Introduction
Navigating the landscape of radiopharmaceutical clinical trials in Brazil offers a compelling opportunity for sponsors eager to leverage the region’s regulatory efficiency and diverse patient population.
With the increasing prevalence of chronic diseases, navigating compliance requirements can be daunting for sponsors.
What challenges do sponsors face in navigating local regulations while ensuring safety and success?
This article delves into strategic approaches that can enhance trial efficiency and effectiveness in Brazil.
By collaborating effectively, sponsors can enhance trial efficiency and position themselves for success in a competitive market.
Define Radiopharmaceuticals and Their Role in Clinical Trials
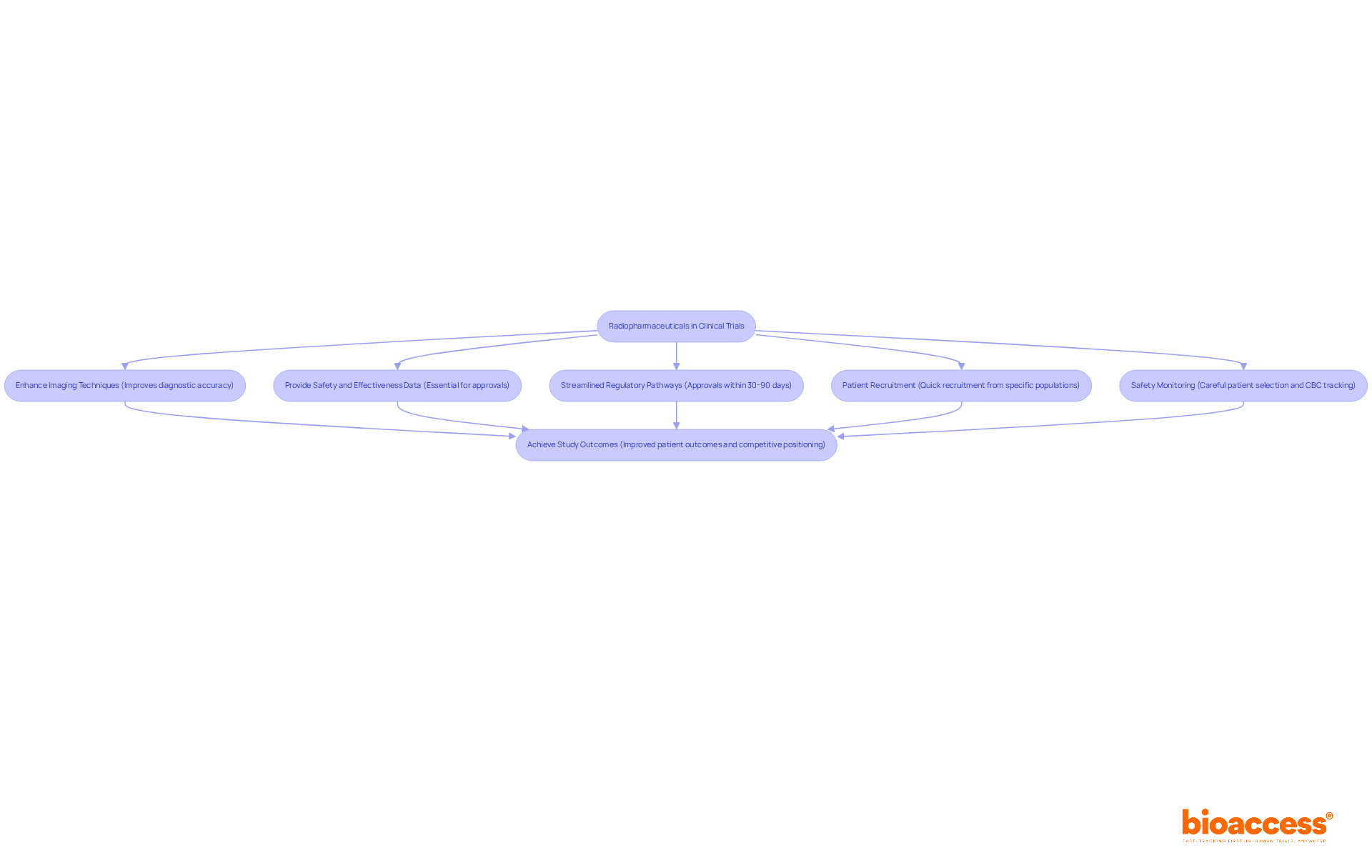
Radiopharmaceuticals are not just agents; they are pivotal tools in the fight against cancer, shaping the future of diagnostic imaging and therapeutic interventions. Their role in research is crucial, as they provide essential safety and effectiveness data needed for approvals. In Brazil, the rising prevalence of chronic diseases has intensified the demand for these agents, compelling study sponsors to understand their applications in the radiopharma clinical trial Brazil and the oversight frameworks established by ANVISA.
