Introduction
While Chile offers a promising landscape for clinical research, the path to successful trial execution is fraught with regulatory challenges. The country has emerged as a frontrunner in the clinical research landscape, particularly in the MedTech, Biopharma, and Radiopharma sectors, thanks to its streamlined regulatory framework and efficient approval processes. Organizations looking to conduct clinical trials in this vibrant market stand to gain significant advantages, including:
- Faster patient recruitment
- Lower costs compared to other regions
However, despite these advantages, many organizations find the approval process in Chile to be a significant hurdle. Failure to understand these complexities can lead to delays and increased costs, undermining the benefits of conducting trials in Chile.
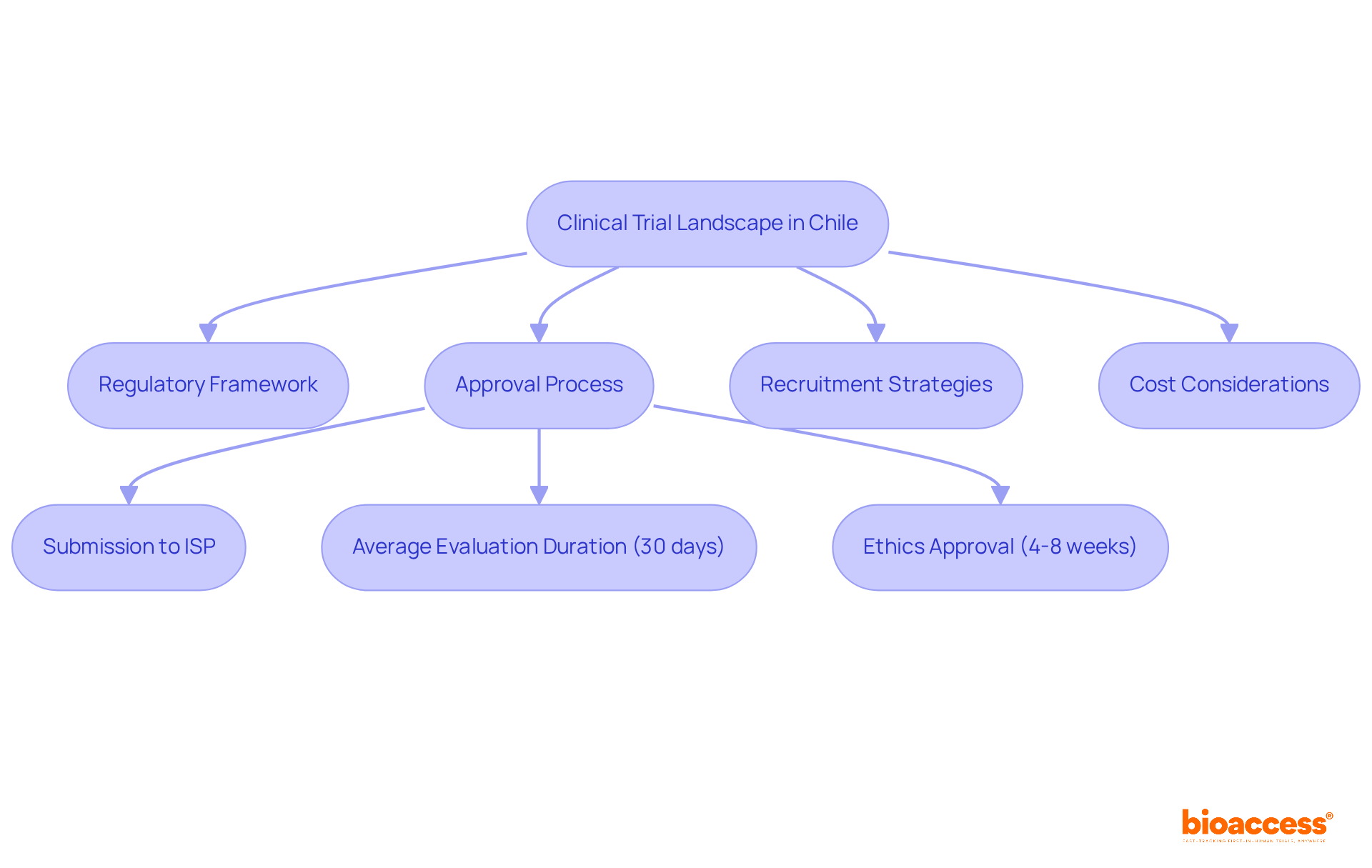
Understand the Clinical Trial Landscape in Chile
While many nations vie for prominence in clinical research, this nation stands out as a leader in the MedTech, Biopharma, and Radiopharma sectors. The regulatory framework is optimized, with the Instituto de Salud Pública (ISP) acting as the main authority supervising clinical studies. This framework facilitates rapid approvals, boasting an average evaluation duration of approximately 30 business days post-submission. Such efficiency is supported by a strong healthcare system and a varied patient demographic, making the region particularly appealing for first-in-human studies.
