Introduction
First-in-Human (FIH) trials represent a pivotal moment in medical innovation, bridging the gap between theory and practice. With Latin America’s unique regulatory advantages and cost efficiencies, conducting FIH studies in this region presents a golden opportunity for MedTech and Biopharma companies aiming to accelerate their market entry.
Many organizations struggle to adapt to the regulatory landscape and operational challenges inherent in FIH trials. How can they tap into local expertise to turn these challenges into opportunities?
This article explores essential steps and strategic insights from bioaccess Chile, guiding companies in transforming potential hurdles into milestones of success.
Define First-in-Human Trials and Their Importance
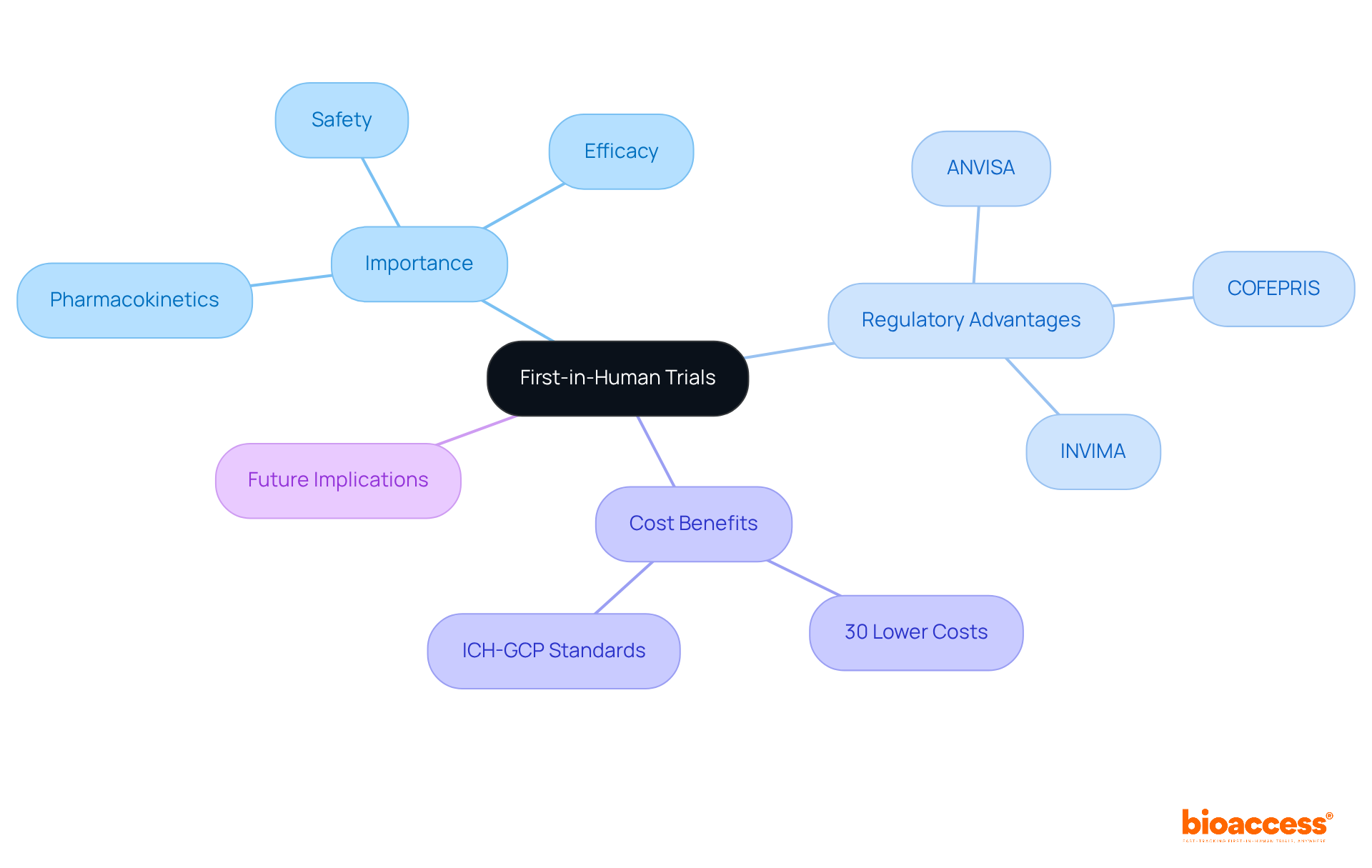
First-in-Human (FIH) studies represent a pivotal moment in clinical research, where the safety and efficacy of new medical devices and pharmaceuticals are put to the test on human subjects for the first time. These evaluations are essential for assessing the safety, tolerability, and pharmacokinetics of investigational products. They play a crucial role in bridging the gap between preclinical studies and later phases, allowing researchers to collect vital data on how humans respond to new therapies.
