Introduction
Choosing the right Contract Research Organization (CRO) for first-in-human (FIH) trials in Chile is not just important; it’s a pivotal decision that can shape the future of biopharma and MedTech innovations. With its unique regulatory landscape, cost efficiency, and rapid patient recruitment capabilities, Chile presents a compelling opportunity for early-stage clinical research. Yet, finding a CRO that understands local regulations and has the specialized expertise for compliance can be quite challenging.
What steps can organizations take to ensure they choose the right CRO for their trials?
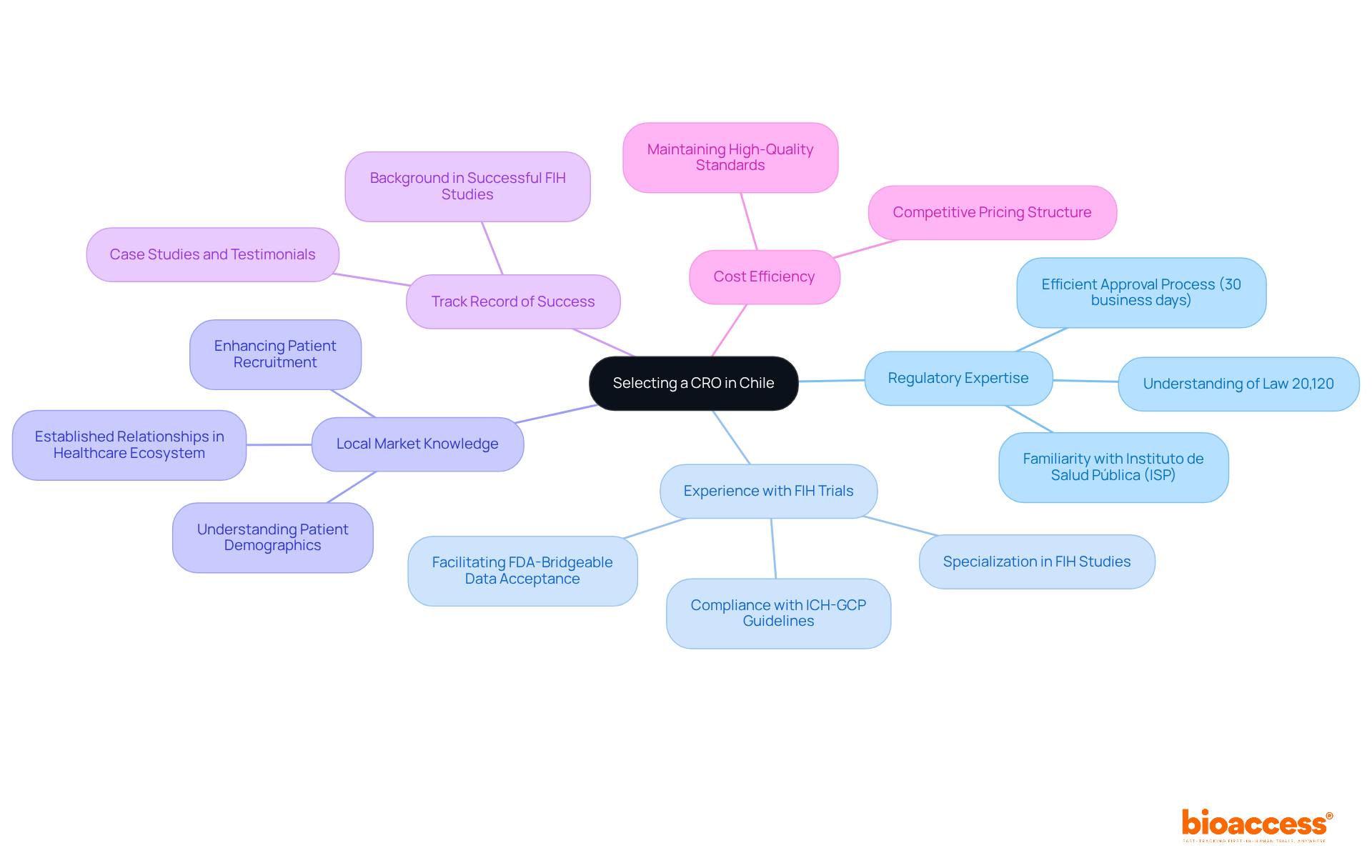
Identify Key Criteria for Selecting a CRO in Chile
Navigating the complexities of CRO Chile first-in-human trials requires a strategic approach to selecting the right Contract Research Organization. When choosing a CRO for FIH trials, consider these key criteria:
- Regulatory Expertise: Ensure the CRO possesses a comprehensive understanding of Chilean regulations, particularly Law 20,120, which governs clinical studies. Familiarity with local regulatory authorities such as the Instituto de Salud Pública (ISP) is essential for navigating the approval process efficiently, as the ISP typically reviews applications within 30 business days.
