Introduction
Navigating the intricate landscape of EU IVDR compliance presents a formidable challenge for Medtech innovators, particularly as the 2025 deadline approaches. With stringent regulations designed to ensure the safety and effectiveness of in vitro diagnostic devices, companies must not only grasp the new standards but also strategically position themselves to meet them. This article unveils ten essential insights that will empower innovators to streamline their compliance processes, enhance product quality, and ultimately secure a competitive edge in the evolving medical technology market.
How can Medtech firms effectively adapt to these changes and thrive amidst the regulatory complexities?
bioaccess®: Accelerate EU IVDR Compliance for Medtech Innovations
bioaccess® leverages its extensive expertise in early-stage clinical research to expedite for . By harnessing the and the diverse patient populations of the Balkans, bioaccess® achieves . This rapid turnaround is crucial for businesses striving to navigate stringent regulatory requirements while sustaining their competitive edge in the market. Given that the average time to market for medical products is significantly impacted by , bioaccess®’s strategy not only accelerates the process but also enhances the likelihood of successful market entry.
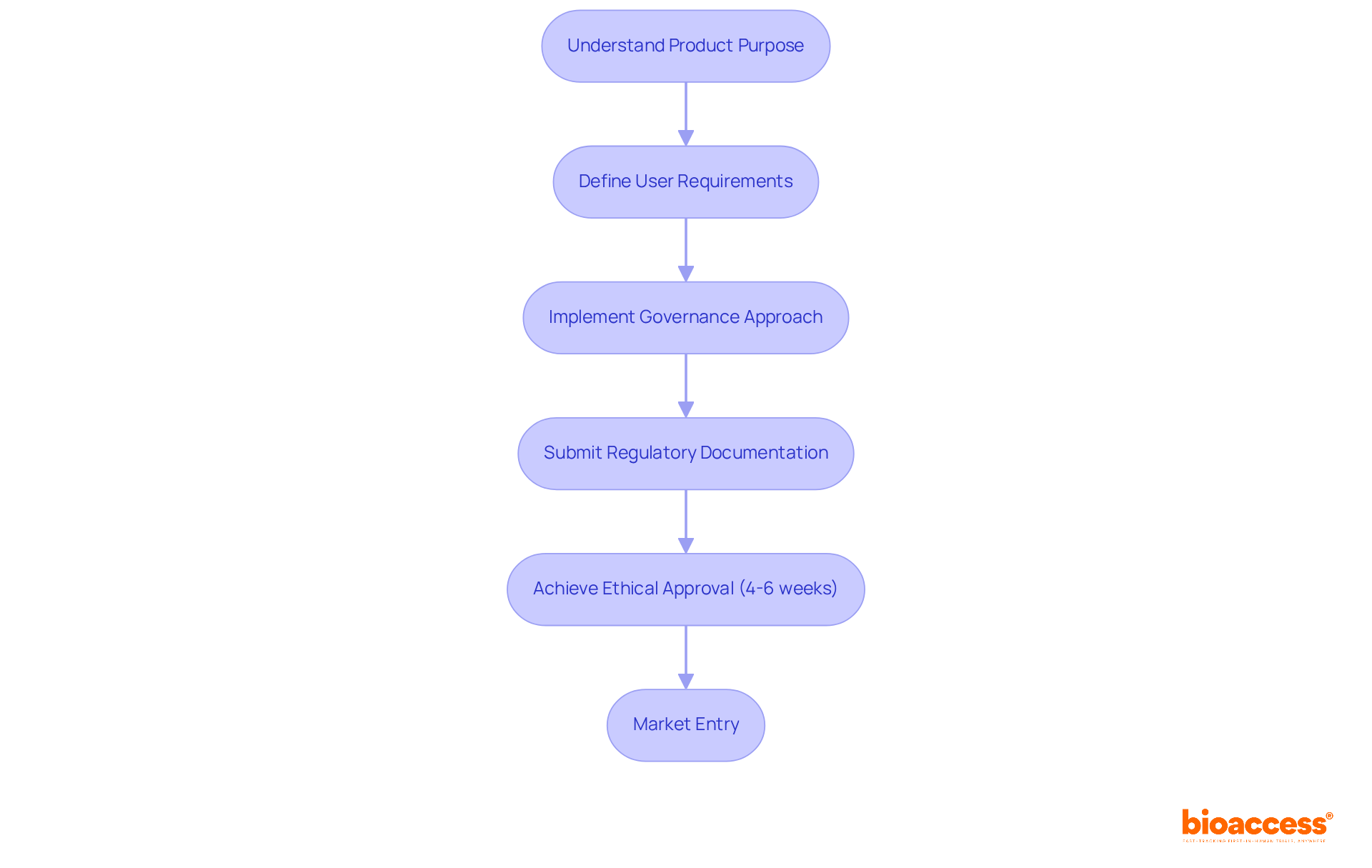
Industry leaders emphasize that a well-defined governance approach, beginning with a and user requirements, is vital for compliance and overall business success. As the transition period for the extends until 2027/2028, must proactively engage in adherence efforts to prevent market delays. The recent case study of Avantec Vascular, which selected bioaccess™ for its of an innovative vascular device in Latin America, exemplifies how bioaccess supports companies in effectively navigating regulatory submissions and site activations. This collaboration highlights the strategic advantage of partnering with bioaccess® to ensure .
In this evolving landscape, bioaccess® positions itself as a strategic ally, assisting Medtech firms in adeptly maneuvering through the complexities of EU IVDR compliance with both agility and confidence.
