Introduction
For MedTech startups in Chile, the regulatory landscape is a double-edged sword, presenting both formidable challenges and promising opportunities. Understanding the intricate requirements set forth by the Instituto de Salud Pública (ISP) is essential for achieving successful market entry and ensuring compliance. Yet, many startups struggle to navigate the intricate web of regulations, which can delay their entry into the market.
What strategies can startups use to simplify their regulatory journey and take advantage of the benefits of conducting clinical trials in Latin America?
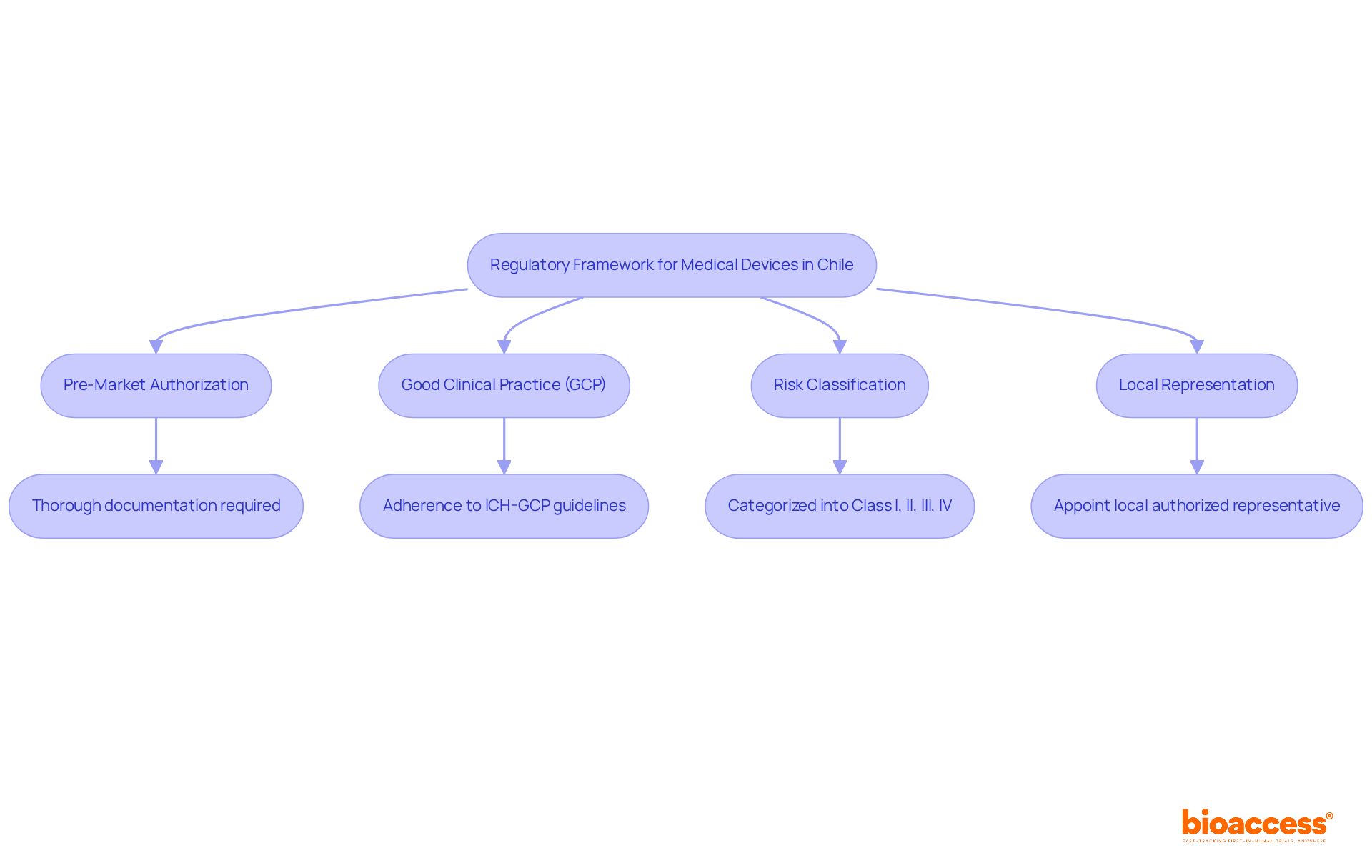
Understand the Regulatory Framework for Medical Devices in Chile
Navigating the medical device regulatory approval in Chile can be a daunting task for MedTech startups, yet understanding these complexities is crucial for success. The regulatory framework for medical devices in Chile is primarily governed by the Instituto de Salud Pública (ISP), which oversees the medical device regulatory approval in Chile, including the registration and approval processes. Key regulations include:
- Pre-Market Authorization: All medical devices must receive pre-market authorization from the ISP before they can be marketed in Chile. This means providing thorough documentation that clearly shows the device’s safety and effectiveness.
