Introduction
Understanding the complexities of bioburden testing is essential for ensuring the safety and efficacy of medical devices. As regulatory standards like ISO 11737 evolve, manufacturers must not only adhere to these guidelines but also master the nuances of microbial load assessment. This article provides a comprehensive, step-by-step guide to bioburden testing, empowering professionals to enhance their processes and safeguard product integrity.
How can manufacturers effectively navigate these standards to prevent costly errors and ensure compliance in an increasingly scrutinized industry?
Understand Bioburden Testing Fundamentals
is crucial in quantifying viable microorganisms present on or within medical instruments before sterilization. This assessment is essential for ensuring and compliance with regulatory standards. Understanding key terms is vital:
- Bioburden: This refers to the total count of viable microorganisms, including bacteria and fungi, found on a product.
- : This denotes the presence of undesirable microorganisms that can compromise and efficacy.
- Sterilization: This is the comprehensive process designed to eliminate all forms of microbial life, including spores.
The significance of assessing microbial load cannot be overstated, as it directly impacts the effectiveness of and the overall safety of medical instruments. Recent cases of in medical instruments underscore the necessity for stringent microbial load examination protocols. Investigations into these contamination incidents have shown that lapses in can result in product variability, loss of potency, and even manufacturing delays. Statistics reveal that s can lead to substantial disruptions, including prolonged shutdowns in manufacturing operations. Effective can avert these issues, ensuring that products adhere to the rigorous cleanliness standards essential for patient safety. By understanding these fundamentals, manufacturers can navigate the complexities of more effectively, enhancing the safety of their medical instruments. Furthermore, monitoring trends in microbial load results over time is critical for maintaining process control and ensuring compliance with , which aids manufacturers in assessing contamination risks throughout the .
Familiarize with ISO 11737 Standards and Requirements
The series of standards known as is crucial for outlining the criteria for of medical products, ensuring safety and compliance with regulations. The key components include:
- -1: This standard specifies the methods for determining the , detailing . It emphasizes the importance of accurate sample handling to reflect the true microbial load, which is essential for validating .
- : This section focuses on the based on data related to microbial load. Manufacturers must demonstrate the efficacy of their sterilization techniques through consistent bioburden evaluations, ensuring that products are free from viable microorganisms.
- : This standard addresses the , which is vital for ensuring the safety of medical equipment according to . It outlines the necessary procedures to detect endotoxins, which can pose significant risks to patient health.
