Introduction
The regulatory submission landscape for clinical trials in Chile is fraught with complexities that can hinder progress for MedTech and Biopharma companies. Mastering the essential components of this process allows stakeholders to streamline compliance efforts. It also enables them to leverage Chile’s strategic advantages for early-stage clinical research. Yet, with these complex regulations and potential pitfalls, what steps can organizations take to ensure smooth submissions and timely approvals? This article explores the intricacies of the regulatory framework and required documentation. It also outlines best practices to overcome common challenges, equipping stakeholders with the knowledge to navigate this critical landscape successfully.
Understand the Regulatory Landscape in Chile
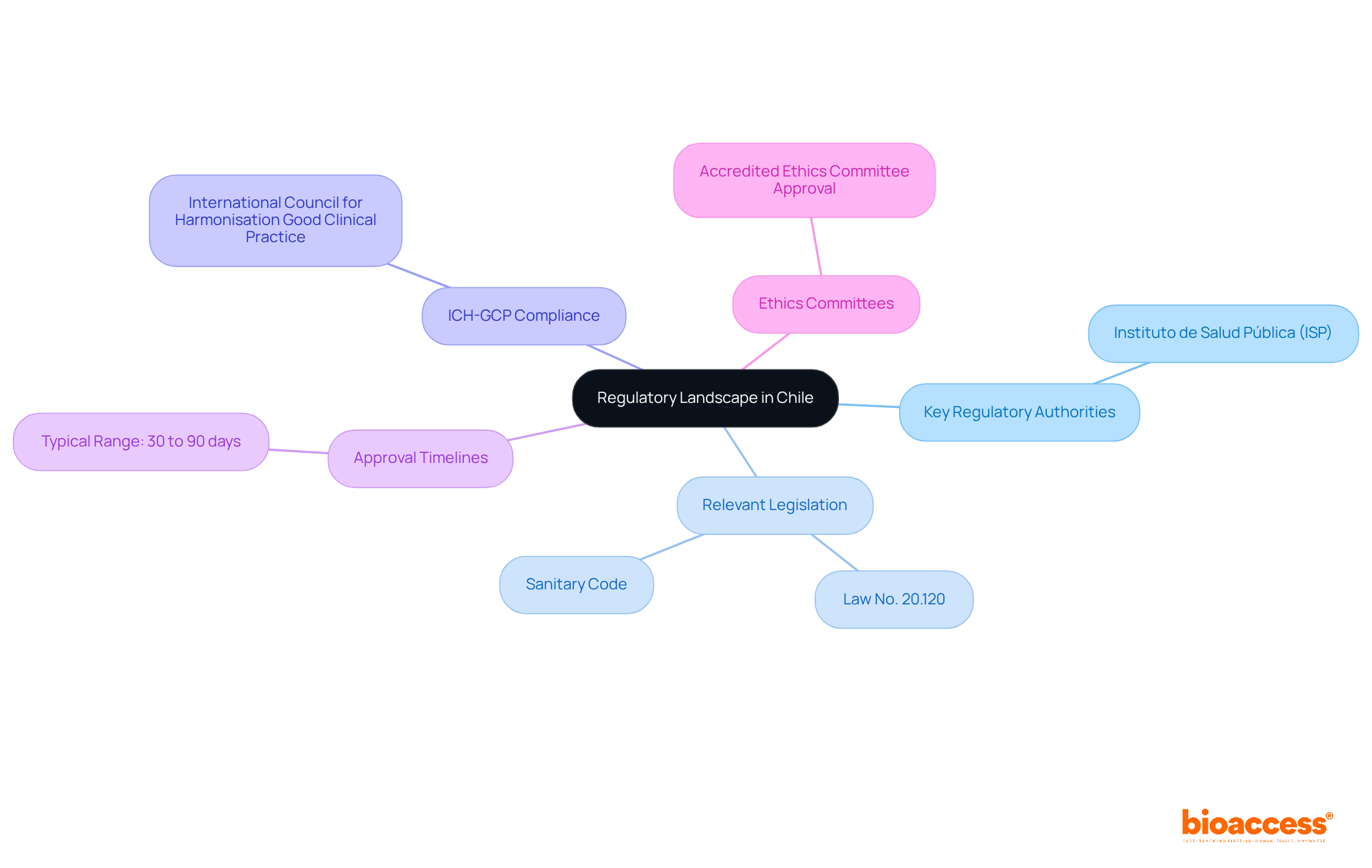
Navigating the regulatory submission Chile clinical trial process can be a daunting task, yet understanding its essential components is crucial for success. To effectively navigate this landscape, stakeholders must consider several key elements:
- Key Regulatory Authorities: The Instituto de Salud Pública (ISP) is the main entity supervising research studies in Chile. Understanding their guidelines and requirements is essential, as they oversee the approval of applications for studies.