- Experience with FIH Trials: Opt for a CRO that specializes in CRO Chile first-in-human studies. Their expertise will be invaluable in designing protocols that comply with both local and international standards, ensuring adherence to ICH-GCP guidelines and facilitating FDA-bridgeable data acceptance.
- Local Market Knowledge: A CRO with established relationships within the local healthcare ecosystem can enhance patient recruitment and site management. Comprehending the intricacies of the Chilean healthcare system and patient demographics is essential for successful execution of studies.
- Track Record of Success: Evaluate the CRO’s background in executing successful CRO Chile first-in-human studies. Request case studies or testimonials from previous clients to assess their effectiveness and reliability in delivering high-quality clinical data.
- Cost Efficiency: Analyze the CRO’s pricing structure. A CRO that offers competitive rates while maintaining high-quality standards will help preserve your startup’s capital, which is vital for sustaining operations and achieving regulatory milestones.
Choosing the right CRO not only streamlines your trial process but also positions your study for success in a competitive landscape.
Understand the Regulatory Landscape for FIH Trials in Chile
Understanding the regulatory landscape for CRO Chile first-in-human trials is crucial for successful clinical research. Navigating this landscape involves several key steps, particularly when leveraging the insights provided by bioaccess® through its Global Trial Accelerators™ program:
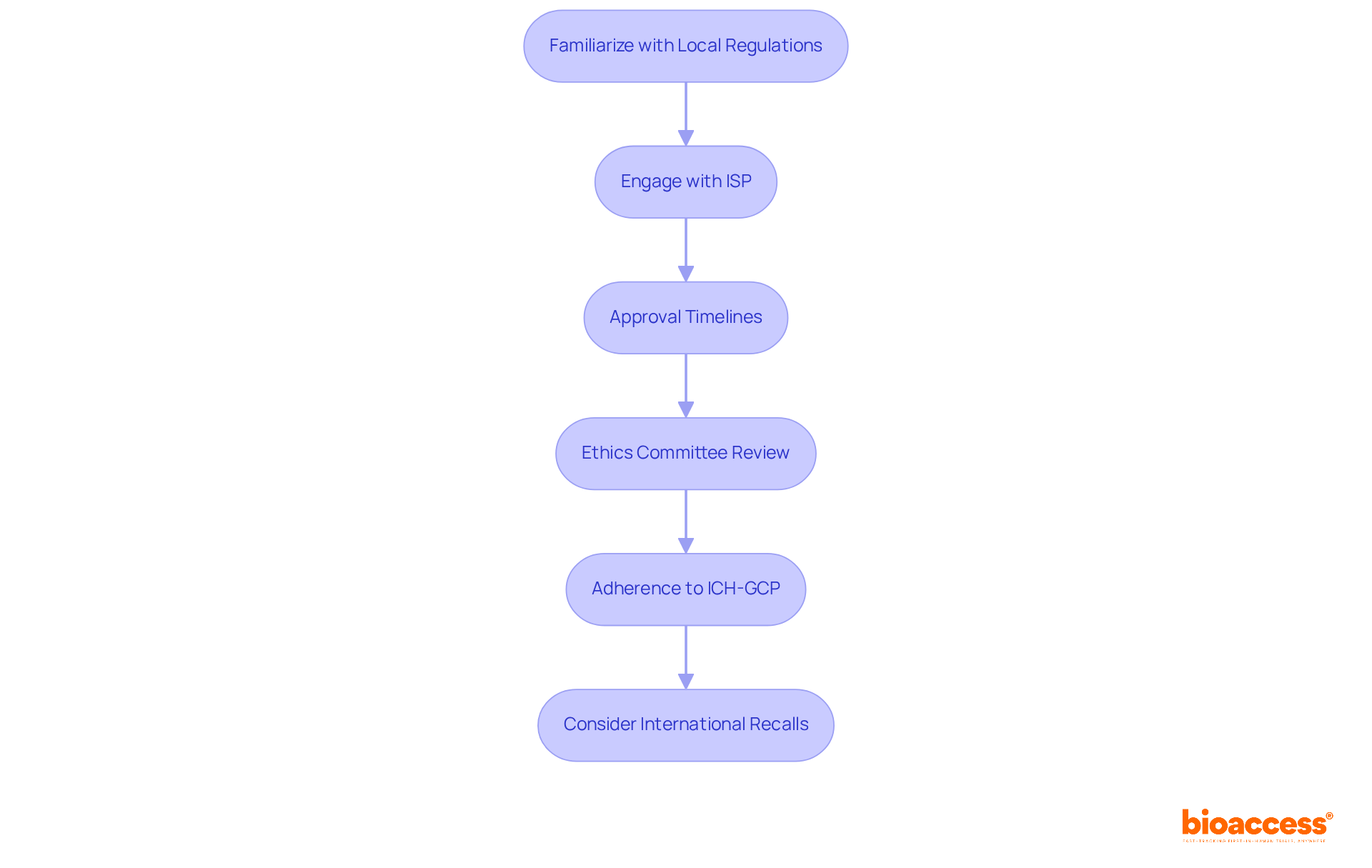
- Familiarize Yourself with Local Regulations: The primary regulation governing clinical studies in Chile is Law 20,120, which outlines the requirements for conducting human research. Grasping this regulation is crucial for adherence and effective execution of studies, particularly in relation to the distinct characteristics of the Chilean market.
