Introduction
For researchers in Chile, the path to obtaining ethics committee approval for clinical trials is fraught with challenges that can impact the success of their studies. This process ensures compliance with ethical standards and plays a crucial role in the success of clinical research in the region. However, navigating these complexities can lead to significant delays and frustration for researchers.
With varying timelines, documentation requirements, and regulatory nuances, how can organizations effectively streamline their submissions to avoid setbacks? By understanding the key steps and strategies for securing ethics committee approval, researchers can confidently navigate this complex landscape.
Understand the Ethics Committee Approval Process in Chile
In the realm of clinical research, obtaining ethics committee approval for Chile clinical trials is not just a formality; it’s a pivotal step that can determine the success of first-in-human studies. The Scientific Ethics Committee (CEC) serves as the primary body responsible for reviewing research protocols to ensure adherence to ethical standards. Here’s a detailed overview of the process:
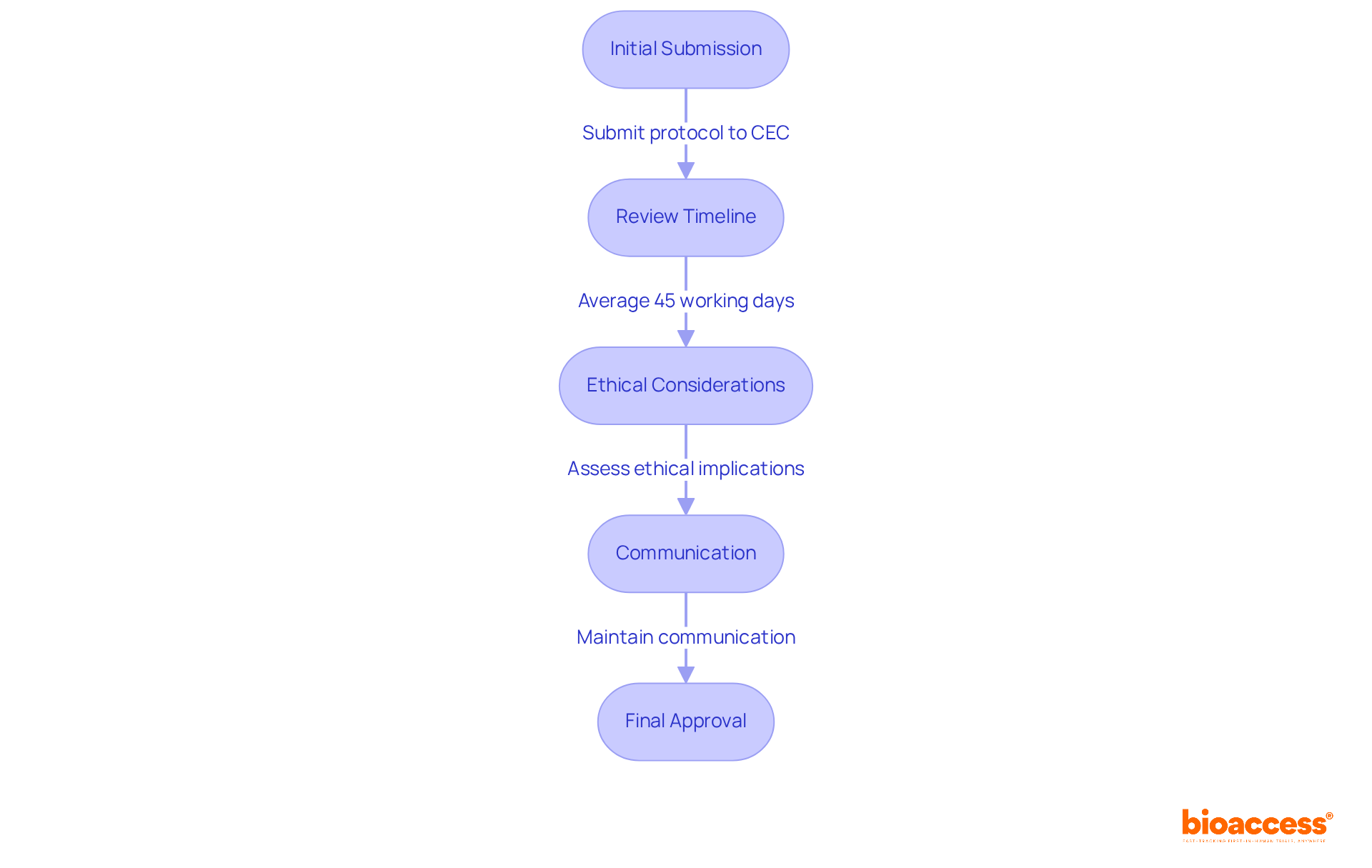
- Initial Submission: Researchers must submit their clinical study protocol to an accredited CEC. This submission should include comprehensive details about the study’s objectives, methodology, and potential risks to participants, ensuring clarity and thoroughness to facilitate the review.