Understanding the EU IVDR: Key Objectives and Scope
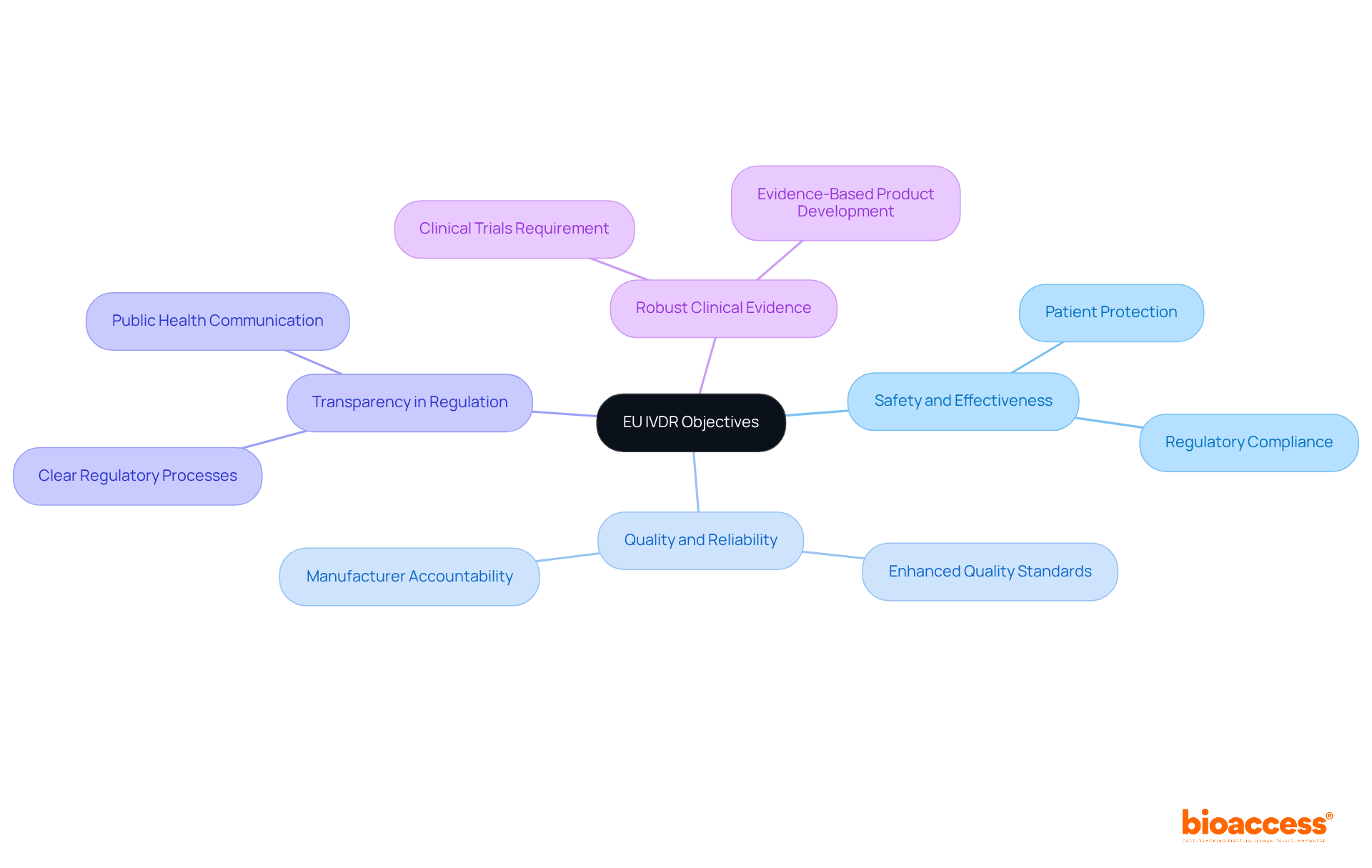
The eu ivdr aims to ensure the safety and effectiveness of in vitro diagnostic tools for patients. Its primary objectives include enhancing the quality and reliability of IVDs, increasing transparency in the , and requiring manufacturers to provide robust to substantiate their claims. The regulation covers a wide range of instruments, from simple tests to complex diagnostics, and enforces strict adherence measures designed to protect public health.
As of 2025, compliance rates for IVDs under the are anticipated to increase substantially, demonstrating the industry’s adjustment to these stringent standards. , such as Ana Criado, Director of Compliance Affairs at bioaccess, and Katherine Ruiz, an expert in compliance for medical devices and , stress that the new regulation not only aligns the compliance framework across the EU but also imposes increased responsibility on producers to prove that their products satisfy rigorous safety and performance standards. Ana Criado observes, “The signify a major change in oversight expectations, urging manufacturers to emphasize .” This shift is crucial for maintaining market access and ensuring that IVDs contribute positively to patient outcomes.
The execution of the eu ivdr signifies a crucial moment for the industry, requiring that manufacturers stay informed about ongoing updates and . With the evolving landscape, companies that proactively engage with these regulations will be better positioned to navigate the complexities of the market and enhance their product offerings. Katherine Ruiz states, “Keeping up with is crucial for companies seeking to thrive in the competitive IVD market.” Furthermore, producers need to recognize the transition phases for legacy devices, which permit certain products to stay available in the market under particular conditions, along with the heightened documentation and regulatory demands established by the new rules. The gradual implementation of the will also play a significant role in shaping the regulatory landscape, making it essential for companies to stay updated on its progress.
IVDR vs. IVDD: Critical Differences Every Innovator Must Know
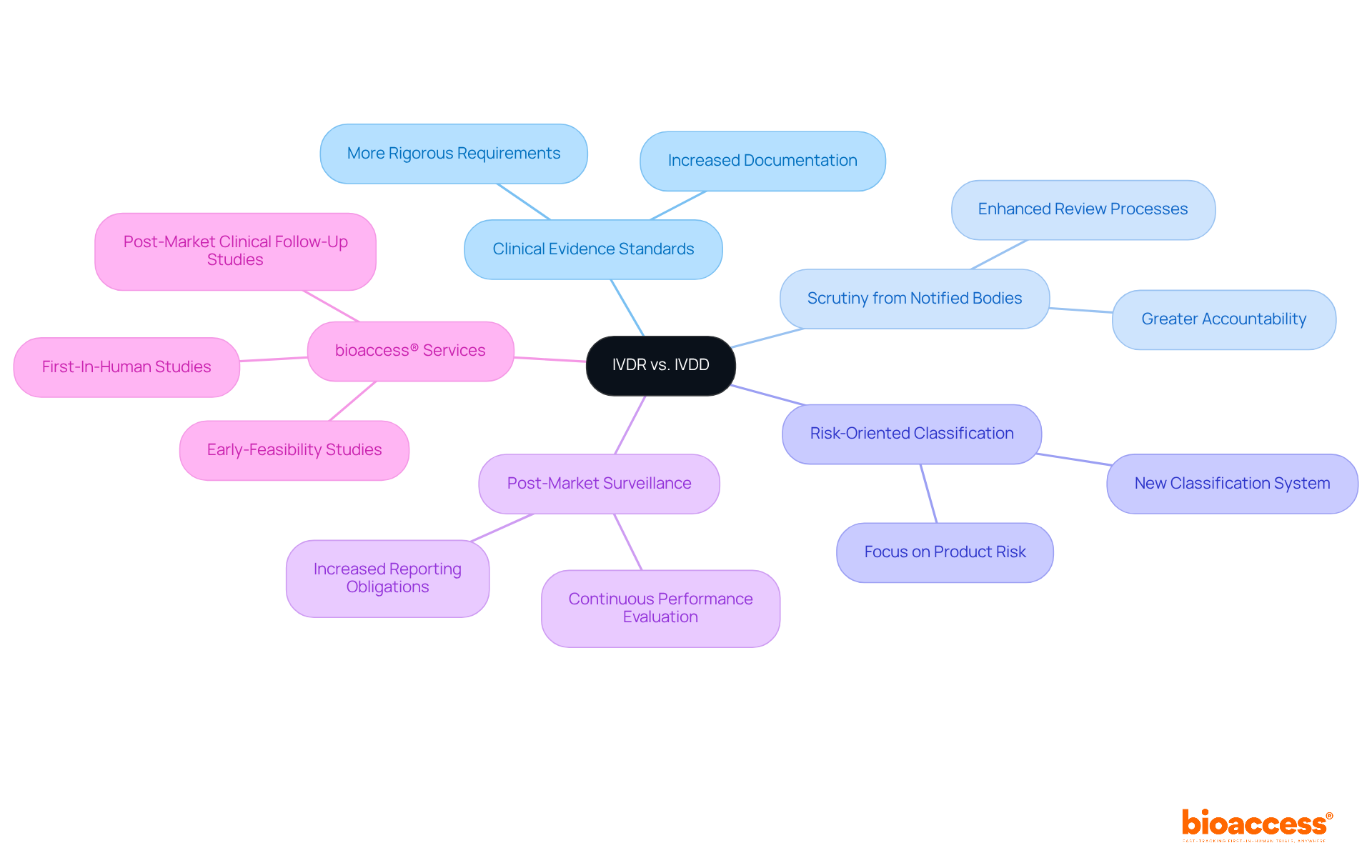
The transition from the In Vitro Diagnostic Directive (IVDD) to the new regulations marks a pivotal moment that Medtech innovators must grasp to ensure compliance and avert potential pitfalls. The EU IVDR imposes more rigorous standards for , increases scrutiny from Notified Bodies, and introduces a novel risk-oriented classification system for products. Furthermore, it places a greater emphasis on and continuous performance evaluation, areas that were less stringent under the EU IVDR.
