Introduction
Navigating the FDA 510(k) submission process presents a significant challenge for medical device developers, with a notable 15% of applications facing rejection due to common pitfalls. This article explores essential practices that can greatly enhance the chances of a successful submission. By focusing on critical aspects such as:
- Understanding substantial equivalence
- Preparing comprehensive documentation
- Implementing rigorous performance testing
- Avoiding frequent mistakes
Innovators can better position themselves for success. What strategies can you employ to not only meet FDA requirements but also streamline your path to market approval?
Understand the FDA 510(k) Process and Substantial Equivalence
The outlines the procedure that serves as a crucial premarket submission for verifying a by demonstrating to a legally marketed product, known as a predicate. Understanding is vital; it requires that your product shares the same intended use and technological attributes as the predicate. To navigate this process effectively, consider the following steps:
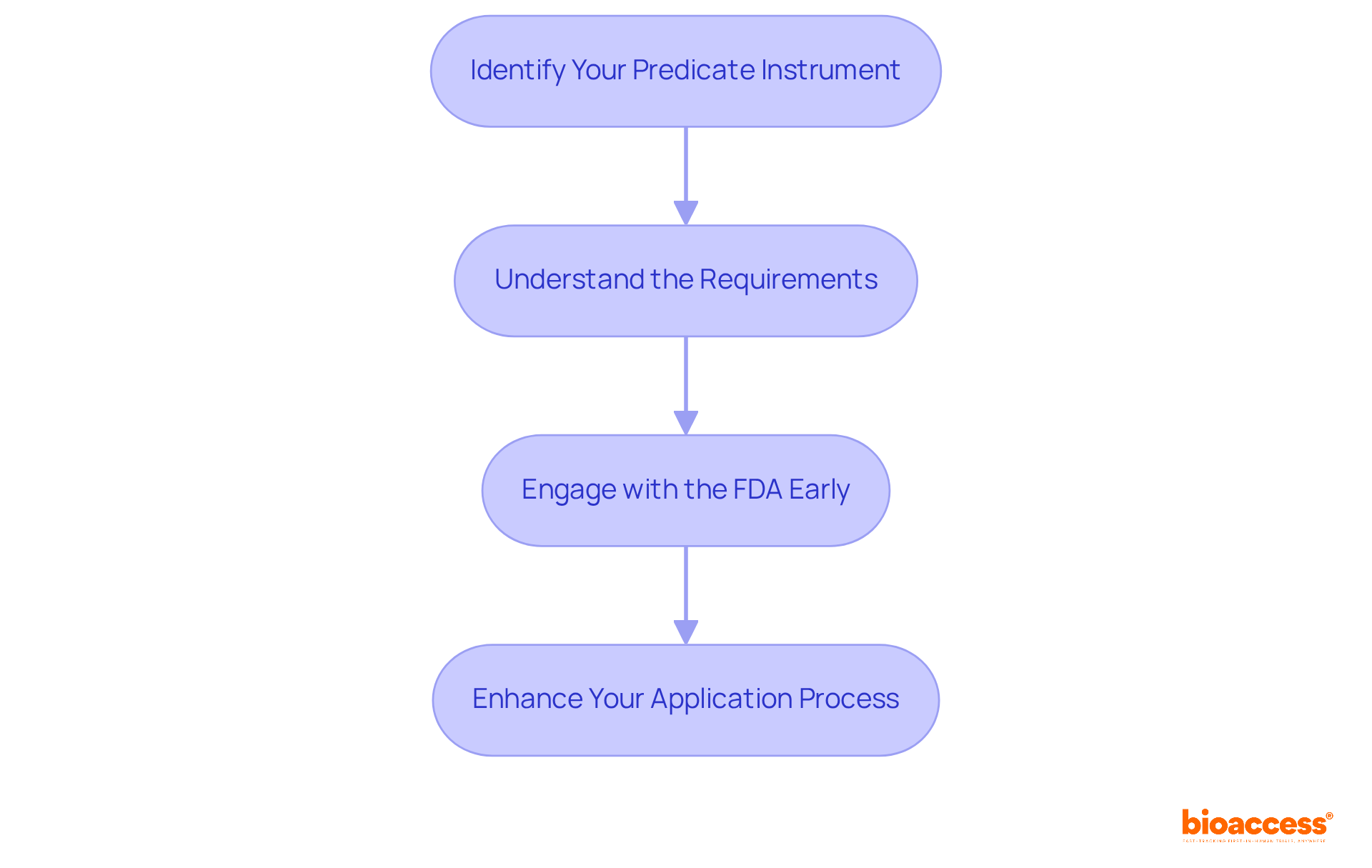
- Identify Your : Conduct thorough research to find a that closely aligns with your product. The highlights that the is an invaluable resource for identifying previously authorized products.