- Engage with the Instituto de Salud Pública (ISP): The ISP is the regulatory authority responsible for approving clinical evaluations. Submissions must include a comprehensive clinical study application, which typically consists of the study protocol, informed consent forms, and approvals from an accredited ethics committee. Additionally, manufacturers must notify the ISP of any product withdrawal due to health risks, which is crucial for compliance. Leveraging insights can streamline this process, ensuring that all necessary documentation meets regulatory expectations.
- Approval Timelines: The approval process generally takes about 30 business days; however, delays can arise if submissions are incomplete or overly complex. The average approval duration for research studies in Chile can surpass six months. Preparing for this schedule is essential to prevent delays in study commencement, and our organization can offer strategies to accelerate this process.
- Ethics Committee Review: Before submitting to the ISP, ensure that your study has been reviewed and approved by an accredited ethics committee. Since this step is mandatory, getting early approval can save you time in the long run. Establishing a three-step ethical framework is essential for addressing significant ethical concerns in research studies, and bioaccess® can assist in navigating these requirements effectively.
- Adherence to ICH-GCP: All studies must comply with International Council for Harmonisation – Good Clinical Practice (ICH-GCP) standards. This compliance guarantees the safeguarding of the rights, safety, and well-being of participants in the study, which is crucial in clinical research. It is also essential to acknowledge that liability waivers are prohibited, highlighting the legal obligations involved in conducting studies. Bioaccess® provides resources to help ensure adherence to these standards.
- Consider International Recalls: Be aware that international recalls can influence regulatory decisions in Chile, as the ISP reviews safety actions taken by foreign regulators. This aspect is vital for maintaining compliance and ensuring participant safety. Utilizing insights from this source can aid in anticipating and addressing these regulatory challenges.
By leveraging these insights, you can navigate the complexities of CRO Chile first-in-human studies and ensure a smoother path to success.