Recent updates to have refined the requirements, enhancing the clarity and applicability of the standards. For instance, the 2018 update of -1 introduced new guidelines for sample preparation and microbial recovery techniques, ensuring that testing accurately represents the entire batch of devices. Key aspects of these updates include improved methods for microbial recovery, enhanced validation requirements, and clearer guidelines for sample handling as specified by .
Compliance with these standards is essential, as statistics indicate that adherence can significantly reduce the risk of audit deficiencies and product recalls, which can lead to substantial financial losses. As Rick L’Amie observes, “Failure to accurately evaluate microbial load could result in insufficient processing controls and data supporting the sterile claim of your product, which can ultimately lead to audit shortcomings and even product recall.”
Familiarizing yourself with these standards will not only help ensure that your are compliant and effective but also enhance the overall safety and reliability of medical devices available in the market. is crucial to verify their continuous efficiency, guaranteeing that your procedures remain robust and dependable over time.
Execute the Bioburden Testing Process Step-by-Step
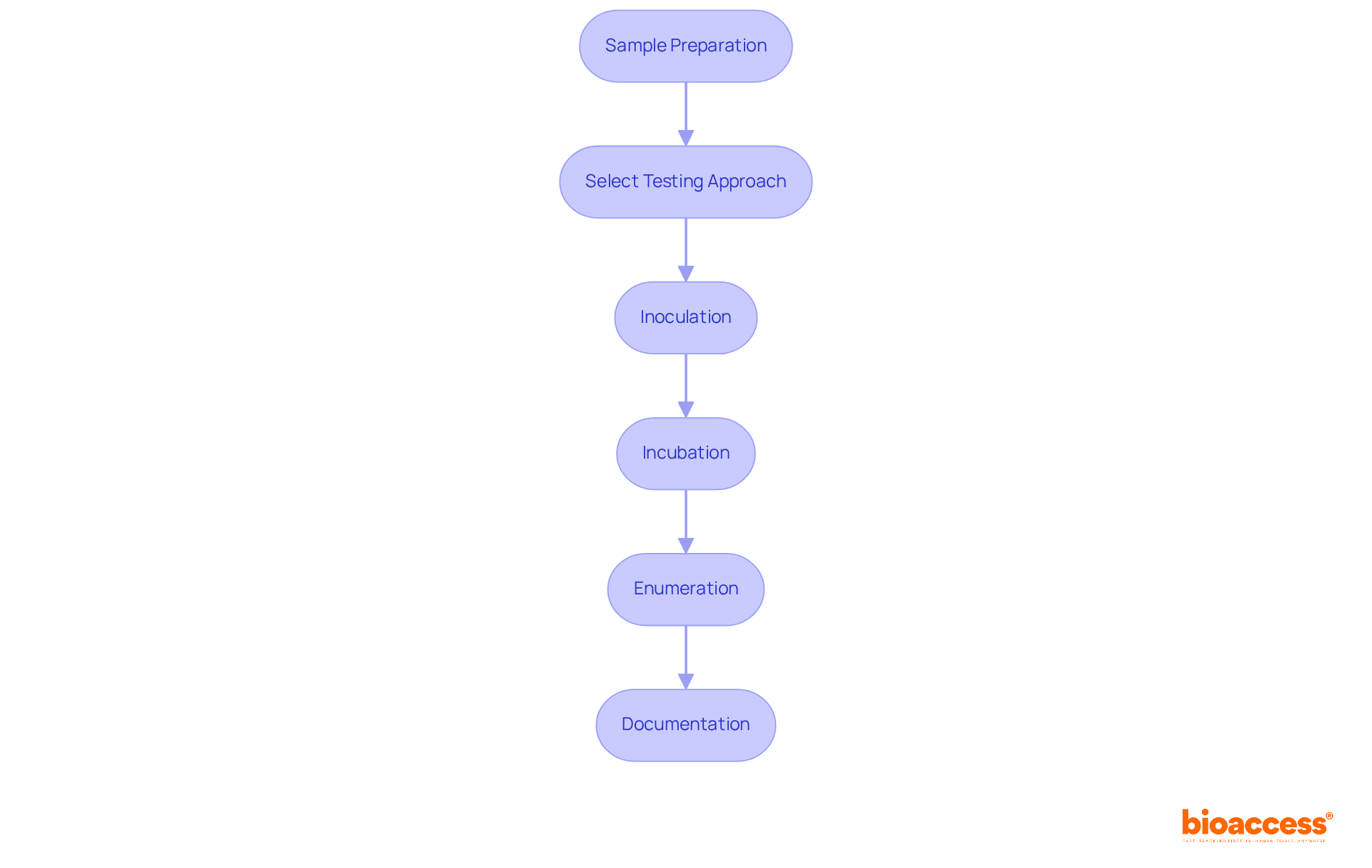
To effectively execute the , follow these essential steps:
- : Employ sterile techniques to collect samples from the medical device. This may involve cutting, disassembling, or flushing the device to prevent contamination. is crucial, particularly those that pose the greatest microbiological challenge. Be aware that sources of contamination can include personnel handling, environmental exposure, and water used in production.
- Select : Choose a suitable technique for quantifying microorganisms, such as the membrane filtration technique or the plate count technique. Ensure that the selected approach is validated during the suitability analysis to guarantee accurate results.
- Inoculation: If necessary, inoculate the sample with a known amount of a test organism to confirm the testing procedure. This step is vital for verifying the effectiveness of the chosen method.
- Incubation: under . Routine monitoring during this phase is recommended, even if contamination risks seem low.
- Enumeration: After incubation, count the number of colonies formed to assess the microbial load. This quantification is essential for compliance with the , which mandate that meets established .
- Documentation: Meticulously record all findings, including any deviations from standard procedures. Setting an expiry date for microbial load results is crucial due to potential changes in manufacturing conditions over time.
By adhering to these steps, you ensure that your is thorough and compliant with , ultimately supporting product safety and regulatory compliance. Routine evaluation is critical for maintaining efficient sterilization methods, as illustrated in various case studies, including ‘Bioburden Analysis in Medical Device Manufacturing.’ This emphasizes the significance of in guaranteeing cleanliness and adherence.