With bioaccess®’s —including , , and Post-Market Clinical Follow-Up Studies—companies can adeptly navigate these compliance challenges. Katherine Ruiz, an expert in regulatory matters for medical instruments and in vitro diagnostics in Colombia, further bolsters bioaccess®’s capacity to assist innovators in achieving adherence and expediting their .
Key Components of EU IVDR Compliance for Medical Devices
To achieve compliance with the EU IVDR, Medtech innovators must prioritize several critical components:
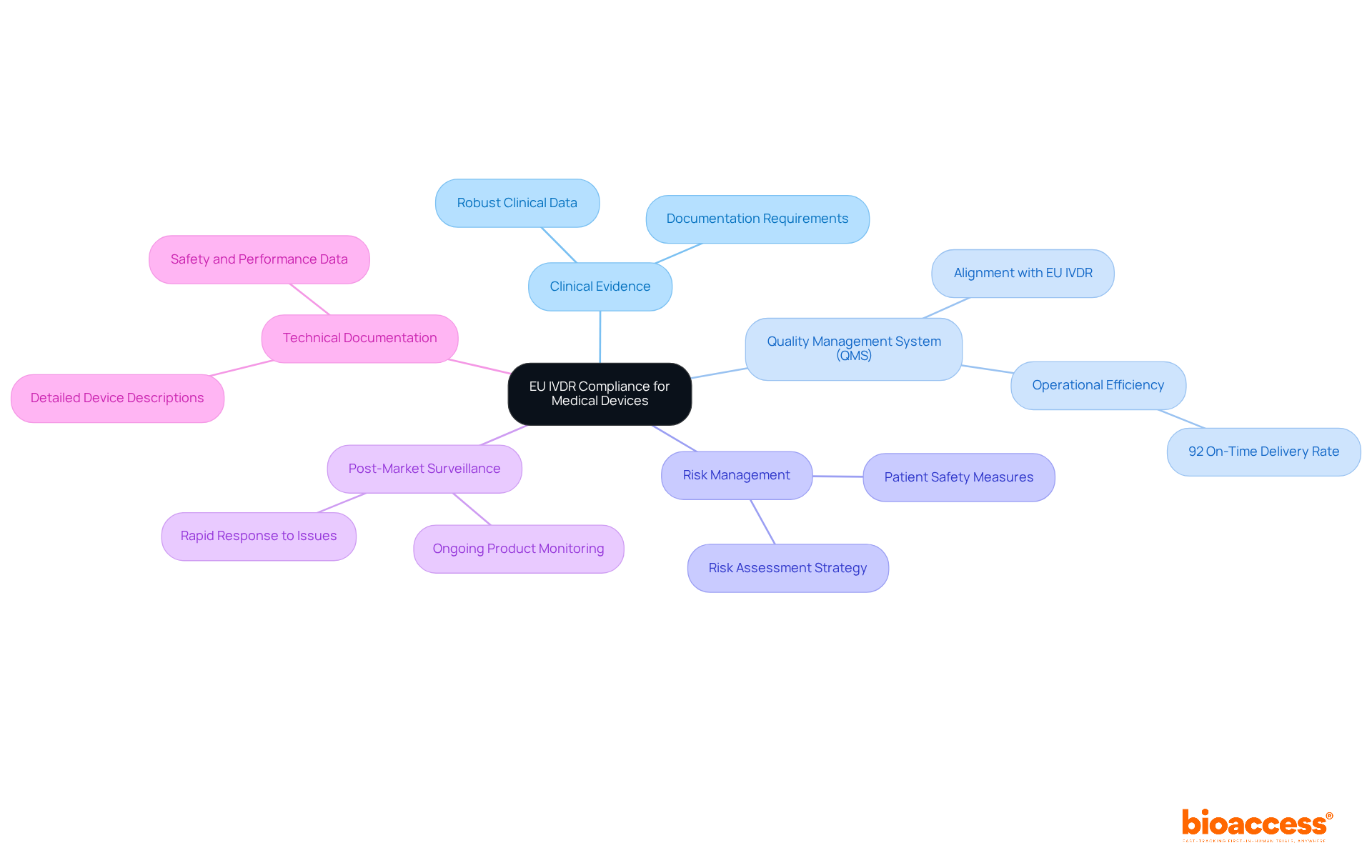
- : A robust body of clinical data is essential to demonstrate the safety and performance of the device. This evidence must be meticulously gathered and documented, as it plays a pivotal role in .
- Implementing a comprehensive (QMS) that aligns with EU IVDR requirements is crucial for ensuring consistent product quality. Organizations that implement effective QMS practices can anticipate enhanced and adherence rates, with studies suggesting that companies with developed systems achieve a 92% on-time delivery rate.
- Risk Management: A thorough must be integrated throughout the product lifecycle. This involves recognizing possible risks related to the equipment and applying strategies to reduce them, which is essential for ensuring and adhering to regulatory standards.
- : Ongoing observation of product performance and safety following market entry is required under the EU IVDR. This continuous alertness assists manufacturers in reacting quickly to any problems that occur, guaranteeing that the product stays safe and effective for users.
- : Detailed is necessary to support adherence claims and facilitate regulatory review. This documentation should include descriptions of equipment, labeling, safety and performance data, and , all organized in a manner that is readily accessible for auditors.
The effective execution of these components not only guarantees adherence to the regulations but also improves the overall quality and dependability of medical products in the market. As the landscape of Medtech evolves, maintaining a strong focus on and robust quality management practices will be essential for innovators aiming to thrive in this competitive environment.