In oncology, radiopharmaceuticals significantly enhance imaging techniques, leading to more accurate diagnoses and personalized treatment plans. For instance, PSMA-targeted imaging has shown promise in prostate cancer studies, allowing for better patient stratification and monitoring. Integrating radiopharmaceuticals into studies boosts diagnostic accuracy and generates robust data for submissions to authorities like ANVISA.
Moreover, Brazil’s operational environment offers unique advantages for radiopharma clinical trial Brazil research. Streamlined regulatory pathways enable approvals within 30 to 90 days in the context of the radiopharma clinical trial Brazil, greatly expediting the process. This efficiency is further enhanced by the ability to quickly recruit patients, especially among specific populations, such as those in ‘watch and wait’ cohorts for prostate cancer. By leveraging these factors, sponsors can improve their study outcomes in the context of the radiopharma clinical trial Brazil while adhering to ICH-GCP standards and ensuring that all radiopharmaceuticals are administered within the necessary timeframes, thus preventing dosing delays.
The proactive management of patient guidance materials and adverse event plans tailored for radiation-specific effects underscores the commitment to safety and efficacy in these studies. As Dr. Alicia K. Morgans, MD, MPH, emphasizes, careful patient selection and safety monitoring, including complete blood count tracking, are essential when utilizing radiopharmaceuticals. As the landscape of cancer treatment progresses, the role of radiopharmaceuticals becomes increasingly crucial. This integration not only enhances patient outcomes but also positions sponsors favorably in the competitive landscape of clinical research. Furthermore, ECG supervision, which