Introduction
Startups in the MedTech and Biopharma sectors face daunting challenges in clinical research, particularly in the ever-evolving landscape of Chile. CROs like bioaccess® are essential allies, providing specialized services that simplify the clinical trial process and help ensure compliance with local regulations. However, with the rapid growth of the CRO market and the increasing demand for efficient trial management, understanding how to effectively collaborate with these organizations could be the key to unlocking success in this competitive arena.
Define Contract Research Organization (CRO)
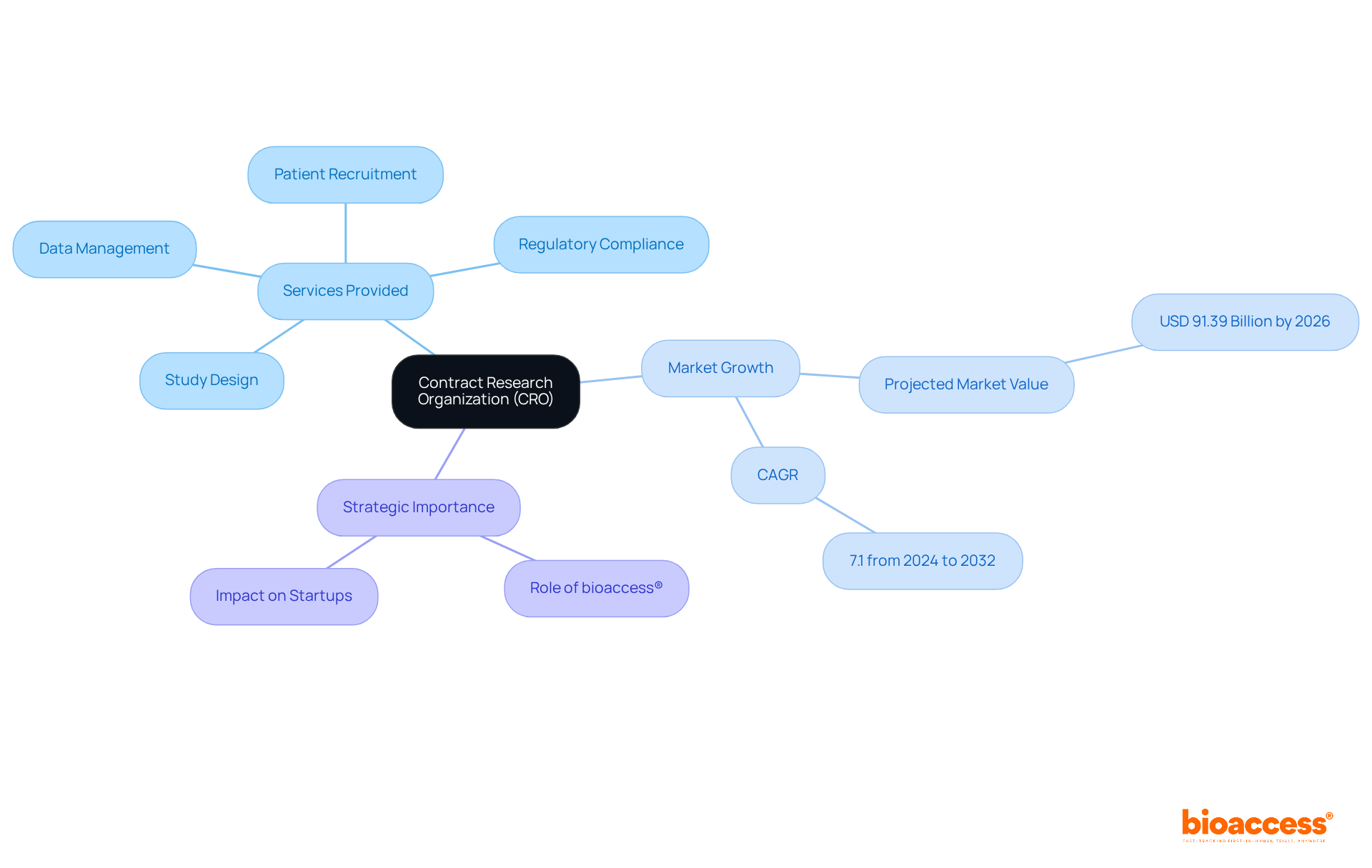
Navigating the complexities of clinical research can be daunting for many startups, but Contract Research Organizations (CROs) like bioaccess® offer essential support. A CRO is a specialized service provider that delivers outsourced assistance for research studies to pharmaceutical, biotechnology, and medical device companies. In Chile, a contract research organization, like bioaccess®, is vital in facilitating clinical development. They oversee key aspects of clinical studies, such as:
- Study design
- Regulatory compliance
- Patient recruitment
- Data management
Bioaccess® leverages local expertise and resources to help sponsors navigate the complex compliance landscape, ensuring adherence to guidelines established by the Instituto de Salud Pública (ISP) and other relevant authorities.