- Good Clinical Practice (GCP): Adherence to ICH-GCP guidelines is essential for carrying out clinical studies. This ensures that trials are ethically conducted and that data integrity is maintained.
- Risk Classification: Medical devices are categorized into four groups (Class I, II, III, and IV) according to their risk level, which affects the compliance requirements and approval timelines. For instance, Class I devices may have a simpler registration process compared to Class III devices, which require more extensive clinical data.
- Local Representation: Companies must appoint a local authorized representative in Chile to facilitate communication with the ISP and manage compliance submissions.
For any MedTech startup, securing medical device regulatory approval in Chile is key to successfully navigating the Chilean market. Without a firm grasp of these regulatory requirements, startups risk costly setbacks that could jeopardize their market entry and growth potential.
Navigate the Medical Device Registration Process
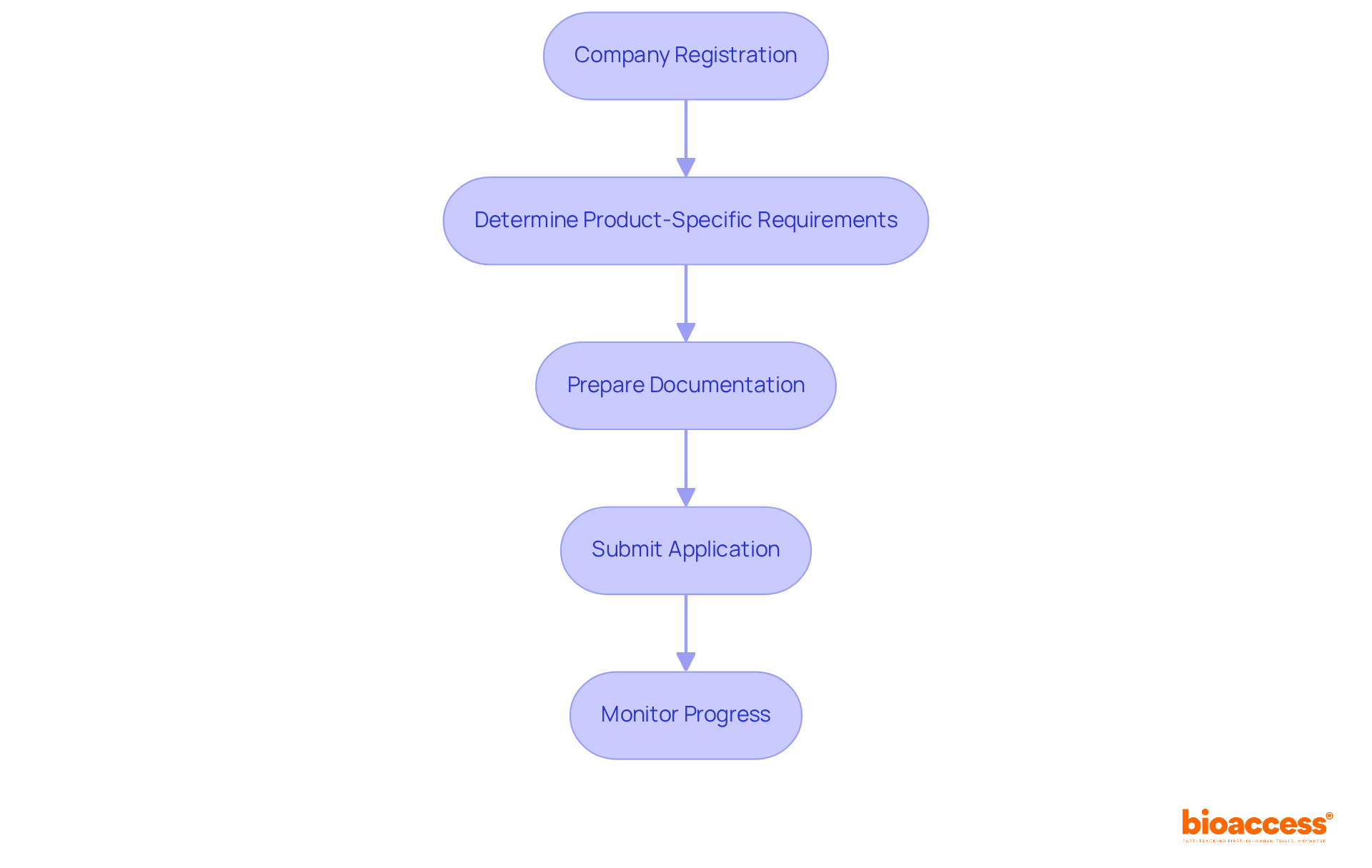
Navigating the medical device regulatory approval Chile process can be daunting, but understanding it is crucial for success. To successfully register a medical device in Chile, follow these steps:
- Company Registration: Ensure your company is legally registered in Chile. This is a prerequisite for any compliance activities.