- Understand the Requirements: Familiarize yourself with the , which include intended use, technological characteristics, and performance data. Notably, 85% of 510(k) applications received a Substantially Equivalent decision by September 2022, highlighting the importance of meeting these criteria. Conversely, 15% of submissions did not receive this decision, underscoring the challenges applicants face.
- Engage with the FDA Early: can clarify uncertainties regarding your product’s classification and the necessary documentation. Engaging early can significantly simplify the review process, as evidenced by a reduction in by 5.9%, bringing it to approximately 168.9 days in 2024. The facilitates the approval of around 3,000 medical devices for marketing each year, emphasizing the significance of this system.
By mastering these elements, you can enhance your application process and improve your chances of obtaining .
Prepare Comprehensive Documentation for 510(k) Submission
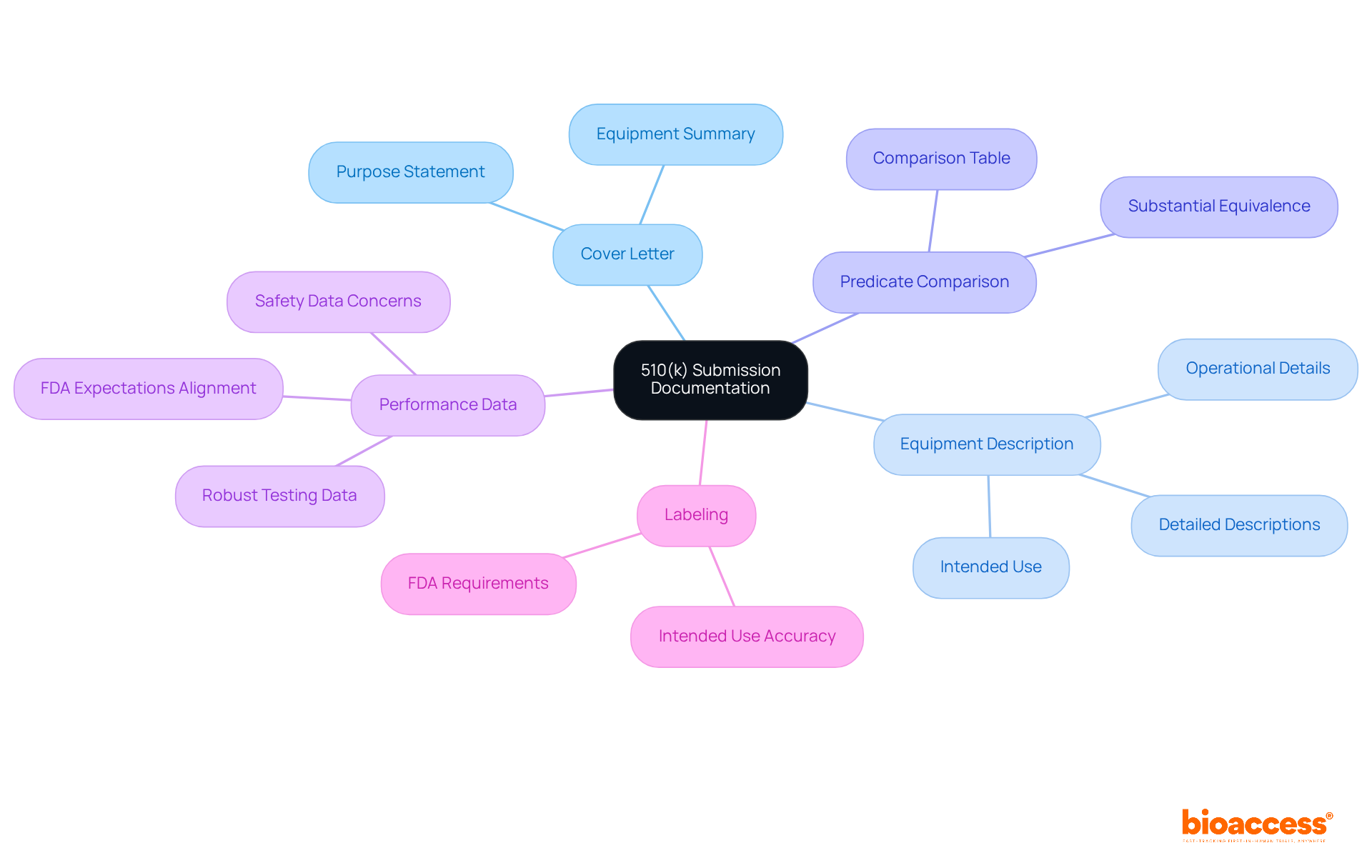
A successful application following relies heavily on the quality and completeness of your documentation. Key components include:
- Cover Letter: Clearly state the purpose of your submission and provide a summary of the equipment.