- Relevant Legislation: Important laws include Law No. 20.120, which governs human research, and the Sanitary Code, which provides the general framework for health-related regulations. A comprehensive grasp of these laws is essential to guarantee adherence throughout the study process.
- ICH-GCP Compliance: All studies must adhere to the International Council for Harmonisation Good Clinical Practice (ICH-GCP) standards. This compliance ensures that studies are conducted ethically and that the data generated is dependable and suitable for a regulatory submission Chile clinical trial.
- Approval Timelines: The authorization process for clinical studies in Chile typically ranges from 30 to 90 days. This relatively swift turnaround can facilitate the faster commencement of studies compared to other regions, making Chile an appealing choice for first-in-human research.
- Ethics Committees: Before presenting to the ISP, studies must secure approval from an accredited Ethics Committee (Comité de Ética). This step is essential for ensuring the protection of participants’ rights and welfare.
By mastering these essential components, stakeholders can streamline their compliance efforts and expedite their clinical trials in Chile, leveraging the country’s strategic advantages for early-stage clinical trials.
Prepare Required Documentation for Submission
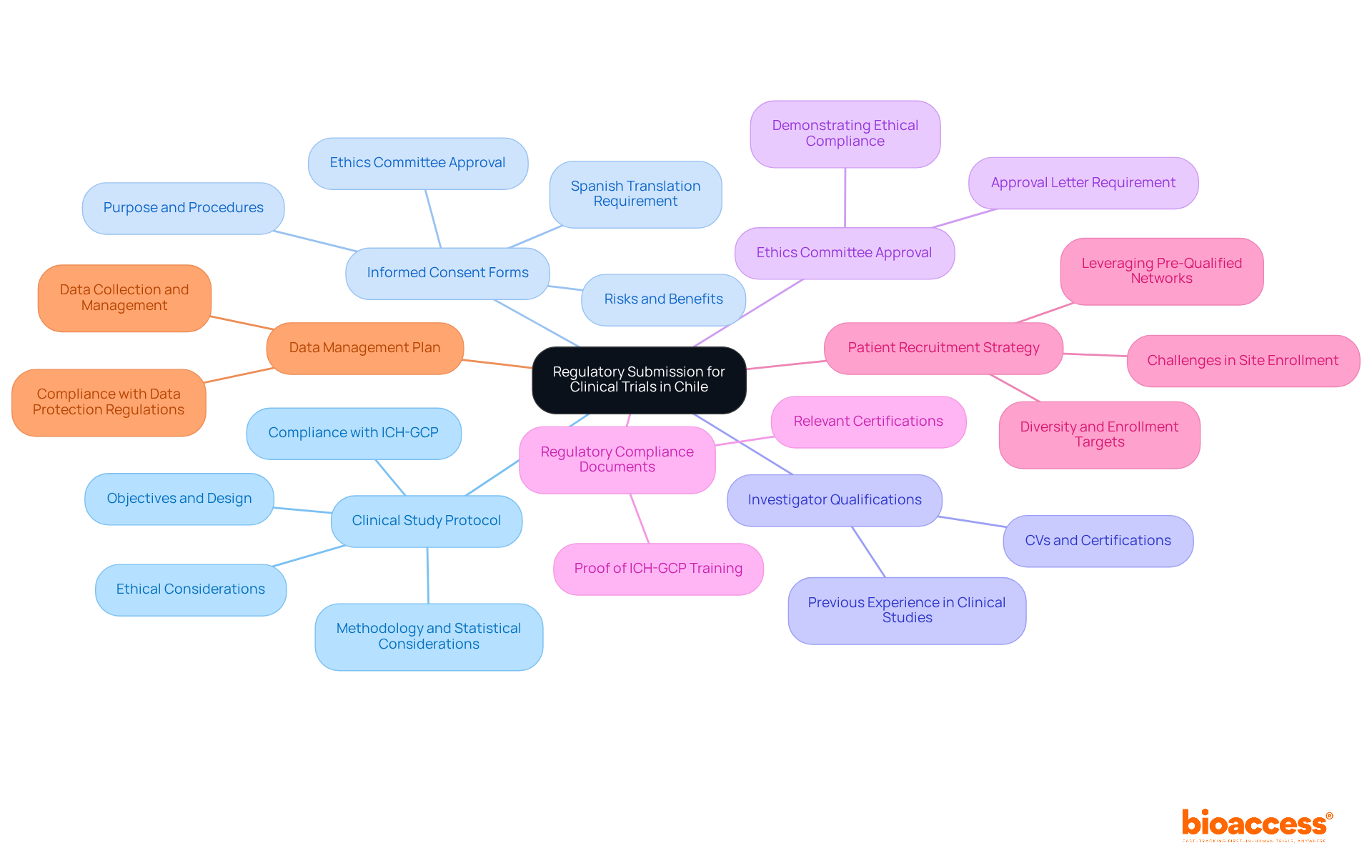
Navigating the regulatory submission Chile clinical trial landscape requires meticulous preparation and a clear understanding of essential documentation for clinical research success. To ensure a successful regulatory submission in Chile, it is crucial to compile the following essential documents:
- Clinical Study Protocol: This document must detail the study’s objectives, design, methodology, statistical considerations, and ethical considerations, adhering strictly to ICH-GCP standards. A well-structured protocol is vital for the regulatory submission Chile clinical trial and should reflect the specific requirements of the Chilean regulatory authority, the Instituto de Salud Pública (ISP).
- Informed Consent Forms: These forms must clearly articulate the study’s purpose, procedures, risks, and benefits to potential participants. They should be translated into Spanish and obtain approval from an accredited Ethics Committee prior to filing.