Contract research organizations in Chile are crucial for streamlining the approval process, especially for startups seeking to conduct first-in-human (FIH) trials efficiently and cost-effectively. With a focus on ICH-GCP compliance and FDA-bridgeable data acceptance, bioaccess® can significantly reduce approval timelines, often achieving regulatory clearance in as little as 15 to 30 calendar days in Panama and 30 to 90 days in Chile. This swift turnaround is essential for startups that need to generate human trial data rapidly to secure funding and advance their products. For instance, Axoft successfully utilized bioaccess®’s LATAM pathway to achieve their first four human implants in record time, showcasing the effectiveness of this approach.
Looking ahead to 2026, the contract research organization Chile environment features an increasing number of specialized organizations that cater to the distinct requirements of early-stage studies. The healthcare CRO market is projected to reach USD 91.39 billion by 2026, with a compound annual growth rate (CAGR) of 7.1% from 2024 to 2032. These contract research organizations not only deliver vital services but also cultivate cooperative partnerships that improve study management and patient recruitment initiatives. By leveraging the advantageous regulatory framework and the varied patient demographic in Chile, bioaccess® serves as a contract research organization in Chile and is positioned as a strategic ally for firms aiming to accelerate their development processes. With bioaccess® as a partner, startups can not only meet regulatory demands but also accelerate their innovations, ultimately transforming patient care in Chile and beyond.
Explore Services Offered by CROs in Clinical Trials
In the rapidly evolving landscape of clinical research, the contract research organization Chile, such as bioaccess®, is pivotal in addressing the unique challenges faced by MedTech, Biopharma, and Radiopharma companies. They offer a comprehensive range of services tailored to the distinct needs of these sectors, significantly enhancing the clinical research process, particularly for radiopharmaceutical and theranostic studies involving Lu-177, Ac-225, and Ga-68. Key offerings include:
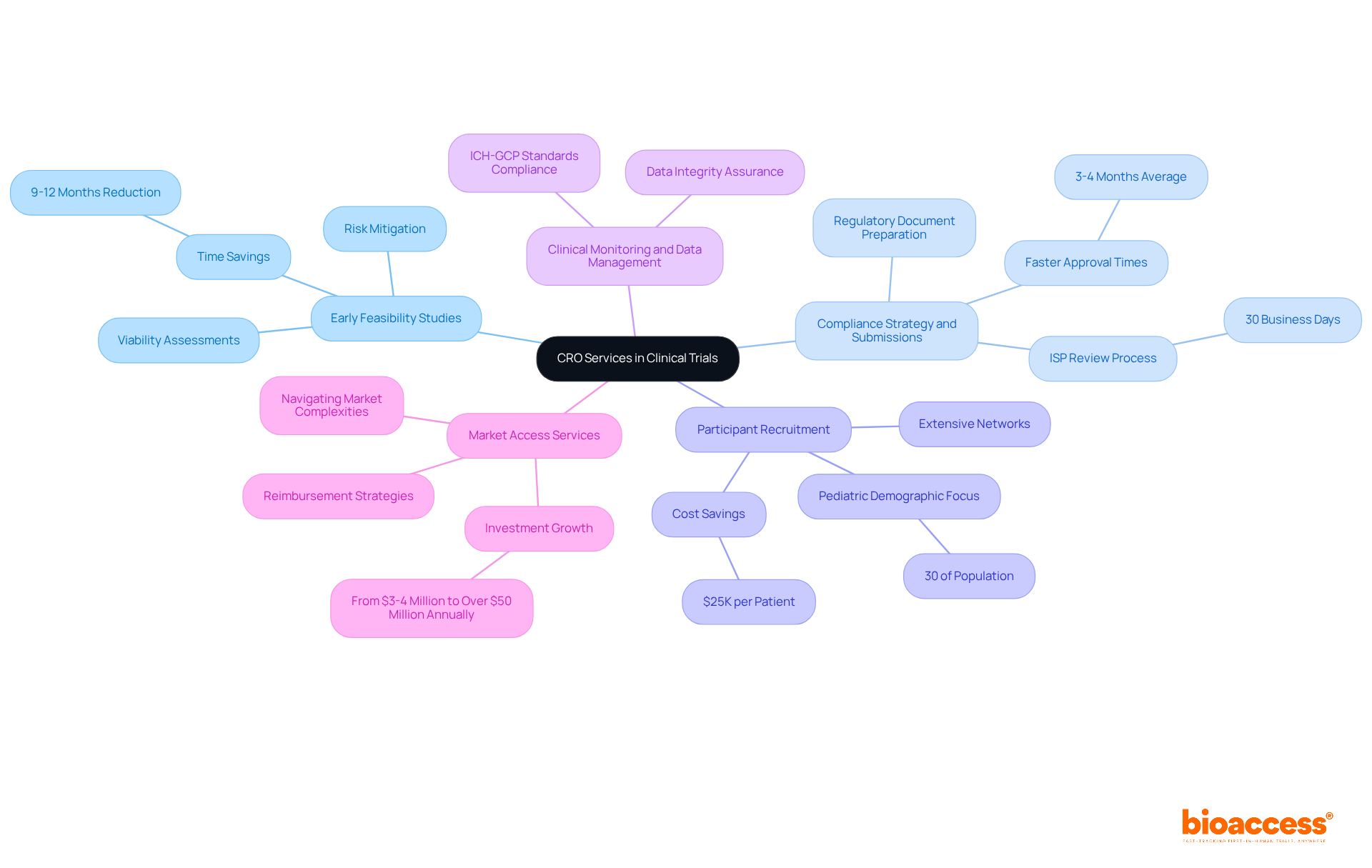
- Early Feasibility Studies (EFS): These critical assessments evaluate the viability of medical devices or drugs prior to larger trials. This enables sponsors to make informed decisions and mitigate risks early in the development process. Companies like GlySure have reported significant time savings, indicating reductions in development time by 9 to 12 months due to accelerated approvals.
- Compliance Strategy and Submissions: In Chile, contract research organizations play a crucial role in preparing and submitting compliance documents to the Instituto de Salud Pública (ISP), ensuring adherence to local regulations. The ISP typically reviews applications within 30 business days. With optimized procedures, the average time for regulatory approval in Chile is approximately 3-4 months, significantly faster than in many other regions.
- Participant Recruitment: Utilizing extensive networks, specialized organizations excel in attracting individuals for clinical studies, which is crucial for achieving enrollment goals and schedules. This capability is particularly important in Chile, where the pediatric demographic constitutes 30% of the population, presenting unique opportunities for targeted studies. By utilizing pre-negotiated site contracts, contract research organizations in Chile can save $25K per patient.
- Clinical Monitoring and Data Management: CROs take charge of operations, making sure they stick to ICH-GCP standards while handling data collection and analysis. This meticulous oversight is crucial for maintaining the integrity of the study and ensuring that the data generated is of regulatory-grade quality.
- Market Access Services: These services support companies in navigating the complexities of entering the Chilean market, including reimbursement strategies and compliance pathways. With the growing investment in research studies in Latin America, which has risen from $3-4 million to over $50 million annually in the Andean region, comprehending market access is crucial for successful commercialization.
By leveraging bioaccess®’s expertise, companies can not only streamline their clinical trials but also enhance their chances of success in a competitive market.