Evaluate CRO Experience and Capabilities in FIH Trials
Choosing the right CRO Chile first-in-human trials is crucial, as the wrong choice can lead to costly delays and setbacks. When evaluating a CRO, consider these critical aspects:
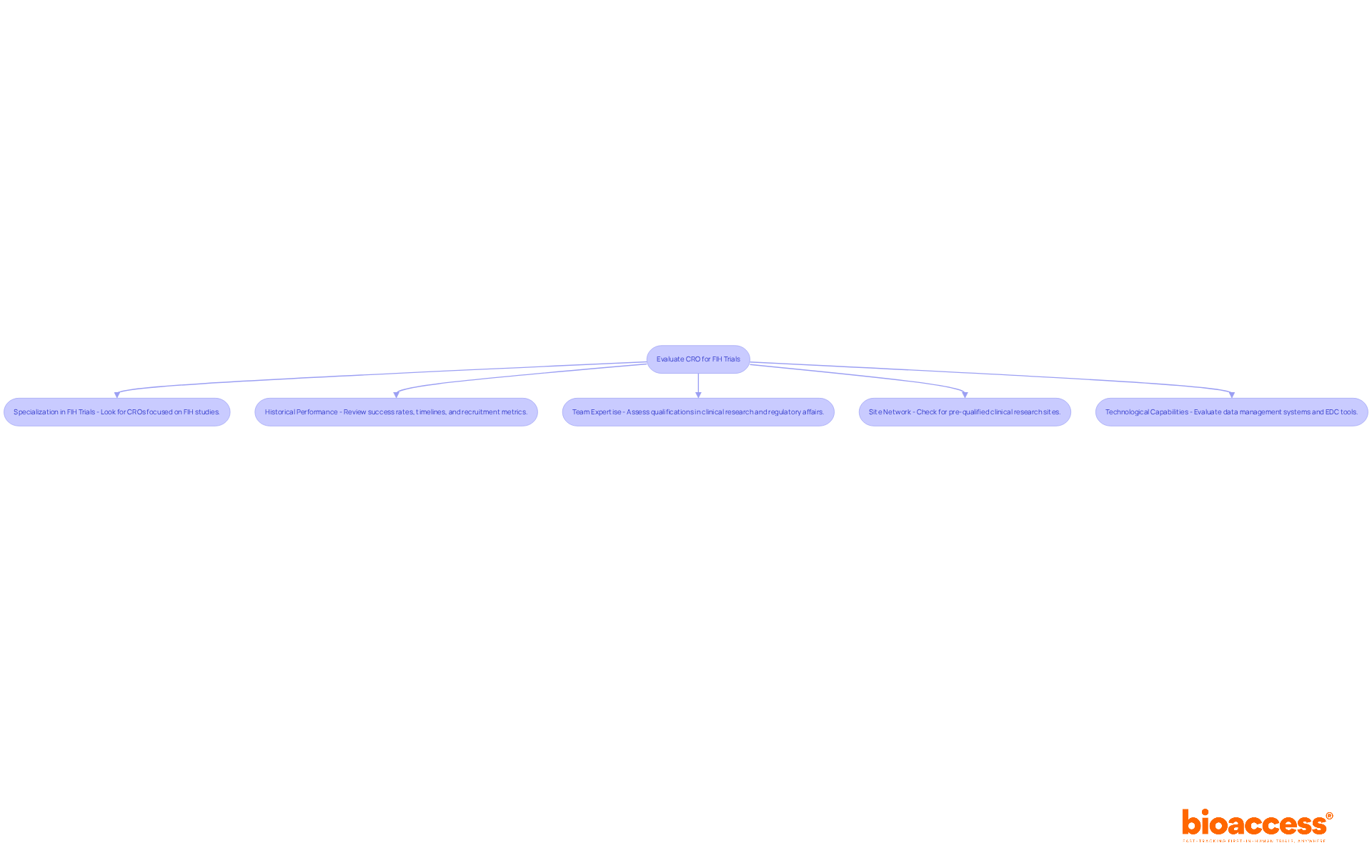
- Specialization in FIH Trials: Choosing a CRO that specializes exclusively in FIH studies is essential for your success. This organization is the only CRO globally established from the outset for first-in-human clinical studies, referred to as CRO Chile first-in-human, guaranteeing extensive expertise in optimal practices and frequent obstacles linked to early-phase studies, which can greatly influence the success of your research.
- Historical Performance: Ask for detailed data on the CRO’s past FIH studies-success rates, timelines, and patient recruitment metrics can tell you a lot about their capabilities. Successful CROs in Chile have shown the ability to recruit patients efficiently, often meeting recruitment goals within 30-60 days, which is essential for upholding project timelines.