- : Include detailed descriptions of the equipment, its intended use, and how it operates.
- Predicate Comparison: Create a comparison table that highlights similarities and differences between your item and the predicate, emphasizing .
- : Provide robust data from that supports your claims of , ensuring it aligns with FDA expectations. , raising questions about the adequacy of safety data. This makes thorough documentation even more critical.
- Labeling: Ensure that your and accurately reflects the product’s intended use.
Additionally, staying updated on recent changes in is essential. The aim to improve the efficiency of the related to the clearance pathway. , enhancing the quality of your submission. By carefully preparing these documents, you can avoid common pitfalls, such as incomplete s and absent sterilization validation, facilitating a smoother review experience.
Implement Rigorous Performance Testing and Safety Protocols
To ensure compliance with , implementing rigorous and safety protocols is essential. Here’s a structured approach:
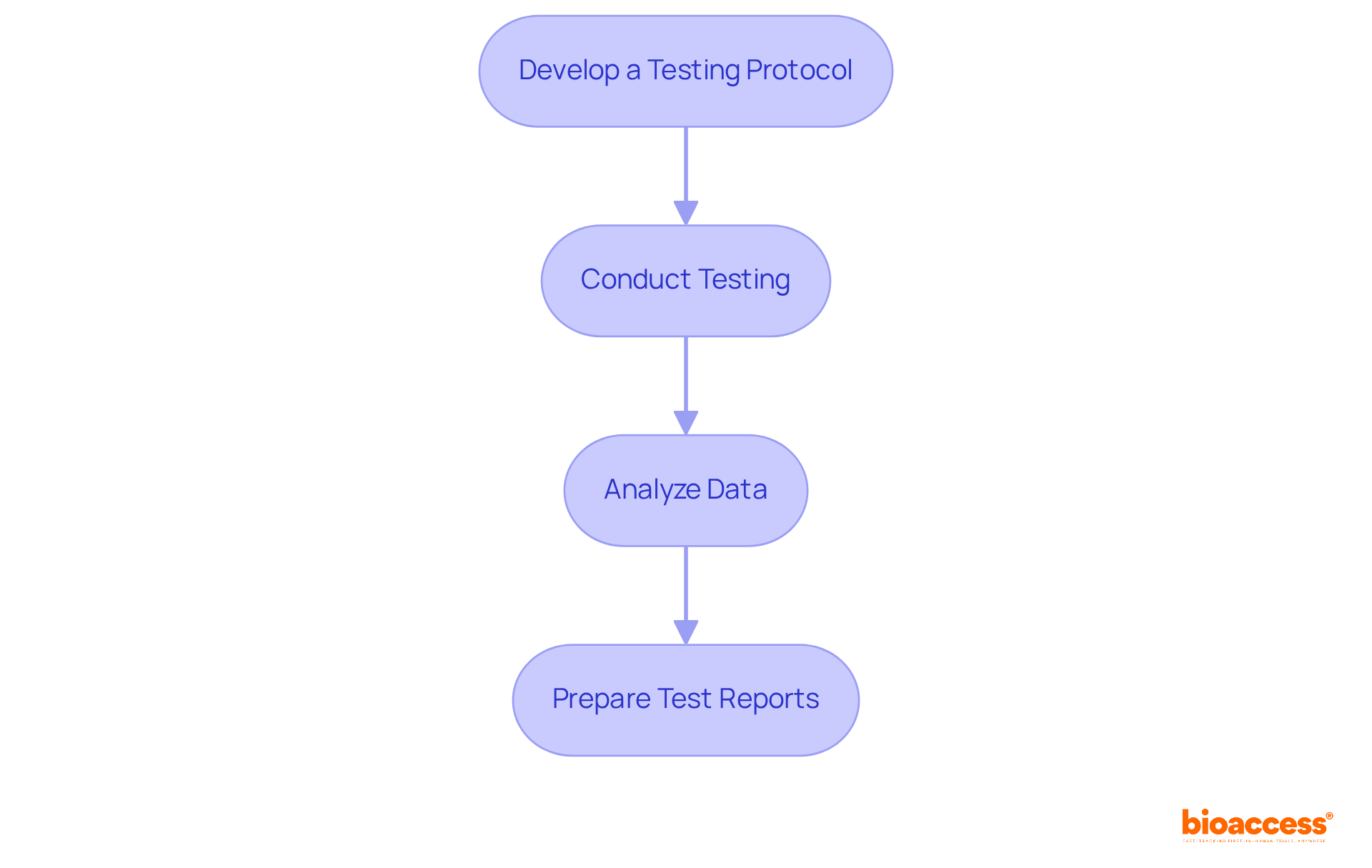
- Develop a Testing Protocol: Clearly outline the specific tests your device will undergo, including . The protocol should be comprehensive, detailing methodologies and operational parameters, and must adhere to . By leveraging bioaccess’s expertise, you can ensure that your protocol aligns with , which is crucial for .
- Conduct Testing: Execute the tests as per the established protocol, meticulously documenting all results. Employ established standards to confirm your testing methods, ensuring that all procedures are clearly defined and comprehended by the staff engaged in the testing activities. Maintaining