- Investigator Qualifications: Documentation demonstrating the qualifications and experience of the principal investigator and co-investigators is essential. This encompasses CVs, pertinent certifications, and evidence of previous experience in clinical studies, particularly in the MedTech or Biopharma fields.
- Before the regulatory submission Chile clinical trial, it is mandatory to obtain approval from an accredited Ethics Committee. This letter must be included in your regulatory submission Chile clinical trial package to demonstrate ethical compliance.
- Regulatory Compliance Documents: Include necessary documents that showcase compliance with local regulations, such as proof of ICH-GCP training for the research team and any other relevant certifications.
- Patient Recruitment Strategy: Outline your strategy for recruiting participants, detailing how you will ensure diversity and meet enrollment targets. This is especially crucial in Chile, where patient recruitment can greatly influence study timelines. Did you know that many trials face significant delays due to recruitment challenges, which can jeopardize timelines and outcomes? Notably, challenges in site enrollment have been identified as a leading cause for clinical trials being halted, with 55% of trials terminating early due to poor enrollment. Leveraging pre-qualified networks can enhance recruitment efficiency, potentially achieving 50% faster enrollment.
- Data Management Plan: Provide a comprehensive plan detailing how data will be collected, managed, and analyzed, ensuring compliance with Chilean data protection regulations.
Meticulous preparation of these documents enhances the likelihood of a smooth process and timely approval for the regulatory submission Chile clinical trial. Did you know that 50% of new drug applications get approved on the first try? While 73% were ultimately accepted after resubmissions, this underscores the importance of high-quality applications. Furthermore, involving biostatisticians early in the application process can significantly enhance the quality of your documentation and help prevent the median delay of 435 days linked to requests necessitating resubmissions. Moreover, employing bioaccess® data from studies can aid in FDA IDE and PMA processes, as the FDA accepts GCP-compliant information from outside the US, boosting your application’s credibility. Ultimately, the quality of your regulatory submission for the Chile clinical trial can determine the success of your clinical trial, making thorough preparation not just beneficial, but essential.
