
What are the common challenges in the approval process for Class II medical devices?
Common challenges include the need for comprehensive data validating safety and effectiveness in the 510(k) application, potential requests for additional information during the review process, and the 32% likelihood of submissions failing to meet minimum acceptability checks.
What services does bioaccess provide to support the clinical trial process?
bioaccess offers extensive clinical trial management services, including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting, all crucial for maintaining regulatory compliance.
How long does the 510(k) review process typically take?
The 510(k) review process typically aims for a decision within 90 days, although delays may occur due to incomplete submissions or the need for further clarification.
List of Sources
- bioaccess®: Accelerating Clinical Research for Class II Medical Devices
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- bioaccessla.com (https://bioaccessla.com/news)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC6113340)
- 6 Important Quotes from Medtech Leaders (https://mddionline.com/business/6-important-quotes-from-medtech-leaders)
- 7 quotes from 2023 to guide you on the medtech market (https://tiinatyni.com/blogi/7-quotes-from-2023-to-guide-you-on-the-medtech-market)
- Understanding Class II Medical Devices: Definition and Classification
- greenlight.guru (https://greenlight.guru/blog/what-exempt-means-medical-devices-regulatory)
- US FDA vs EU Medical Device Classification Guide (https://emergobyul.com/news/us-fda-and-eu-risk-classification-medical-devices)
- FDA Class II Medical Device Regulations and Compliance – Ketryx Compliance Framework (https://ketryx.com/blog/fda-class-ii-medical-device-regulations-and-compliance)
- What is a Class 2 Medical Device in the US? [+Examples] (https://greenlight.guru/blog/class-2-medical-device)
- FDA Authorization of Medical Devices (https://jamanetwork.com/journals/jama/fullarticle/1817798)
- Navigating the Approval Process for Class II Medical Devices
- thefdagroup.com (https://thefdagroup.com/blog/510k-explained)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC8920055)
- emergobyul.com (https://emergobyul.com/news/how-long-fda-review-process-510k-medical-device-submissions)
- 5 Medical Device Regulatory Approval Statistics You Need to Know – Arrotek | Medical Device Innovation (https://arrotek.com/5-medical-device-regulatory-approval-statistics-you-need-to-know)
- medtechdive.com (https://medtechdive.com/news/fda-turnaround-510k-record-approval-waits/687476)
- The Role of Clinical Trials in Class II Medical Device Development
- Case Study 28 : Registration of Class II Medical Device in Australia, Brazil, China, India, Pakistan and Russia (https://globalregulatorypartners.com/case_studies/case-study-28-registration-of-class-ii-medical-device-in-australia-brazil-china-india-pakistan-and-russia)
- Quotes About Medical Research (https://changingthepresent.org/pages/quotes-about-medical-research)
- qualtrics.com (https://qualtrics.com/blog/research-quotes)
- 30 Quotes About the Future of Healthcare: Expert Takes (https://deliberatedirections.com/quotes-future-of-healthcare)
- Post-Market Surveillance: Ensuring Safety for Class II Medical Devices
- 7 Key Insights on Post-Market Surveillance under ANVISA | bioaccess® (https://bioaccessla.com/blog/7-key-insights-on-post-market-surveillance-under-anvisa)
- Global Perspectives: Class II Medical Device Regulations Around the World
- US FDA vs EU Medical Device Classification Guide (https://emergobyul.com/news/us-fda-and-eu-risk-classification-medical-devices)
- Global Regulatory Challenges for Medical Devices: Impact on Innovation and Market Access (https://mdpi.com/2076-3417/14/20/9304)
- rimsys.io (https://rimsys.io/blog/fda-class-ii-medical-devices)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S0020138323005946)
- Ethical Considerations in Class II Medical Device Research and Approval
- journalofethics.ama-assn.org (https://journalofethics.ama-assn.org/article/patient-centered-ethical-approach-medical-device-innovation/2010-02)
- Challenges in Innovating Class II Medical Devices: Regulatory Hurdles and Market Access
- Global Regulatory Challenges for Medical Devices: Impact on Innovation and Market Access (https://mdpi.com/2076-3417/14/20/9304)
- 6 Important Quotes from Medtech Leaders (https://mddionline.com/business/6-important-quotes-from-medtech-leaders)
- 37 Quotes on Health Care and Health Tech from 2016 (https://medium.com/@r_scott_munro/37-quotes-on-health-care-and-health-tech-from-2016-e34576edfc1f)
- atsindustrialautomation.com (https://atsindustrialautomation.com/case_studies/achieving-micron-level-accuracy-in-medical-device-manufacturing)
- Future Trends in Class II Medical Devices: Innovations and Regulatory Changes
- mdpi.com (https://mdpi.com/2079-9292/13/3/498)
- 50+ AI in Healthcare Statistics 2024 · AIPRM (https://aiprm.com/ai-in-healthcare-statistics)
- binariks.com (https://binariks.com/blog/artificial-intelligence-ai-healthcare-market)
- pathwaymedtech.com (https://pathwaymedtech.com/future-medical-technology-trends)
- 30 Quotes About the Future of Healthcare: Expert Takes (https://deliberatedirections.com/quotes-future-of-healthcare)
- Key Takeaways: Essential Facts About Class II Medical Devices
- Medical Devices Statistics and Facts (2026) (https://media.market.us/medical-devices-statistics)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S2452302X16300638)
- FDA Authorization of Medical Devices (https://jamanetwork.com/journals/jama/fullarticle/1817798)
- What is a Class 2 Medical Device in the US? [+Examples] (https://greenlight.guru/blog/class-2-medical-device)









 process. Follow the arrows to see how monitoring and communication lead to safety and compliance. Each box represents a critical step in the post-market surveillance process. Follow the arrows to see how monitoring and communication lead to safety and compliance.](https://images.tely.ai/telyai/jfnquvoo-each-box-represents-a-critical-step-in-the-post-market-surveillance-process-follow-the-arrows-to-see-how-monitoring-and-communication-lead-to-safety-and-compliance.webp)