In Latin America, the implementation of FIH studies is particularly beneficial due to efficient regulatory pathways and cost savings. Regulatory authorities such as ANVISA in Brazil, COFEPRIS in Mexico, and INVIMA in Colombia facilitate faster approval timelines, often allowing for regulatory decisions within 30 to 90 days. This quick approval process is a game-changer for startups looking to secure funding and move their projects forward without burning through resources.
Additionally, the expense of executing FIH studies in Latin America can be considerably reduced – up to 30% lower than in conventional markets – while adhering to ICH-GCP standards and ensuring acceptance of FDA-compatible information. This blend of speed and cost-effectiveness establishes Latin America as a strategic center for early-stage studies, allowing innovators to introduce their products to the market more rapidly and effectively.
As clinical research evolves, how can FIH studies meet the growing demand for timely and reliable data that supports regulatory approvals and market access? By leveraging the advantages of FIH studies in Latin America, companies can not only enhance their chances of success but also redefine the future of clinical research.
Explore bioaccess Chile’s Methodology for FIH Trials
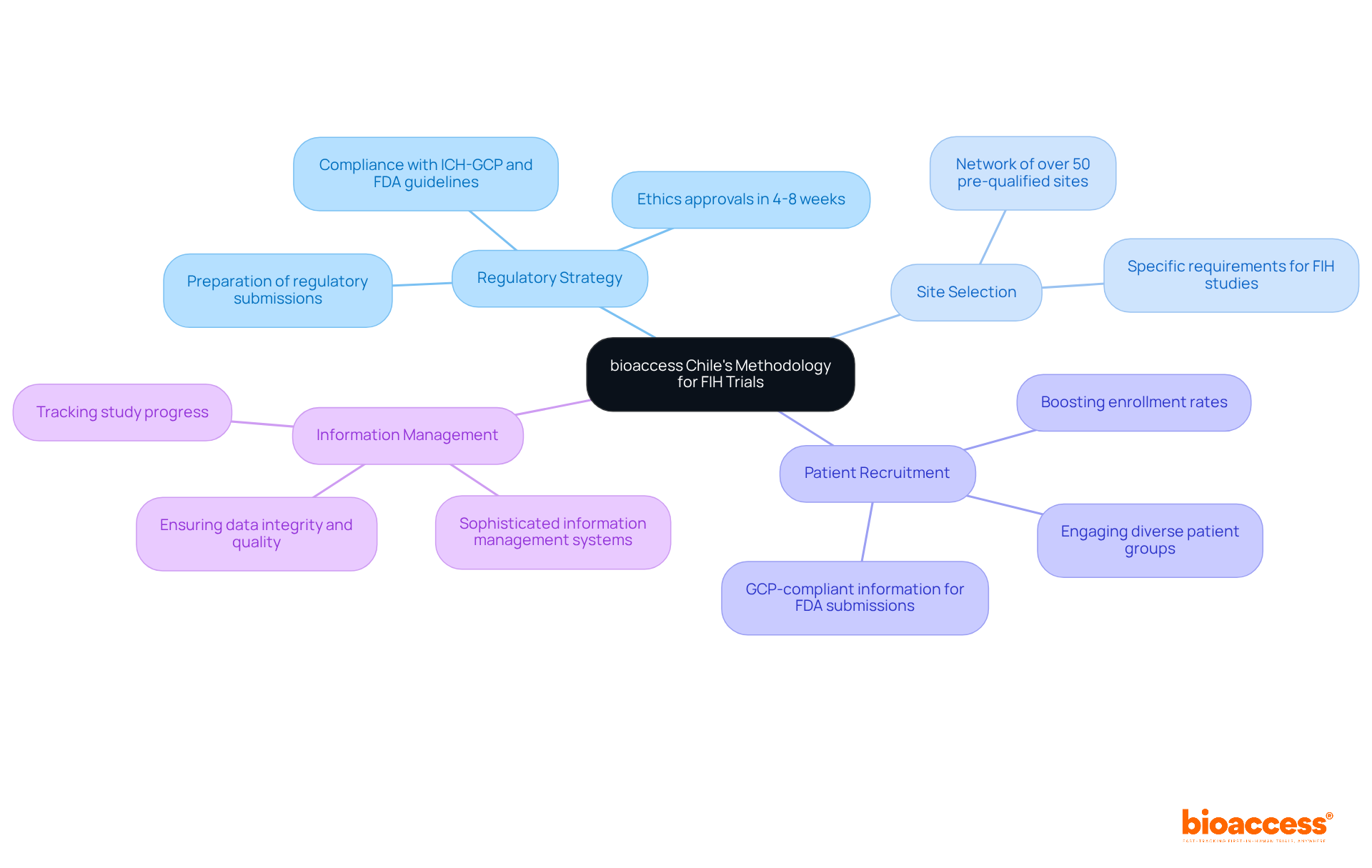
Navigating the complexities of first-in-human studies can be daunting, but a streamlined approach from bioaccess Chile sets it apart in the Medtech landscape. bioaccess utilizes a comprehensive methodology for conducting FIH studies that includes several key components:
- Regulatory Strategy: bioaccess ensures that all studies comply with local regulations and international standards, including ICH-GCP and FDA guidelines. This involves thorough preparation of regulatory submissions to authorities like ANVISA in Brazil and INVIMA in Colombia, which typically see ethics approvals in just 4-8 weeks – significantly faster than the 6+ months often required in the US and EU.
- Site Selection: The organization employs a network of over 50 pre-qualified clinical study sites across Latin America, ensuring that each location is equipped to meet the specific requirements of FIH studies.
- Patient Recruitment: At bioaccess Chile, we focus on engaging diverse patient groups to boost enrollment rates and ensure our studies truly reflect the wider population. This method not only speeds up patient access but also utilizes GCP-compliant information for FDA submissions.
- Information Management: The company utilizes sophisticated information management systems to track study progress and ensure integrity, enabling prompt reporting and analysis. This structured approach accelerates assessments and enhances data quality, ensuring fairness and paving the way for more R&D investments.
Additionally, bioaccess Chile supports early feasibility studies across Colombia, Brazil, and Mexico, further de-risking the development of medical devices. By choosing bioaccess, you’re not just ensuring compliance; you’re investing in a faster, more efficient pathway to market for your innovations.
Implement Key Steps for Successful FIH Trial Execution
To ensure the success of first-in-human (FIH) trials, sponsors must navigate a series of critical steps that can significantly impact their outcomes:
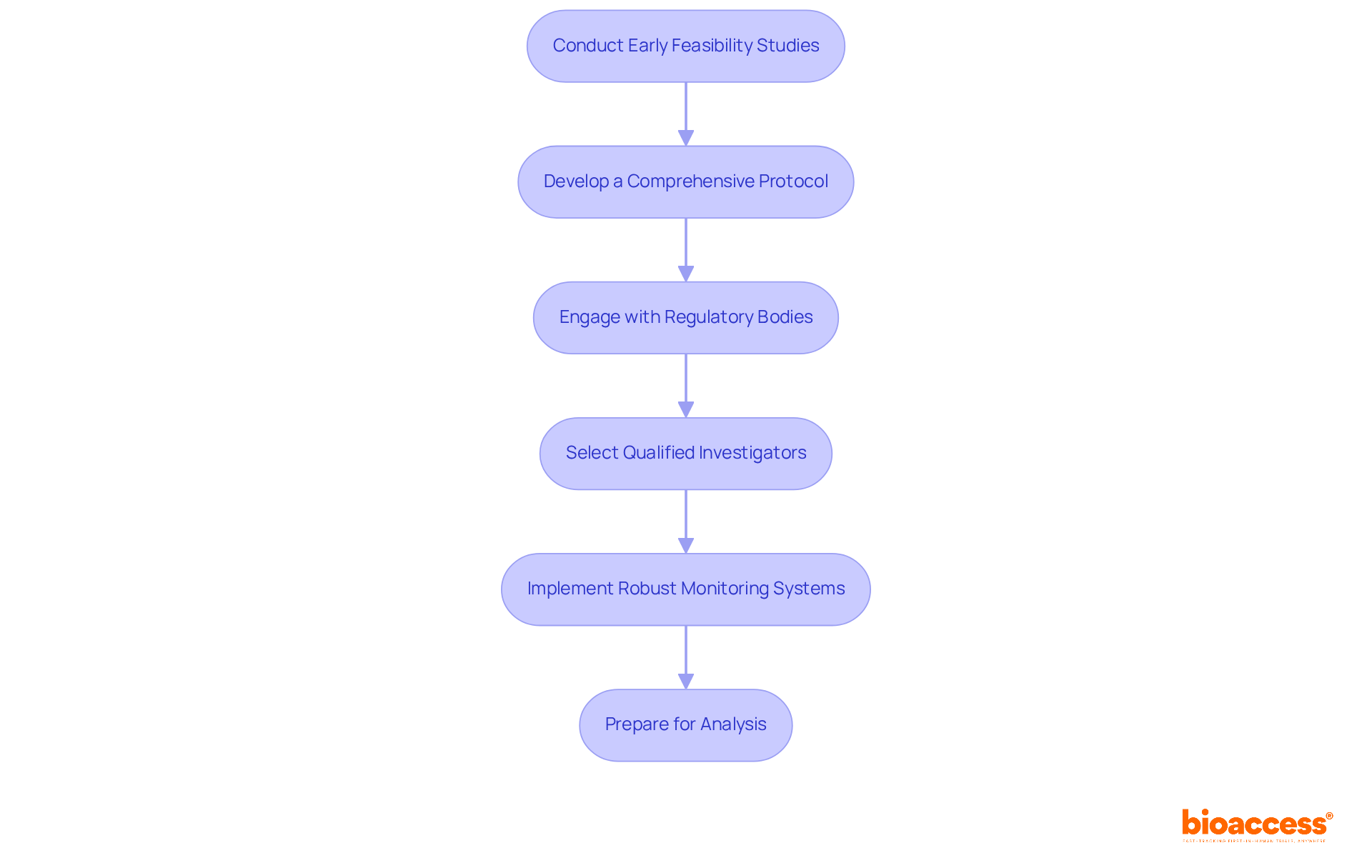
- Conduct Early Feasibility Studies (EFS): Initiate EFS to evaluate the investigational product’s viability in a clinical setting. This step is key because it helps spot potential challenges early and improves the study design. With bioaccess chile, sponsors can utilize our expertise in executing EFS across Colombia, Brazil, and Mexico, ensuring that studies are designed with local insights in mind.
- Develop a Comprehensive Protocol: Create a detailed study protocol that clearly outlines objectives, methodologies, and compliance requirements. Have you ever submitted a protocol to regulatory authorities like ANVISA or INVIMA? It’s crucial to ensure compliance with ICH-GCP standards. Bioaccess Chile facilitates this process, helping sponsors navigate the submission pathways efficiently.
- Engage with Regulatory Bodies: Maintain proactive communication with regulatory authorities like ANVISA and INVIMA to ensure all requirements are met. This engagement facilitates a smoother approval process and helps navigate the regulatory landscape effectively. With bioaccess chile, sponsors benefit from our established relationships with these regulatory bodies, expediting the approval timelines to as little as 4-8 weeks.
- Select Qualified Investigators: Choose experienced investigators who are well-versed in FIH studies. Their knowledge is essential for overseeing patient safety and maintaining information integrity throughout the study. Bioaccess Chile assists in identifying and recruiting qualified investigators who comprehend the intricacies of conducting studies in Latin America.