- Review Timeline: The CEC is mandated to provide a favorable or unfavorable opinion within an average of 45 working days. Furthermore, the average duration from submission to ethics committee approval for clinical trials of medical devices in Chile is exceptionally efficient, typically spanning from one to one and a half months. Understanding these timelines is crucial for effective trial planning and scheduling, allowing researchers to align their project timelines accordingly. Utilizing bioaccess®’s expertise can further accelerate this process, as they enable swift ethics clearances and offer crucial insights into regulatory pathways.
- Ethical Considerations: The committee rigorously assesses the ethical implications of the study. They focus on participant safety, informed consent, and the scientific validity of the research. Understanding these criteria can greatly improve your chances of getting that all-important approval. Notably, Chile has achieved retention rates exceeding 85% in clinical trials, underscoring the effectiveness of its patient engagement strategies. Bioaccess® has successfully supported numerous innovators in navigating these ethical considerations, ensuring compliance with ICH-GCP standards and obtaining ethics committee approval for clinical trials in Chile as set forth by the Ministry of Health (MINSAL).
- Communication: Maintaining open lines of dialogue with the CEC is essential for expediting the review. Researchers should be prepared to address any questions or concerns that may arise during the evaluation, demonstrating responsiveness and commitment to ethical standards. The thorough accreditation of research ethics committees (RECs) by MINSAL guarantees high safety and ethical standards, further enhancing the credibility of the ethics committee system. With bioaccess®’s Global Trial Accelerators™, organizations gain vital insights into market access strategies and regulatory updates. This streamlines communication with the CEC.
By effectively navigating this framework, organizations can utilize the strong regulatory environment and efficient timelines for approvals, positioning themselves favorably in the competitive landscape of clinical studies in Latin America. Conducting clinical studies in this region can be up to 30% less costly than in North America or Europe, making it an appealing choice for organizations seeking budget-friendly solutions.