The commitment to ethical research practices ensures that all studies comply with ICH-GCP standards, instilling confidence among sponsors and stakeholders. Moreover, the country facilitates first-in-human device evaluations 40% quicker than the US FDA IDE pathway, with ethics approval usually granted within 4-8 weeks. This blend of speed and cost-effectiveness positions the region as a strategic advantage for early-stage medical research. Total program expenses for FIH studies range from $250K to $500K, significantly lower than the $500K to $1.2M required in the US.
However, many organizations find the approval process complex, often leading to delays and increased costs. Engaging with local healthcare providers and patient advocacy groups can enhance participant recruitment, which is vital given that recruitment rates can be twice as fast compared to traditional methods. Failing to engage with local healthcare providers can result in slower recruitment rates, jeopardizing study timelines. By leveraging these insights, organizations can not only expedite their research but also enhance their competitive edge in the global market. Furthermore, bioaccess®’s Innovation Runway provides a customized route for MedTech, Biopharma, and Radiopharma startups, speeding up their progress from prototype to development milestones, ensuring they can secure funding and connect with strategic partners effectively.
Identify Regulatory Requirements and Key Agencies
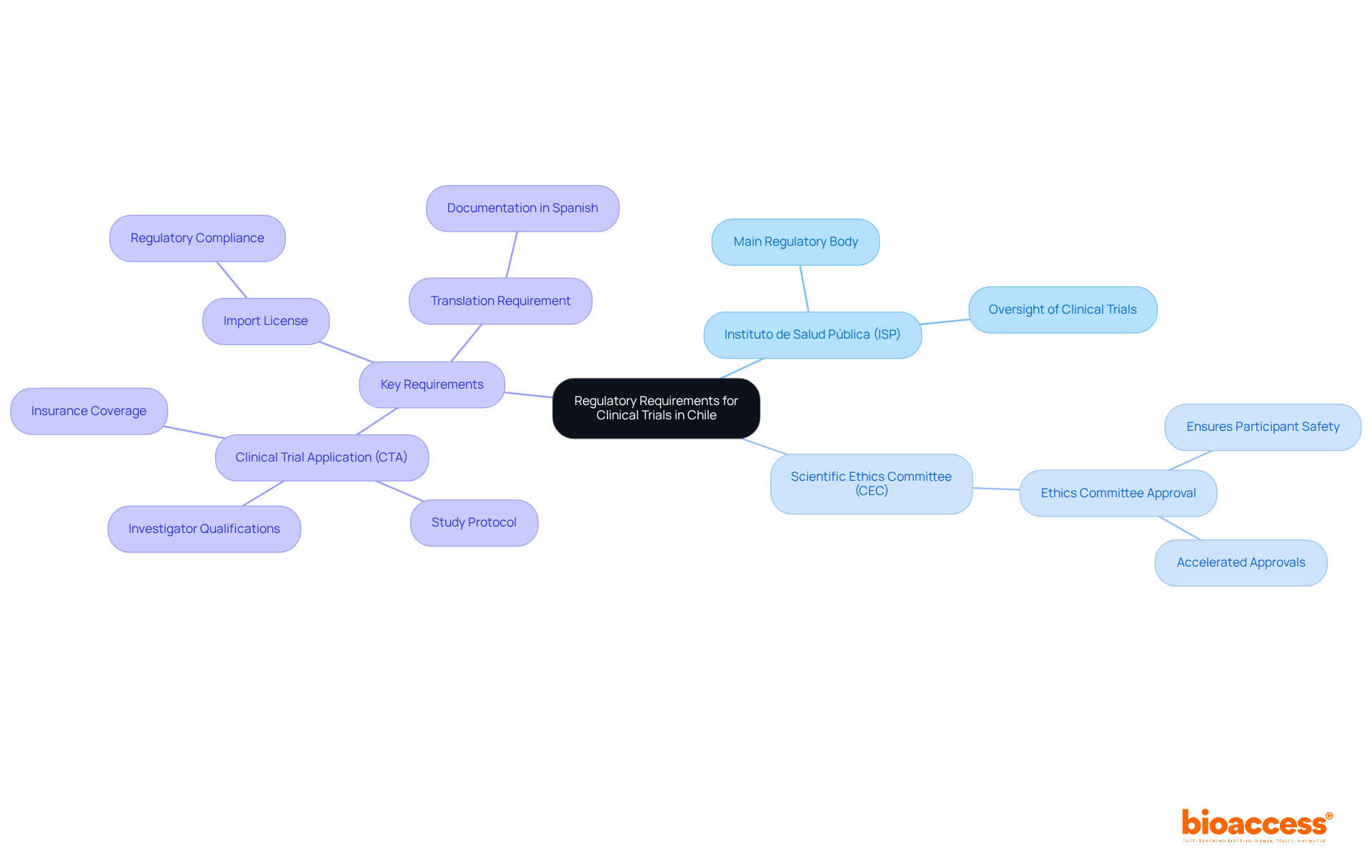
Navigating the regulatory landscape for clinical trial approval in Chile is crucial for successful study execution. The Instituto de Salud Pública (ISP) serves as the main regulatory body overseeing clinical trial approval in Chile. To start a medical study, sponsors need to secure clinical trial approval in Chile from an accredited Scientific Ethics Committee (CEC). This process typically takes 4 to 8 weeks. Afterward, the Clinical Trial Application (CTA) must be submitted to the ISP for clinical trial approval in Chile, which includes the study protocol, ethics approval, and any necessary import licenses for investigational products.