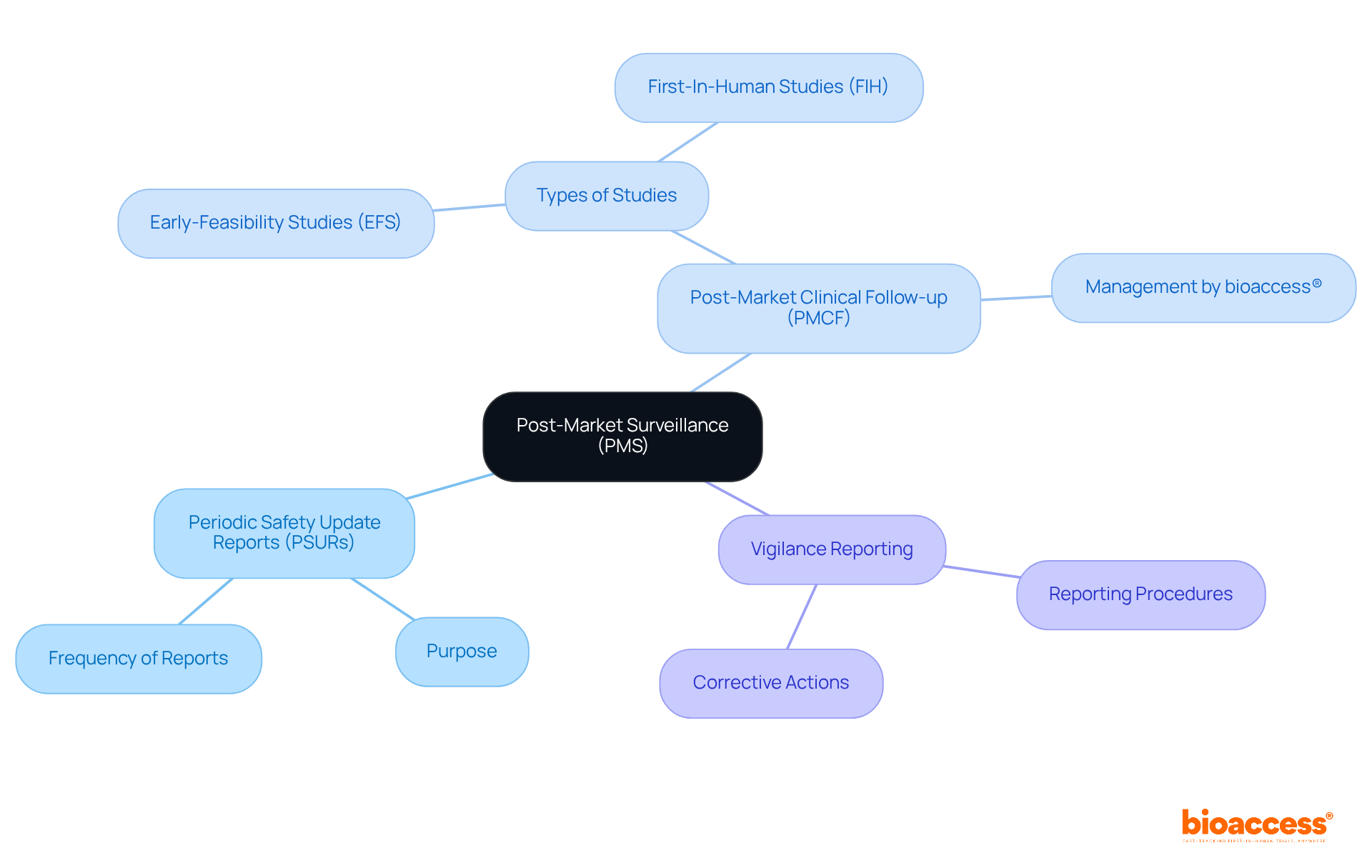
Post-Market Surveillance: A Pillar of IVDR Compliance
(PMS) is a fundamental requirement under the EU IVDR, aimed at ensuring the continued safety and effectiveness of IVDs. Manufacturers must establish a PMS plan that includes:
- : Regular assessments of device performance and safety.
- : Studies conducted to gather further on the product’s performance, expertly managed by bioaccess® with over 20 years of experience in Medtech, including Early-Feasibility Studies (EFS) and .
- : Procedures for reporting adverse events and taking corrective actions.
By adopting a strong PMS strategy, including PMCF studies supported by bioaccess®, manufacturers can proactively tackle potential problems, uphold adherence to industry standards, and guarantee the long-term success of their products.
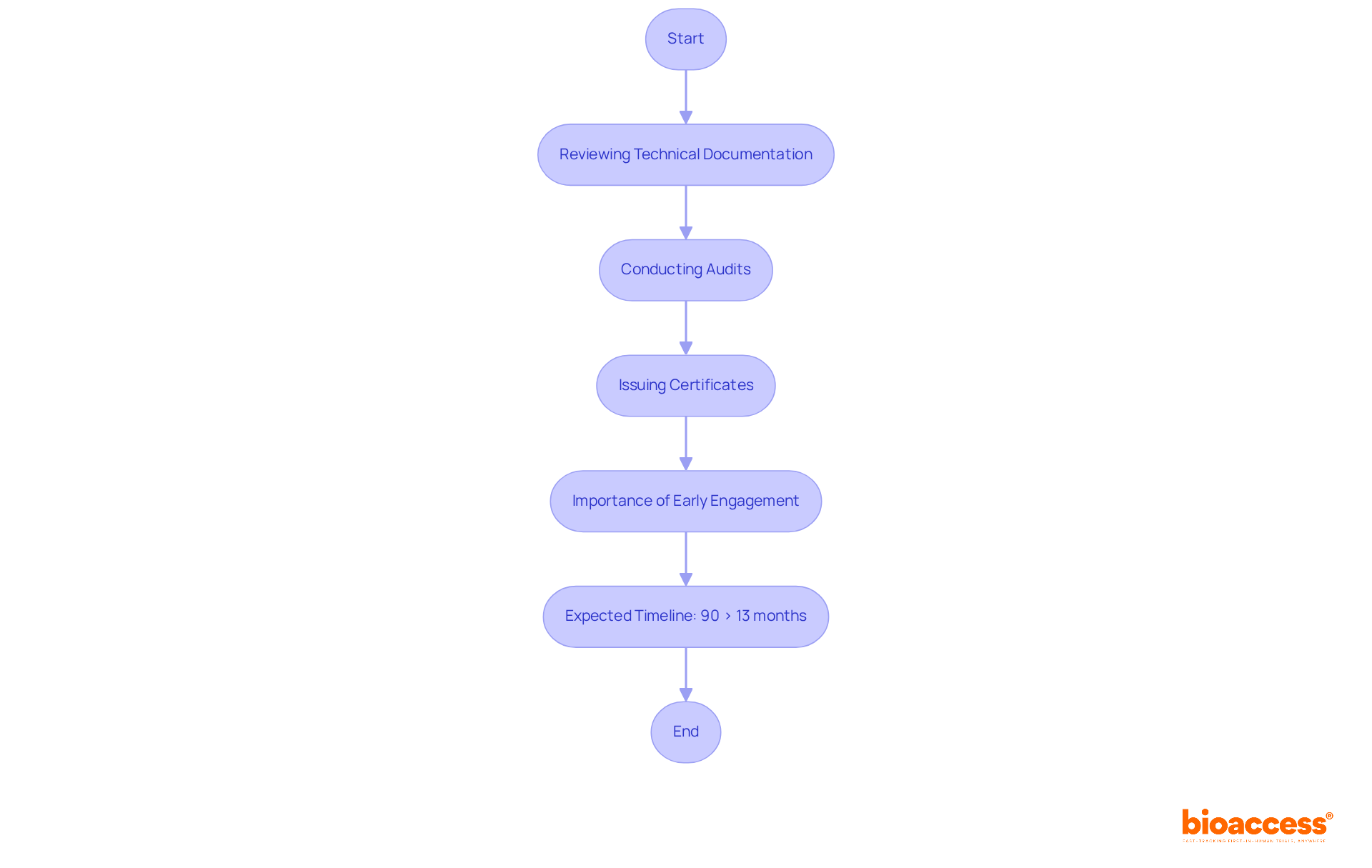
The Role of Notified Bodies in IVDR Certification
play a crucial role in the certification process under the , serving as independent organizations appointed by EU member states to evaluate the conformity of in vitro diagnostic devices (IVDs). Their primary responsibilities include:
- Reviewing : They meticulously assess whether all necessary documentation adheres to regulatory standards, ensuring comprehensive compliance.