Understand the Role of CROs in Chile’s Regulatory Landscape
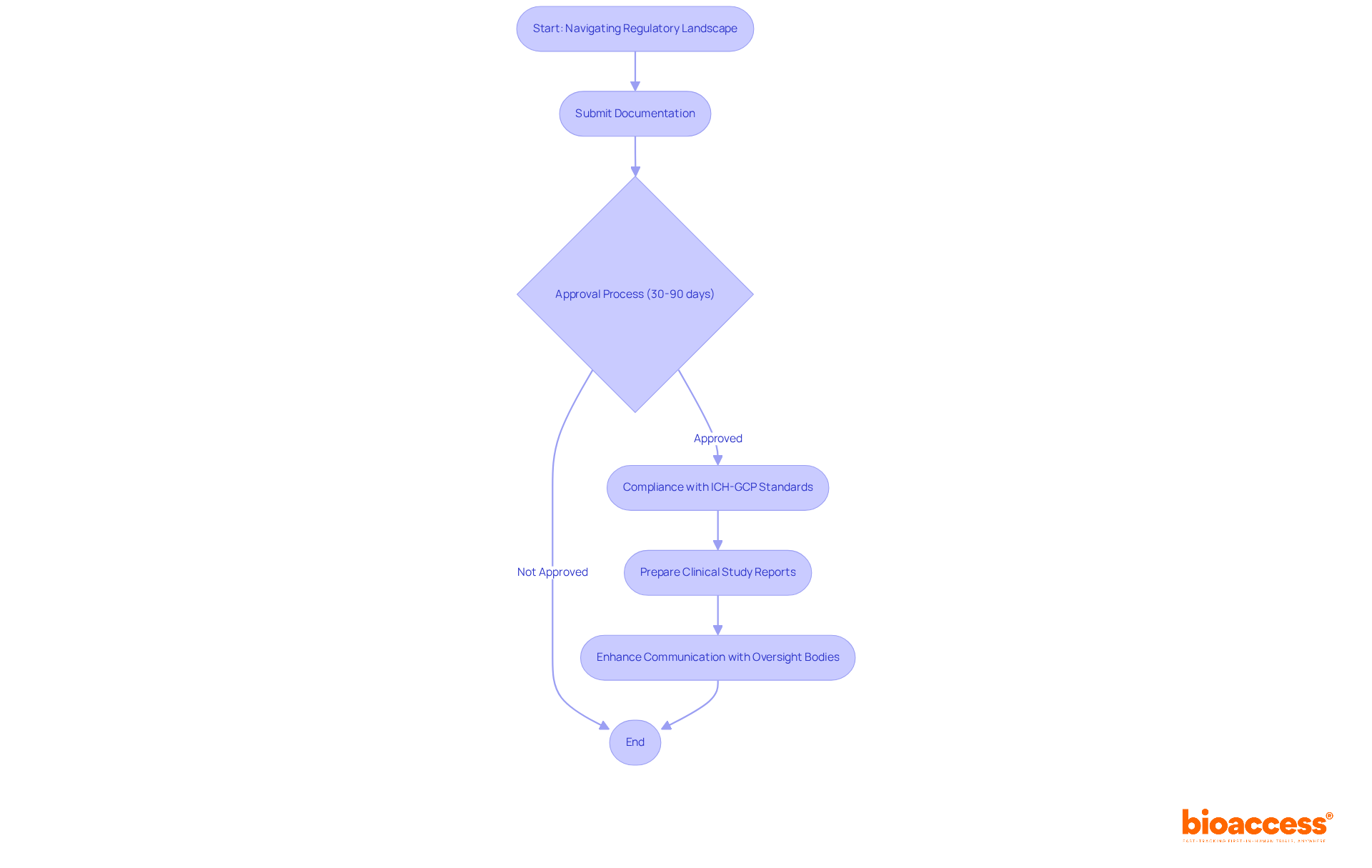
Navigating the regulatory landscape in Chile can be daunting, especially for startups without dedicated resources. Contract research organization Chile, like bioaccess®, plays a crucial role in ensuring that medical studies comply with local regulations overseen by the Instituto de Salud Pública (ISP). They facilitate the submission of necessary documentation for approval, with the typical approval timeframe for trials in Chile ranging from 30 to 90 days, depending on the study’s complexity and the completeness of the submission.