- Team Expertise: Take a close look at the qualifications and experience of the CRO’s team members. Look for professionals with a strong background in clinical research management, regulatory affairs, and specific therapeutic areas relevant to your study. A versatile team at our organization can navigate the complexities of first-in-human studies more effectively.
- Site Network: Having a strong network of pre-qualified clinical research sites is crucial for speeding up patient recruitment and boosting study efficiency. Our organization has established relationships with local hospitals and clinics, facilitating smoother operations and quicker patient enrollment. Approvals in countries like Panama and El Salvador take just 4-8 weeks compared to 6+ months in the US/EU. This network also ensures compliance with local regulatory authorities such as INVIMA and ANVISA.
- Technological Capabilities: Look into the CRO’s technological infrastructure for managing studies. Advanced data management systems and electronic data capture (EDC) tools at our organization streamline processes, enhance data quality, and ensure compliance with ICH-GCP standards, which are essential for regulatory submissions.
By thoroughly assessing these factors, you can choose a CRO Chile first-in-human that is not only well-prepared to manage the complexities of first-in-human studies but also positioned to utilize the region’s strategic benefits in speed, cost efficiency, and regulatory pathways. By making an informed choice, you can leverage the unique advantages of conducting FIH trials in Chile, ensuring a smoother path to success.
Establish Effective Communication with Your Chosen CRO
Effective communication with your CRO Chile first-in-human can make or break your study in Latin America. Here are strategies to enhance this communication:
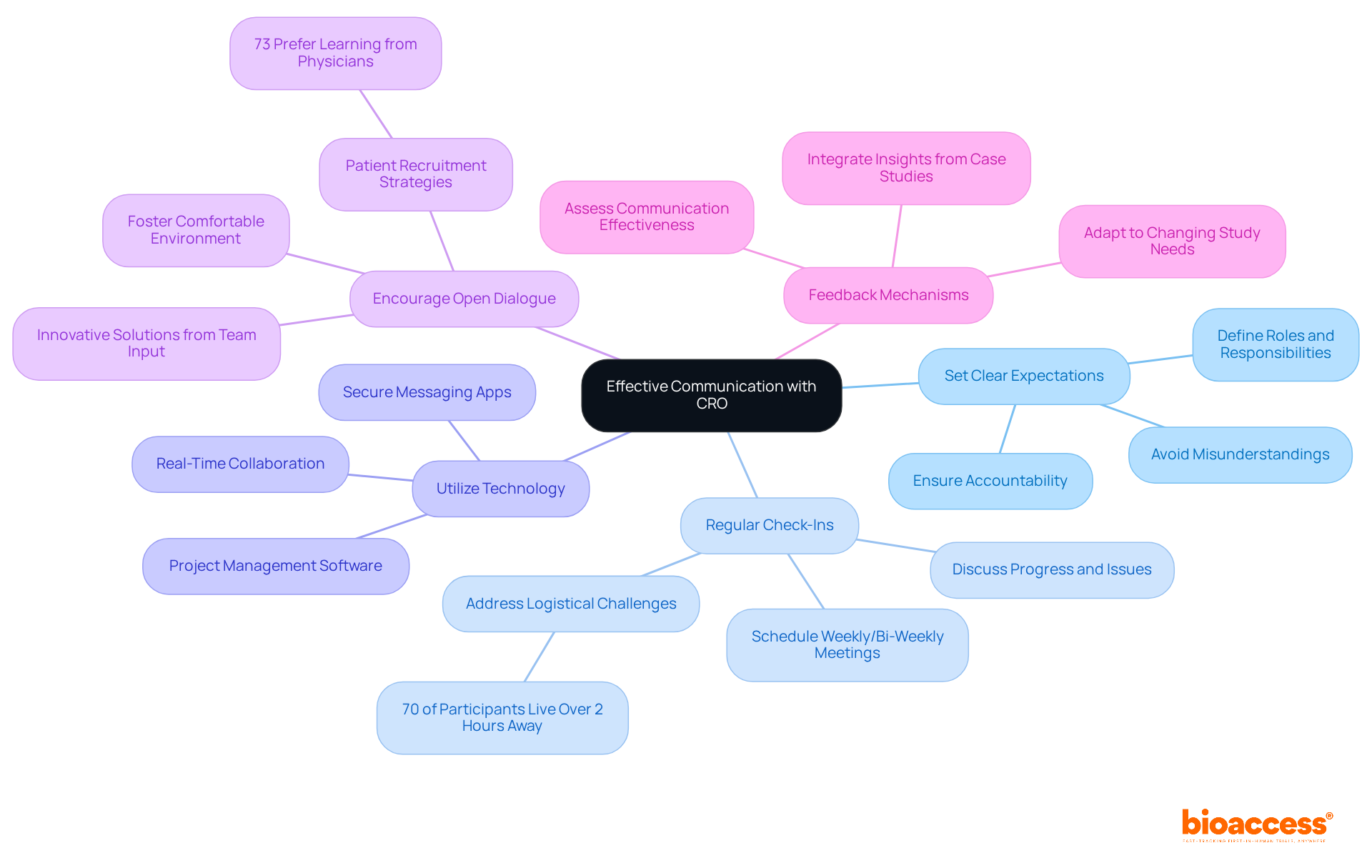
- Set Clear Expectations: Clearly define roles, responsibilities, and expectations at the beginning of the partnership with the organization. This clarity helps avoid misunderstandings and guarantees accountability, which is crucial for navigating the regulatory frameworks of nations like Chile and Brazil. Our organization has established proficiency in expediting Phase I studies with rapid ethics approvals from regulatory bodies such as INVIMA and ANVISA.
- Regular Check-Ins: Schedule consistent meetings – weekly or bi-weekly – to discuss progress, address issues, and provide updates. Regular check-ins keep the momentum going and ensure compliance with ICH-GCP standards and local regulations. Significantly, 70% of potential clinical study participants reside more than two hours from a research center, making these discussions essential for tackling logistical challenges, particularly in the context of streamlined logistics and sponsor support.