- Implement Robust Monitoring Systems: Establish comprehensive monitoring systems to track patient safety, data collection, and compliance with the protocol. This oversight is vital for ensuring the quality and reliability of study results. Bioaccess Chile offers robust monitoring solutions that are customized to the specific needs of first-in-human studies.
- Prepare for Analysis: Plan for analysis from the outset, ensuring that statistical methods align with the study design and objectives. This foresight improves the capacity to draw meaningful conclusions from the experimental results. Bioaccess Chile supports sponsors in the development of data analysis plans that comply with regulatory expectations.
By adhering to these steps, sponsors not only streamline their processes but also enhance their chances of successful outcomes. The strategic benefits of conducting FIH studies in Latin America encompass quicker regulatory approval timelines, cost-effectiveness, and enhanced patient recruitment, establishing it as an optimal region for early-stage research. Ultimately, the right approach can transform the challenges of FIH trials into opportunities for groundbreaking advancements in clinical research.
Overcome Common Challenges in FIH Trials with bioaccess Solutions
Navigating the complexities of first-in-human (FIH) studies presents significant hurdles that can impede clinical progress. Common challenges in these studies include:
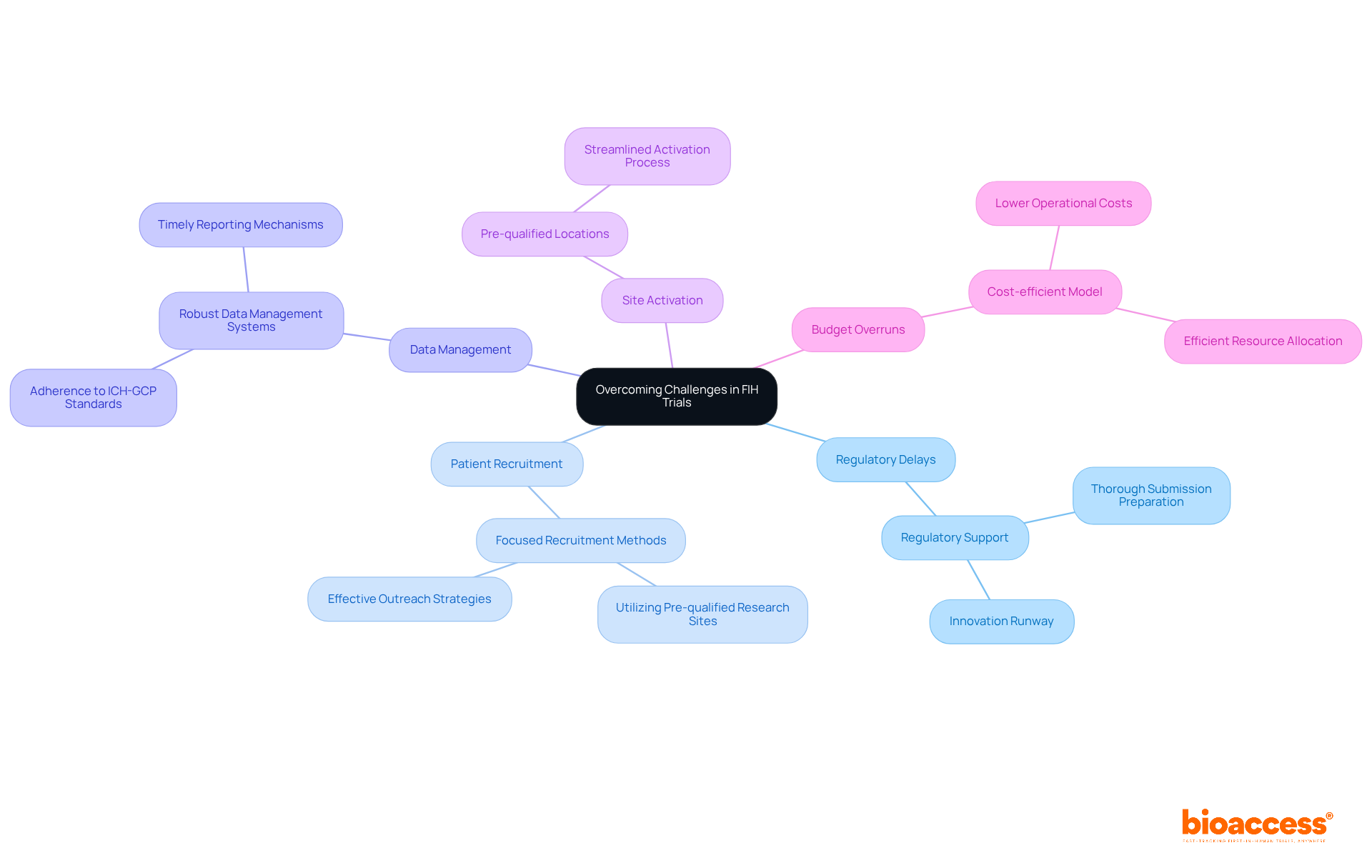
- To tackle regulatory delays, bioaccess chile provides thorough regulatory support, ensuring that all submissions meet the stringent requirements of authorities such as ANVISA and COFEPRIS. This proactive approach can significantly decrease approval timelines, often obtaining regulatory clearance in as little as 30 to 90 days, thanks to our Innovation Runway, specifically designed for FIH studies.