Execute the Regulatory Submission Process
Navigating the regulatory submission Chile clinical trial process can be daunting, especially for MedTech and Biopharma companies facing complex challenges. To successfully execute this process, follow these essential steps:
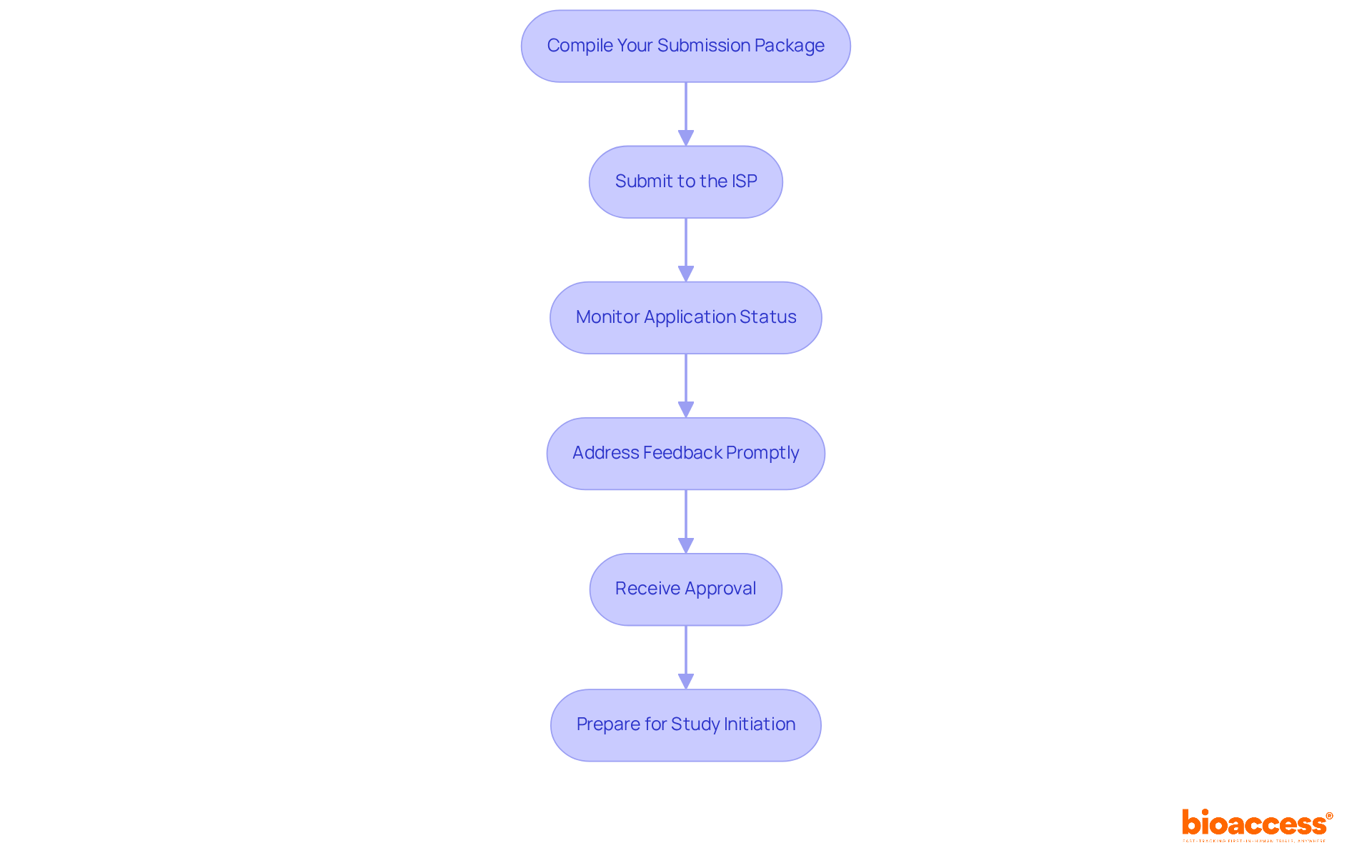
- Compile Your Submission Package: Gather all necessary documents, including clinical trial protocols, informed consent forms, and safety data. Ensure that each document adheres to the regulatory guidelines set by the Instituto de Salud Pública (ISP) to avoid any delays in the review process. A centralized document management system (DMS) can assist in maintaining version control and ensuring compliance throughout the application process.
