Introduction
While Chile offers enticing financial advantages for clinical research, the path to successful study execution is fraught with challenges that demand strategic navigation. Chile has emerged as a beacon for clinical research, particularly for first-in-human studies, with clinical trial costs being 30% to 75% lower than in the U.S. and Europe. This financial landscape allows startups to optimize their budgets and resources effectively.
Despite the financial advantages, many companies struggle to effectively manage the complexities of clinical trials in Chile. To truly harness the potential of Chile’s clinical research landscape, companies must not only recognize these challenges but also develop robust strategies to overcome them.
Understand the Financial Landscape of Clinical Trials in Chile
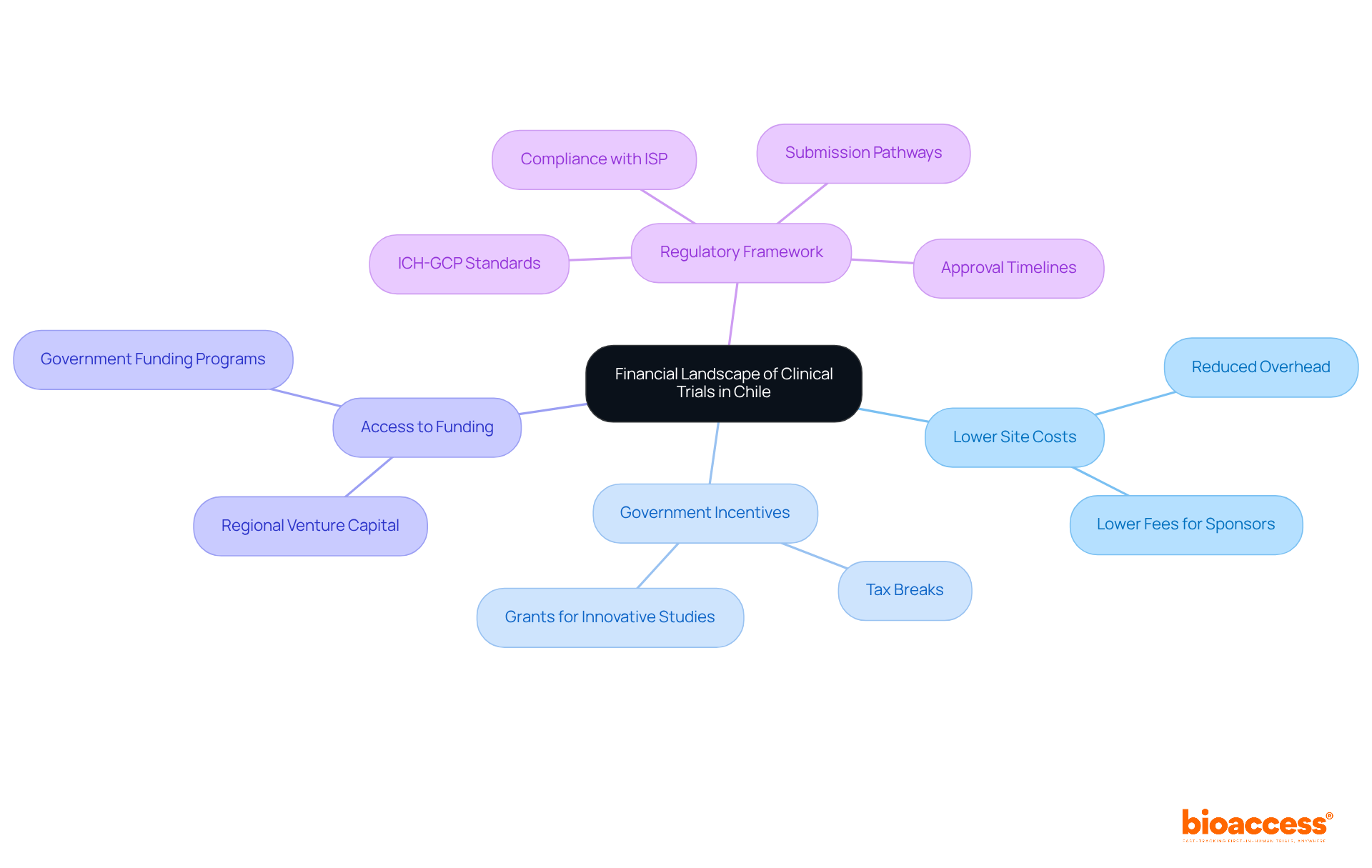
Chile stands out as a prime destination for clinical research, particularly for first-in-human studies, because the clinical trial cost in Chile is significantly lower compared to the U.S. and Europe. The clinical trial cost in Chile can be conducted at levels that are roughly 30% to 75% lower than in developed markets. Several key factors contribute to this cost efficiency:
- Lower Site Costs: Clinical research sites in Chile typically incur reduced overhead, resulting in lower fees for sponsors. This is particularly advantageous for startups looking to maximize their budgets.
