Introduction
Navigating the regulatory landscape of radiopharmaceutical clinical trials in Chile presents significant challenges that can impede success, particularly given the stringent frameworks established by authorities like the Instituto de Salud Pública (ISP). Yet, grasping these regulations is crucial for sponsors who want to improve their trial efficiency and outcomes.
What strategies can be employed to overcome the inherent challenges of trial execution, from regulatory compliance to participant recruitment? This article explores four best practices that not only facilitate successful radiopharma trials in Chile but also leverage the unique advantages of conducting research in Latin America.
Understand Regulatory Frameworks for Radiopharmaceutical Trials in Chile
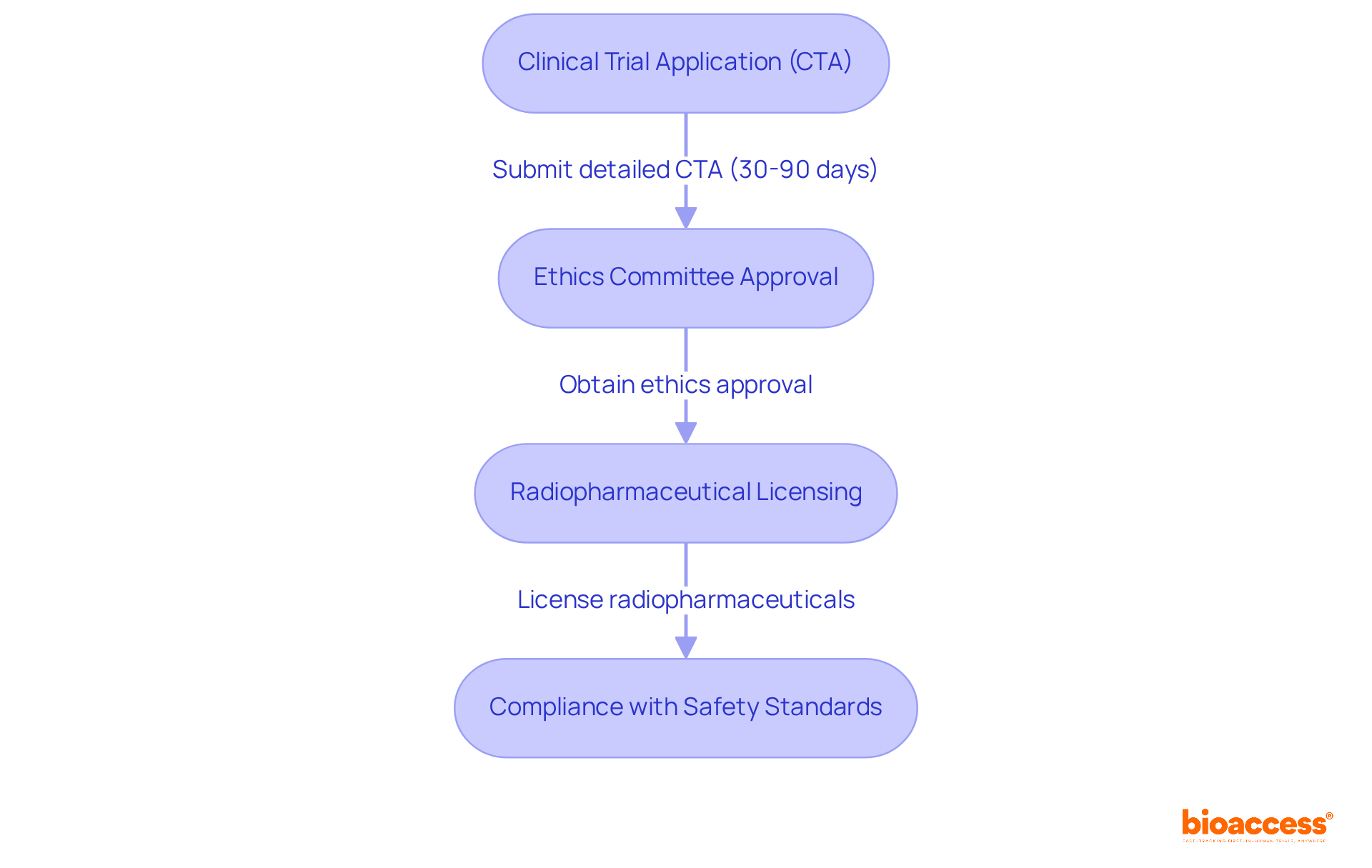
Navigating the regulatory landscape for radiopharma clinical trial Chile can be daunting, yet it is crucial for successful clinical research. Conducting these studies requires a thorough understanding of the regulatory framework established by the Instituto de Salud Pública (ISP). The ISP supervises the validation steps for clinical trials, ensuring adherence to local regulations and international standards such as ICH-GCP. Key steps in this process include:
- Clinical Trial Application (CTA): Submit a detailed CTA to the ISP, which must include the study protocol, informed consent forms, and safety data. The ISP generally assesses applications within 30 business days, though the overall process can take between 30 to 90 days.