Validate and Verify Testing Methods for Compliance
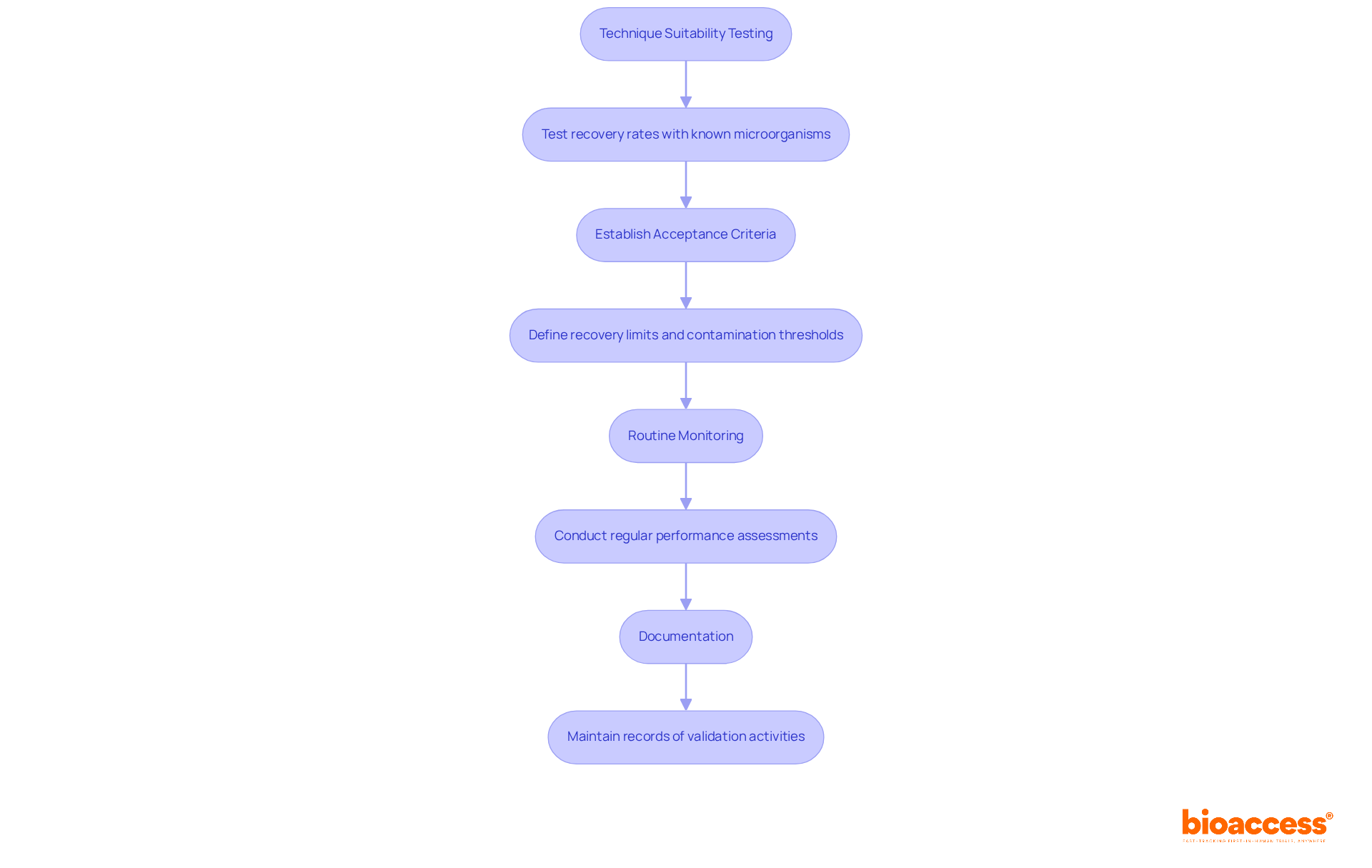
To ensure the reliability and compliance of your , adhere to the following guidelines:
- Technique Suitability Testing: Implement tests to confirm that your chosen approach can accurately recover microorganisms from samples. This involves inoculating known quantities of microorganisms, including two in-house isolates, and assessing recovery rates. For instance, using Bacillus atrophaeus spores can help determine the effectiveness of your extraction protocol, with a recovery target of 100 CFU being ideal. A or higher is advised to verify the lack of inhibitory properties in your assessment methods. If the average recovery percentage from three evaluations is 75%, the correction factor is determined as 100/75 = 1.33.
- Establish Acceptance Criteria: Clearly define , including recovery limits and contamination thresholds. suggest a maximum of 10 CFU/100 mL for acceptable bioburden levels, with a combined TAMC and TYMC result of 4 CFU/10 mL providing a more comprehensive understanding of acceptable limits. This emphasizes the need for .
- Routine Monitoring: Conduct regular checks to ensure that your assessment techniques maintain consistent performance over time. This includes periodic re-validation and performance assessments, which are essential for compliance with evolving regulatory expectations. For instance, a or more is suggested to verify the lack of inhibitory properties in your evaluation techniques.
- Documentation: Maintain , including methodologies, results, and corrective actions taken. This documentation is essential for showcasing adherence to iso 11737 standards and for effectively managing any potential contamination excursions. As emphasized by Sebastian Thoelken, all .
By rigorously validating and verifying your testing methods, you can ensure that your are robust, reliable, and compliant with industry standards.
Conclusion
Understanding and executing bioburden testing in accordance with ISO 11737 standards is crucial for ensuring the safety and efficacy of medical devices. This guide has outlined the fundamental principles of bioburden testing, the specific requirements of ISO 11737, and the step-by-step process necessary for accurate microbial load assessment. By mastering these elements, manufacturers can significantly reduce the risk of contamination and enhance product reliability.