Identify Regulatory Requirements and Key Agencies
Navigating the ethics committee approval process for clinical trials in Chile requires a thorough understanding of the regulatory landscape and the key agencies involved:
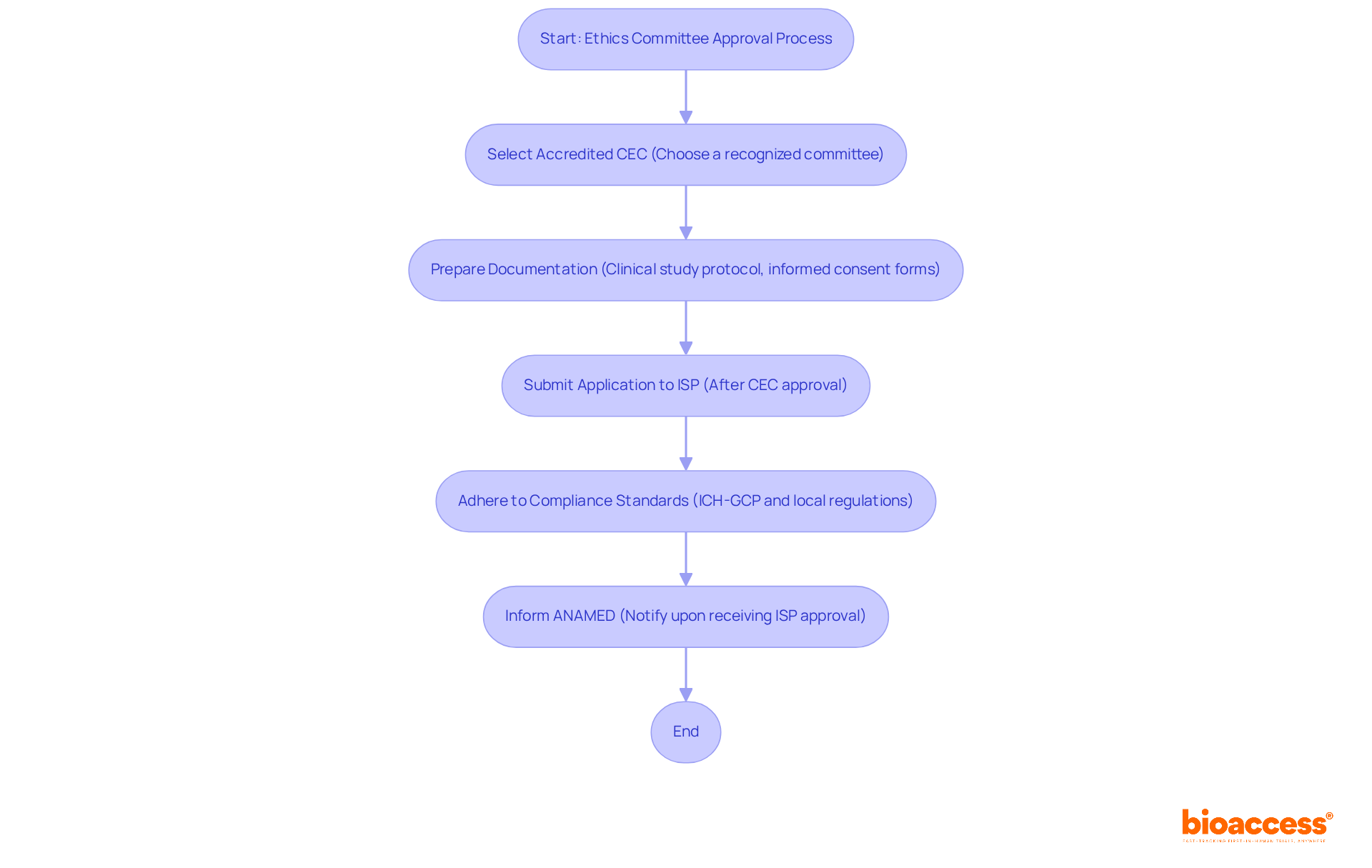
- Instituto de Salud Pública (ISP): The ISP serves as the primary regulatory authority for clinical trials in Chile. All applications must be submitted to the ISP following ethics committee approval for the Chile clinical trial. The ISP typically evaluates applications within 30 business days, providing a clear expectation for the timeline of authorization.
- Scientific Ethics Committees (CECs): These committees are responsible for reviewing and approving research protocols. It’s essential to select a CEC that is accredited and recognized by the ISP to ensure compliance with ethical standards. Delays often stem from inadequate informed consent documentation, which can hinder progress and must be meticulously prepared and translated into Spanish.
- Documentation Requirements: Understand the necessary documentation for submission. This includes the clinical study protocol, informed consent forms, and the qualifications of the investigators. All documents must be accurately translated into Spanish to meet local requirements.
