Introduction
For MedTech companies, mastering the investigational device exemption (IDE) process in Chile is essential for launching innovative medical devices successfully. With the Instituto de Salud Pública (ISP) overseeing this critical regulatory pathway, understanding the specific IDE requirements can significantly enhance the chances of successful clinical trials. Navigating these complexities can lead to significant setbacks for companies.
What strategies can you use to make the IDE submission process smoother and take advantage of Chile’s regulatory benefits for quicker market access?
Understand the Investigational Device Exemption (IDE) Requirements in Chile
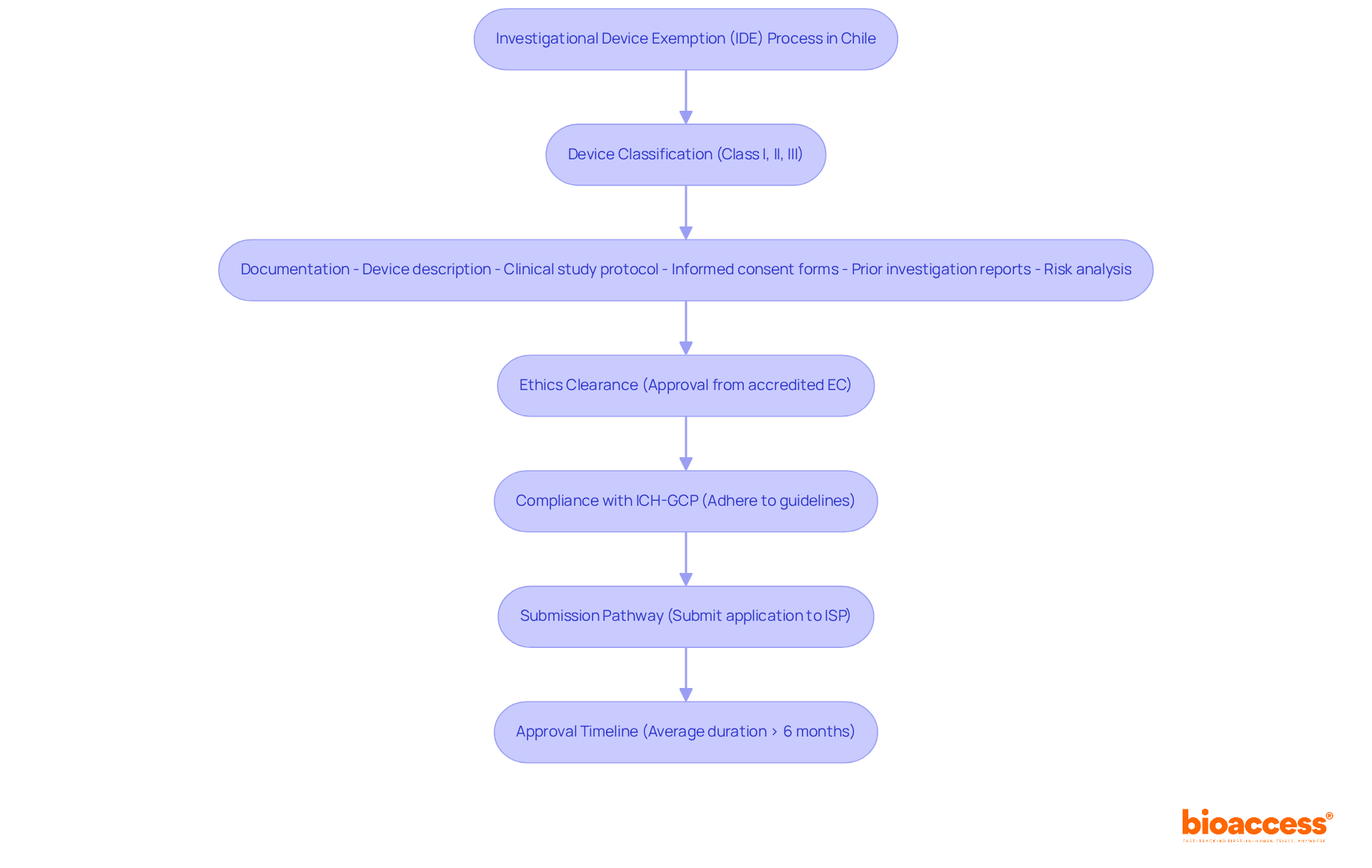
Navigating the investigational device exemption Chile process is not just a regulatory formality; it’s a critical step that can determine the success of your clinical research endeavors. Understanding the specific requirements set forth by the Instituto de Salud Pública (ISP) is crucial. The IDE facilitates the clinical investigation of medical devices to collect essential safety and effectiveness data. Here are the key requirements:
-
Device Classification: Identify the classification of your device (Class I, II, or III), as this determines the IDE requirements. Class I devices may be exempt from the investigational device exemption Chile requirements, while Class II and III devices generally require a complete IDE process.