- Government Incentives: The Chilean government actively supports clinical research through various incentives, including tax breaks and grants for innovative studies, which can further reduce overall study costs.
- Access to Funding: Startups can leverage regional venture capital and government funding programs specifically designed to support healthcare innovations, enhancing their financial viability.
- Regulatory Framework: Navigating the regulatory landscape can be daunting for many sponsors, often leading to delays and increased costs. Compliance with regional regulatory authorities such as the Instituto de Salud Pública (ISP) and adherence to ICH-GCP standards are essential for study approval. Understanding the submission pathways and approval timelines can significantly expedite the process.
When sponsors grasp these financial dynamics and regulatory requirements, they can really optimize their budgets and allocate resources effectively, ensuring they hit their first-in-human milestones without draining their capital reserves. This strategic approach not only facilitates faster study execution but also aligns with the regulatory frameworks established by local authorities, ensuring compliance and expediting the approval process. Have you considered how per-patient expenditures in LATAM, ranging from $15,000 to $35,000, compare to the $40,000 to $75,000 in the US/EU? This stark difference underscores the financial advantages of conducting studies, particularly considering the clinical trial cost in Chile. With insights from bioaccess®, MedTech startups can navigate these financial landscapes more effectively, ensuring successful study outcomes.
Implement Strategic Budgeting and Cost Management Techniques
To thrive in the competitive landscape of clinical trials in Chile, startups must master budgeting and cost management techniques:
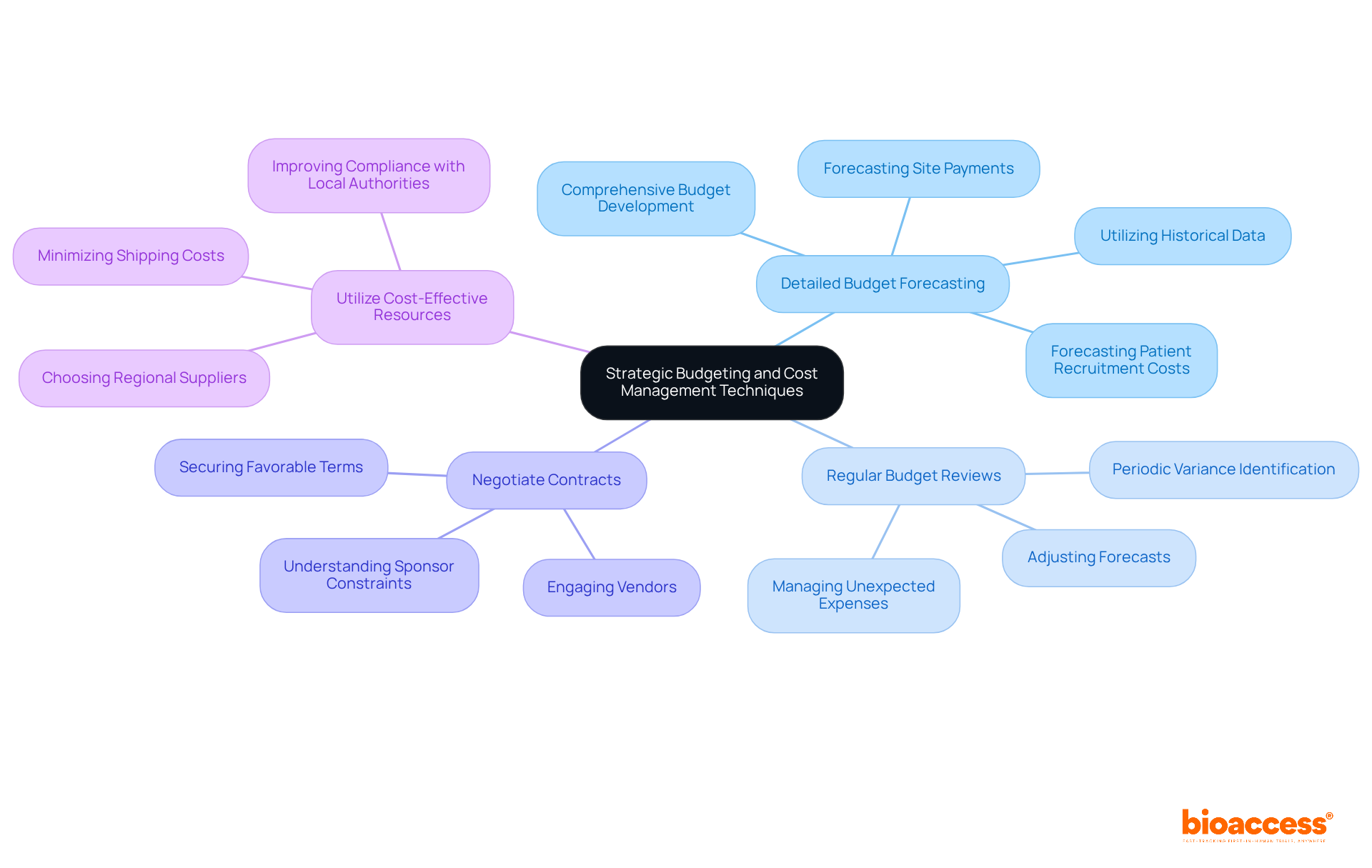