- Ethics Committee Approval: Before commencing any clinical study, securing authorization from a recognized ethics committee is essential. This step is vital for ensuring that the study adheres to ethical standards and safeguards participant rights.
- Radiopharmaceutical Licensing: All radiopharmaceuticals utilized in the study must be licensed and comply with local regulations governing their use and handling. This ensures that the materials are safe and effective for clinical use.
- Compliance with Safety Standards: It is essential to adhere to safety protocols for handling radioactive materials. This involves ensuring that all staff engaged in the study receive adequate training to handle these substances safely.
Navigating the regulatory landscape can be daunting for sponsors, often leading to delays and confusion. By mastering these regulations, sponsors can streamline the approval process and enhance outcomes in the radiopharma clinical trial in Chile. This strategic approach not only increases the probability of successful study execution but also aligns with community health priorities, ultimately enhancing the effectiveness of clinical research in the region.
Implement Early Feasibility Studies to Enhance Trial Success
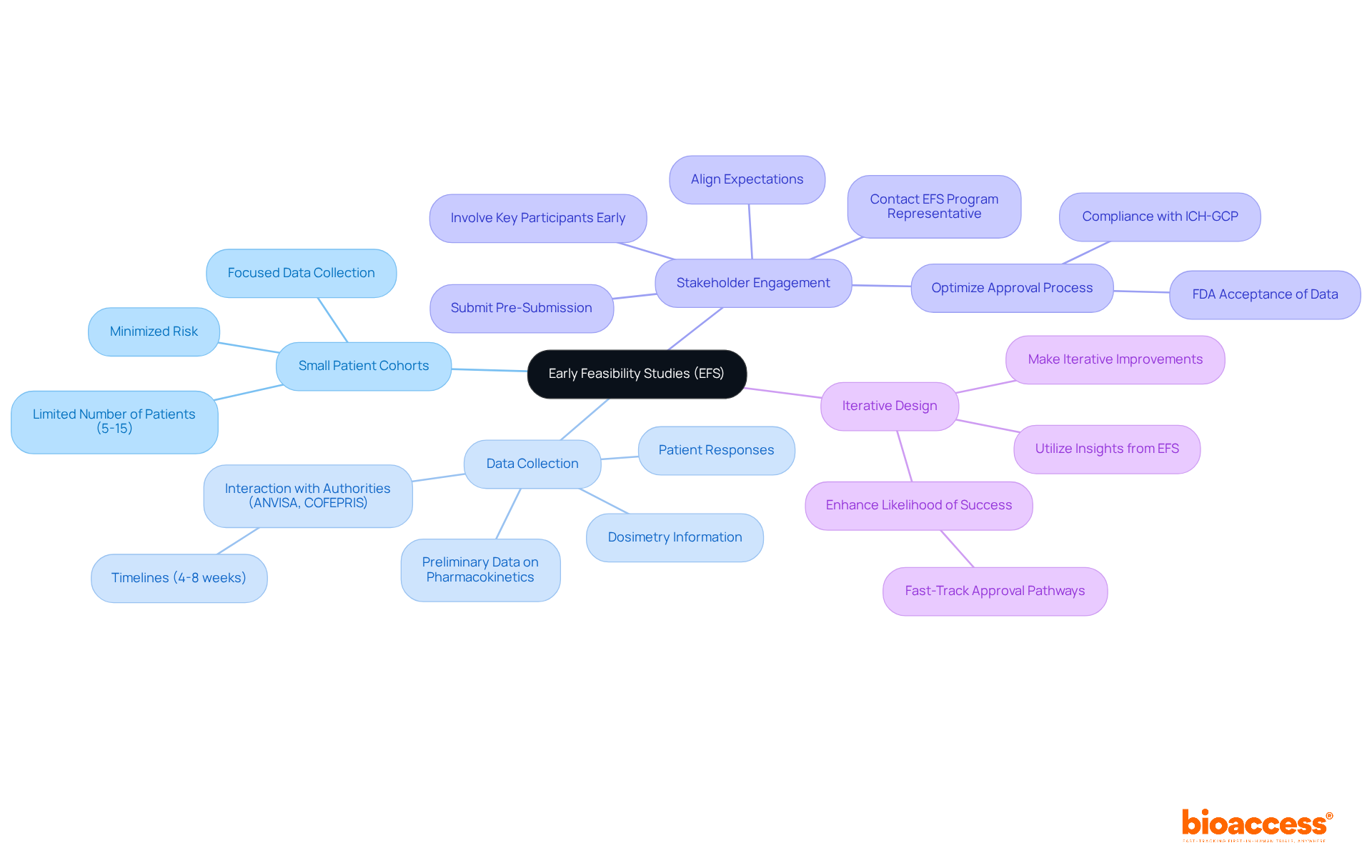
In the complex world of radiopharmaceutical research, early feasibility studies (EFS) are essential for determining the viability of new treatments before they reach full-scale implementation. These studies are designed to identify potential challenges and refine trial protocols, paving the way for a smoother path to securing essential approvals. Key considerations for successful EFS include:
- Small Patient Cohorts: Conduct EFS with a limited number of patients, typically between 5 to 15, to assess the safety and functionality of the radiopharmaceutical. This approach allows for focused data collection while minimizing risk.
- Data Collection: Collect preliminary data on pharmacokinetics, dosimetry, and patient responses. This information is essential for guiding further development and submission processes, especially when interacting with authorities like ANVISA or COFEPRIS, which have timelines of 4-8 weeks for EFS.
- Stakeholder Engagement: Involve key participants, including oversight organizations and clinical sites, early in the development. This partnership assists in aligning expectations and optimizing the approval process, which is crucial for fulfilling the compliance requirements of ICH-GCP and ensuring FDA acceptance of data. Contacting an EFS Program representative and submitting a Pre-Submission can facilitate this alignment.
- Iterative Design: Utilize insights gained from EFS to make iterative improvements to the study design. This adaptability enhances the likelihood of success in subsequent phases, particularly in the context of the fast-track approval pathways available in Latin America.
By adopting EFS, sponsors can effectively reduce risks and enhance their clinical development strategies. Ultimately, embracing EFS not only mitigates risks but also positions sponsors for success in the competitive landscape of clinical research.