-
Documentation: Prepare comprehensive documentation, which must include:
- Device description and intended use
- Clinical study protocol
- Informed consent forms
- Prior investigation reports
- Risk analysis and management plans
-
Ethics Clearance: Obtain consent from an accredited ethics committee (EC) in Chile, which is essential prior to submitting your investigational device exemption Chile request. This step is critical to ensure adherence to ethical guidelines and local health needs, as emphasized by experts in the field.
-
Compliance with ICH-GCP: Ensure that your study adheres to International Council for Harmonisation – Good Clinical Practice (ICH-GCP) guidelines, which are critical for regulatory acceptance. Compliance with these standards not only enhances the credibility of your research but also aligns with the ethical implications of conducting studies in Latin America.
-
Submission Pathway: Familiarize yourself with the submission pathway through the ISP, which involves submitting your investigational device exemption Chile application along with all required documentation for review. Significantly, the ISP has optimized the research authorization procedure, reducing bureaucratic delays by over 30%, greatly accelerating the start of your clinical investigation. This efficiency gives MedTech startups a real edge, helping them connect with strategic acquirers and secure funding faster.
-
Approval Timeline: Be aware that the average approval duration for research studies in Chile can exceed six months. Understanding this timeline is essential for effective project management and planning.
Understanding these requirements is essential. Participant recruitment in Chile can be a significant hurdle for clinical trials, often leading to delays and increased costs. By addressing these challenges, you can enhance your readiness for the IDE process and increase your chances of a successful submission. As Julio G. Martinez-Clark, CEO of bioaccess®, states, “In the dynamic landscape of clinical research, navigating the regulatory framework in Chile is crucial for success.” With bioaccess®’s support, innovative startups can leverage these insights to accelerate their clinical trials and achieve their goals.
Prepare Your IDE Application: Essential Documentation and Steps
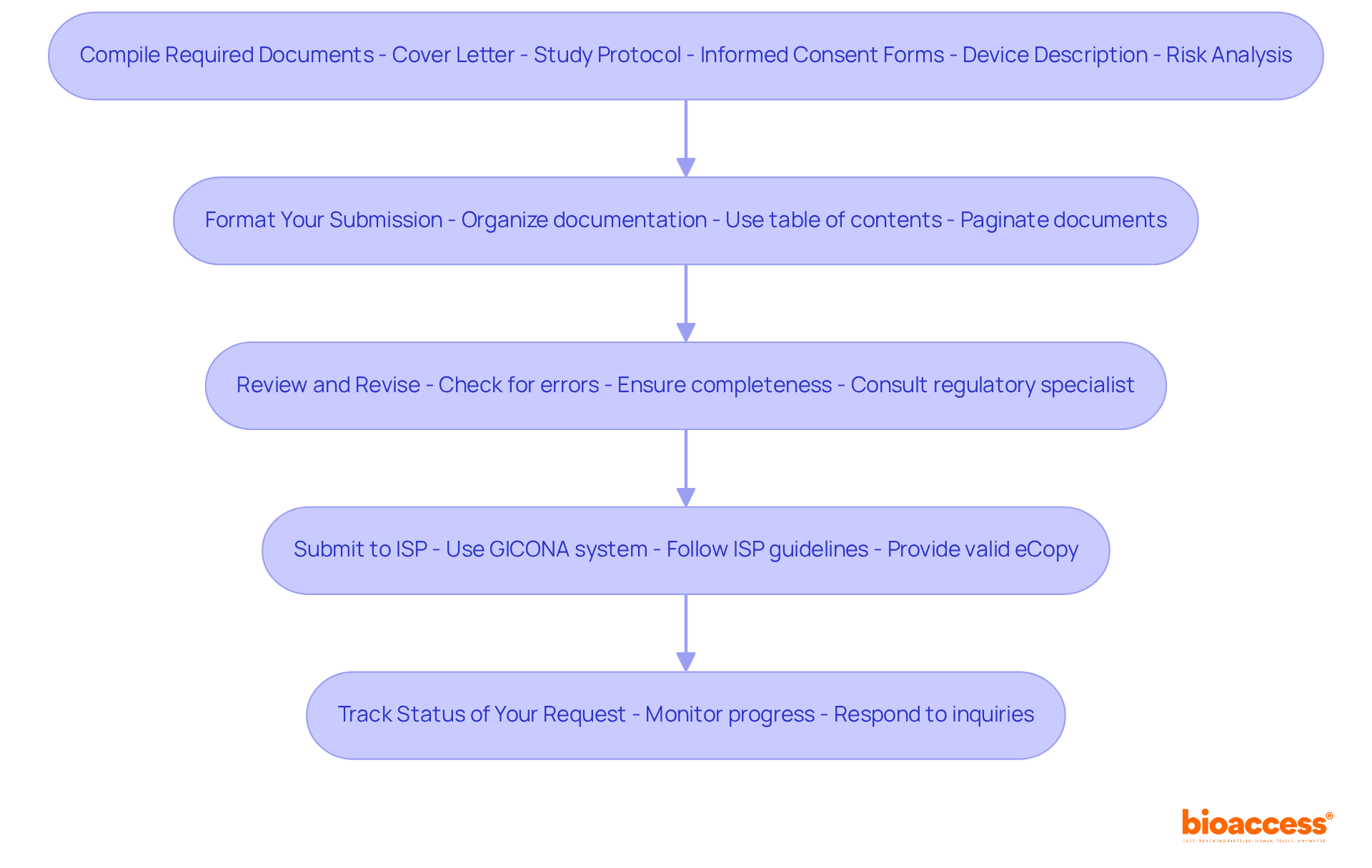