- Detailed Budget Forecasting: Develop a comprehensive budget that encompasses all potential costs, including site fees, patient recruitment, and regulatory submissions. Historical data from previous studies can provide valuable insights for more accurate estimates. For instance, utilizing historical data can assist in forecasting expenses related to site payments and patient recruitment, which are essential for sustaining financial oversight. In Latin America, understanding the clinical trial cost in Chile, which can be 30% lower than in the US/EU, is crucial.
- Regular Budget Reviews: Have you considered implementing periodic budget reviews to identify variances and adjust forecasts accordingly? This proactive strategy aids in managing unexpected expenses and maintaining financial control. As operational burdens rise in 2024, startups may struggle to keep budgets on track without regular reviews, especially in a region where regulatory timelines can be significantly shorter.
- Negotiate Contracts: Engage in negotiations with vendors and service providers to secure favorable terms. The competitive landscape in Chile can be leveraged to obtain better pricing, which is crucial for startups managing the clinical trial cost in Chile on tight budgets. Comprehending a sponsor’s budget constraints can also aid in establishing equitable objectives and requests during discussions, especially when considering the clinical trial cost in Chile and the financial benefits of performing studies in Latin America.
- Utilize Cost-Effective Resources: Choose regional suppliers for clinical trial materials and services to minimize shipping costs and import taxes. Not only does this lower expenses, but it also supports the regional economy, creating a more sustainable operational model. Furthermore, using regional resources can improve compliance with regulatory authorities such as the Instituto de Salud Pública (ISP) and ensure adherence to ICH-GCP standards. Bioaccess® can assist in navigating these local resources effectively.
By implementing these strategies, startups can effectively manage their finances. It keeps studies within budget while navigating the regulatory landscape set by authorities like the ISP and adhering to ICH-GCP standards. Avoiding common pitfalls in budgeting can be the difference between a successful trial and financial strain.