Bioaccess® assists sponsors in navigating these submissions by guiding them through compliance requirements, including adherence to ICH-GCP standards and the preparation of clinical study reports, particularly for a contract research organization in Chile. Furthermore, bioaccess® enhances communication with oversight bodies, aiding in accelerating the approval process and reducing potential delays. This expertise is a game-changer for startups that often lack the resources to navigate these regulatory complexities on their own.
Take Axoft and Newrotex, for instance; they’ve successfully utilized bioaccess®’s LATAM pathway to secure rapid approvals and first-in-human evaluations, highlighting the strategic advantage of conducting early-stage studies in Latin America.
Assess the Impact of CRO Partnerships on Clinical Trial Success
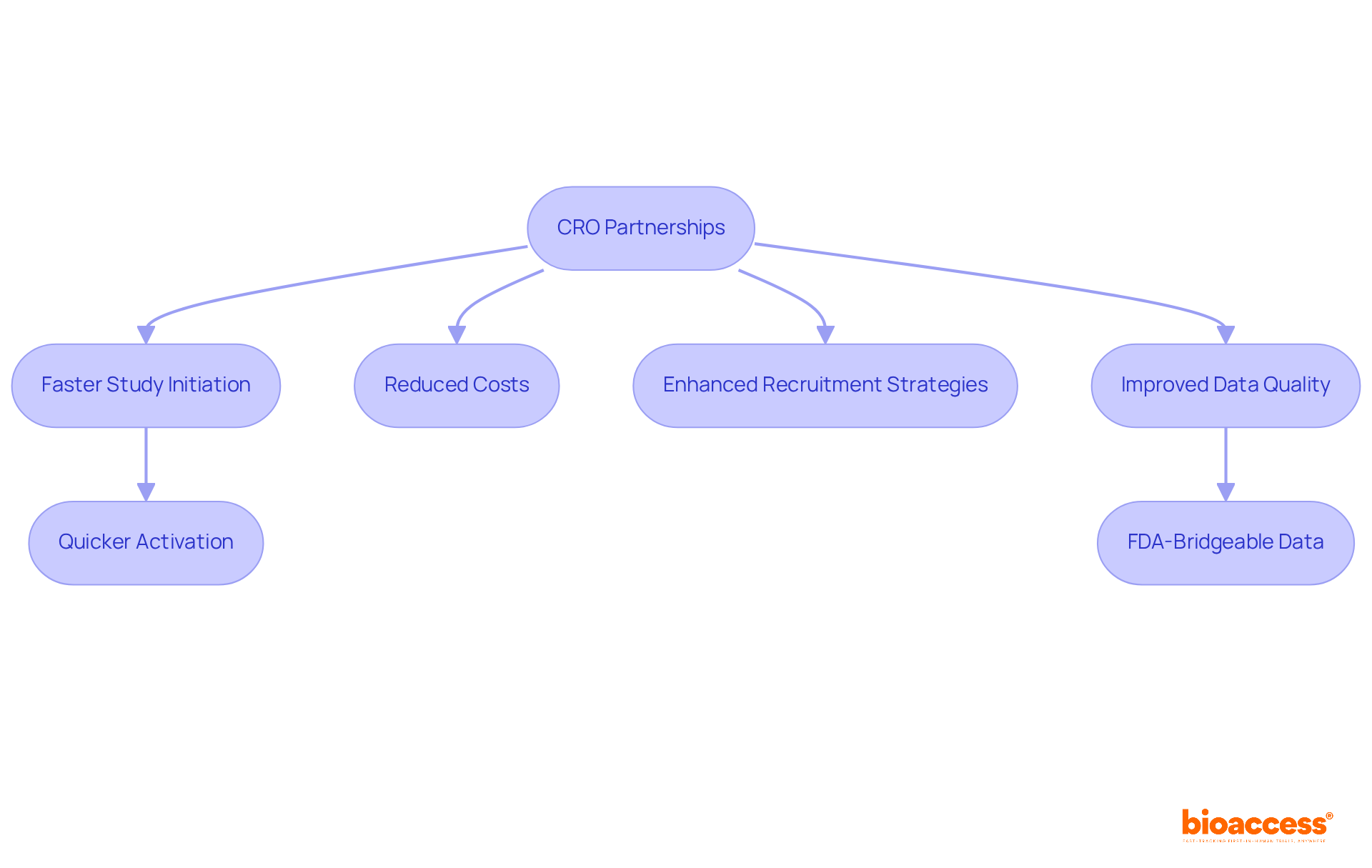
In the competitive landscape of clinical research in Chile, the importance of contract research organizations (CROs) is more critical than ever. Utilizing the specialized knowledge and resources of CROs allows sponsors to speed up study initiation and execution. This leads to faster market access. For instance, bioaccess® has demonstrated that its integrated model-merging U.S. regulatory foundations with execution in Latin America-results in an impressive 40% quicker activation and 30% reduced costs compared to conventional U.S. or EU studies. What if those delays could be minimized? This effectiveness is vital, particularly considering that delays in medical investigations can cost sponsors roughly $800,000 daily in lost income.
Furthermore, organizations like bioaccess® offer invaluable insights into local patient populations, enhancing recruitment strategies and ensuring that trials accurately reflect the target demographic. This partnership speeds up development and boosts data quality, which is essential for securing funding and approvals. Startups that engage with a contract research organization in Chile are therefore better positioned to reach their first-in-human milestones and advance their programs effectively.
In Chile, the regulatory framework, guided by the Instituto de Salud Pública (ISP), provides a streamlined pathway for trials conducted by a contract research organization. With approval timelines often ranging from 30 to 90 days, the advantages of CRO partnerships become even more pronounced. By ensuring compliance with ICH-GCP standards and providing FDA-bridgeable data, bioaccess® exemplifies how strategic CRO collaborations can lead to successful clinical outcomes and foster innovation in the MedTech and Biopharma sectors. The right collaborations can not only save time and money but also redefine the future of clinical research in the region.