- Submit to the ISP: Applications must be submitted electronically through the ISP’s online portal. Familiarize yourself with the specific guidelines provided by the ISP, as compliance with these requirements is essential for a smooth process. Keep in mind that depending on your product type, you might need to coordinate advisory committee reviews or inspections. Furthermore, understanding the pathways of ANVISA, INVIMA, and COFEPRIS can offer insights into broader market access strategies in Latin America.
- Monitor Application Status: After sending your application, maintain regular communication with the ISP to track your progress. Be prepared to respond quickly to any inquiries or requests for additional information from the governing authority, as this can greatly influence the approval timeline. Typically, a regulatory submission for a clinical trial in Chile may require between 30 to 90 days for approval, based on the intricacy of the study.
- Address Feedback Promptly: If the ISP requests modifications or additional documentation, respond quickly and comprehensively. Delays in addressing feedback can lead to extended approval timelines, which may impede your study’s progress. Common pitfalls include underestimating the time required for revisions and failing to provide complete documentation.
- Receive Approval: Upon approval, you will receive an official notification from the ISP. Keep this documentation organized for your records and for any subsequent steps in the research process.
- Prepare for Study Initiation: With approval obtained, commence preparations for study initiation. This includes site selection, patient recruitment, and training your research team to ensure compliance with ICH-GCP standards. Utilizing bioaccess®’s services can improve your study’s efficiency, as they offer access to pre-qualified research sites and swift patient recruitment capabilities, taking advantage of Chile’s favorable compliance environment and varied patient population.
By understanding these steps and potential pitfalls, you position your research for success in a competitive landscape.
Troubleshoot Common Submission Challenges
Even with meticulous planning, the regulatory submission Chile clinical trial process can present unexpected hurdles that require strategic solutions. Here are common issues and how to troubleshoot them:
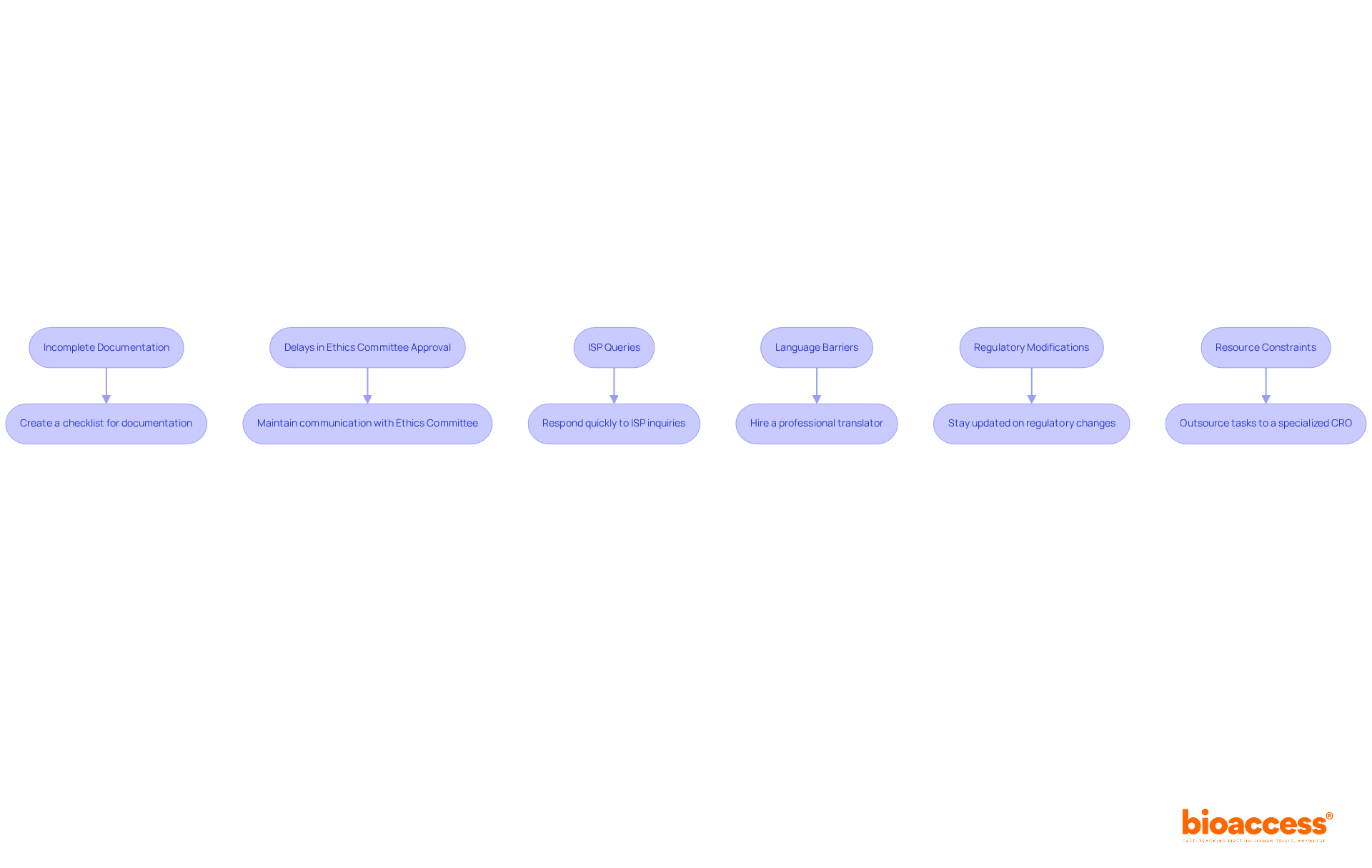
- Incomplete Documentation: One of the most frequent reasons for delays is incomplete documentation. To prevent this, create a checklist based on the necessary documents, including QMS certificates, research data, and labels in Spanish. Ensure each item is examined prior to submission. Taking this proactive step can really help cut down on delays.
- Delays in Ethics Committee Approval in Chile highlight the significant challenge of patient recruitment in clinical trials, as up to 80% of studies related to regulatory submission Chile clinical trial do not enroll on time due to insufficient patient participation. If the Ethics Committee takes longer than expected to review your application, keep the lines of communication open. This can help speed up their decision.