Navigate Regulatory Requirements to Optimize Trial Costs
Navigating Chile’s regulatory landscape can be a daunting task for startups aiming to conduct clinical research. Chile’s regulatory framework for research studies is overseen by the Instituto de Salud Pública (ISP), which enforces adherence to ICH-GCP standards. Startups can significantly reduce trial costs by implementing these strategic approaches:
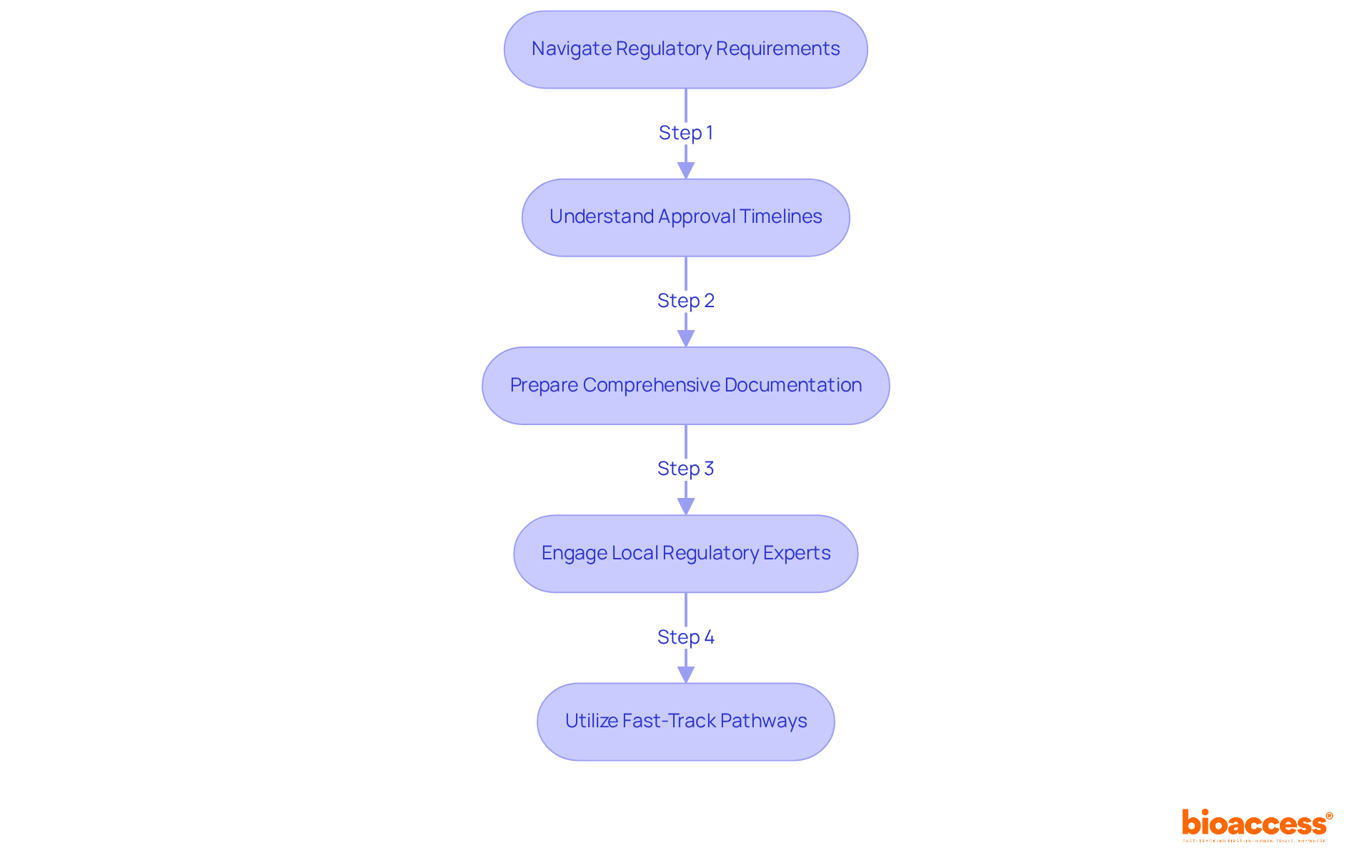
- Understand Approval Timelines: The average approval time for clinical trials in Chile is approximately 30 business days. Planning submissions with this timeline in mind can help mitigate potential delays. Delays in approval can lead to increased timelines and a higher clinical trial cost in Chile for startups.