Navigating the complexities of IDE submissions can be daunting, yet it is crucial for advancing clinical research effectively. Preparing your IDE software involves several critical steps and documentation requirements. Follow these guidelines to ensure a thorough and compliant submission:
-
Compile Required Documents: Gather all necessary documents, including:
- Cover Letter: Introduce your application and summarize the purpose of the IDE.
- Study Protocol: Detail the study design, objectives, methodology, and statistical analysis plan.
- Informed Consent Forms: Ensure these are clear and compliant with local regulations, including a statement about the availability of trial descriptions on ClinicalTrials.gov.
- Device Description: Provide a comprehensive description of the device, including its intended use, design specifications, mechanism of action, and any prior testing results.
- Risk Analysis: Include a detailed risk assessment and management plan, outlining potential risks and mitigation measures.
-
Format Your Submission: Organize your documentation clearly and logically. Use a table of contents and paginate all documents for easy navigation.
-
Review and Revise: Make sure to review all your documents carefully to catch any errors and ensure everything is complete. Consider having a regulatory specialist or legal consultant examine your documentation prior to filing to ensure adherence to local laws, including ICH-GCP standards and the stipulations of ANVISA, INVIMA, and COFEPRIS.
-
Submit to ISP: Once your documentation is complete, submit it through the ISP’s GICONA system. Ensure that you adhere to any specific guidelines for presenting provided by the ISP, including the requirement for a valid eCopy.
-
Track Status of Your Request: After sending your materials, monitor the progress of your inquiry through the ISP portal and be ready to respond quickly to any questions or requests for further information.
By leveraging the investigational device exemption Chile, you can take advantage of the swift approval timelines in countries like Panama, El Salvador, and Chile to significantly accelerate your path to market for your innovative medical device. This expedited process not only facilitates faster access to the market but also allows you to save significantly on costs, with potential savings of $25K per patient through pre-negotiated site contracts. Ultimately, by streamlining your IDE submission process, you not only enhance your chances of approval but also position your innovation for success in a competitive market.