includes central assessment and cardiologist involvement at sites, reinforces the dedication to safety and compliance throughout the study process. With bioaccess®’s Innovation Runway, study sponsors can accelerate their journey to approval and funding success, ensuring they do not run out of runway before achieving critical milestones.
Explore Brazil’s Regulatory Framework for Radiopharmaceutical Trials
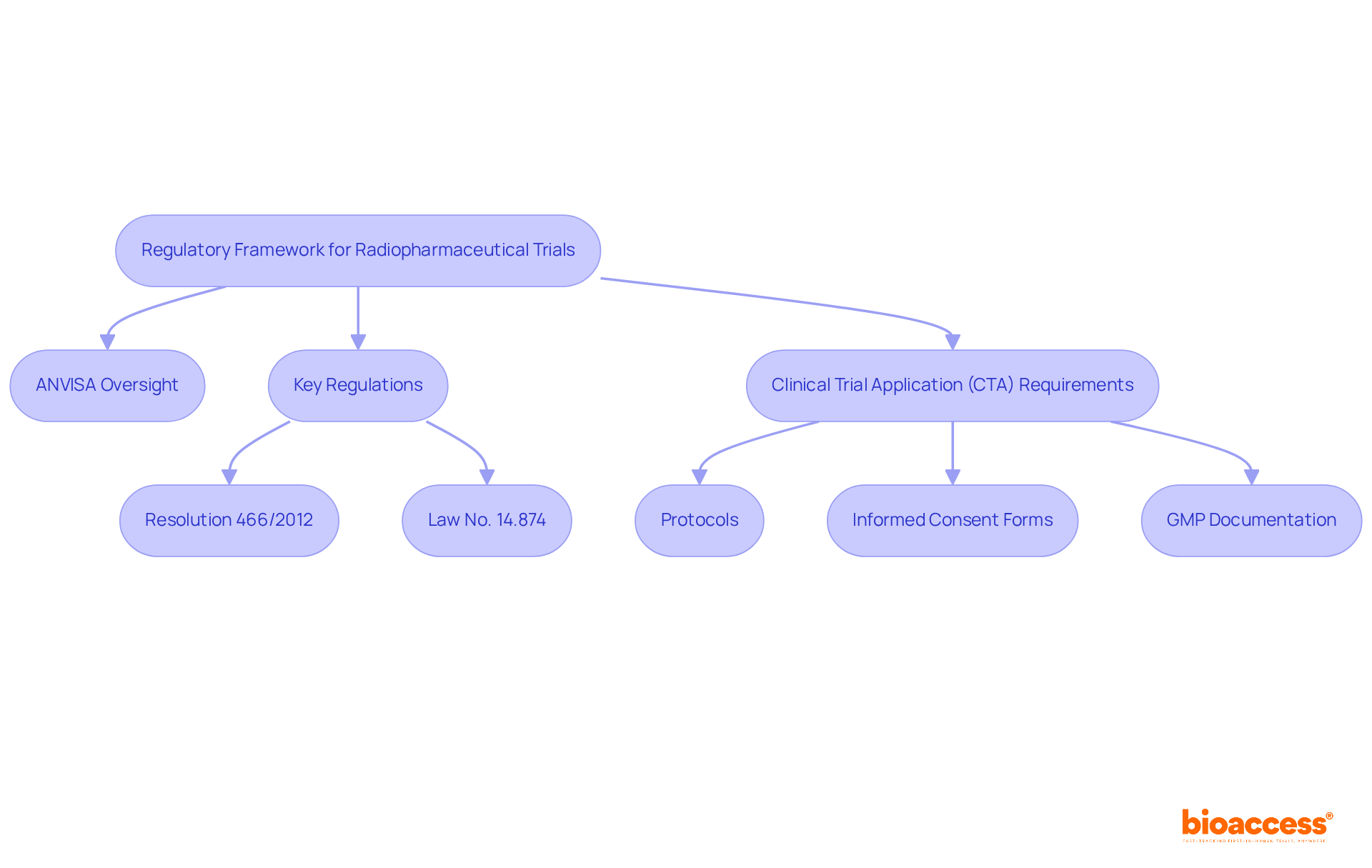
Navigating the regulatory landscape for radiopharmaceutical studies is crucial for the success of a radiopharma clinical trial in Brazil. In this region, ANVISA (Agência Nacional de Vigilância Sanitária) oversees the approval and management of research activities. Key regulations include:
- Resolution 466/2012, which emphasizes ethical considerations and participant protection.
- Recent updates introduced by Law No. 14.874, which significantly streamline the approval process.
These updates require sponsors to submit a Clinical Trial Application (CTA) that includes comprehensive protocols, informed consent forms, and documentation demonstrating adherence to Good Manufacturing Practices (GMP).
Recent reforms have aimed to shorten the average review period for medical research applications to about 90 days, enhancing the region’s appeal as a hub for research initiatives. Understanding these regulations is key to ensuring compliance and getting studies off the ground quickly. Have you considered how the new framework integrates ethical and health assessments? This allows for simultaneous evaluations by local ethics committees and ANVISA, further accelerating the approval process. This compliance environment not only promotes efficiency but also positions the country as a strategic center for radiopharma clinical trials in the radiopharmaceutical sector.