data integrity throughout this procedure is crucial, as it ensures that all generated data is accurate, consistent, and reliable. With bioaccess’s support, you can expedite this process, as they are built specifically for startups and understand the urgency of generating .
- Analyze Data: Thoroughly evaluate the results to confirm that your device meets the necessary safety and performance criteria. This analysis should identify any deviations and assess their impact, preparing you to provide a comprehensive assessment in your report. Be aware that nearly 32 percent of applications under FDA 510 k guidance failed the initial acceptance for review check in the year leading up to September 2022, a slight improvement from the 35 percent failure rate in 2021, highlighting the importance of thorough preparation. Bioaccess’s experience with successful can provide valuable insights during this phase.
- Prepare Test Reports: Compile detailed reports of all testing conducted, including methodologies, results, and conclusions. This documentation is essential for showing adherence to FDA regulations and should feature a summarizing the complete procedure. Additionally, utilizing a Performance Qualification Checklist can help ensure that all critical aspects of the PQ process are addressed. By collaborating with bioaccess, you can boost the reliability of your proposal, as they specialize in generating FDA-ready tailored for startups.
By following these steps, you can substantiate your claims of safety and effectiveness, significantly enhancing the credibility of your proposal. Comprehensive preparation and documentation can help prevent delays and extra expenses related to the submission process.