Navigate the Submission Process: Timelines and Regulatory Interactions
Navigating the investigational device exemption Chile application process can be daunting, but with the right strategy, you can turn challenges into opportunities. Successfully managing this critical phase of the investigational device exemption Chile requires meticulous planning and a clear understanding of the associated timelines. Here’s how to effectively approach this process, leveraging insights from bioaccess® to enhance your strategy:
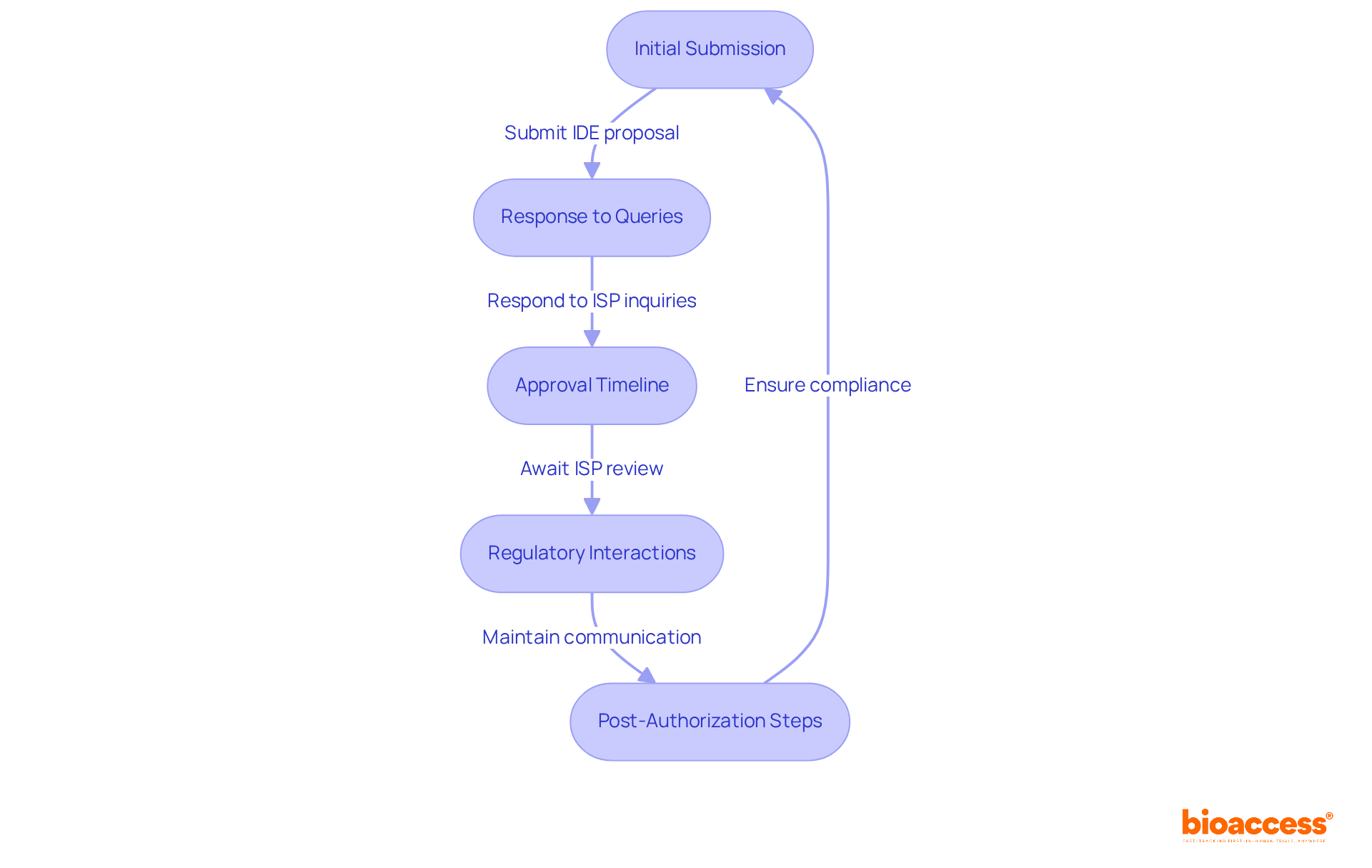
- Initial Submission: Prepare your IDE proposal, ensuring that all documentation is translated into Spanish, as this is a mandatory requirement for submission. Submit it to the Instituto de Salud Pública (ISP) via the GICONA system for the investigational device exemption Chile. Expect an initial review period of around 30 days, during which the ISP will assess the completeness of your submission. Utilizing bioaccess®’s Global Trial Accelerators™ can provide you with the latest regulatory updates and market access strategies tailored for the Latin American landscape.