Develop Effective Recruitment Strategies for Clinical Trials
Recruiting participants for the radiopharma clinical trial in Chile presents unique challenges that require strategic solutions. What strategies can effectively enhance participant recruitment? Here are some targeted approaches:
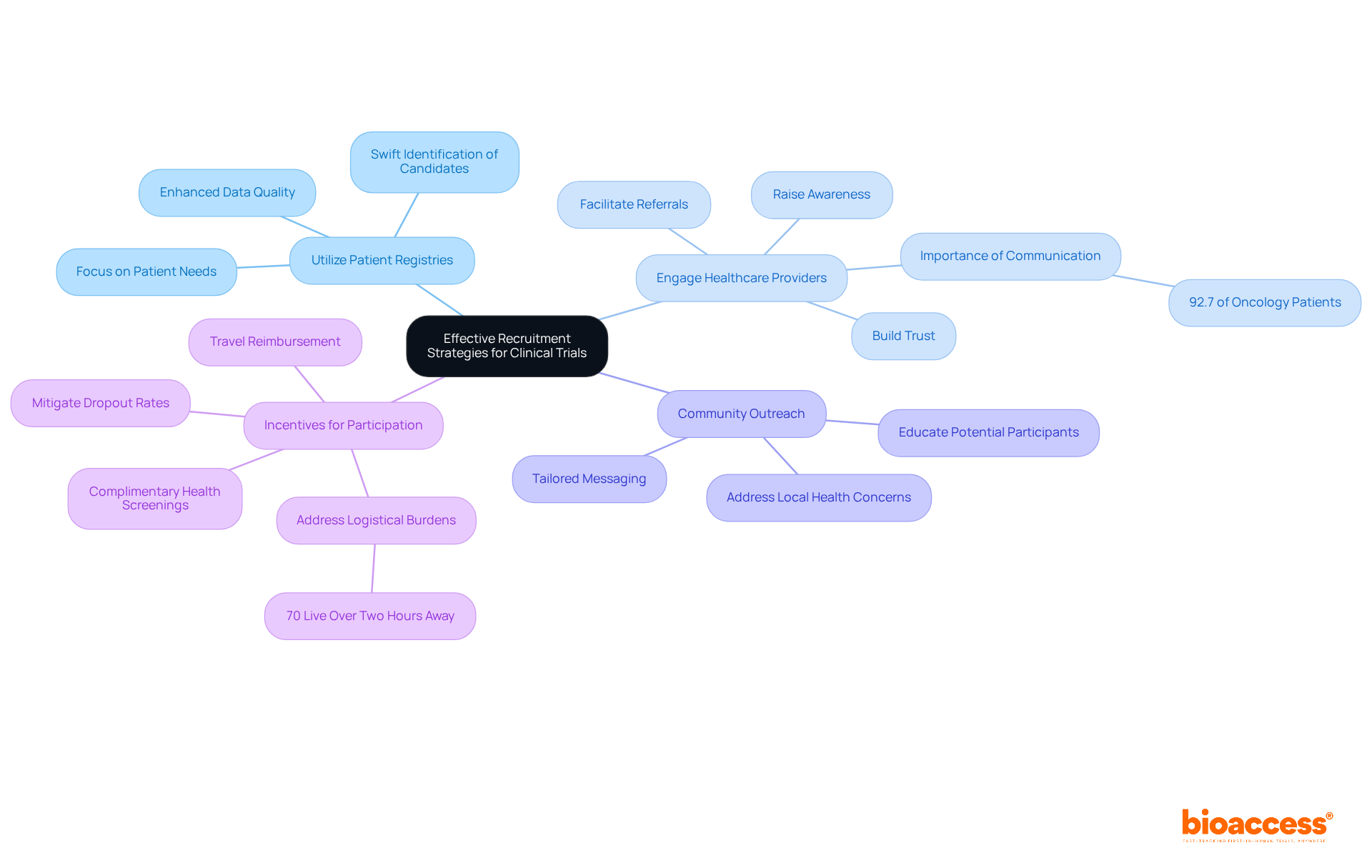
- Utilize Patient Registries: Leverage existing patient registries to swiftly identify eligible candidates, streamlining the recruitment effort. This method not only accelerates participant identification but also enhances the quality of data collected. As noted in case studies, focusing on patient needs can significantly improve recruitment outcomes.