- ISP Queries: If the Instituto de Salud Pública (ISP) requests clarification or additional information, respond as quickly and thoroughly as possible. Delays in communication can prolong the regulatory submission Chile clinical trial approval process, so ensure that your team is prepared to efficiently address any inquiries.
- Language Barriers: Ensure that all documents are translated into Spanish, as required by the ISP. Consider hiring a professional translator with experience in research related to medical trials to avoid misinterpretations that could lead to compliance issues.
- Stay informed about any modifications in regulations or filing requirements related to regulatory submission Chile clinical trial. Regularly check the ISP’s website and subscribe to relevant newsletters to receive updates. For example, Law 14.874/24, enacted in May 2024, aims to streamline the evaluation process for studies in Brazil, which may also affect regional practices.
- Resource Constraints: If your team is overwhelmed, consider outsourcing certain tasks to a specialized CRO like bioaccess®, which can offer expertise in compliance processes and early feasibility study management. This can help ensure that your submission is handled efficiently and in compliance with ICH-GCP standards.
By anticipating these challenges and leveraging expert resources, you can navigate the regulatory landscape more effectively and accelerate your clinical research success.
Conclusion
Navigating the regulatory submission process in Chile is a critical step for MedTech and Biopharma companies aiming to introduce innovative therapies efficiently. Understanding the regulatory landscape and preparing the necessary documentation are crucial steps. When executed effectively, stakeholders can navigate the complexities of clinical trials with confidence and speed.
Key elements discussed include:
- The importance of familiarizing oneself with the Instituto de Salud Pública (ISP) guidelines
- Adhering to ICH-GCP standards
- Preparing comprehensive documentation such as clinical study protocols and informed consent forms
Navigating the submission process can be daunting, especially with challenges like incomplete documentation and Ethics Committee delays. Addressing these issues is crucial for ensuring timely submissions and successful study initiation. By leveraging resources like specialized contract research organizations, companies can enhance their regulatory compliance and streamline their processes.
In the end, the regulatory submission landscape in Chile offers unique advantages that can be leveraged for success, such as relatively swift approval timelines and a favorable environment for patient recruitment. By embracing these opportunities and implementing best practices in regulatory submissions, stakeholders can not only expedite their clinical trials but also contribute to transformative healthcare solutions.
Frequently Asked Questions
What is the main regulatory authority overseeing clinical trials in Chile?
The main regulatory authority is the Instituto de Salud Pública (ISP), which supervises research studies and oversees the approval of applications for clinical trials.