Implement Effective Strategies for Conducting Trials in Brazil
To navigate the complexities of radiopharmaceutical trials in Brazil, sponsors must adopt strategic approaches that leverage local insights and expertise:
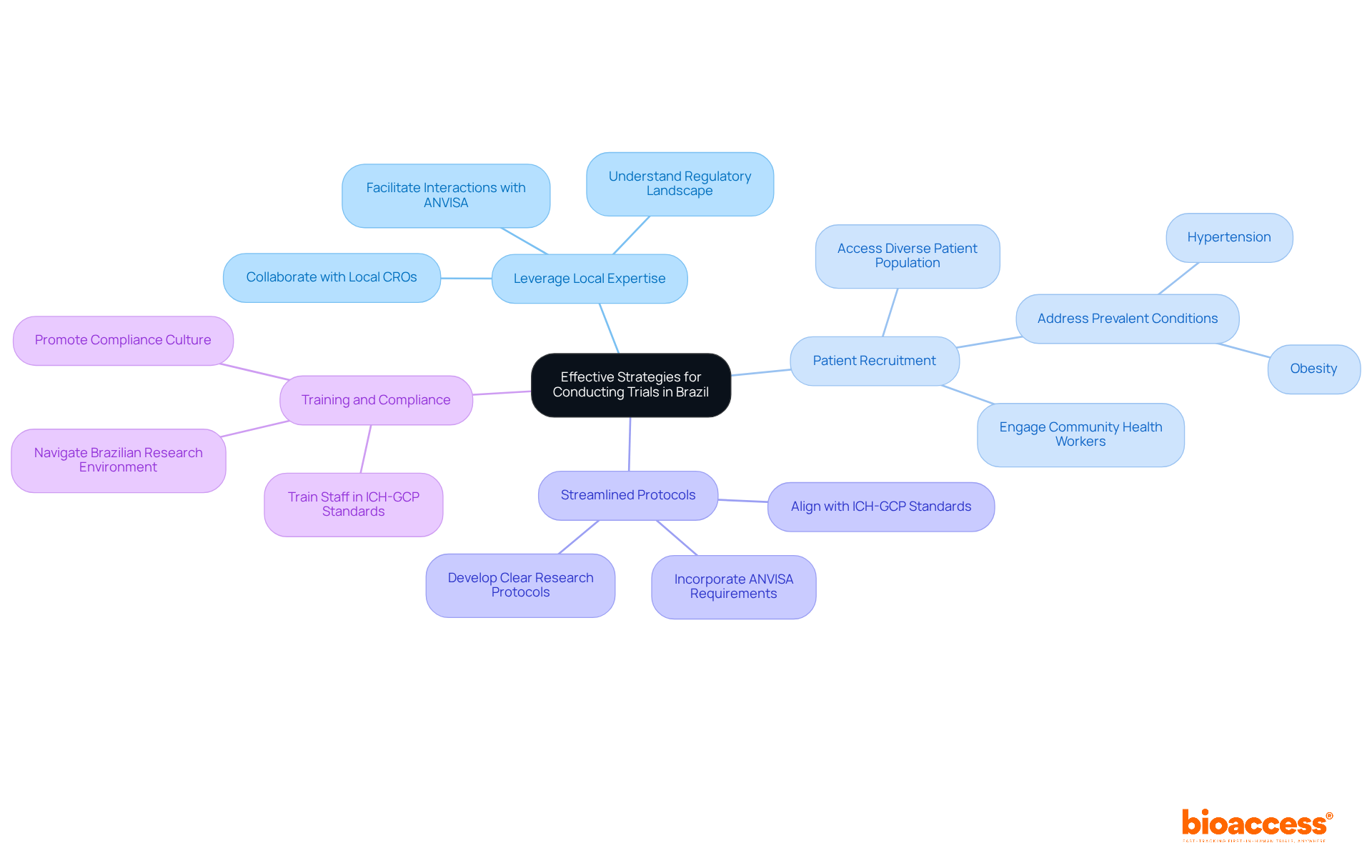
- Leverage Local Expertise: Collaborating with local CROs like bioaccess® provides invaluable insights into the regulatory landscape, facilitating smoother interactions with ANVISA, the regulatory authority in the region. This collaboration not only streamlines the approval process but is also vital for ensuring prompt study initiation.