Key insights include:
- The critical role of microbial load assessment in validating sterilization processes
- The importance of adhering to updated ISO standards
- The necessity of rigorous validation and documentation practices
Each step in the bioburden testing process, from sample preparation to method verification, contributes to a comprehensive approach that safeguards patient health and maintains regulatory compliance.
Ultimately, the commitment to effective bioburden testing not only protects consumers but also fosters trust in the medical device industry. Embracing these practices ensures that products meet the highest cleanliness standards, thereby minimizing the likelihood of audit deficiencies and product recalls. As the landscape of healthcare continues to evolve, prioritizing bioburden testing will remain a cornerstone of quality assurance in the production of safe and effective medical devices.
Frequently Asked Questions
What is bioburden testing?
Bioburden testing is the evaluation process that quantifies the viable microorganisms present on or within medical instruments before they undergo sterilization.
Why is bioburden evaluation important?
Bioburden evaluation is essential for ensuring product safety and compliance with regulatory standards, as it directly impacts the effectiveness of sterilization techniques and the overall safety of medical instruments.
What does the term “bioburden” refer to?
Bioburden refers to the total count of viable microorganisms, including bacteria and fungi, found on a product.
What is microbial contamination?
Microbial contamination denotes the presence of undesirable microorganisms that can compromise the safety and efficacy of a product.
What is the definition of sterilization?
Sterilization is the comprehensive process designed to eliminate all forms of microbial life, including spores.
What are the consequences of inadequate microbial control?
Inadequate microbial control can lead to product variability, loss of potency, manufacturing delays, and substantial disruptions, including prolonged shutdowns in manufacturing operations.
How can effective microbial load testing benefit manufacturers?
Effective microbial load testing can prevent issues related to microbial contamination, ensuring that products meet rigorous cleanliness standards essential for patient safety.
Why is it important to monitor trends in microbial load results over time?
Monitoring trends in microbial load results over time is critical for maintaining process control and ensuring compliance with ISO 11737, which helps manufacturers assess contamination risks throughout the product lifecycle.
List of Sources
- Understand Bioburden Testing Fundamentals
- How Bioburden Testing Supports Device Safety | NABI (https://nabi.bio/bioburden-testing-for-medical-devices)
- Bioburden Testing: Everything You Need to Know | Test Labs (https://testlabsuk.com/blog/bioburden-testing-everything-you-need-to-know)
- Case Studies of Microbial Contamination in Biologic Product Manufacturing (https://americanpharmaceuticalreview.com/Featured-Articles/36755-Case-Studies-of-Microbial-Contamination-in-Biologic-Product-Manufacturing)
- Familiarize with ISO 11737 Standards and Requirements
- ISO 11737-1: Understanding Bioburden Testing Principles – CMDC Labs (https://cmdclabs.com/iso-11737-1-understanding-bioburden-testing-principles)
- Five Aspects of the ISO 11737-1:2018 | Nelson Labs (https://nelsonlabs.com/five-aspects-of-the-iso-11737-12018-updates-you-need-to-know)
- ISO 11737 Bioburden Test | Microchem Laboratory (https://microchemlab.com/test/iso-11737-bioburden-test)
- ANSI/AAMI/ISO 11737-1:2018 – Determination of a Population of Microrganisms on Products for Sterilization (https://blog.ansi.org/ansi/ansi-aami-iso-11737-1-2018-sterilization)
- Medical Device Bioburden Testing | QA Consulting, Inc. (https://qaconsultinginc.com/bioburden-basics-for-sterile-medical-devices)
- Execute the Bioburden Testing Process Step-by-Step
- Bioburden Testing: Everything You Need to Know | Test Labs (https://testlabsuk.com/blog/bioburden-testing-everything-you-need-to-know)
- bioaccessla.com (https://bioaccessla.com/blog/10-essential-insights-on-bioburden-testing-for-clinical-research)
- Validate and Verify Testing Methods for Compliance
- Q&A from European GMP Conference: “Bioburden – Regulatory Expectations and Practical Experiences” – GMP Journal (https://gmp-journal.com/current-articles/details/q-a-from-european-gmp-conference-bioburden-regulatory-expectations-and-practical-experiences.html)
- Method Suitability (Bacteriostasis/Fungistasis) (https://nelsonlabs.com/testing/method-suitability-bacteriostasis-fungistasis)
- How to Validate a Bioburden Analysis on a Medical Device (MD)? – SuperMicrobiologists (https://supermicrobiologists.com/en/how-to-validate-a-bioburden-analysis-on-a-medical-device-md)
- ARL Bio Pharma | Method Suitability and its Importance to the Sterility Test Method (https://arlok.com/news/Method-Suitability-and-its-Importance-to-the-Sterility-Test-Method)
- Bioburden Method Suitability: A Practical Solution to Screening for Inhibition – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10512993)

Leave a Reply