Key regulatory requirements include:
- Ethics Committee Approval: This is crucial for ensuring participant safety and ethical compliance, reflecting the commitment to high ethical standards in research. With bioaccess®’s Global Trial Accelerators™, sponsors can benefit from accelerated ethics approvals, often achieved within 4-8 weeks, significantly reducing time to market.
- Clinical Trial Application (CTA): The CTA must encompass comprehensive documentation, including the study protocol, qualifications of the investigators, and proof of insurance coverage for participants.
- Import License: Necessary for any investigational products being brought into the region, ensuring adherence to local regulations.
- Translation Requirement: All documentation must be translated into Spanish, as this is a mandatory requirement for submission.
Grasping these requirements can really help you navigate the clinical trial approval process in Chile and reduce delays. Proactive communication with the ISP and the CEC can further streamline operations, enhancing the feasibility of clinical trial approval in Chile’s robust healthcare landscape. Significantly, the nation executed over 150 health-related studies in 2025, highlighting its robust healthcare system and dedication to promoting medical research. By taking advantage of early-stage studies in Latin America, companies can benefit from cost-effectiveness and quick patient recruitment, leading to improved research results. By leveraging the strategic advantages of early-stage studies in Latin America, companies can not only enhance their research outcomes but also contribute to the region’s growing reputation in medical innovation.