- Patient Recruitment: Brazil’s extensive public healthcare system offers access to a varied patient population, crucial for strong clinical evaluations. Engaging community health workers can significantly enhance recruitment efforts, ensuring a broader representation of participants across various demographics, including those affected by prevalent conditions such as hypertension and obesity. How can you ensure your recruitment strategies are as effective as possible?
- Streamlined Protocols: Developing clear and concise research protocols that align with local regulations minimizes delays in approval. By adhering to ICH-GCP standards and incorporating specific requirements from ANVISA, sponsors can expedite the regulatory process and enhance compliance.
- Training and Compliance: Ensuring that all staff involved in the study are well-trained in ICH-GCP standards and local regulations is crucial for maintaining compliance and data integrity. This training not only promotes a culture of excellence but also equips the team to effectively navigate the intricacies of the Brazilian research environment.
By embracing these strategies, sponsors can not only enhance their trial outcomes but also position themselves advantageously in the competitive landscape of radiopharma clinical trial Brazil and radiopharmaceutical research in Latin America.
Analyze Cost-Effective Approaches for Radiopharmaceutical Trials
Why are sponsors increasingly turning to Brazil for radiopharma clinical trial studies? Conducting studies in this region offers significant cost advantages compared to other areas. Here are key strategies to enhance cost-effectiveness:
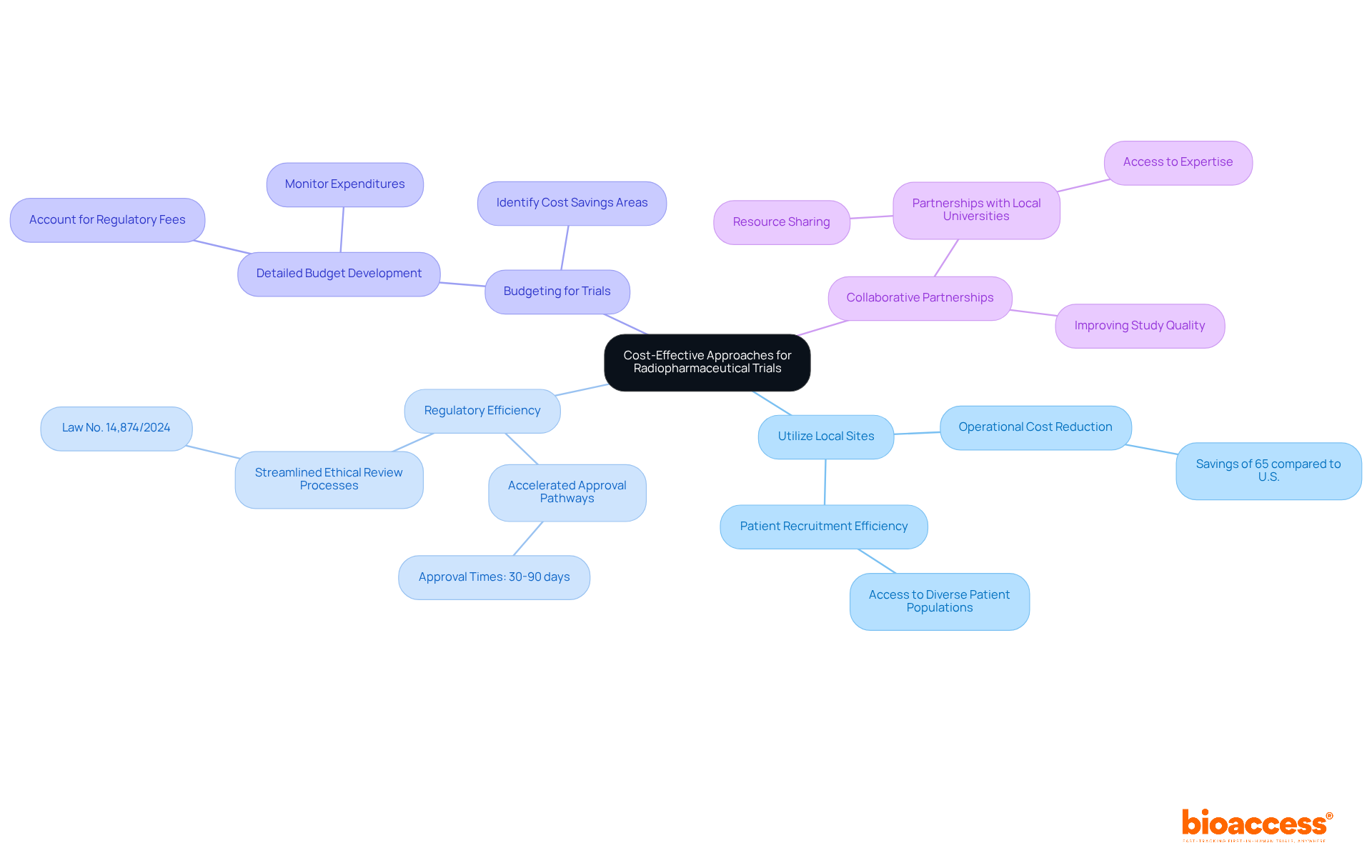
- Utilize Local Sites: By engaging local clinical research sites, sponsors can cut operational costs by about 65% compared to the U.S. This approach not only lowers expenses but also boosts patient recruitment efficiency, as local sites are better positioned to access diverse patient populations.
- Regulatory Efficiency: The country’s accelerated approval pathways, including streamlined ethical review processes under Law No. 14,874/2024, can significantly shorten approval times, often reducing them to 30-90 days. This efficiency not only cuts costs but also accelerates study initiation, making Brazil a top choice for radiopharma clinical trial sponsors.