What legislation governs human research in Chile?
Key legislation includes Law No. 20.120, which specifically governs human research, and the Sanitary Code, which provides a general framework for health-related regulations.
What compliance standards must clinical trials in Chile adhere to?
Clinical trials must comply with the International Council for Harmonisation Good Clinical Practice (ICH-GCP) standards to ensure ethical conduct and the reliability of generated data.
What is the typical approval timeline for clinical studies in Chile?
The authorization process for clinical studies in Chile typically ranges from 30 to 90 days, allowing for a faster commencement of studies compared to other regions.
Is approval from an Ethics Committee required before submitting to the ISP?
Yes, studies must secure approval from an accredited Ethics Committee (Comité de Ética) before presenting their application to the ISP to ensure participant rights and welfare are protected.
How can stakeholders streamline their compliance efforts for clinical trials in Chile?
By mastering the essential components such as understanding regulatory authorities, relevant legislation, ICH-GCP compliance, and approval processes, stakeholders can expedite their clinical trials and leverage Chile’s strategic advantages for early-stage studies.
List of Sources
- Understand the Regulatory Landscape in Chile
- Trends in clinical trials performed in Chile (https://scielo.cl/article_plus.php?pid=S0034-98872021000100110&tlng=en&lng=es)
- Number of clinical trials registered in the Chilean Institute of Public… (https://researchgate.net/figure/Number-of-clinical-trials-registered-in-the-Chilean-Institute-of-Public-Health-ISP_tbl1_351504068)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOork5qcO2ZeE5015qChLM7L8UOh9biERdV2VzPLNncXEQn1BHlOj)
- 4 Best Practices for First in Human Biopharma Trials in Chile | bioaccess® (https://bioaccessla.com/blog/4-best-practices-for-first-in-human-biopharma-trials-in-chile)
- Clinical Cancer Research in South America and Potential Health Economic Impacts (https://mdpi.com/2227-9032/11/12/1753)
- Prepare Required Documentation for Submission
- Statistical Support for Regulatory Submissions | PharPoint (https://pharpoint.com/resources/statistical-support-regulatory-submissions-discussions)
- Recognizing Statistical Problems in Reports of Clinical Trials: a Readers’ Aid: Part 33 of a Series on Evaluation of Scientific Publications – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11741548)
- Statistical Challenges with Site Enrollment in Clinical Trials | Rho (https://rhoworld.com/statistical-challenges-with-site-enrollment-in-clinical-trials)
- Statistical Issues and Recommendations for Clinical Trials Conducted During the COVID-19 Pandemic (https://tandfonline.com/doi/full/10.1080/19466315.2020.1779122)
- Execute the Regulatory Submission Process
- Master Early Phase Clinical Trials in Chile: Key Strategies and Insights | bioaccess® (https://bioaccessla.com/blog/master-early-phase-clinical-trials-in-chile-key-strategies-and-insights)
- Navigate Clinical Trial Approval in Chile: 4 Essential Steps | bioaccess® (https://bioaccessla.com/blog/navigate-clinical-trial-approval-in-chile-4-essential-steps)
- A Helpful Guide To The Regulatory Submission Process (https://kivo.io/news/regulatory-submission-process)
- Master Phase 1 Clinical Trials in Chile: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/master-phase-1-clinical-trials-in-chile-a-step-by-step-guide)
- Troubleshoot Common Submission Challenges
- Common Challenges Faced in Clinical Trials in LATAM and How to Overcome Them (https://linkedin.com/pulse/common-challenges-faced-clinical-trials-latam-how-overcome-them-xknec)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- How Chile Is Shaping Medical Device Clinical Trials In Latin America (https://meddeviceonline.com/doc/how-chile-is-shaping-medical-device-clinical-trials-in-latin-america-0001)
- Common Pitfalls in Regulatory Submissions: Practical Tips to Avoid Delays or Rejections (https://linkedin.com/pulse/common-pitfalls-regulatory-submissions-practical-tips-naima-fouayzi–9obwe)
- Best Practices for Medical Device Regulatory Approval in Chile – cms.bioaccessla.com (https://cms.bioaccessla.com/blog/best-practices-for-medical-device-regulatory-approval-in-chile)

Leave a Reply