- Engage Healthcare Providers: Establish strong relationships with local healthcare providers to facilitate referrals and raise awareness of the study among potential participants. Effective communication with these providers can significantly enhance patient engagement and trust in the study process. Research indicates that 92.7% of oncology patients consider it crucial to discuss clinical studies with their doctors, highlighting the importance of provider engagement.
- Community Outreach: Implement outreach programs within communities to educate potential participants about the benefits and safety of engaging in radiopharmaceutical studies. Tailoring messages to address local health concerns can improve participation rates. This approach aligns with research showing that community engagement is key to raising awareness about the study.
- Incentives for Participation: Offer incentives such as travel reimbursement or complimentary health screenings to encourage participation and mitigate dropout rates. Financial support can alleviate logistical burdens, particularly for patients in rural areas. Many potential participants face significant logistical hurdles, particularly those residing in rural areas. Addressing these challenges is crucial, as an estimated 70% of potential participants live more than two hours away from study centers.
Implementing these strategies helps sponsors enhance patient recruitment and ensures studies are completed on time. This method not only conforms to compliance standards established by organizations like ANVISA but also takes advantage of the unique benefits of conducting studies in Latin America, where approval pathways can be more efficient and cost-effective. Ultimately, these strategies can transform the landscape of clinical research in Chile, particularly in the field of radiopharma clinical trial Chile, paving the way for more effective studies and better patient outcomes.
Leverage U.S. Regulatory Standards for Faster Trial Execution in Latin America
Navigating the intricate landscape of regulatory compliance can be daunting for many sponsors, yet aligning radiopharma clinical trial Chile with U.S. standards offers a pathway to expedited execution and enhanced data quality. Key strategies include:
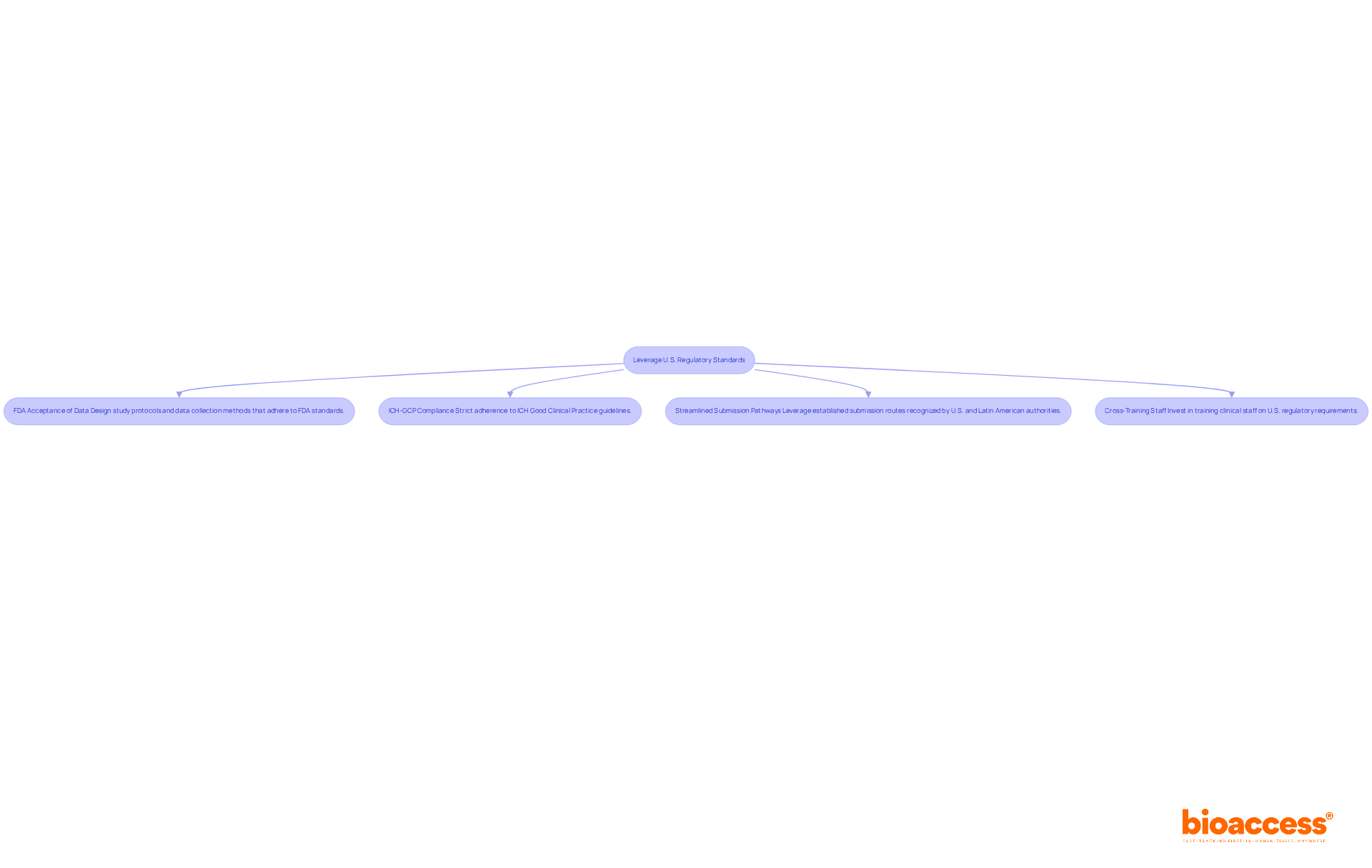
- FDA Acceptance of Data: Design study protocols and data collection methods that adhere to FDA standards. This alignment not only enhances the credibility of the data submitted for approval but also facilitates smoother interactions with U.S. oversight bodies.