- Utilize Technology: Using technology can really streamline processes and improve how we manage data. Leverage project management software, shared document platforms, and secure messaging apps to facilitate real-time communication and collaboration. This integration is essential for meeting FDA-bridgeable data acceptance requirements, which our organization is well-equipped to handle.
- Encourage Open Dialogue: Foster an environment where team members feel comfortable sharing concerns and suggestions. Open dialogue can lead to innovative solutions and enhance study outcomes, especially in patient recruitment strategies tailored to the local population. Research shows that 73% of patients prefer to learn about clinical research opportunities from their physician’s office, highlighting the significance of effective communication, a principle emphasized in our operations.
- Feedback Mechanisms: Implement feedback loops to assess the effectiveness of communication strategies. Consistently seek input from both your team and the CRO to identify areas for enhancement, ensuring that the partnership evolves to meet the changing needs of the study. Integrating insights from successful case studies, such as those emphasizing quality collaborations between sponsors and CROs, can offer practical examples of how effective communication has resulted in successful research outcomes.
By enhancing communication, you not only streamline processes but also improve participant recruitment and study success. The success of your trial, specifically the CRO Chile first-in-human, hinges on how well you communicate and collaborate with bioaccess®.
Conclusion
Choosing the right Contract Research Organization (CRO) for first-in-human (FIH) trials in Chile is not just important; it’s a decision that can make or break your clinical research success. Evaluating a CRO requires careful consideration of several factors:
- Regulatory expertise
- Experience with FIH trials
- Local market knowledge
- A proven track record
- Cost efficiency
Each of these elements plays a crucial role in ensuring that the selected CRO can navigate the complexities of the Chilean healthcare landscape effectively.
Key insights highlighted in this guide emphasize the importance of understanding the regulatory landscape, including compliance with Law 20,120 and the role of the Instituto de Salud Pública (ISP). What criteria will you prioritize to ensure your research objectives are met? Additionally, the necessity for effective communication and collaboration between sponsors and CROs is underscored, as regular check-ins and open dialogue can enhance study outcomes and streamline processes.