- Compliance Standards: Adherence to ICH-GCP guidelines and local regulations established by the ISP is crucial. By grasping these standards, researchers can navigate the approval process more effectively, minimizing delays. Furthermore, upon receiving ISP consent, researchers must swiftly inform the Agencia Nacional de Medicamentos (ANAMED) regarding the ethics committee approval for their clinical trial in Chile.
By adhering to these steps and maintaining a comprehensive understanding of the regulatory framework, researchers can improve their chances of obtaining timely ethics committee approval for their clinical trials in Chile, while also considering the socio-political context that may influence the ethics committee approval Chile clinical trial operations.
Prepare and Submit Required Documentation
Obtaining ethics committee approval for a clinical trial in Chile is a critical step in the clinical research process, and meticulous preparation is key to success. Follow these steps to ensure your submission is complete and compliant:
-
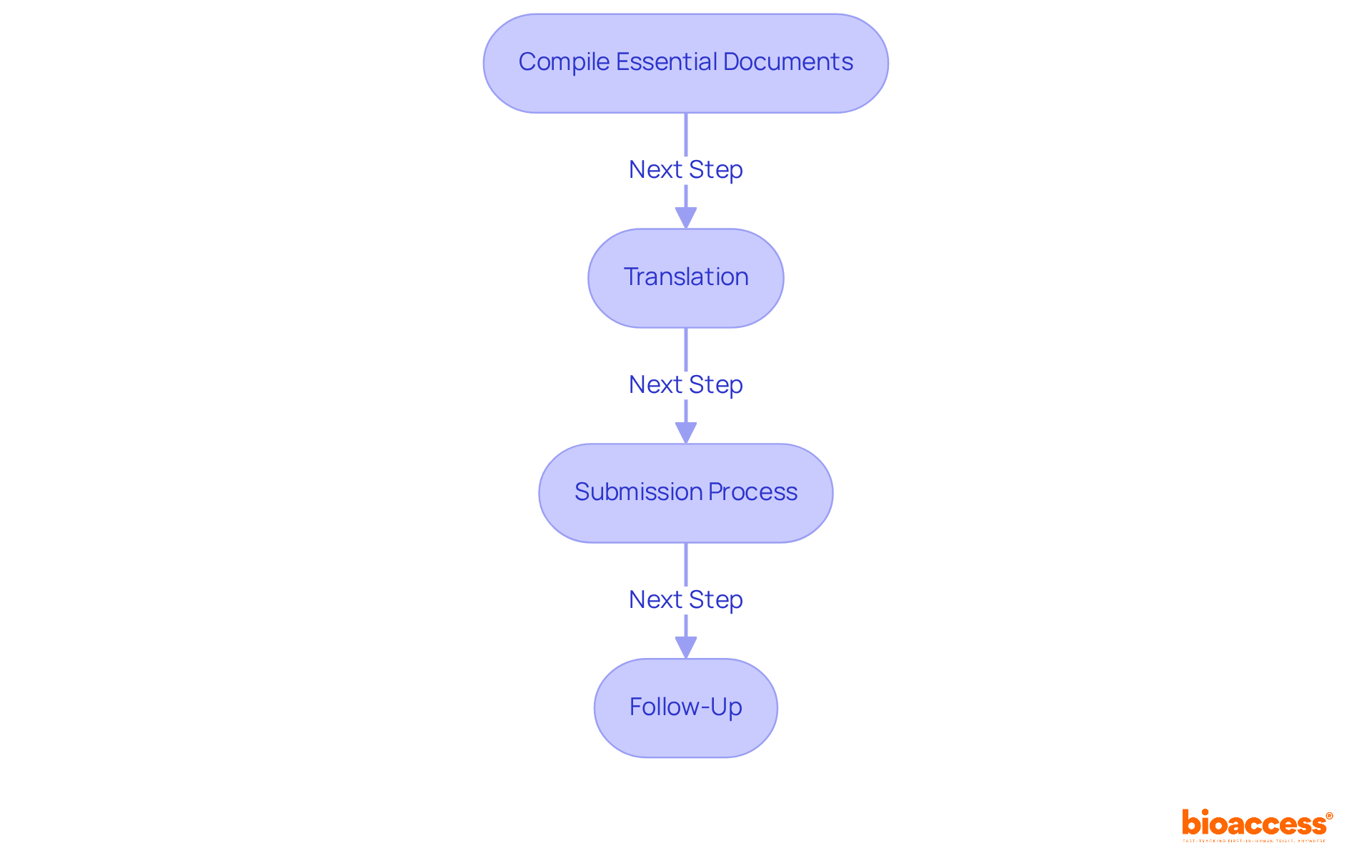
Compile Essential Documents: Gather all necessary documents, including:
- Clinical study protocol
- Informed consent forms
- Investigator’s curriculum vitae
- Any previous ethical approvals (if applicable)
-
Translation: Ensure that all documents are accurately translated into Spanish, as this is a mandatory requirement for submission to the Comité Ético Científico (CEC) and the Instituto de Salud Pública (ISP). Accurate translation is crucial, as it ensures that all regulatory requirements are met and facilitates clear communication with the review committees.