Prepare and Submit Required Documentation
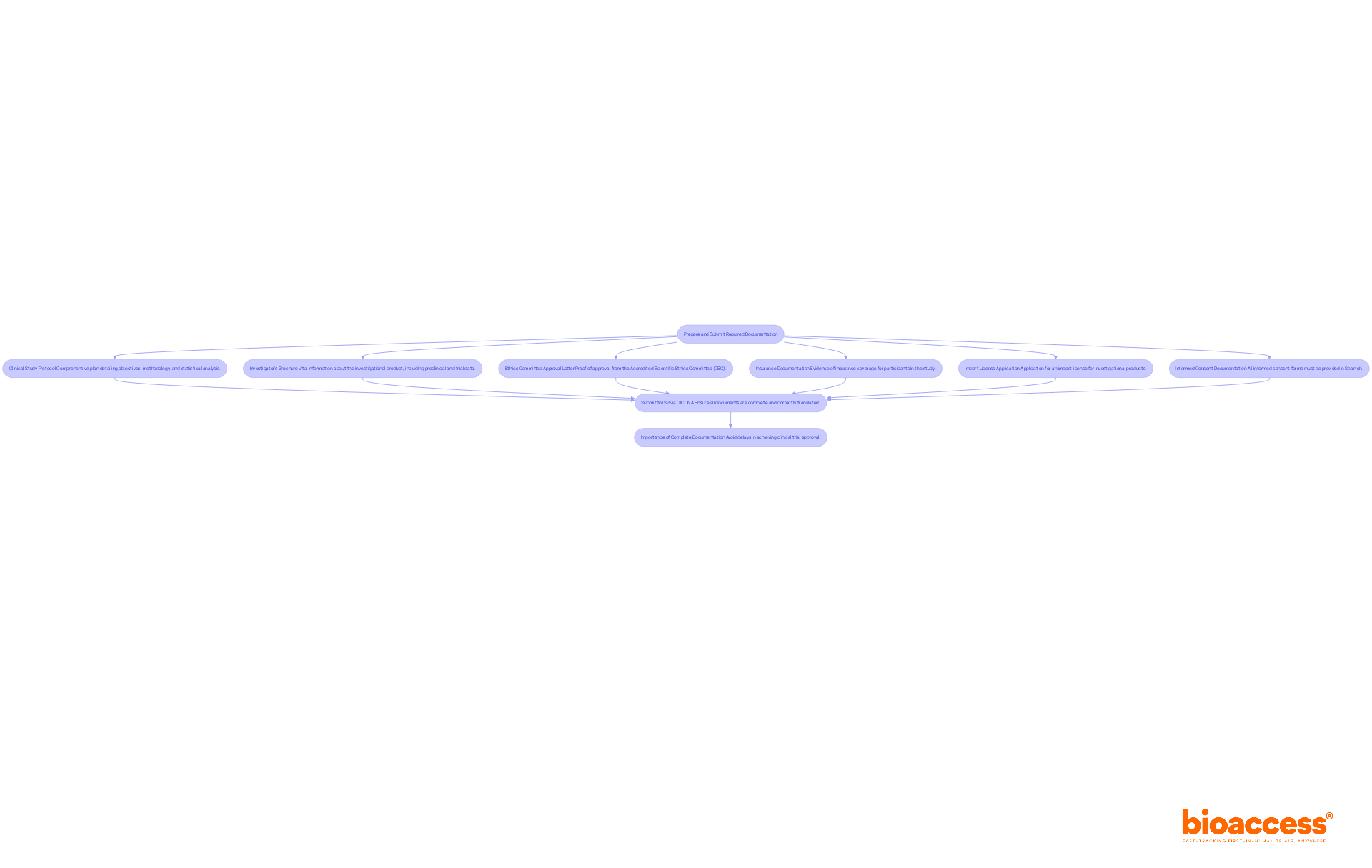
Navigating the regulatory landscape for clinical trial approval in Chile can be daunting, but understanding the necessary documentation is crucial for success. To prepare for submission, sponsors must compile a comprehensive set of documents that meet the Instituto de Salud Pública (ISP) requirements. The following steps outline the necessary documentation:
- Clinical Study Protocol: A comprehensive plan detailing the study’s objectives, methodology, and statistical analysis, ensuring adherence to ICH-GCP standards, which are essential for regulatory acceptance in Chile.