- Prepare Comprehensive Documentation: Ensure that all necessary documents, including the clinical research protocol, informed consent forms, and investigator brochures, are meticulously prepared to meet ISP requirements. This thorough preparation is critical for successful approval.
- Engage Local Regulatory Experts: Collaborating with local regulatory consultants, such as bioaccess®, can facilitate smoother interactions with the ISP and help navigate bureaucratic hurdles effectively. Without local expertise, startups risk facing bureaucratic challenges that could hinder their progress. Bioaccess® has successfully assisted numerous clients in achieving timely approvals, leveraging its extensive experience in the region.
- Utilize Fast-Track Pathways: Leverage Chile’s fast-track approval processes for innovative therapies, which can significantly reduce time to market and enhance the overall efficiency of study execution. For example, bioaccess® has assisted clients in accelerating their studies through these pathways, demonstrating the potential for quicker patient enrollment and lower clinical trial cost in Chile.
With bioaccess® guiding them through regulatory obligations, startups can minimize delays and costs, paving the way for a faster market entry.
Leverage Local Partnerships for Cost-Effective Trial Execution
Navigating the complexities of clinical study execution in Chile can be daunting for startups, but strategic regional collaborations offer a powerful solution. Startups should consider:
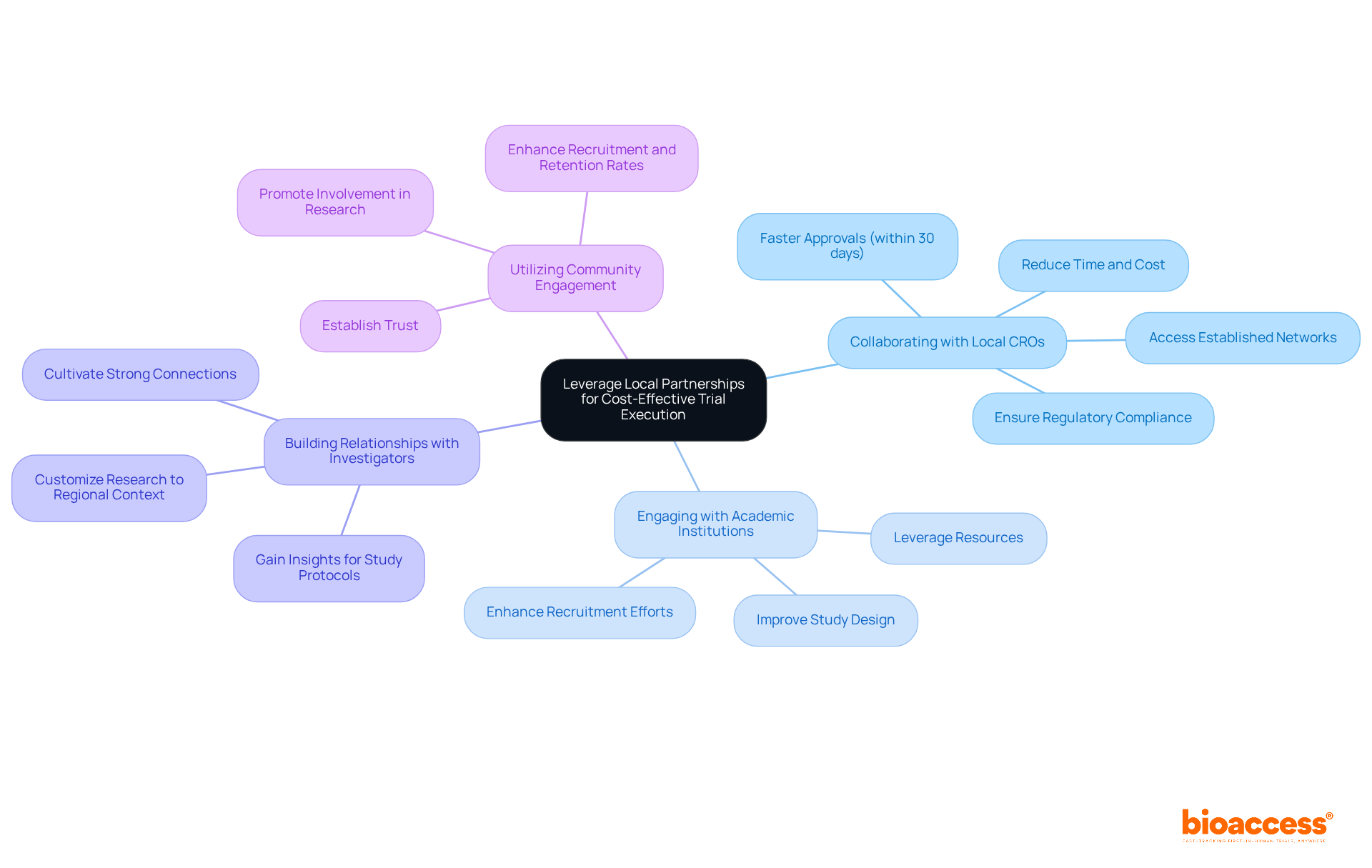
- Collaborating with Local CROs: Partnering with local Contract Research Organizations (CROs) like bioaccess® provides access to established networks, significantly reducing the time and cost associated with site selection and patient recruitment. Local CROs are well-versed in the regulatory landscape governed by the Instituto de Salud Pública (ISP), ensuring compliance with ICH-GCP standards and facilitating faster approvals, often within 30 business days.
- Engaging with Academic Institutions: Collaborating with universities and research institutions allows startups to leverage their resources, including extensive patient databases and research expertise. This collaboration can enhance recruitment efforts and improve study design, ultimately leading to more efficient execution of research.
- Building Relationships with Investigators: Cultivating strong connections with regional researchers who possess expertise in conducting medical studies is essential. Their insights can improve study protocols and enhance operational execution, ensuring that research is customized to the regional context and regulatory requirements.
- Utilizing Community Engagement: Interacting with regional communities is crucial for establishing trust and promoting involvement in research studies. Effective community outreach can lead to quicker recruitment and enhanced retention rates, which are vital for the success of research studies.