-
Submission Process: Submit your compiled documents to the chosen CEC for review. Adhere to their specific submission guidelines, which may vary between committees. Navigating the submission process can be complex and time-consuming, often leading to delays if not handled correctly. The ethics committee approval for clinical trials in Chile typically lasts around 4-8 weeks, as the ISP generally reviews applications within this timeframe. By leveraging bioaccess®’s expertise, you can streamline your submission process and potentially reduce approval times.
-
Follow-Up: After submission, maintain proactive communication with the CEC to address any questions or requests for additional information promptly. By maintaining proactive communication, how might you expedite the review process and showcase your dedication to ethical standards? As highlighted by Julio G. Martinez-Clark, CEO of bioaccess®, “Mastering the clinical trial approval system in this region necessitates a comprehensive grasp of the regulatory framework and requirements, particularly when navigating the intricacies of FIH and early feasibility studies.
Navigate the Review Process and Address Challenges
Navigating the ethics committee approval in Chile for clinical trial can be a daunting task, but with the right strategies, it becomes manageable. Here are essential steps to effectively manage the review process and address potential challenges:
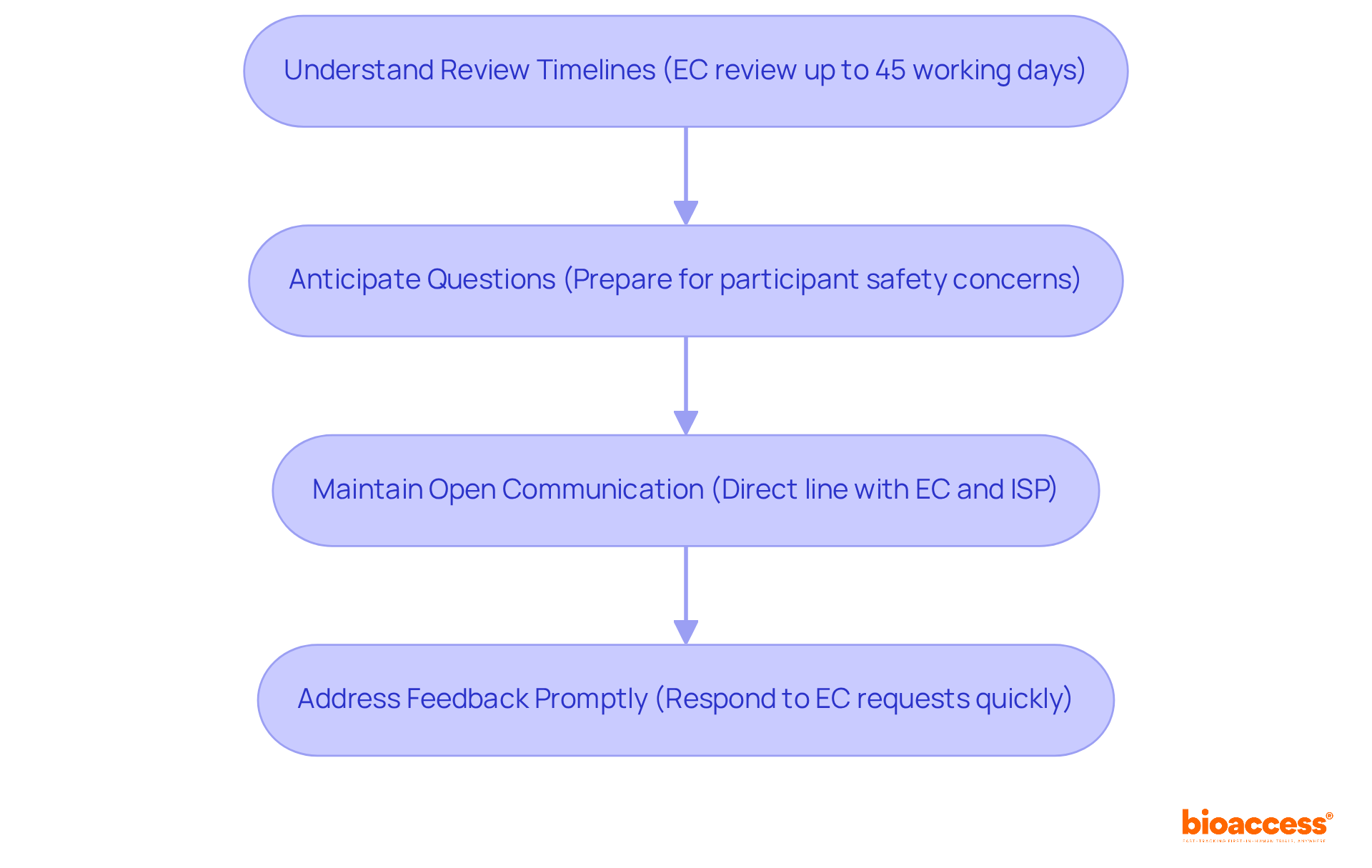
- Understand Review Timelines: The Ethical-Scientific Evaluation Committee (EC) typically has up to 45 working days to review your submission. Make sure to plan your project timeline to fit this review period, ensuring that you remain on track for your clinical trial objectives.
- Anticipate Questions: Familiarize yourself with common concerns related to participant safety and ethical considerations. Preparing for potential questions or requests for clarification from the EC can streamline the review and enhance your submission’s quality. What challenges do you foresee in addressing these concerns?