- Investigator’s Brochure: This document offers vital information about the investigational product, including preclinical and trial data, which is critical for the ethics committee’s evaluation.
- Ethics Committee Approval Letter: Proof of approval from the Accredited Scientific Ethics Committee (CEC) is mandatory before submitting to the ISP, ensuring that ethical standards are upheld.
- Insurance Documentation: Evidence of insurance coverage for participants in the study is required to protect their welfare and comply with local regulations.
- Import License Application: If applicable, submit an application for an import license for any investigational products, which typically requires 2-4 weeks for ISP authorization.
- Informed Consent Documentation: All informed consent forms must be provided in Spanish, in accordance with Law 20.120, to ensure participant understanding and compliance with local regulations.
Once all documents are prepared, they should be submitted to the ISP through their online platform, GICONA. It’s essential to have all your documents complete, correctly translated into Spanish, and aligned with the requirements to avoid any delays in achieving clinical trial approval in Chile. The assessment period for research related to clinical trial approval in Chile usually spans around 30 days. Have you ever faced delays due to incomplete documentation or misunderstandings of regulations? These common issues can create significant hurdles for sponsors. By leveraging bioaccess®’s expertise, sponsors can navigate these complexities more efficiently, leading to faster approvals and reduced costs. Imagine cutting down your study timelines and costs by up to 30%-that’s what bioaccess® can help you achieve in navigating these regulatory frameworks in Latin America.
Navigate the Review Process and Address Challenges
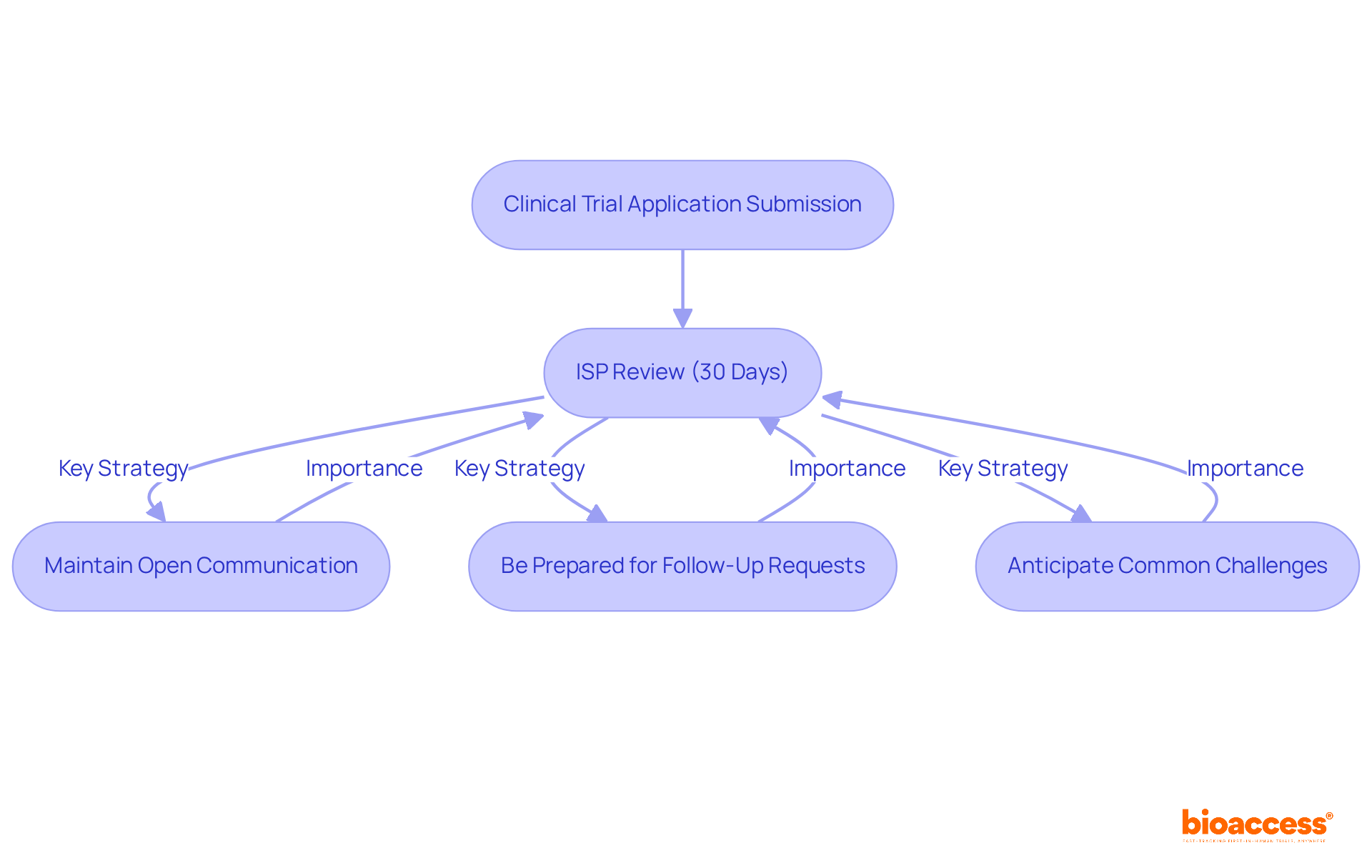
Navigating the complexities of the Clinical Trial Application process is crucial for sponsors aiming for clinical trial approval in Chile‘s evolving regulatory landscape. Upon submission of the Clinical Trial Application, the Instituto de Salud Pública (ISP) undertakes a comprehensive review, typically lasting around 30 days. During this critical phase, sponsors must be ready to respond to any inquiries or requests for additional information from the ISP. Here are effective strategies to navigate this process:
- Maintain Open Communication: Establish a direct line of communication with the ISP to clarify any questions or concerns that may arise during the review. This proactive approach can help mitigate misunderstandings and streamline the approval process.
- Be Prepared for Follow-Up Requests: The ISP may require additional documentation or clarifications. Having these materials readily available can significantly expedite the review timeline.
- Anticipate Common Challenges: Delays in the review process can lead to significant setbacks for sponsors, impacting timelines and resources. Address these proactively by ensuring that all submissions are thorough and compliant with ICH-GCP standards and local regulations. Annette Rid emphasizes that establishing a three-step ethical framework is essential for addressing significant ethical concerns in research studies.
Furthermore, it is essential to recognize that there has been a 20% reduction in the number of medical studies registered in the country following the implementation of Law 20.850, which underscores the changing regulatory environment. By mastering the ISP review process, sponsors can not only avoid delays but also enhance their chances of achieving clinical trial approval in Chile, positioning themselves for success in a competitive landscape. This strategic preparation not only enables smoother interactions with regulatory bodies but also aligns with bioaccess®’s mission to accelerate clinical development in Latin America. By leveraging the region’s regulatory pathways and expertise, we support MedTech and Biopharma companies in their first-in-human initiatives. Testimonials from our clients, such as Dr. Martinez-Clark, highlight how bioaccess® has successfully navigated these regulatory challenges, ensuring efficient trial management and compliance, ultimately leading to faster patient recruitment and reduced costs.
Conclusion
For MedTech, Biopharma, and Radiopharma companies, the clinical trial approval process in Chile offers both challenges and significant opportunities. The regulatory framework, spearheaded by the Instituto de Salud Pública (ISP), is designed to facilitate swift approvals, making Chile an attractive destination for early-stage clinical research. With an average evaluation period of approximately 30 business days and a commitment to ethical research practices, the country stands out as a leader in the region.