- ICH-GCP Compliance: Strict adherence to International Council for Harmonisation (ICH) Good Clinical Practice (GCP) guidelines is essential. This ensures that trials are conducted ethically and that the data generated meets high-quality standards, which is crucial for acceptance by authorities.
- Streamlined Submission Pathways: Leverage established submission routes recognized by both U.S. and Latin American authorities, such as ANVISA in Brazil and COFEPRIS in Mexico. This approach minimizes redundancy in documentation and accelerates the approval process, with timelines in Brazil now capped at 90 days for new drug submissions.
- Cross-Training Staff: Invest in training clinical staff on U.S. regulatory requirements. This ensures that all study activities are aligned with best practices, enhancing compliance and operational efficiency. Comprehending the subtleties of U.S. regulations can lead to improved preparation for FDA submissions and enhance overall study outcomes.
By embracing these strategies, sponsors not only position themselves for success but also pave the way for future innovations in clinical research.
Conclusion
Navigating the intricate regulatory landscape of radiopharmaceutical clinical trials in Chile can be daunting, yet it holds the key to unlocking groundbreaking advancements in healthcare. By focusing on the regulatory frameworks set by the Instituto de Salud Pública (ISP), sponsors can streamline the approval process and ensure compliance with both local and international standards. Early feasibility studies are essential for assessing new treatments’ viability, while effective recruitment strategies boost participant engagement and lead to successful trial outcomes.
Mastering the regulatory landscape, implementing early feasibility studies, and developing targeted recruitment strategies are key practices for success. Grasping submission pathways and compliance requirements, including ICH-GCP and FDA standards, positions sponsors for success in clinical research. Leveraging the unique advantages of conducting trials in Latin America, like expedited approval timelines and cost efficiency, can significantly enhance clinical studies’ effectiveness.
Embracing these best practices not only mitigates risks but also fosters innovation and improves patient outcomes in radiopharmaceutical trials. By prioritizing these strategies, sponsors can not only enhance trial success but also drive transformative change in patient care across Chile and beyond.
Frequently Asked Questions
What is the role of the Instituto de Salud Pública (ISP) in radiopharmaceutical trials in Chile?
The ISP supervises the validation steps for clinical trials, ensuring adherence to local regulations and international standards such as ICH-GCP.