- Conducting Audits: perform thorough evaluations of the manufacturer’s (QMS) to confirm compliance with relevant regulations.
- Issuing Certificates: They are tasked with granting , an essential step for market access within the EU.
Engaging with early in the development process is vital. This proactive approach simplifies the certification journey and enables manufacturers to identify potential at an early stage. As we approach 2025, the landscape of operating under the is evolving, marked by a significant increase in applications and certifications. This trend underscores the and demand for IVDs in the market. Manufacturers are strongly encouraged to initiate their promptly, as delays can adversely impact product timelines. Notably, 90% of report that product certificates under the regulation require over 13 months for processing, with 75% of rejections attributed to incomplete applications. As emphasized by Emergo by UL, manufacturers are encouraged to commence their journeys now.
In this context, the expertise of professionals like Ana Criado, Director of Compliance Affairs and a consultant with extensive experience in regulatory frameworks, is invaluable. Her background includes significant contributions at INVIMA, where she played a key role in developing compliance policies, alongside her academic positions as a professor at leading Colombian universities. Collaborating with specialists such as Ana and Katherine Ruiz, who focuses on regulatory issues for medical equipment and in vitro diagnostics in Colombia, can significantly enhance a manufacturer’s understanding and implementation of compliance strategies.
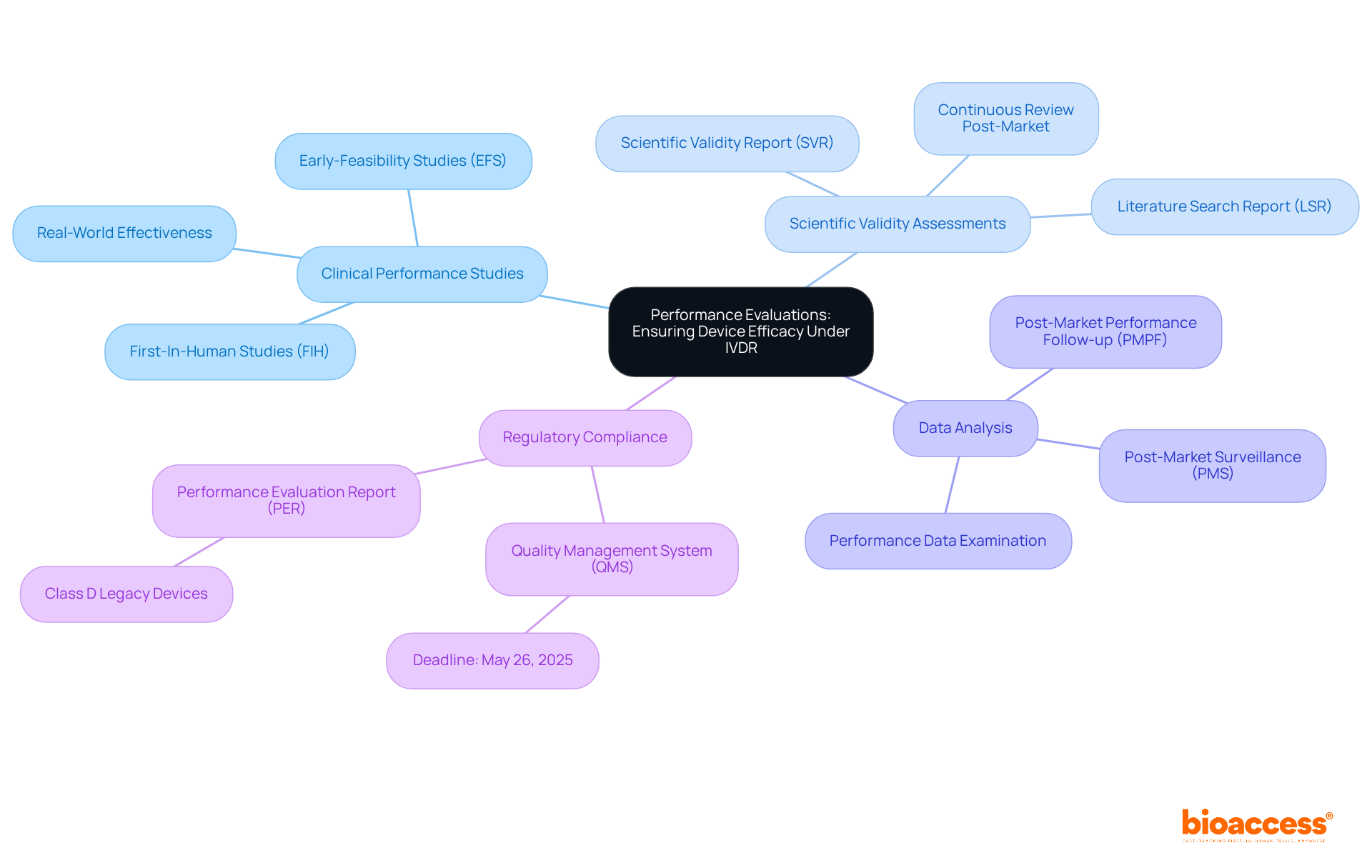
Performance Evaluations: Ensuring Device Efficacy Under IVDR
, emphasizing the demonstration of clinical performance and safety of in vitro diagnostics (IVDs). Manufacturers must conduct comprehensive evaluations that encompass several critical elements:
- : These trials evaluate the device’s effectiveness in real-world settings, providing essential evidence of its reliability and safety. With Bioaccess’s expertise in managing (EFS) and , manufacturers can ensure that their are executed with the highest standards of quality and adherence.
- : This process involves confirming that the device’s intended purpose is supported by robust scientific data, ensuring that the analyte is appropriately linked to the clinical condition it aims to detect. Bioaccess provides comprehensive support in reviewing and offering feedback on study documents to comply with country requirements, which is vital for establishing .
- Data Analysis: A meticulous examination of performance data is crucial to verify adherence to eu ivdr standards, including the assessment of both analytical and clinical performance. Bioaccess’s project management and monitoring services guarantee that data collection and analysis are conducted efficiently and effectively.
Moreover, stricter requirements apply to higher-risk IVDs, necessitating a more rigorous approach to performance evaluations. As Dr. Yupei Xiao emphasizes, “Performance assessment is not merely a pre-market necessity; producers must consistently refresh evidence to represent the most recent data, ensuring ongoing safety and effectiveness.”