- Budgeting for Trials: Developing a detailed budget is crucial. Sponsors should account for all potential expenses, including regulatory fees, site costs, and patient compensation. By closely monitoring expenditures, sponsors can identify areas for cost savings and optimize resource allocation.
- Collaborative Partnerships: Forming partnerships with local universities and research institutions can further reduce costs. These collaborations enable resource sharing and access to expertise, improving study quality while conserving financial resources.
By embracing these strategies, sponsors can not only save costs but also enhance the quality and speed of their clinical trials in Brazil.
Conclusion
Navigating the integration of radiopharmaceuticals into clinical trials is not just an opportunity; it’s a necessity for advancing cancer treatment in Brazil. Understanding Brazil’s regulatory frameworks is crucial for study sponsors. It allows them to navigate the complexities of trials involving these vital agents, ultimately enhancing patient outcomes and speeding up approval processes.
Key strategies for success include:
- Leveraging local expertise to streamline interactions with ANVISA
- Optimizing patient recruitment through Brazil’s diverse healthcare system
- Adhering to stringent compliance protocols
The recent regulatory updates not only facilitate quicker approvals but also highlight the importance of ethical considerations in trial management. Moreover, cost-effective approaches, such as utilizing local sites and forming partnerships with academic institutions, further bolster the feasibility and efficiency of these trials.
With the growing demand for innovative cancer treatments, we must recognize the critical role radiopharmaceuticals play in clinical research. By adopting these strategies, sponsors can take full advantage of Brazil’s unique position in the radiopharma trial landscape. The successful integration of radiopharmaceuticals will not only enhance patient care but also redefine the future of cancer research in Brazil.
Frequently Asked Questions
What are radiopharmaceuticals and their significance in clinical trials?
Radiopharmaceuticals are pivotal tools in cancer treatment, essential for diagnostic imaging and therapeutic interventions. They provide crucial safety and effectiveness data required for regulatory approvals in clinical trials.