- Determine Product-Specific Requirements: Identify the specific requirements for your device based on its classification. This includes understanding the necessary documentation and testing data required for submission.
- Prepare Documentation: Compile all required documents, including:
- Quality Management System (QMS) certificates
- Clinical data supporting safety and efficacy
- Labels and instructions for use in Spanish
- A Free Sale Certificate or export certificate from the country of origin.
- Submit Application: Access the GICONA system to submit your registration request. Ensure all documents are complete and correctly formatted to avoid delays.
- Monitor Progress: After submission, maintain communication with your local representative and the ISP (Instituto de Salud Pública) to track the status of your application and respond promptly to any requests for additional information.
With bioaccess®’s expertise, startups can simplify their registration process, achieving rapid approvals in just 4-8 weeks, a stark contrast to the 6+ months often seen in the US and EU. This efficiency not only accelerates market access but also allows for significant cost savings-up to $25K per patient-enabling companies to reinvest in R&D or reach their next funding milestone. Following these procedures allows startups to effectively navigate the compliance framework for medical device regulatory approval Chile. This approach also enables them to capitalize on the strategic advantages of conducting studies in Latin America.
Conduct Compliant First-in-Human Clinical Trials
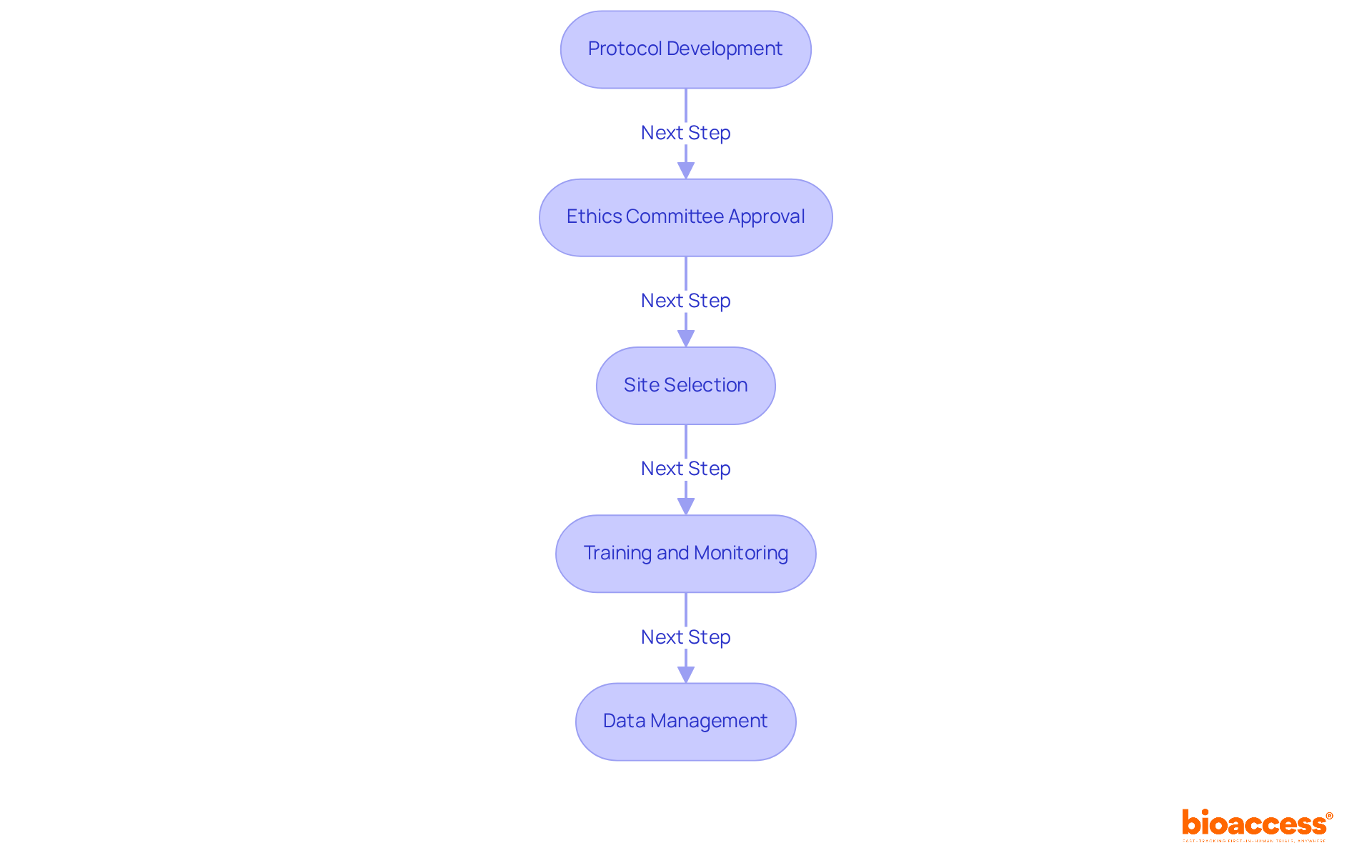
Navigating the complexities of first-in-human (FIH) clinical trials in Chile can be daunting for startups, but adhering to best practices is crucial for success. To conduct compliant FIH clinical trials, it is essential to follow these best practices:
- Protocol Development: Create a comprehensive clinical trial protocol that aligns with ICH-GCP guidelines. This protocol should clearly outline the study’s objectives, methodology, and statistical analysis plans, ensuring that all aspects are meticulously detailed to facilitate regulatory review.
- Ethics Committee Approval: Securing endorsement from an accredited ethics committee (EC) is a crucial step before commencing any study. This endorsement ensures that ethical standards are met and safeguards the rights and welfare of participants, which is vital in any research endeavor.
- Site Selection: Choose clinical research locations that possess the necessary experience and infrastructure to conduct FIH studies effectively. Sites should have a proven track record of compliance with standards and be equipped to handle the specific requirements of FIH studies.
- Training and Monitoring: All personnel involved in the study must receive thorough training on Good Clinical Practice (GCP) and the specific requirements of the protocol. Regular monitoring should be carried out to ensure compliance with both the protocol and legal standards throughout the study duration.
- Data Management: Establish a robust data management system to maintain the integrity and confidentiality of research data. This includes secure data storage solutions and conducting regular audits to ensure compliance with data protection laws and ethical standards.
By adopting these optimal approaches, startups can greatly improve the quality of their studies in Chile, thus enhancing their likelihood of obtaining approval efficiently. Without a commitment to these best practices, the path to regulatory approval may become increasingly challenging and uncertain.