- Response to Queries: Be ready for potential inquiries from the ISP. They may request further information or clarification on specific aspects of your submission. Prompt responses are essential to maintain the review timeline and demonstrate your commitment to compliance. Without thorough preparation, you risk delays that could derail your project. Bioaccess® can assist in ensuring that your documentation meets all necessary standards.
- Approval Timeline: Once your application is deemed complete, the ISP will conduct a thorough review. The overall timeline for the investigational device exemption Chile can vary from 2 to 4 months, depending on the complexity of your device and the detail of your submission. Recognizing that the evaluation timeframe for the investigational device exemption Chile generally spans around 30 days can assist you in planning effectively. By leveraging bioaccess®, you can turn potential delays into streamlined progress, ensuring you are well-prepared for each phase.
- Regulatory Interactions: Maintain open lines of communication with the ISP throughout the process. Regularly check for updates on your application status and proactively address any concerns raised by the regulatory body. Effective communication can significantly streamline the endorsement process. Furthermore, obtaining ethics committee consent is a vital step that should not be overlooked. Bioaccess® provides insights into optimal practices for these interactions, boosting your chances of a seamless evaluation process.
- Post-Authorization Steps: Upon receiving consent, ensure adherence to any additional requirements established by the ISP, including monitoring and reporting obligations during the clinical trial. Staying compliant is vital for keeping your regulatory standing and ensuring the safety of participants. Leveraging bioaccess®’s resources can help you stay informed about ongoing compliance requirements and best practices.
Mastering these steps not only accelerates your clinical trial efforts but also positions you for success in the competitive Latin American market.
Troubleshoot Common Challenges in IDE Submission
Navigating the IDE application process is fraught with challenges that can hinder your clinical research efforts. Here are some common issues and how to troubleshoot them:
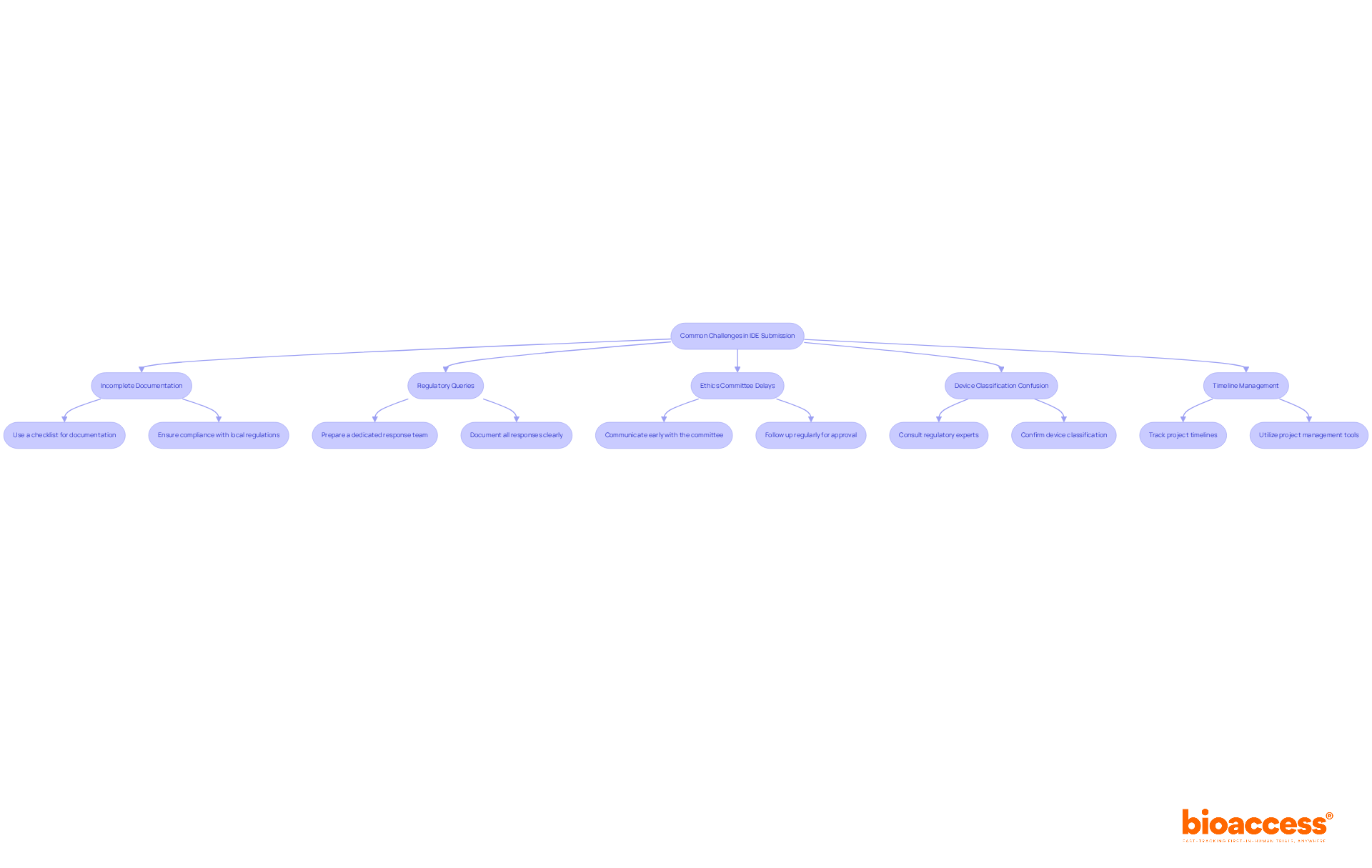
- Incomplete Documentation: One of the most frequent reasons for delays is incomplete documentation. Make sure you include all required documents and format them according to the guidelines from local regulatory authorities like ANVISA in Brazil or COFEPRIS in Mexico. Use a checklist to verify that nothing is missing before submission, as over 60% of FDA warning letters in 2022 cited inadequate documentation practices as a key issue. At bioaccess®, we emphasize the importance of meticulous documentation to enable quicker endorsements and support the acceleration of Phase I trials with FDA/EMA-ready clinical data.