By adhering strictly to these requirements, manufacturers can validate their assertions, ensuring that their products not only meet regulatory expectations but also deliver real-world benefits to patients. Engaging in proactive data gathering and maintaining robust post-market monitoring further enhances adherence and safety throughout the product’s lifecycle. Additionally, with the impending deadline of May 26, 2025, for the implementation of (QMS) and the submission of (PER) for Class D legacy products, it is imperative for manufacturers to act swiftly to ensure compliance. —, analytical performance, and clinical performance—must be clearly addressed to meet compliance standards.
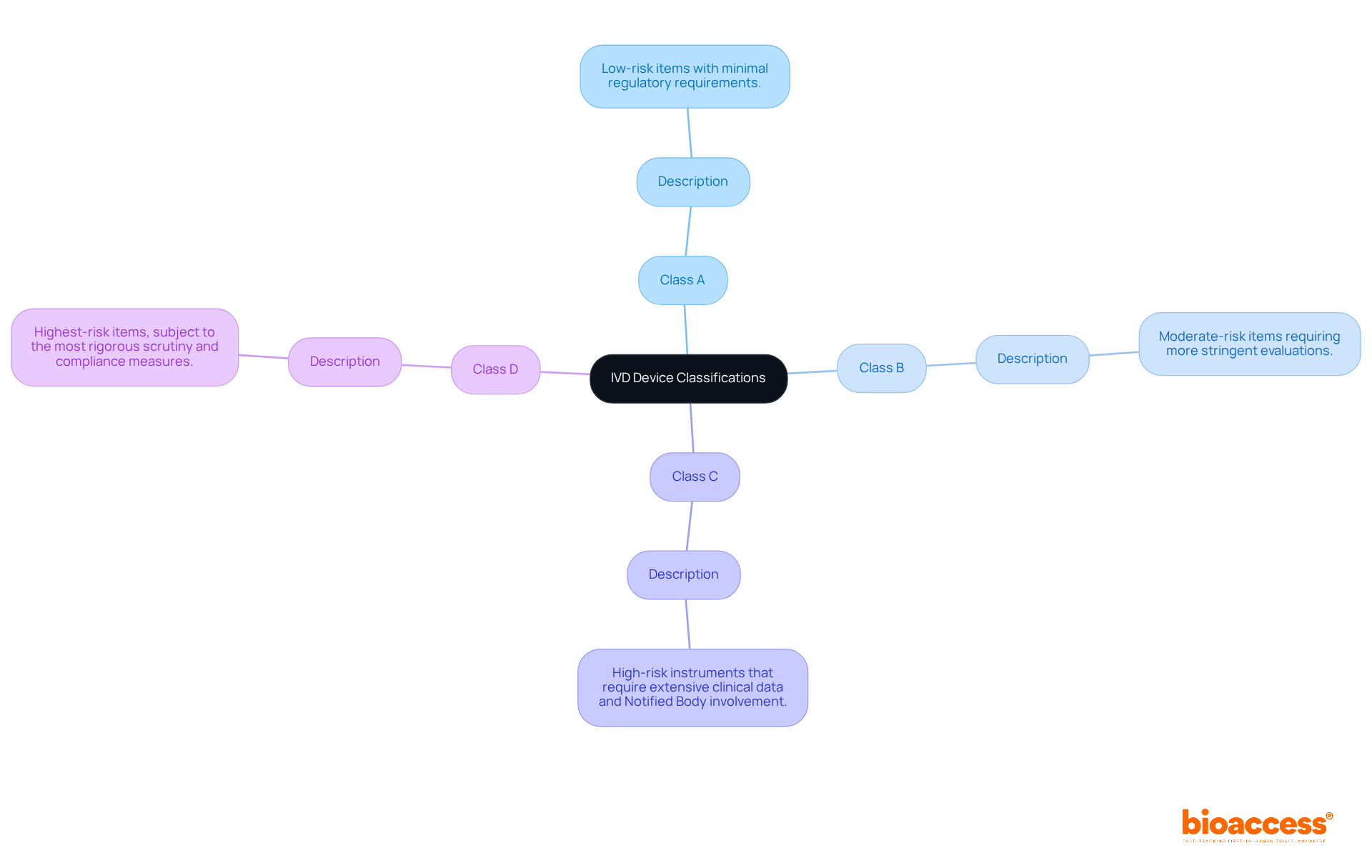
Categorizing Your IVD Device: Navigating IVDR Classifications
Under the IVDR, IVD devices are classified into four categories based on risk:
- Class A: Low-risk items with minimal regulatory requirements.
- Class B: Moderate-risk items requiring more stringent evaluations.
- Class C: that require extensive clinical data and .
- Class D: Highest-risk items, subject to the most rigorous scrutiny and compliance measures.
In Colombia, the oversight framework is managed by (Colombia National Food and Drug Surveillance Institute), which plays a vital role in inspecting and supervising health products, including . As a Level 4 health authority acknowledged by the Pan American Health Organization/World Health Organization, ensures that items meet . Understanding these classifications within the EU , along with ‘s , is essential for manufacturers to ensure they meet the specific requirements linked to their product category.
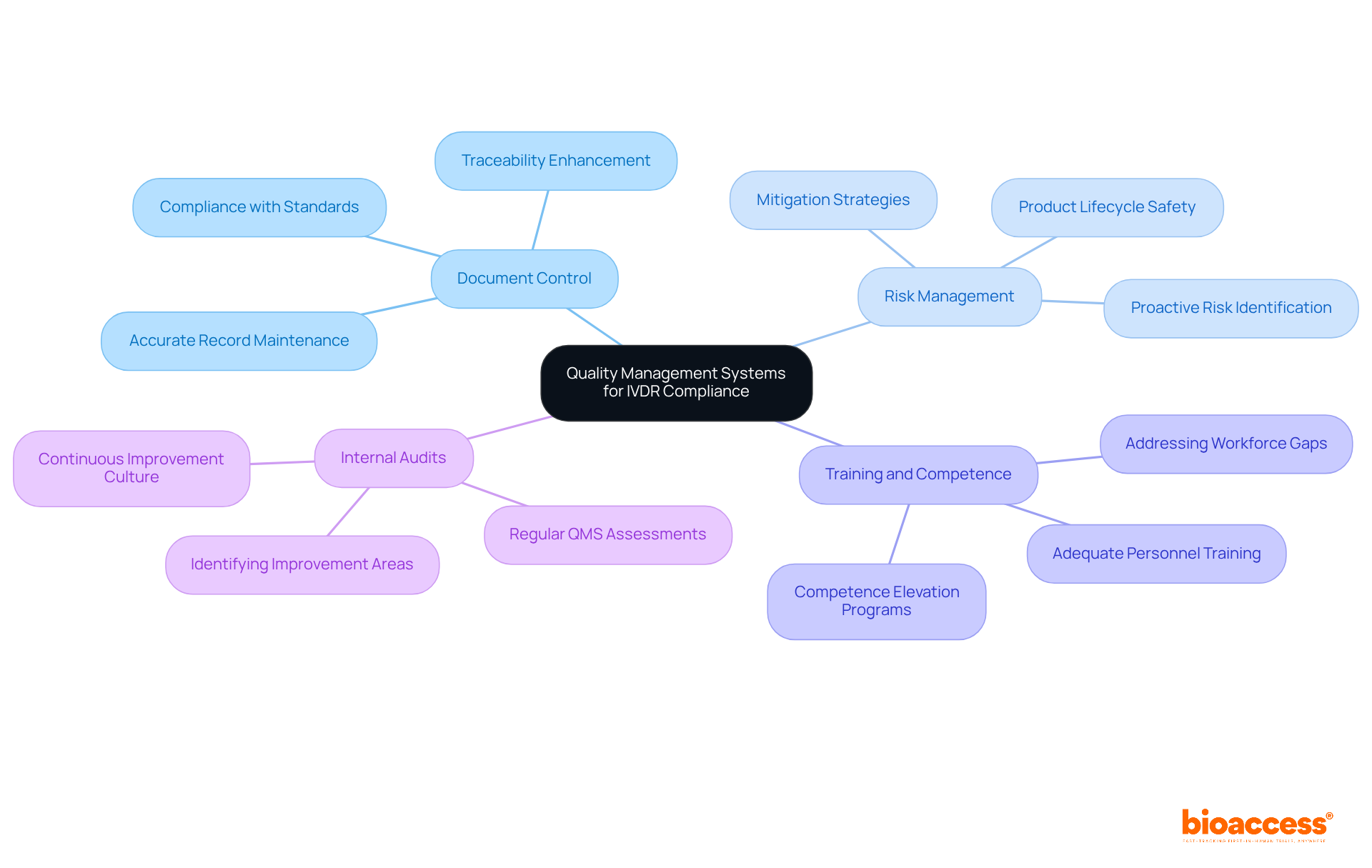
Quality Management Systems: Essential for IVDR Compliance
A is essential for adherence to the , which ensures that manufacturers consistently provide safe and effective devices. Essential components of an effective QMS include:
- Document Control: Accurate maintenance of records related to all processes and changes is crucial. Implementing effective can significantly enhance traceability and adherence to standards.