- Patient Recruitment Challenges: Have you ever considered that 41% of U.S. adults are unaware of trials? Bioaccess Chile utilizes focused recruitment methods and capitalizes on its vast network of over 50 pre-qualified research sites throughout Latin America to boost patient enrollment. A notable 85% of participants were uninformed about participation options, emphasizing the necessity for effective outreach. Our approach has successfully accelerated recruitment, allowing startups to reach clinical milestones faster.
- Implementing robust data management systems that adhere to ICH-GCP standards allows bioaccess chile to maintain data integrity and facilitate timely reporting, reducing the risk of data-related issues.
- Site Activation Delays: By utilizing pre-qualified locations and streamlining the site activation process, bioaccess chile ensures that studies can commence quickly, minimizing delays.
- Budget overruns can be managed effectively with bioaccess Chile’s cost-efficient model, which includes lower operational costs in Latin America while achieving high-quality results. Additionally, recruitment delays can cost sponsors between $600,000 and $8 million per day, emphasizing the importance of efficient patient recruitment strategies.
These challenges can result in significant financial implications and extended timelines for clinical trials. By tackling these challenges head-on, bioaccess chile enables sponsors to concentrate on what truly matters and achieve successful trial outcomes. Client testimonials from our partners highlight the effectiveness of our strategies in overcoming these challenges, reinforcing our commitment to accelerating clinical development in Latin America.
Conclusion
Successfully navigating First-in-Human trials is not just a necessity; it’s a strategic imperative for companies aiming to lead in medical innovation. Bioaccess Chile stands at the forefront of this endeavor, offering unique advantages that Latin America provides, including expedited regulatory processes and cost efficiencies. These factors allow companies to accelerate their journey from concept to clinical application, ensuring that their groundbreaking therapies reach the market more swiftly.
The article highlights several key aspects essential for successful FIH trials:
- Early feasibility studies
- Comprehensive protocol development
- Proactive engagement with regulatory bodies
- Selection of qualified investigators
Bioaccess Chile’s robust methodologies, including effective patient recruitment strategies and advanced data management systems, address common challenges that can hinder progress in clinical research. Achieving regulatory approvals in as little as 30 to 90 days underscores the compelling advantages of conducting trials in Latin America.
In conclusion, embracing the opportunities presented by FIH trials in Latin America not only enhances the chances of successful outcomes but also fosters innovation within the MedTech and Biopharma sectors. By collaborating with specialized organizations like Bioaccess Chile, sponsors can navigate the complexities of clinical research with confidence. Successfully navigating these trials can position companies as leaders in medical innovation, paving the way for future breakthroughs. The path to medical innovation is paved with the successful execution of FIH trials, and those who seize this opportunity will redefine the future of healthcare.
Frequently Asked Questions
What are First-in-Human (FIH) trials?
First-in-Human (FIH) trials are clinical studies that evaluate the safety and efficacy of new medical devices and pharmaceuticals on human subjects for the first time. These trials are crucial for assessing safety, tolerability, and pharmacokinetics of investigational products.
Why are FIH trials important in clinical research?