Conclusion
Startups in clinical research face daunting challenges that can hinder their progress. Navigating the complexities of clinical research in Chile is significantly enhanced by the vital role played by Contract Research Organizations (CROs) like bioaccess®. These specialized entities provide essential support to pharmaceutical, biotechnology, and medical device companies, streamlining the clinical development process and ensuring compliance with local regulations. CROs help startups navigate the complexities of study design, patient recruitment, and data management, making clinical trials faster and more efficient, which is crucial for bringing innovative solutions to market.
What strategic advantages do CROs offer, especially for early-stage studies? Key insights from the article highlight that with expedited approval timelines ranging from 30 to 90 days, CROs not only help in meeting regulatory demands but also enhance the quality of data collected. This is instrumental in securing funding and advancing product development. Companies like Axoft have successfully leveraged these partnerships to achieve remarkable results, showcasing the effectiveness of CROs in navigating the regulatory landscape and accelerating trial processes.
CROs are not just operational support; they are key players reshaping clinical research in Latin America. The significance of CROs in Chile extends beyond mere operational support; they are pivotal in transforming the clinical research landscape in Latin America. As the market for healthcare CRO services continues to grow, stakeholders are urged to consider the immense benefits of partnering with specialized organizations. By doing so, they can capitalize on the favorable regulatory environment and diverse patient demographics, ultimately driving innovation and improving patient care. Partnering with CROs could be the key to unlocking the full potential of clinical trials in Chile.
Frequently Asked Questions
What is a Contract Research Organization (CRO)?
A Contract Research Organization (CRO) is a specialized service provider that offers outsourced support for clinical research studies to pharmaceutical, biotechnology, and medical device companies.
What services do CROs like bioaccess® provide?
CROs like bioaccess® provide essential services including study design, regulatory compliance, patient recruitment, and data management for clinical studies.
How does bioaccess® assist startups in Chile with clinical development?