- : Proactively identifying and mitigating risks throughout the product lifecycle is essential for maintaining product safety and efficacy.
- Training and Competence: Ensuring that personnel are adequately trained and competent is fundamental. Data indicates that a significant portion of the Medtech workforce lacks sufficient training, highlighting the need for robust .
- : Conducting regular assessments of the QMS helps identify areas for improvement, fostering a culture of continuous enhancement.
By creating a thorough QMS, manufacturers can not only enhance product quality but also simplify their regulatory efforts, ultimately enabling easier market access under the .
Transitioning to IVDR: Strategies for Medtech Innovators
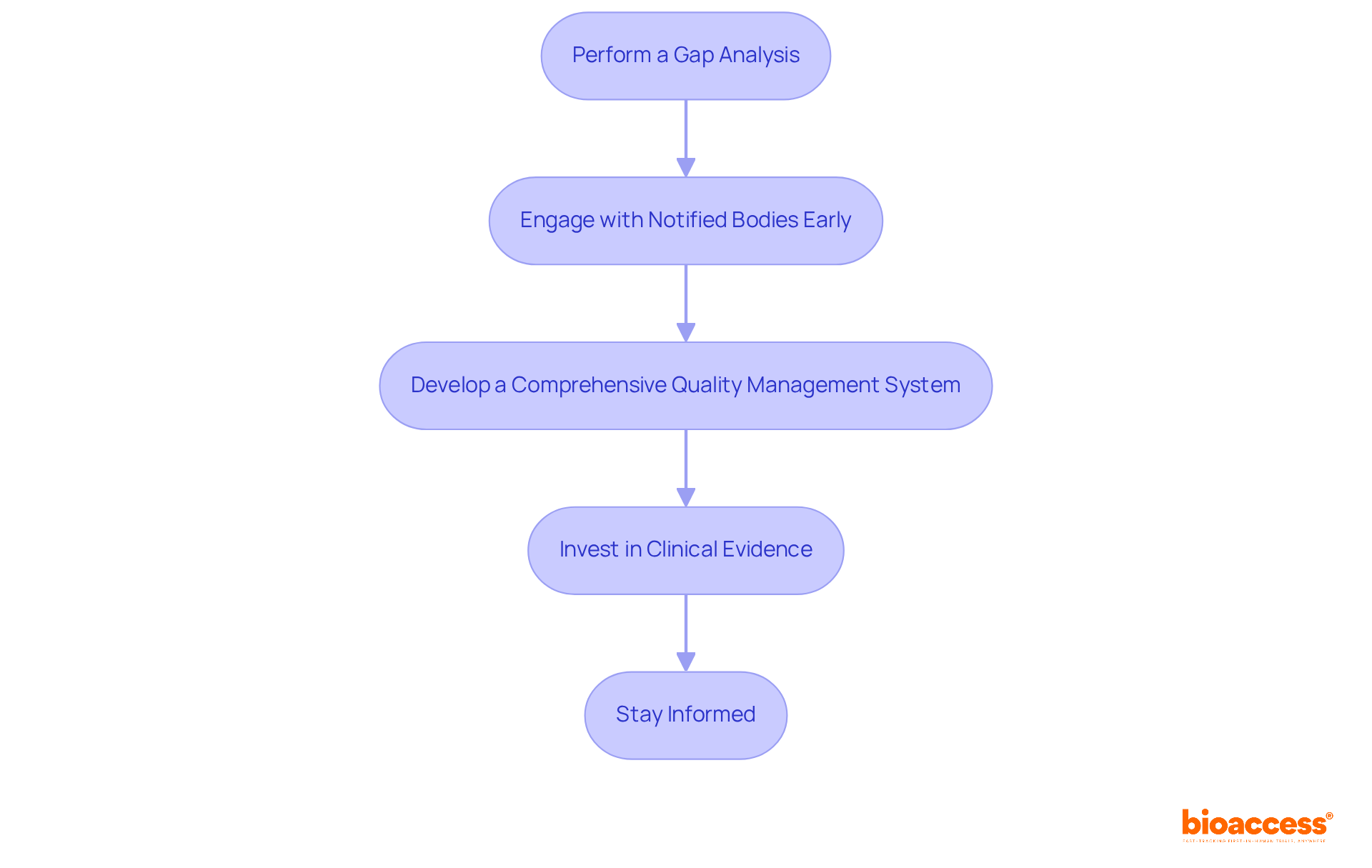
Shifting to the new regulations necessitates meticulous planning and execution. Medtech innovators can adopt the following strategies to ensure :
- Perform a Gap Analysis: Assess your current adherence status against to pinpoint areas for improvement. This analysis is critical, as successful examples have demonstrated that early identification of gaps can streamline the transition process and mitigate risks. Incorporating a Performance Evaluation Plan (PEP) within this analysis offers a structured approach to gathering .
- Engage with Notified Bodies Early: Initiate dialogue with Notified Bodies to clarify their expectations and requirements. Early engagement is linked to smoother compliance processes, allowing for proactive adjustments to meet regulatory standards. Under the new regulations, it is anticipated that 80-90% of IVD products will require the involvement of notified bodies, making this step indispensable.