How do radiopharmaceuticals enhance oncology research?
In oncology, radiopharmaceuticals improve imaging techniques, leading to more accurate diagnoses and personalized treatment plans, such as PSMA-targeted imaging in prostate cancer studies, which aids in patient stratification and monitoring.
What advantages does Brazil offer for radiopharmaceutical clinical trials?
Brazil provides streamlined regulatory pathways that enable approvals within 30 to 90 days, along with efficient patient recruitment, particularly among specific populations, enhancing study outcomes while adhering to ICH-GCP standards.
What is the importance of patient guidance materials and adverse event plans in radiopharmaceutical studies?
Proactive management of patient guidance materials and tailored adverse event plans for radiation-specific effects reflects a commitment to safety and efficacy in radiopharmaceutical studies.
What safety measures are emphasized when using radiopharmaceuticals?
Careful patient selection and safety monitoring, including tracking complete blood counts, are essential for ensuring patient safety during the use of radiopharmaceuticals.
How does ECG supervision contribute to the safety of radiopharmaceutical studies?
ECG supervision, involving central assessment and cardiologist involvement at study sites, reinforces safety and compliance throughout the clinical trial process.
How can study sponsors accelerate their journey to approval for radiopharmaceuticals in Brazil?
Utilizing platforms like bioaccess®’s Innovation Runway can help study sponsors expedite approval and funding success, ensuring they meet critical milestones without delays.
List of Sources
- Define Radiopharmaceuticals and Their Role in Clinical Trials
- Phase 3 Prostate Cancer Case Study: Radiopharm Imaging at Scale (https://precisionformedicine.com/blog/phase-3-prostate-cancer-case-study-radiopharm-imaging-at-scale)
- Clinical Experience With Radiopharmaceuticals | Targeted Oncology – Immunotherapy, Biomarkers, and Cancer Pathways (https://targetedonc.com/view/clinical-experience-with-radiopharmaceuticals)
- Explore Brazil’s Regulatory Framework for Radiopharmaceutical Trials
- How Brazil’s New Law Is Redefining The Country’s Clinical Research (https://clinicalleader.com/doc/how-brazil-s-new-law-is-redefining-the-country-s-clinical-research-0001)
- New regulations for clinical research in Brazil | Licks Attorneys (https://lickslegal.com/post/new-regulations-for-clinical-research-in-brazil)
- Implement Effective Strategies for Conducting Trials in Brazil
- Unlocking Brazil’s Clinical Trial Opportunity: A Strategic Roadmap for Healthcare Leaders (https://lek.com/insights/life-sciences-pharma/unlocking-brazils-clinical-trial-opportunity-strategic-roadmap)
- Current scenario and future perspectives of clinical research in Brazil: a national survey – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10898894)
- Brazil Clinical Trial Patient Recruitment Services Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trial-patient-recruitment-services-market/brazil)
- CLINICAL TRIALS SCENARIO IN BRAZIL- STUDY AND SPONSOR PROFILES (https://ispor.org/heor-resources/presentations-database/presentation/ispor-19th-annual-european-congress/clinical-trials-scenario-in-brazil-study-and-sponsor-profiles)
- Analyze Cost-Effective Approaches for Radiopharmaceutical Trials
- Brazil Clinical Trials Market Size, Share | Analysis Report [2032] (https://fortunebusinessinsights.com/brazil-clinical-trials-market-112310)
- The Ultimate Guide to Clinical Trial Costs in 2025 (https://sofpromed.com/ultimate-guide-clinical-trial-costs)
- Unlocking Brazil’s Clinical Trial Opportunity: A Strategic Roadmap for Healthcare Leaders (https://lek.com/insights/life-sciences-pharma/unlocking-brazils-clinical-trial-opportunity-strategic-roadmap)
- Best Practices For First In Human Trials In Brazil: Strategies For Success | bioaccess® (https://bioaccessla.com/blog/best-practices-for-first-in-human-trials-in-brazil-strategies-for-success)