- Regulatory Queries: Have you ever faced questions from regulatory authorities? If so, responding promptly and thoroughly is crucial. Delays in communication can extend the review process significantly. Prepare a dedicated team to handle queries efficiently, ensuring that all responses are clear and well-documented to facilitate a smoother review. Our experience in Latin America allows us to navigate these queries effectively, ensuring timely responses and compliance with ICH-GCP standards.
- Ethics Committee Delays: If your ethics committee endorsement is postponed, communicate with them early in the process. Provide them with all necessary information and follow up regularly to ensure timely approval. Taking this proactive approach can significantly reduce potential delays in your IDE timeline. With bioaccess®, you can leverage our established relationships with ethics committees to expedite this process, enhancing patient recruitment and overall efficiency.
- Device Classification Confusion: Misunderstanding the classification of your device can lead to incorrect entries. Consult with regulatory experts to confirm the classification and ensure that you are following the correct IDE requirements. This step is crucial, as incorrect classifications can lead to significant delays in the approval process. Our experts at bioaccess® understand the nuances of device classification in Latin America, ensuring you stay compliant with local regulations.
- Timeline Management: Keep track of all project timelines and set internal deadlines to ensure that you stay on schedule. Utilize project management tools to monitor progress and identify potential bottlenecks early. By managing timelines effectively, you can enhance the efficiency of your IDE process and increase the likelihood of a successful outcome. Bioaccess® provides comprehensive support in timeline management, ensuring that your clinical programs move forward without unnecessary delays.
By addressing these challenges head-on, you can not only streamline your IDE submission but also enhance the overall success of your clinical programs in Latin America.
Conclusion
For MedTech companies, navigating the Investigational Device Exemption (IDE) process in Chile is not just a regulatory hurdle; it’s a critical pathway to success in clinical trials. Mastering this regulatory pathway speeds up the timeline for bringing innovative medical devices to market and boosts the success rate of clinical research.
Key elements of the IDE process include:
- Understanding device classification
- Preparing comprehensive documentation
- Obtaining ethics clearance
- Ensuring compliance with ICH-GCP guidelines
Each of these steps plays a critical role in facilitating a smooth application process, ultimately leading to quicker approvals and reduced costs. However, many companies struggle with the complexities of regulatory compliance, which can delay their progress. Staying in touch with regulatory authorities like the Instituto de Salud Pública (ISP) can help you tackle common challenges head-on, ensuring that any potential roadblocks are addressed promptly.