- Develop a (QMS) that aligns with the , focusing on documentation, risk management, and staff training. A robust QMS is vital for maintaining compliance and ensuring product safety throughout the lifecycle.
- Invest in : Prioritize the to substantiate device efficacy and safety claims. As the eu ivdr mandates rigorous , having strong is essential for successful market entry. bioaccess® offers comprehensive , including feasibility studies, , project management, review processes, and reporting, to support this critical phase.
- Stay Informed: Consistently update your knowledge of regulatory changes and industry best practices to ensure continuous compliance. This proactive strategy not only aids in fulfilling regulatory requirements but also positions your organization as a leader in the Medtech sector. Collaborating with bioaccess, a vetted CRO and consulting partner for U.S. medical device companies in Colombia, can provide invaluable insights and support throughout this process.
By implementing these strategies, Medtech innovators can adeptly navigate the transition to eu ivdr, ensuring compliance while maintaining a competitive edge in the evolving regulatory landscape.
Conclusion
bioaccess® plays a pivotal role in guiding Medtech innovators through the intricate landscape of EU IVDR compliance. By emphasizing the importance of robust clinical evidence, effective quality management systems, and proactive engagement with Notified Bodies, the article underscores the necessity for manufacturers to adapt swiftly to the evolving regulatory environment. Understanding the nuances between the IVDR and its predecessor, the IVDD, is crucial for ensuring successful market entry and maintaining competitive advantage.
Key arguments presented include:
- The significance of post-market surveillance in safeguarding patient safety and product efficacy.
- The critical nature of comprehensive performance evaluations that align with regulatory expectations.
- The early identification of compliance gaps and the establishment of a solid QMS to streamline the transition process, ultimately enhancing product quality and regulatory adherence.
As the deadline for full compliance approaches, Medtech innovators are called to action to prioritize their transition strategies. By leveraging resources like bioaccess® and staying informed about regulatory changes, companies can not only meet compliance requirements but also position themselves as leaders in the Medtech sector. Embracing these insights will pave the way for successful navigation of the EU IVDR landscape, ensuring that innovations contribute positively to patient outcomes while adhering to the highest standards of safety and effectiveness.
Frequently Asked Questions
What is bioaccess® and how does it assist Medtech innovators?
bioaccess® is a company that leverages its expertise in early-stage clinical research to expedite EU regulatory compliance for Medtech innovators. It helps businesses achieve ethical approvals in 4-6 weeks, facilitating quicker market entry while navigating stringent regulatory requirements.
What is the significance of the EU IVDR for in vitro diagnostic tools?
The EU IVDR aims to ensure the safety and effectiveness of in vitro diagnostic (IVD) tools. Its primary objectives include enhancing quality and reliability, increasing transparency in the regulatory process, and requiring manufacturers to provide robust clinical evidence for their products.
How does bioaccess® improve the time to market for medical products?
By utilizing regulatory efficiencies found in Latin America and diverse patient populations in the Balkans, bioaccess® accelerates the approval process, which is crucial for Medtech companies facing regulatory hurdles under the EU IVDR.
What are the critical differences between the EU IVDR and the previous IVDD?
The EU IVDR imposes more rigorous standards for clinical evidence, increases scrutiny from Notified Bodies, introduces a risk-oriented classification system, and emphasizes post-market surveillance and continuous performance evaluation compared to the IVDD.
What role does governance play in compliance with EU IVDR?
A well-defined governance approach is vital for compliance and business success. It begins with a clear understanding of the product’s intended purpose and user requirements, which is essential for navigating regulatory processes effectively.
What is the anticipated impact of the EU IVDR on compliance rates by 2025?
Compliance rates for IVDs under the new regulations are expected to increase substantially by 2025, as the industry adjusts to the stringent standards set by the EU IVDR.
How does bioaccess® support companies during clinical trials?
bioaccess® offers expedited clinical trial management services, including Early-Feasibility, First-In-Human, and Post-Market Clinical Follow-Up Studies, helping companies navigate compliance challenges effectively.
What should manufacturers know about the transition phases for legacy devices under the EU IVDR?
Manufacturers need to recognize that transition phases allow certain legacy devices to remain on the market under specific conditions, while also adhering to heightened documentation and regulatory demands established by the new regulations.
Why is it important for companies to stay informed about compliance changes in the IVD market?
Keeping up with compliance changes is crucial for companies to maintain market access, enhance their product offerings, and thrive in the competitive IVD market.
What is the role of the EUDAMED platform in the regulatory landscape?
The gradual implementation of the EUDAMED platform will significantly shape the regulatory landscape, making it essential for companies to stay updated on its progress and understand their obligations under the new regulations.
List of Sources
- bioaccess®: Accelerate EU IVDR Compliance for Medtech Innovations
- 7 quotes from 2023 to guide you on the medtech market (https://tiinatyni.com/blogi/7-quotes-from-2023-to-guide-you-on-the-medtech-market)
- mediacc.de (https://mediacc.de/en/medizinprodukte-wiki/zertifizierungsstand-bei-medizinprodukten-auswertung-der-eu-kommission-von-august-2025)
- Understanding the EU IVDR: Key Objectives and Scope
- aristo-group.com (https://aristo-group.com/en/mdr-and-ivdr-what-to-consider-from-2025)
- iconplc.com (https://iconplc.com/insights/regulatory-intelligence/eu-regulations)
- Key Components of EU IVDR Compliance for Medical Devices
- qualio.com (https://qualio.com/blog/eu-ivdr-everything-you-need-to-know)
- llcbuddy.com (https://llcbuddy.com/data/medical-quality-management-systems-statistics)
- Quality Statistics and Why Your QMS Matters | Steelhead Technologies (https://gosteelhead.com/quality-statistics-and-why-your-qms-matters)
- greenlight.guru (https://greenlight.guru/blog/ivdr-in-vitro-diagnostic-medical-device)
- Quality Management Software Market Size & Trends [2024-2032] (https://snsinsider.com/reports/quality-management-software-market-3168)
- The Role of Notified Bodies in IVDR Certification
- emergobyul.com (https://emergobyul.com/news/european-notified-body-survey-shows-uptick-mdr-ivdr-applications)
- Performance Evaluations: Ensuring Device Efficacy Under IVDR
- greenlight.guru (https://greenlight.guru/blog/performance-evaluation-report)
- seleon.com (https://seleon.com/en/performance-evaluation-according-to-ivdr-scientific-validity)
- lfhregulatory.co.uk (https://lfhregulatory.co.uk/performance-evaluation-under-ivdr-2025)
- medloft.net (https://medloft.net/performance-evaluation-under-eu-2017-746-ivdr)
- emergobyul.com (https://emergobyul.com/news/eu-releases-guidance-ivd-performance-studies-finally)
- Quality Management Systems: Essential for IVDR Compliance
- What Are the Key Elements of a Quality Management System (QMS)? (https://arenasolutions.com/resources/articles/key-elements-of-a-quality-management-system)
- Transitioning to IVDR: Strategies for Medtech Innovators
- greenlight.guru (https://greenlight.guru/blog/ivdr-in-vitro-diagnostic-medical-device)

Leave a Reply