Implement Effective Patient Recruitment and Site Selection Strategies
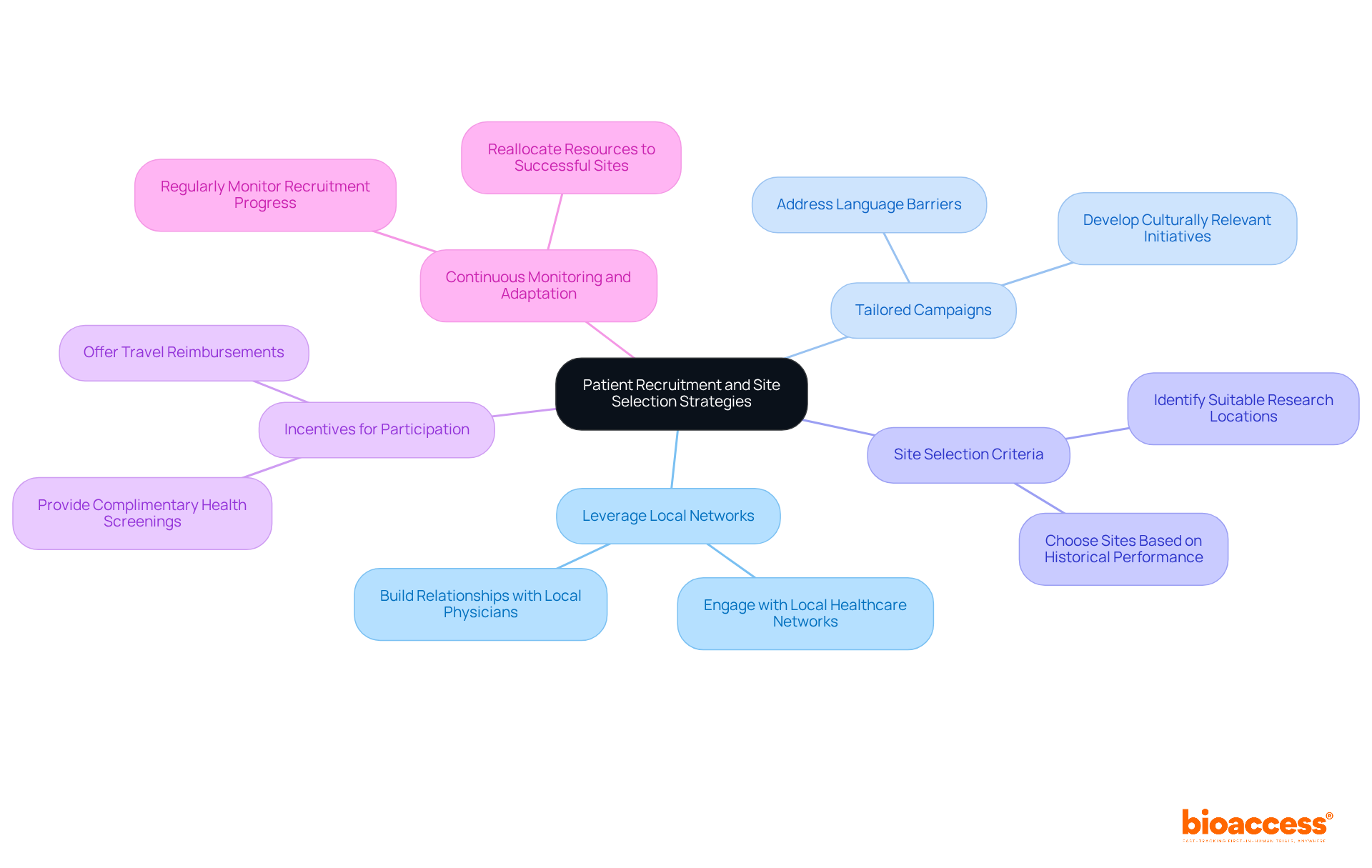
In Chile, the challenge of timely patient recruitment for clinical trials is a pressing concern that demands strategic solutions. To optimize patient recruitment and site selection, consider the following strategies:
- Leverage Local Networks: With established connections in local healthcare networks, bioaccess® empowers MedTech and Biopharma companies to navigate the Chilean landscape effectively. Engaging with local healthcare networks and community organizations can help identify potential participants. Building strong relationships with local physicians promotes referrals and significantly enhances recruitment efforts.
- Tailored Campaigns: Understanding local cultures is crucial for bioaccess® in successfully attracting participants to clinical trials. Developing culturally relevant hiring initiatives that resonate with the local population can improve participant engagement and trust. Addressing language barriers and utilizing culturally appropriate messaging is essential.
- Site Selection Criteria: Startups can rely on bioaccess®’s expertise to identify the most suitable research locations, expediting their studies. Choosing research sites based on historical performance, patient demographics, and logistical capabilities is vital. Sites with established patient databases can accelerate recruitment, as they already have access to potential participants.
- Incentives for Participation: Offering incentives such as travel reimbursements or complimentary health screenings can encourage enrollment and improve retention rates among participants. This approach aligns with bioaccess®’s commitment to fostering patient engagement in clinical trials.