Bioaccess® helps startups navigate the complex compliance landscape in Chile, ensuring adherence to guidelines established by the Instituto de Salud Pública (ISP) and other relevant authorities, which facilitates clinical development.
What are the benefits of using a CRO for first-in-human (FIH) trials?
CROs streamline the approval process for FIH trials, allowing startups to conduct studies efficiently and cost-effectively while generating human trial data rapidly to secure funding.
What is the typical timeline for regulatory clearance when working with bioaccess®?
Bioaccess® can achieve regulatory clearance in as little as 15 to 30 calendar days in Panama and 30 to 90 days in Chile, significantly reducing approval timelines for clinical trials.
How does bioaccess® ensure compliance with regulatory requirements?
Bioaccess® focuses on ICH-GCP compliance and FDA-bridgeable data acceptance, which helps ensure that studies meet necessary regulatory standards.
What is the projected growth of the healthcare CRO market in Latin America?
The healthcare CRO market is projected to reach USD 91.39 billion by 2026, with a compound annual growth rate (CAGR) of 7.1% from 2024 to 2032.
How do CROs like bioaccess® improve study management and patient recruitment?
CROs cultivate cooperative partnerships that enhance study management and patient recruitment initiatives, leveraging the advantageous regulatory framework and diverse patient demographics in Chile.
Can you provide an example of a successful collaboration with bioaccess®?
Axoft successfully utilized bioaccess®’s LATAM pathway to achieve their first four human implants in record time, demonstrating the effectiveness of this approach for startups.
What advantages does Latin America offer for early-stage clinical trials?
Latin America provides speed, cost efficiency, and favorable regulatory pathways for early-stage clinical trials, making it an attractive region for MedTech, Biopharma, and Radiopharmaceutical companies.
List of Sources
- Define Contract Research Organization (CRO)
- Healthcare Contract Research Organization Market Share 2032 (https://polarismarketresearch.com/industry-analysis/healthcare-contract-research-organization-market)
- Contract Research Organization Services Market Report 2026-2031, By Type, Therapeutic Area, and Geo (https://marketsandmarkets.com/Market-Reports/contract-research-organization-service-market-167410116.html)
- Contract Research Organisations: Market and Trends | Novotech CRO (https://novotech-cro.com/faq/contract-research-organisations-market-and-trends)
- Contract Research Organization Market Forecast, 2026-2033 (https://coherentmarketinsights.com/industry-reports/contract-research-organization-market)
- Explore Services Offered by CROs in Clinical Trials
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Clinical Trials in Latin America (https://languageconnections.com/clinical-trials-in-latin-america)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Clinical Trials Quotes (2 quotes) (https://goodreads.com/quotes/tag/clinical-trials)
- Quotes About Medical Research (https://changingthepresent.org/pages/quotes-about-medical-research)
- Understand the Role of CROs in Chile’s Regulatory Landscape
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Clinical Trials in Latin America (https://languageconnections.com/clinical-trials-in-latin-america)
- Clinical Trials in Latin America: How CROs in Chile are Driving Innovation in 2023 – BOPAL (https://bopalcro.com/clinical-trials-in-latin-america-how-cros-in-chile-are-driving-innovation-in-2023)
- Assess the Impact of CRO Partnerships on Clinical Trial Success
- Critical CRO Selection Affects Trial Timeline and Revenue | Rubix LS posted on the topic | LinkedIn (https://linkedin.com/posts/rubixls_key-considerations-for-selecting-the-right-activity-7443276905430118401-Tv4E)
- Worldwide Clinical Trials’ Annual Survey Reveals 2018 Trends in CRO/Sponsor Partnerships (https://worldwide.com/newsroom/worldwide-clinical-trials-annual-survey-reveals-2018-trends-cro-sponsor-partnerships)
- Clinical Trial Roles: Sponsor, CRO, and Site Responsibilities | IntuitionLabs (https://intuitionlabs.ai/articles/sponsor-cro-site-roles)
- CRO Quotes About the Benefits of CRO (https://rich-page.com/cro/cro-beat-seo-with-great-tweetable-cro-quotes)

Leave a Reply