What is required for a Clinical Trial Application (CTA) in Chile?
A CTA must include the study protocol, informed consent forms, and safety data. The ISP typically assesses applications within 30 business days, but the overall process may take between 30 to 90 days.
Why is Ethics Committee Approval necessary before starting a clinical study?
Securing authorization from a recognized ethics committee is essential to ensure that the study adheres to ethical standards and safeguards participant rights.
What regulations must be followed regarding radiopharmaceuticals in clinical trials?
All radiopharmaceuticals used in the study must be licensed and comply with local regulations governing their use and handling to ensure safety and effectiveness for clinical use.
What safety standards must be complied with when handling radioactive materials in trials?
It is crucial to adhere to safety protocols for handling radioactive materials, which includes ensuring that all staff involved in the study receive adequate training for safe handling.
How can sponsors streamline the approval process for radiopharmaceutical trials in Chile?
By mastering the regulatory landscape and compliance requirements, sponsors can reduce delays and confusion, ultimately enhancing study execution and aligning with community health priorities.
What are the advantages of conducting early-stage clinical trials in Latin America?
Latin America offers advantages such as speed, cost efficiency, and effective patient recruitment, along with streamlined regulatory pathways, making it a strategic location for early-stage clinical trials.
List of Sources
- Understand Regulatory Frameworks for Radiopharmaceutical Trials in Chile
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Chile ISP Introduces Regulatory Reliance Framework for Pharmaceutical GMP Inspections (https://regask.com/chile-isp-introduces-regulatory-reliance-framework-for-pharmaceutical-gmp-inspections)
- Master Early Phase Clinical Trials in Chile: Key Strategies and Insights | bioaccess® (https://bioaccessla.com/blog/master-early-phase-clinical-trials-in-chile-key-strategies-and-insights)
- Implement Early Feasibility Studies to Enhance Trial Success
- Early Feasibility Studies (EFS) Program (https://fda.gov/medical-devices/investigational-device-exemption-ide/early-feasibility-studies-efs-program)
- Early Feasibility Studies | MED Institute (https://medinstitute.com/blog/early-feasibility-studies)
- Estimation of Clinical Trial Success Rates and Related Parameters – CanceRx: New Approaches to Commercializing Biomedical Research (https://cancerx.mit.edu/638)
- Feasibility In the Age of International Clinical Trials | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/feasibility-age-international-clinical-trials)
- Develop Effective Recruitment Strategies for Clinical Trials
- Patient Recruitment for Clinical Trials: Strategies That Actually Work (https://kapsuletech.com/blog/patient-recruitment-clinical-trials)
- Enrollment in Clinical Trials: Statistics and Patient Recruitment Strategies | Power (https://withpower.com/guides/enrollment-in-clinical-trials-statistics-and-patient-recruitment-strategies)
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- 3 Patient Recruitment Strategies in Clinical Trials (https://clariness.com/resource/3-patient-recruitment-strategies-in-clinical-trials)
- 10 Inspiring Patient Experience Quotes | Relias (https://relias.com/blog/patient-experience-quotes)
- Leverage U.S. Regulatory Standards for Faster Trial Execution in Latin America
- 7 Takeaways from FDA’s “Clinical Data in Med Device Submissions” (https://blog.cognition.us/fda-clinical-data-med-device-submissions)
- Can regulatory reform unearth LATAMs untapped potential for drug development? (https://clinicaltrialsarena.com/news/can-regulatory-reform-unearth-latams-untapped-potential-for-drug-development)
- US FDA to monitor clinical trial data in real time in pilot program aimed at speeding approvals (https://reuters.com/legal/litigation/us-fda-monitor-clinical-trial-data-real-time-pilot-program-aimed-speeding-2026-04-28)
- Analysis of US Food and Drug Administration new drug and biologic approvals, regulatory pathways, and review times, 1980–2022 – Scientific Reports (https://nature.com/articles/s41598-024-53554-7)

Leave a Reply