- Continuous Monitoring and Adaptation: Regularly monitoring progress in hiring is crucial. If certain sites are underperforming, reallocating resources to more successful sites can enhance overall efficiency in participant acquisition. With ongoing support from bioaccess®, startups can adapt their hiring strategies to meet the evolving demands of clinical trials.
In Chile, it is essential to understand that up to 80% of clinical studies do not enroll on time due to insufficient patient participation. Many clinical studies face significant delays due to inadequate patient participation, which can jeopardize timelines and outcomes. The typical approval period for research studies can surpass six months, highlighting the need for implementing effective participant engagement strategies. Furthermore, the Instituto de Salud Pública (ISP) is the principal authority responsible for approving study applications. By comprehending and adhering to ISP guidelines, startups can streamline their approval processes and enhance compliance, ultimately leading to more successful trials. By leveraging local expertise and understanding regulatory requirements, startups can transform their recruitment strategies and drive successful clinical outcomes.
Conclusion
For MedTech startups, understanding the regulatory landscape in Chile is not just beneficial; it’s crucial for survival in a competitive market. A solid grasp of the regulatory framework – like pre-market authorization, risk classification, and local representation – is essential for a successful market entry. By adhering to these guidelines, companies can mitigate risks and enhance their chances of obtaining timely approvals.
The article highlights several key strategies for achieving compliance and ensuring efficient registration processes. Understanding the specific requirements for each device classification, preparing comprehensive documentation, and following best practices for conducting first-in-human clinical trials are pivotal steps. Many MedTech startups struggle with participant recruitment in Chile, which can hinder their clinical studies. Moreover, implementing effective patient recruitment and site selection strategies can significantly impact the success of clinical studies.
By following these best practices, startups can not only navigate the regulatory maze but also seize the unique opportunities that Latin America offers. This adherence not only leads to smoother approvals but also opens doors to faster market entry. The insights provided in this article serve as a valuable roadmap for navigating the complexities of medical device regulations in Chile. Navigating these regulations effectively can be the difference between success and stagnation in the rapidly evolving MedTech industry.
Frequently Asked Questions
What is the primary regulatory authority for medical devices in Chile?
The primary regulatory authority for medical devices in Chile is the Instituto de Salud Pública (ISP), which oversees the regulatory approval processes, including registration.
What is required for pre-market authorization of medical devices in Chile?
All medical devices must receive pre-market authorization from the ISP, which requires thorough documentation demonstrating the device’s safety and effectiveness before it can be marketed in Chile.
What guidelines must be followed for conducting clinical studies involving medical devices in Chile?
Clinical studies must adhere to the International Council for Harmonisation Good Clinical Practice (ICH-GCP) guidelines to ensure ethical conduct and data integrity during trials.
How are medical devices classified in Chile, and why is this classification important?
Medical devices in Chile are classified into four groups (Class I, II, III, and IV) based on their risk level. This classification affects compliance requirements and approval timelines, with Class I devices typically having a simpler registration process compared to Class III devices, which require more extensive clinical data.
Do companies need a local representative to navigate the regulatory process in Chile?
Yes, companies must appoint a local authorized representative in Chile to facilitate communication with the ISP and manage compliance submissions.
Why is understanding the regulatory framework essential for MedTech startups in Chile?
Understanding the regulatory framework is crucial for MedTech startups to successfully navigate the Chilean market, as a lack of knowledge about these requirements can lead to costly setbacks that jeopardize market entry and growth potential.
List of Sources
- Conduct Compliant First-in-Human Clinical Trials
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Master First-in-Human Clinical Trials in Latin America: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/master-first-in-human-clinical-trials-in-latin-america-a-step-by-step-guide)
- First-in-Human Trial Participants: Not a Vulnerable Population, but Vulnerable Nonetheless – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC2692671)
- Implement Effective Patient Recruitment and Site Selection Strategies
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- Clinical Trials Patient Recruitment in Latin America | H Clinical (https://hclinical.com/patient-recruitment)
- Keys to Maximize Patient Recruitment (https://linical.com/articles-research/keys-to-maximize-patient-recruitment)
- Patient Recruitment Strategies for Clinical Trials (https://freyrsolutions.com/blog/patient-recruitment-strategies-for-clinical-trials)

Leave a Reply