Avoid Common Pitfalls in 510(k) Submissions
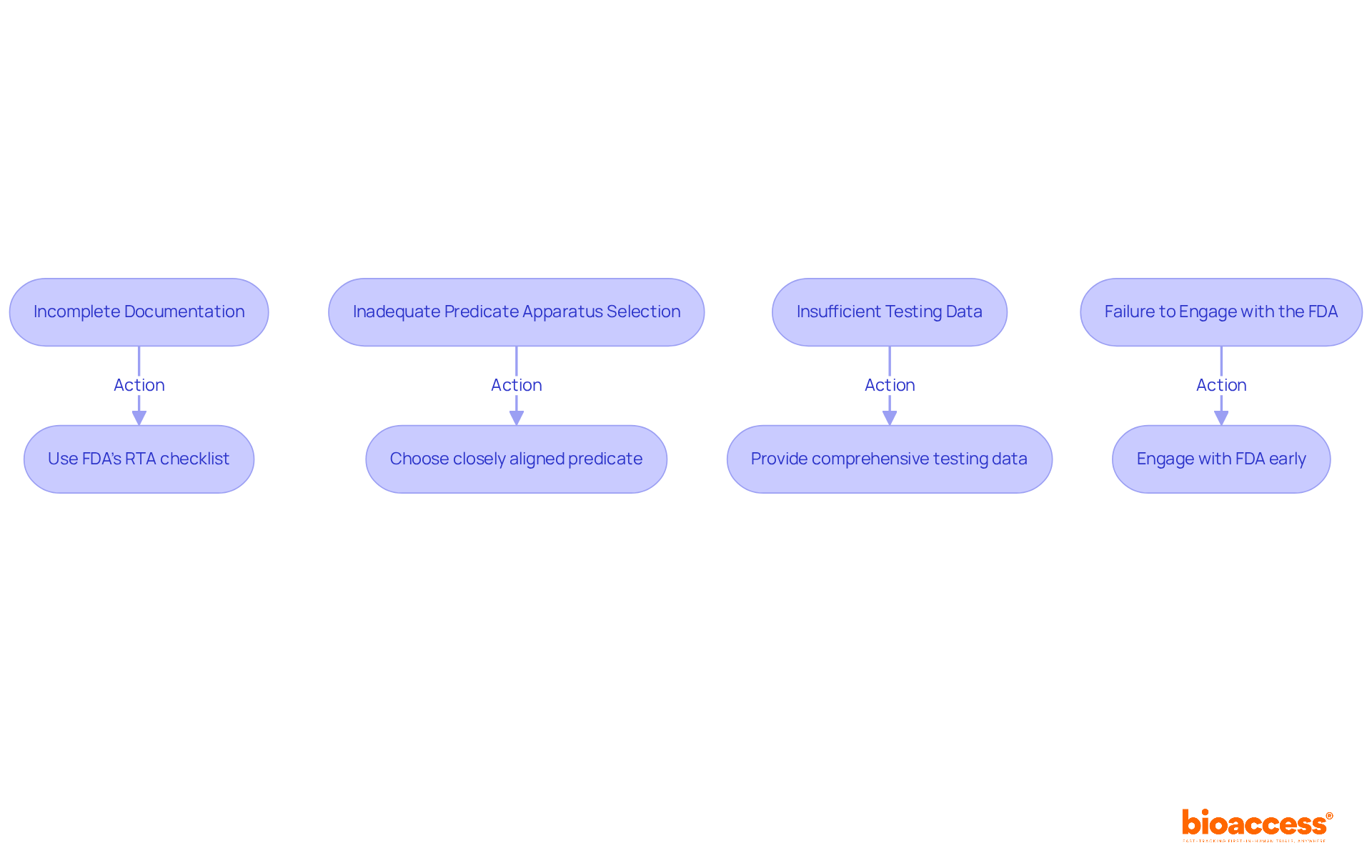
Many entries encounter delays or rejections due to . To avoid these issues, consider the following:
- Incomplete Documentation: Ensure that all required documents are included and complete. Use the FDA’s to verify your submission.
- Inadequate : Carefully choose a predicate apparatus that closely aligns with your product. Avoid selecting devices that are too dissimilar, as this can lead to .
- Insufficient Testing Data: Provide . Avoid vague statements and ensure that all claims are backed by solid evidence.
- : Don’t hesitate to reach out to the FDA for guidance. Early engagement can clarify expectations and help you avoid missteps.
By being aware of these pitfalls and proactively addressing them, you can significantly improve your chances of a successful submission following the .
Conclusion
Mastering the FDA 510(k) submission process is crucial for any medical device manufacturer looking to successfully launch their product. Understanding the complexities of substantial equivalence, preparing detailed documentation, conducting thorough performance testing, and steering clear of common pitfalls can significantly boost a company’s chances of obtaining timely FDA clearance.
Key insights from this article highlight the necessity of:
- Identifying a suitable predicate device
- Ensuring documentation is both comprehensive and precise
- Engaging with the FDA early in the process
These practices not only streamline submissions but also reduce the risk of delays or rejections, which can be both costly and time-consuming.
Ultimately, adhering to these best practices transcends mere compliance; it fosters trust in medical products that prioritize safety and efficacy. By committing to excellence in every facet of the 510(k) submission, manufacturers can play a vital role in a healthcare landscape that values innovation while upholding regulatory standards. Embrace these strategies to navigate the FDA 510(k) process effectively and set the stage for successful medical device introductions.
Frequently Asked Questions
What is the FDA 510(k) process?