- Maintain Open Communication: Establishing a direct line of communication with both the EC and the Instituto de Salud Pública (ISP) is vital. This proactive strategy enables faster responses to inquiries and assists in resolving issues as they emerge, ultimately speeding up the authorization timeline.
- Address Feedback Promptly: If the EC requests modifications or additional information, respond promptly and thoroughly. Exhibiting your dedication to ethical standards is crucial as it can significantly accelerate the timeline for ethics committee approval for a clinical trial in Chile. How quickly can you implement feedback to keep your project on track?
By mastering these steps, you not only enhance your chances of approval but also contribute to the integrity of clinical research in Chile.
Conclusion
Navigating the ethics committee approval process in Chile is a pivotal challenge that can determine the success of clinical trials, particularly for first-in-human studies. By understanding the intricacies of this approval process, researchers can effectively navigate the regulatory landscape and position themselves for success in a competitive environment.
This article outlines a structured approach to obtaining ethics committee approval, emphasizing the importance of thorough documentation, clear communication, and adherence to regulatory requirements. Key steps include:
- Submitting a comprehensive clinical study protocol
- Understanding review timelines
- Maintaining proactive engagement with the Scientific Ethics Committees (CECs) and the Instituto de Salud Pública (ISP)
Moreover, the significance of ethical considerations and compliance with ICH-GCP standards cannot be overstated, as they enhance the likelihood of approval.