FIH trials are essential for bridging the gap between preclinical studies and later phases of clinical research. They allow researchers to gather vital data on human responses to new therapies, which is critical for advancing medical innovations.
How do regulatory authorities in Latin America facilitate FIH trials?
Regulatory authorities such as ANVISA in Brazil, COFEPRIS in Mexico, and INVIMA in Colombia provide efficient regulatory pathways that enable faster approval timelines, often achieving regulatory decisions within 30 to 90 days.
What are the cost advantages of conducting FIH trials in Latin America?
The cost of executing FIH trials in Latin America can be significantly lower-up to 30% compared to conventional markets-while still adhering to ICH-GCP standards and ensuring FDA-compatible information.
How does the speed of FIH trials in Latin America benefit startups?
The quick approval process in Latin America allows startups to secure funding and advance their projects more rapidly without depleting their resources, making it a strategic advantage for early-stage clinical trials.
What compliance standards are necessary for FIH trials in Latin America?
FIH trials in Latin America must comply with ICH-GCP standards, ensuring that the trials meet international quality and ethical guidelines while also facilitating acceptance of data by the FDA.
How can companies leverage FIH studies in Latin America for market access?
By taking advantage of the speed, cost efficiency, and favorable regulatory pathways in Latin America, companies can enhance their chances of success in obtaining timely and reliable data that supports regulatory approvals and market access.
List of Sources
- Define First-in-Human Trials and Their Importance
- Medical Research Quotes (22 quotes) (https://goodreads.com/quotes/tag/medical-research)
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)
- Beyond the Safety Check: Why First-in-Human Trials Demand a New Approach in 2026 | BioPharm International (https://biopharminternational.com/view/safety-first-human-trials-new-approach)
- Quotes About Medical Research (https://changingthepresent.org/pages/quotes-about-medical-research)
- Clinical trials started by trial phase globally 2018-2024| Statista (https://statista.com/statistics/1249010/clinical-trials-started-by-trial-phase-worldwide?srsltid=AfmBOoqkpLJT831A9DYrCeeIu0Ns5GcxVKUfLGwppVa0RGTrtv9Rd3aa)
- Explore bioaccess Chile’s Methodology for FIH Trials
- Best Practices For First-In-Human Clinical Trials In Brazil | bioaccess® (https://bioaccessla.com/blog/best-practices-for-first-in-human-clinical-trials-in-brazil)
- Latin America Clinical Trials Boom: $4.62B in 2025, $7.94B by 2034 (https://linkedin.com/pulse/latin-america-clinical-trials-boom-462b-2025-794b-2034-iaqhc)
- Latin America Clinical Trials Market to Apex USD 9.17 Bn by 2035 (https://towardshealthcare.com/insights/latin-america-clinical-trials-market-sizing)
- Latin America Clinical Trials Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trials-market/latin-america)
- Implement Key Steps for Successful FIH Trial Execution
- Clinical Trials Quotes (2 quotes) (https://goodreads.com/quotes/tag/clinical-trials)
- Early Feasibility Studies for Cardiovascular Devices in the United States: JACC State-of-the-Art Review (https://jacc.org/doi/10.1016/j.jacc.2020.10.019)
- Using Epidemiological Data to Inform Clinical Trial Feasibility Assessments: A Case Study | Stroke (https://ahajournals.org/doi/10.1161/STROKEAHA.122.041650)
- New York Times Article on Clinical Trials Features Quote from the Alliance for Aging Research – Alliance for Aging Research (https://agingresearch.org/blog/new-york-times-article-on-clinical-trials-features-quote-from-the-alliance-for-aging-research)
- Quotes About Medical Research (https://changingthepresent.org/pages/quotes-about-medical-research)
- Overcome Common Challenges in FIH Trials with bioaccess Solutions
- Enrollment in Clinical Trials: Statistics and Patient Recruitment Strategies | Power (https://withpower.com/guides/enrollment-in-clinical-trials-statistics-and-patient-recruitment-strategies)
- What clinical trial statistics tell us about the state of research today (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- SCIRP Open Access (https://scirp.org/journal/paperinformation?paperid=128354)
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)

Leave a Reply