Startups often struggle with the complexities of clinical study execution, leading to delays and increased costs. By forming strategic partnerships, they can navigate these challenges more effectively, ensuring timely and cost-efficient study execution. Embracing these collaborative strategies not only streamlines operations but also positions startups to thrive in the evolving MedTech and Biopharma landscape.
Conclusion
Navigating the complexities of clinical trials can be daunting for startups, especially in a foreign market like Chile, where opportunities abound. Chile offers a unique advantage for clinical trials, particularly for first-in-human studies, due to its significantly lower costs compared to developed markets. Startups can navigate the financial landscape, use smart budgeting techniques, and build local partnerships to streamline their clinical trial processes while staying compliant and efficient. These strategies are essential for achieving successful study outcomes while maintaining financial stability.
The article outlines several best practices that can enhance cost management in clinical trials. Key strategies include:
- Detailed budget forecasting
- Regular budget reviews
- Negotiating favorable contracts with vendors
Additionally, understanding the regulatory framework and engaging local experts can expedite approval processes and lower overall costs. Collaborating with local CROs, academic institutions, and community stakeholders further streamlines execution and fosters trust, ultimately enhancing patient recruitment and retention.
In conclusion, embracing these strategies not only positions startups for success but also redefines the landscape of clinical research in Latin America. By adopting these best practices, MedTech and Biopharma startups can navigate the complexities of early-stage clinical research and accelerate their timeline to market, making Chile an ideal choice for innovative clinical studies.
Frequently Asked Questions
Why is Chile considered a prime destination for clinical research, particularly for first-in-human studies?
Chile is considered a prime destination for clinical research due to significantly lower clinical trial costs, which can be 30% to 75% lower than in the U.S. and Europe. This cost efficiency is attractive for sponsors, especially startups.
What factors contribute to the lower clinical trial costs in Chile?
Key factors contributing to lower clinical trial costs in Chile include lower site costs due to reduced overhead, government incentives such as tax breaks and grants, access to regional venture capital and funding programs, and a supportive regulatory framework.
What types of government incentives are available for clinical research in Chile?