The strategic advantages of conducting FIH trials in Chile are clear:
- Expedited approval timelines
- Cost efficiency
- Strong patient recruitment capabilities
By leveraging the insights and criteria outlined in this article, you can position yourself for success in the competitive realm of early-stage clinical trials. By embracing these considerations, you can ensure smoother operations and improve the quality and reliability of clinical research outcomes in Latin America.
Frequently Asked Questions
What is the importance of regulatory expertise when selecting a CRO in Chile for FIH trials?
Regulatory expertise is crucial as the CRO must have a comprehensive understanding of Chilean regulations, particularly Law 20,120, which governs clinical studies. Familiarity with local regulatory authorities like the Instituto de Salud Pública (ISP) is essential for efficiently navigating the approval process, as the ISP typically reviews applications within 30 business days.
Why is experience with first-in-human (FIH) trials significant when choosing a CRO?
Experience with FIH trials is significant because a specialized CRO will have the expertise necessary to design protocols that comply with both local and international standards. This ensures adherence to ICH-GCP guidelines and facilitates the acceptance of FDA-bridgeable data.
How does local market knowledge impact the selection of a CRO in Chile?
Local market knowledge is important as a CRO with established relationships within the local healthcare ecosystem can enhance patient recruitment and site management. Understanding the intricacies of the Chilean healthcare system and patient demographics is essential for the successful execution of studies.
What should be considered regarding the track record of success of a CRO?
When evaluating a CRO, it is important to assess their background in executing successful FIH studies. Requesting case studies or testimonials from previous clients can help determine their effectiveness and reliability in delivering high-quality clinical data.
How does cost efficiency factor into the selection of a CRO?
Cost efficiency is a key consideration, as analyzing the CRO’s pricing structure can reveal whether they offer competitive rates while maintaining high-quality standards. This is vital for preserving your startup’s capital, which is essential for sustaining operations and achieving regulatory milestones.
How does choosing the right CRO contribute to the success of a clinical trial in Chile?
Choosing the right CRO streamlines the trial process and positions your study for success in a competitive landscape. A well-selected CRO can enhance regulatory compliance, improve patient recruitment, and ensure high-quality data collection, all of which are critical for the success of FIH trials.
List of Sources
- Identify Key Criteria for Selecting a CRO in Chile
- Key Factors in CRO Selection | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/key-factors-cro-selection)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Beyond the Safety Check: Why First-in-Human Trials Demand a New Approach in 2026 | BioPharm International (https://biopharminternational.com/view/safety-first-human-trials-new-approach)
- The Value Of A High-Performing Regulatory Function Within A CRO (https://clinicalleader.com/doc/the-value-of-a-high-performing-regulatory-function-within-a-cro-0001)
- Understand the Regulatory Landscape for FIH Trials in Chile
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Drug & Medical Device Litigation Laws and Regulations Report 2026 Chile (https://iclg.com/practice-areas/drug-and-medical-device-litigation/chile)
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Evaluate CRO Experience and Capabilities in FIH Trials
- First-in-Human Clinical Trial CRO | bioaccess Latin America (https://bioaccessla.com/first-in-human-cro)
- Quotes About Medical Research (https://changingthepresent.org/pages/quotes-about-medical-research)
- A cross-sectional study on the first-in-human trials of anticancer drugs in Japan and the United States and the probability of approval – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC12474682)
- Women in Clinical Trials: 5 Quotes To Inspire Action (https://3blmedia.com/news/women-clinical-trials-5-quotes-inspire-action)
- Establish Effective Communication with Your Chosen CRO
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- Tips For Improving Communication Between Sponsors And CROs (https://clinicalleader.com/doc/tips-for-improving-communication-between-sponsors-and-cros-0001)
- Back to (Communication) Basics: Reducing Site Burden and Establishing a Sponsor/CRO-of-Choice Relationship with Investigative Sites (https://globalforum.diaglobal.org/issue/june-2024/back-to-communication-basics-reducing-site-burden-and-establishing-a-sponsor-cro-of-choice-relationship-with-investigative-sites)
- Enrollment and Retention: A Strategic Imperative for Clinical Trial Success – ConfidenceResearch (https://confidenceresearch.com/enrollment-and-retention-a-strategic-imperative-for-clinical-trial-success)

Leave a Reply