Securing timely ethics committee approval in Chile streamlines the clinical trial process and demonstrates a strong commitment to ethical research practices. Organizations looking to conduct clinical studies in Latin America must master the ethics committee approval process to leverage advantages like cost efficiency and rapid patient recruitment. Mastering these strategies ensures that clinical trials not only proceed smoothly but also uphold the highest standards of research integrity in the region.
Frequently Asked Questions
What is the role of the Scientific Ethics Committee (CEC) in Chile for clinical trials?
The CEC is responsible for reviewing research protocols in clinical trials to ensure adherence to ethical standards, focusing on participant safety, informed consent, and the scientific validity of the research.
What is the process for obtaining ethics committee approval in Chile?
Researchers must submit their clinical study protocol to an accredited CEC, including comprehensive details about the study’s objectives, methodology, and potential risks to participants.
How long does the ethics committee review process typically take in Chile?
The CEC is required to provide a favorable or unfavorable opinion within an average of 45 working days. The typical duration from submission to approval for clinical trials of medical devices is about one to one and a half months.
What ethical considerations does the CEC assess during the review?
The committee evaluates participant safety, informed consent processes, and the scientific validity of the proposed research to ensure compliance with ethical standards.
How can researchers improve their chances of obtaining ethics committee approval?
Researchers can enhance their chances by thoroughly addressing ethical implications in their submissions and maintaining open communication with the CEC to respond to any questions or concerns.
What are the benefits of conducting clinical studies in Chile?
Conducting clinical studies in Chile can be up to 30% less costly than in North America or Europe, and Chile has a strong regulatory environment with efficient approval timelines, making it a strategic choice for early-stage clinical trials.
How does Bioaccess® assist researchers in navigating the ethics committee approval process?
Bioaccess® provides expertise in accelerating the ethics clearance process, ensuring compliance with ICH-GCP standards, and offering insights into regulatory pathways to facilitate communication with the CEC.
What is the significance of patient engagement in Chile’s clinical trials?
Chile has achieved retention rates exceeding 85% in clinical trials, indicating effective patient engagement strategies that contribute to successful study outcomes.
List of Sources
- Understand the Ethics Committee Approval Process in Chile
- How Chile Is Shaping Medical Device Clinical Trials In Latin America (https://meddeviceonline.com/doc/how-chile-is-shaping-medical-device-clinical-trials-in-latin-america-0001)
- Clinical Trials in Latin America (https://languageconnections.com/clinical-trials-in-latin-america)
- Navigate Clinical Trial Approval in Chile: 4 Essential Steps | bioaccess® (https://bioaccessla.com/blog/navigate-clinical-trial-approval-in-chile-4-essential-steps)
- Identify Regulatory Requirements and Key Agencies
- Navigate Clinical Trial Approval in Chile: 4 Essential Steps | bioaccess® (https://bioaccessla.com/blog/navigate-clinical-trial-approval-in-chile-4-essential-steps)
- Trends in clinical trials performed in Chile (https://scielo.cl/article_plus.php?pid=S0034-98872021000100110&tlng=en&lng=es)
- Navigate Biopharma Clinical Trials in Chile: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/navigate-biopharma-clinical-trials-in-chile-a-step-by-step-guide)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Prepare and Submit Required Documentation
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- National medical malpractice data reveals a persistent trend in clinical documentation errors (https://rmf.harvard.edu/News-and-Blog/Press-Releases-Home/Press-Releases/2024/November/2024-Candello-report-For-the-Record)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Navigate the Review Process and Address Challenges
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- How the Scientific Ethical Committees work in Chile (https://linkedin.com/pulse/how-scientific-ethical-committees-work-chile-bopalcro-sigce)
- Conducting First-in-Human Clinical Trials in Chile: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/conducting-first-in-human-clinical-trials-in-chile-a-step-by-step-guide)

Leave a Reply