The FDA 510(k) process is a premarket submission procedure that verifies a medical product’s safety and effectiveness by demonstrating substantial equivalence to a legally marketed product, known as a predicate.
What does substantial equivalence mean?
Substantial equivalence means that your product shares the same intended use and technological attributes as a predicate device that has already been legally marketed.
How can I identify my predicate instrument?
You can identify your predicate instrument by conducting thorough research, utilizing the FDA 510(k) database, which is a valuable resource for finding previously authorized products.
What are the key requirements for substantial equivalence?
The key requirements for substantial equivalence include having the same intended use, similar technological characteristics, and relevant performance data.
What percentage of 510(k) applications received a Substantially Equivalent decision by September 2022?
By September 2022, 85% of 510(k) applications received a Substantially Equivalent decision.
What challenges do applicants face in the 510(k) process?
Approximately 15% of submissions did not receive a Substantially Equivalent decision, indicating challenges in meeting the necessary criteria.
How can engaging with the FDA early benefit my application process?
Early engagement with the FDA through pre-submission meetings can clarify uncertainties regarding product classification and documentation, potentially simplifying the review process and reducing the average review duration.
What was the average review duration for the FDA 510(k) process in 2024?
The average review duration for the FDA 510(k) process in 2024 was approximately 168.9 days.
How many medical devices does the FDA 510(k) guidance facilitate for marketing each year?
The FDA 510(k) guidance facilitates the approval of around 3,000 medical devices for marketing each year.
List of Sources
- Understand the FDA 510(k) Process and Substantial Equivalence
- 2024 Medtech FDA Approval Volume Trends Down (https://mddionline.com/medical-device-regulations/2024-medtech-fda-approval-volume-trends-down)
- 5 Medical Device Regulatory Approval Statistics You Need to Know – Arrotek | Medical Device Innovation (https://arrotek.com/5-medical-device-regulatory-approval-statistics-you-need-to-know)
- 510(k) Statistical Patterns (https://mddionline.com/business/510-k-statistical-patterns)
- The 510(k) Third Party Review Program: Promise and Potential – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10465388)
- Prepare Comprehensive Documentation for 510(k) Submission
- Tips for Preparing Successful FDA 510(k) Submissions (https://propharmagroup.com/thought-leadership/preparing-successful-fda-510k-submission-tips)
- FDA 510(k) Explained: Medical Device Premarket Notification | IntuitionLabs (https://intuitionlabs.ai/articles/fda-510k-premarket-notification-process)
- How To Avoid Being Part of the 69% of 510(k) Submissions That Get Rejected the First Time (https://greenlight.guru/blog/how-to-avoid-510-k-submission-rejected)
- Decision Speed and Success Rates for Medical Devices (https://linkedin.com/pulse/decision-speed-success-rates-medical-devices-katrina-rogers)
- Implement Rigorous Performance Testing and Safety Protocols
- Performance Qualification: A Step-by-Step Guide for Pharma Experts (https://zamann-pharma.com/2024/08/21/performance-qualification-a-step-by-step-guide-for-pharma-expert)
- 5 Medical Device Regulatory Approval Statistics You Need to Know – Arrotek | Medical Device Innovation (https://arrotek.com/5-medical-device-regulatory-approval-statistics-you-need-to-know)
- Top 20 Safety Quotes To Improve Your Safety Culture (https://blog.safetyculture.com/industry-trends/top-20-safety-quotes-improve-safety-culture)
- FDA Performance Testing Requirements: Complete Bench, Software, and Biocompatibility Guide (https://complizen.ai/post/fda-performance-testing-requirements-complete-bench-software-and-biocompatibility-guide)
- Avoid Common Pitfalls in 510(k) Submissions
- 10 Reasons FDA Submissions Fail — Even When You Think You’re Ready (https://complizen.ai/post/10-reasons-fda-submissions-fail-even-when-you-think-you-re-ready)
- 6 Mistakes to Avoid When Submitting a 510(k) to the FDA (https://thefdagroup.com/blog/6-mistakes-to-avoid-when-submitting-a-510k-to-the-fda)
- 60% of Compliance Failures Start with Poor Documentation (https://qualitysystemsnow.com.au/latestnews/b/60-of-compliance-failures-start-with-poor-documentation)
- 7 Common 510(k) Mistakes and How to Avoid Them (https://greenlight.guru/blog/common-510k-mistakes)

Leave a Reply