The Chilean government offers various incentives for clinical research, including tax breaks and grants aimed at innovative studies, which can help reduce the overall costs of conducting clinical trials.
How can startups access funding for clinical trials in Chile?
Startups can access funding through regional venture capital and government funding programs specifically designed to support healthcare innovations, which can enhance their financial viability for clinical trials.
What is the regulatory framework for conducting clinical trials in Chile?
The regulatory framework in Chile requires compliance with the Instituto de Salud Pública (ISP) and adherence to ICH-GCP standards. Understanding submission pathways and approval timelines is essential for study approval.
How can sponsors optimize their budgets when conducting clinical trials in Chile?
Sponsors can optimize their budgets by understanding financial dynamics and regulatory requirements, allowing for effective resource allocation to hit first-in-human milestones without exhausting capital reserves.
How do per-patient expenditures in Chile compare to those in the U.S. and Europe?
Per-patient expenditures in Latin America, including Chile, range from $15,000 to $35,000, compared to $40,000 to $75,000 in the U.S. and Europe, highlighting the financial advantages of conducting studies in Chile.
What role does bioaccess® play in navigating the financial landscape of clinical trials in Chile?
Insights from bioaccess® help MedTech startups navigate the financial landscapes effectively, ensuring successful study outcomes by optimizing budgets and understanding the regulatory environment.
List of Sources
- Understand the Financial Landscape of Clinical Trials in Chile
- Trends in clinical trials performed in Chile (https://scielo.cl/article_plus.php?pid=S0034-98872021000100110&tlng=en&lng=es)
- Chile Clinical Trial Management System Market Size, Trends & Forecast Analysis (2025-2033) (https://reedintelligence.com/insights/clinical-trial-management-system-market/chile)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOoqs38GZHJgcHiGuq3l1mJ6EOqPFlA_xvgkVOi4V8PVjFF_S2NKn)
- Chile Clinical Trial Supply & Logistics Market Size & Outlook, 2030 (https://grandviewresearch.com/horizon/outlook/clinical-trial-supply-logistics-market/chile)
- Costs & Timelines | bioaccess® (https://bioaccessla.com/costs-and-timelines)
- Implement Strategic Budgeting and Cost Management Techniques
- How Rising Operating Costs Are Impacting LATAM Research Sites in 2024 (https://integrait.co/latam-clinical-research-sites-operating-costs-2024)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOoqY-XIdCuCM7cCI0sdrhRlG1DZP2FyMTX8Pi45ix1E8_PTyijgE)
- Clinical Trials Budgeting and Forecasting: Six Simple Steps to Immediately Improve Accuracy (https://clinicaltrialsarena.com/news/clinical-trials-budgeting-and-forecasting-six-simple-steps-to-immediately-improve-accuracy-5018849-2)
- Strategies For Efficient Clinical Trial Budget Management (https://advarra.com/blog/strategies-for-efficient-clinical-trial-budget-management)
- Navigate Regulatory Requirements to Optimize Trial Costs
- Clinical Trials in Latin America (https://languageconnections.com/clinical-trials-in-latin-america)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Navigate Biopharma Clinical Trials in Chile: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/navigate-biopharma-clinical-trials-in-chile-a-step-by-step-guide)
- Leverage Local Partnerships for Cost-Effective Trial Execution
- Best Practices for Clinical Trial Outsourcing in Chile | bioaccess® (https://bioaccessla.com/blog/best-practices-for-clinical-trial-outsourcing-in-chile)
- What is a Contract Research Organization in Chile? | bioaccess® (https://bioaccessla.com/blog/what-is-a-contract-research-organization-in-chile)
- How Chile Is Shaping Medical Device Clinical Trials In Latin America — bioaccess™: LATAM CRO Experts | Julio G. Martinez-Clark (https://linkedin.com/posts/juliomartinezclark_how-chile-is-shaping-medical-device-clinical-activity-7259074399159349248-2aFu)
- Contract Research Organization Market Forecast, 2026-2033 (https://coherentmarketinsights.com/industry-reports/contract-research-organization-market)
- Local Partnerships Are Key to Building Community Trust (https://aamchealthjustice.org/news/viewpoint/local-partnerships-key)

Leave a Reply