In this guide, we’ve explored the essential steps for conducting successful clinical trials in Chile, including:
- Understanding the landscape
- Identifying regulatory requirements
- Preparing necessary documentation
- Navigating the review process
Engaging with local healthcare providers and utilizing resources like bioaccess®’s Innovation Runway can significantly enhance participant recruitment and streamline the approval process, ultimately positioning organizations for success in a competitive market.
By embracing these insights, organizations can not only enhance their research outcomes but also solidify their position in the evolving landscape of medical innovation. As the demand for innovative medical solutions continues to rise, navigating the complexities of clinical trial approval in Chile will empower organizations to contribute to the region’s growing reputation in medical innovation.
Frequently Asked Questions
What makes Chile a leader in clinical research for MedTech, Biopharma, and Radiopharma sectors?
Chile stands out due to its optimized regulatory framework, efficient approval processes, and a strong healthcare system with a diverse patient demographic, making it particularly appealing for first-in-human studies.
What is the role of the Instituto de Salud Pública (ISP) in Chile’s clinical trials?
The ISP is the main authority supervising clinical studies in Chile, ensuring that all trials comply with ethical research practices and ICH-GCP standards.
How long does it typically take to receive approval for clinical studies in Chile?
The average evaluation duration for clinical study submissions in Chile is approximately 30 business days.
How does Chile’s approval process for first-in-human studies compare to the US FDA IDE pathway?
Chile facilitates first-in-human device evaluations 40% quicker than the US FDA IDE pathway, with ethics approval usually granted within 4-8 weeks.
What are the typical costs associated with first-in-human studies in Chile?
Total program expenses for first-in-human studies in Chile range from $250K to $500K, which is significantly lower than the $500K to $1.2M required in the US.
What challenges do organizations face in the approval process for clinical trials in Chile?
Many organizations find the approval process complex, which can lead to delays and increased costs if not navigated properly.
How can engaging with local healthcare providers enhance participant recruitment in Chile?
Engaging with local healthcare providers and patient advocacy groups can significantly enhance participant recruitment rates, which can be twice as fast compared to traditional methods.
What is bioaccess®’s Innovation Runway, and how does it assist startups in Chile?
bioaccess®’s Innovation Runway provides a customized route for MedTech, Biopharma, and Radiopharma startups, helping them speed up their progress from prototype to development milestones, ensuring they can secure funding and connect with strategic partners effectively.
List of Sources
- Understand the Clinical Trial Landscape in Chile
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- First-in-Human Medical Device Trial in Chile | bioaccess® (https://bioaccessla.com/blog/first-in-human-medical-device-trial-chile)
- Chile First-in-Human Clinical Trial Market Size, Share, Growth & Trends 2025–2033 (https://reedintelligence.com/market-analysis/chile-first-in-human-clinical-trial-market)
- Identify Regulatory Requirements and Key Agencies
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- The Best Places outside U.S. to Run Clinical Trials According to their Regulatory Times | FOMAT (https://fomatmedical.com/blogs-updates/best-places-outside-us-to-run-clinical-trials)
- Prepare and Submit Required Documentation
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Chile (ISP – Instituto de Salud Pública) – PharmaRegulatory.in – India’s Regulatory Knowledge Hub (https://pharmaregulatory.in/country-specific-regulatory-affairs/chile-isp-instituto-de-salud-publica)
- Chile Clinical Trial Regulations | ISP Guide | bioaccess® (https://bioaccessla.com/regulatory-guide/chile)
- Navigate the Review Process and Address Challenges
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- LATAM regulatory timelines and approval benchmarks – PharmaRegulatory.in – India’s Regulatory Knowledge Hub (https://pharmaregulatory.in/latam-regulatory-timelines-and-approval-benchmarks)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)

Leave a Reply