In the competitive landscape of early-stage clinical trials, leveraging the advantages of conducting research in Chile can yield substantial benefits. Without a streamlined approach, companies risk falling behind in the competitive landscape of clinical trials. By streamlining the IDE application process, companies can save time and resources while enhancing patient recruitment efforts. This strategic approach positions organizations to capitalize on the growing opportunities within Latin America’s clinical trial ecosystem, ultimately driving innovation and improving patient outcomes. Embracing a strategic approach to the IDE process can be the difference between leading the market and being left behind.
Frequently Asked Questions
What is the purpose of the Investigational Device Exemption (IDE) in Chile?
The IDE facilitates the clinical investigation of medical devices in Chile to collect essential safety and effectiveness data, playing a critical role in the success of clinical research endeavors.
How is the device classification determined, and why is it important?
The device classification is determined by identifying whether the device is Class I, II, or III. This classification is important as it dictates the specific IDE requirements; Class I devices may be exempt, while Class II and III devices generally require a complete IDE process.
What documentation is required for the IDE application in Chile?
The required documentation includes a device description and intended use, clinical study protocol, informed consent forms, prior investigation reports, and risk analysis and management plans.
Why is ethics clearance necessary for the IDE process?
Ethics clearance is necessary to obtain consent from an accredited ethics committee in Chile, ensuring adherence to ethical guidelines and local health needs before submitting the IDE request.
What are the compliance requirements for conducting clinical studies in Chile?
Studies must comply with the International Council for Harmonisation – Good Clinical Practice (ICH-GCP) guidelines, which enhance the credibility of research and align with ethical implications in Latin America.
What is the submission pathway for the IDE application through the Instituto de Salud Pública (ISP)?
The submission pathway involves submitting the IDE application along with all required documentation to the ISP for review. The ISP has optimized the research authorization procedure, significantly reducing bureaucratic delays by over 30%.
What is the average approval timeline for research studies in Chile?
The average approval duration for research studies in Chile can exceed six months, making it essential for effective project management and planning.
What challenges might be faced during participant recruitment in Chile for clinical trials?
Participant recruitment can be a significant hurdle, often leading to delays and increased costs in clinical trials. Addressing these challenges is crucial for enhancing readiness for the IDE process and increasing submission success rates.
List of Sources
- Understand the Investigational Device Exemption (IDE) Requirements in Chile
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- First-in-Human Medical Device Trial in Chile | bioaccess® (https://bioaccessla.com/blog/first-in-human-medical-device-trial-chile)
- Prepare Your IDE Application: Essential Documentation and Steps
- TOP 25 DOCUMENTATION QUOTES (of 78) | A-Z Quotes (https://azquotes.com/quotes/topics/documentation.html)
- Clinical Trial Requirements In Latin America A Complete Pre Clinical Document Checklist For Sponsors | bioaccess® (https://bioaccessla.com/blog/clinical-trial-requirements-in-latin-america-a-complete-pre-clinical-document-checklist-for-sponsors)
- IDE Application (https://fda.gov/medical-devices/investigational-device-exemption-ide/ide-application)
- Step-by-Step Guide to the IDE Application Package (https://biobostonconsulting.com/step-by-step-guide-to-the-ide-application-package)
- Navigate the Submission Process: Timelines and Regulatory Interactions
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Investigational Device Exemption: Conducting Clinical Studies (https://premier-research.com/perspectives/conducting-clinical-studies-under-an-investigational-device-exemption)
- 7 quotes from 2023 to guide you on the medtech market (https://tiinatyni.com/blogi/7-quotes-from-2023-to-guide-you-on-the-medtech-market)
- Regulatory Jokes: Laughter and Quotes for Professionals | Dr. Verah Oketch posted on the topic | LinkedIn (https://linkedin.com/posts/verahoketch_regulatoryhumor-pharmalife-complianceculture-activity-7397543447412273152-FdfA)
- Troubleshoot Common Challenges in IDE Submission
- IDE Submission Strategy: Critical Review Factors for FDA Approval (https://linkedin.com/pulse/ide-submission-strategy-critical-review-factors-fda-lcpoc)
- Quotes of the week – all about documentation (https://lwn.net/Articles/310569)
- best quotes about software documentation (https://linfo.org/q_documentation.html)
- The Impact of Poor Documentation on Regulatory Compliance – JAF Consulting, Inc. (https://jafconsulting.com/the-impact-of-poor-documentation-on-regulatory-compliance)

Leave a Reply