Introduction
The Global Unique Identifier Database (GUDID) serves as a cornerstone in the realm of medical device regulation, ensuring that products are traceable and safe for patients. For manufacturers navigating this intricate system, significant benefits await, ranging from enhanced compliance to improved patient outcomes. However, the journey to secure GUDID access is laden with challenges, including stringent documentation requirements and potential application pitfalls.
How can manufacturers effectively prepare and streamline their entry into this essential database to circumvent common missteps and ensure compliance?
Understand GUDID and Its Importance for Medical Devices
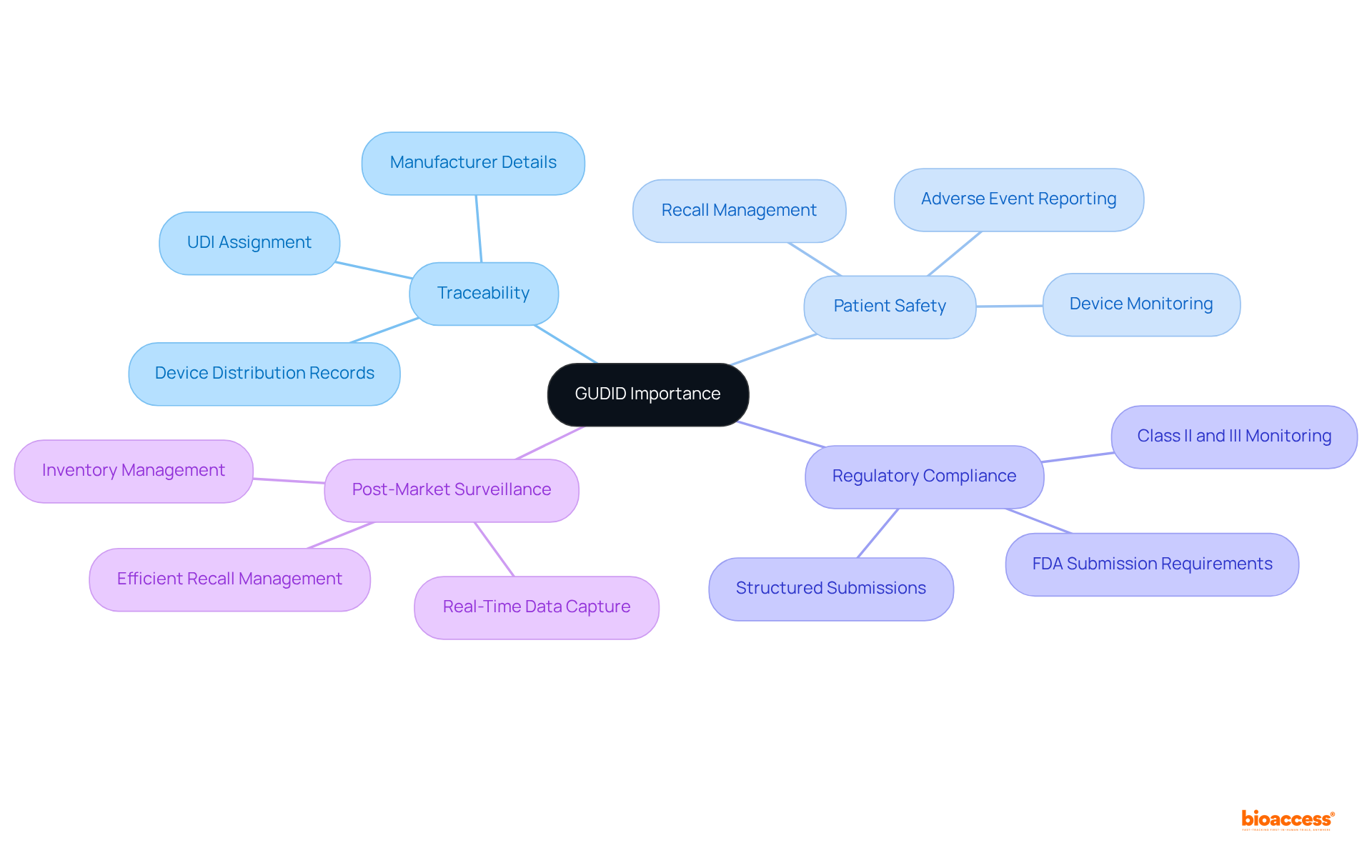
The Global Unique Identifier Database serves as a pivotal system established by the FDA, acting as a comprehensive register for assigned a (UDI). For manufacturers, understanding the is essential as it greatly enhances traceability, bolsters , and ensures for . Each entry in the registry is accompanied by a digital record that contains crucial information, including the item identifier, manufacturer details, and production specifics. This database is instrumental in , facilitating efficient recall management and supporting adverse event reporting. Notably, the FDA requires manufacturers to monitor , which present serious health risks, guaranteeing traceability from production to patient. Familiarizing oneself with the database is a critical step in ensuring that your medical product has , which ultimately improves patient outcomes and safety. Case studies illustrate that the successful implementation of this system results in enhanced inventory management and quicker identification of defective devices, underscoring the importance of this framework in the healthcare sector.
Prepare Required Documentation and Credentials for GUDID Access
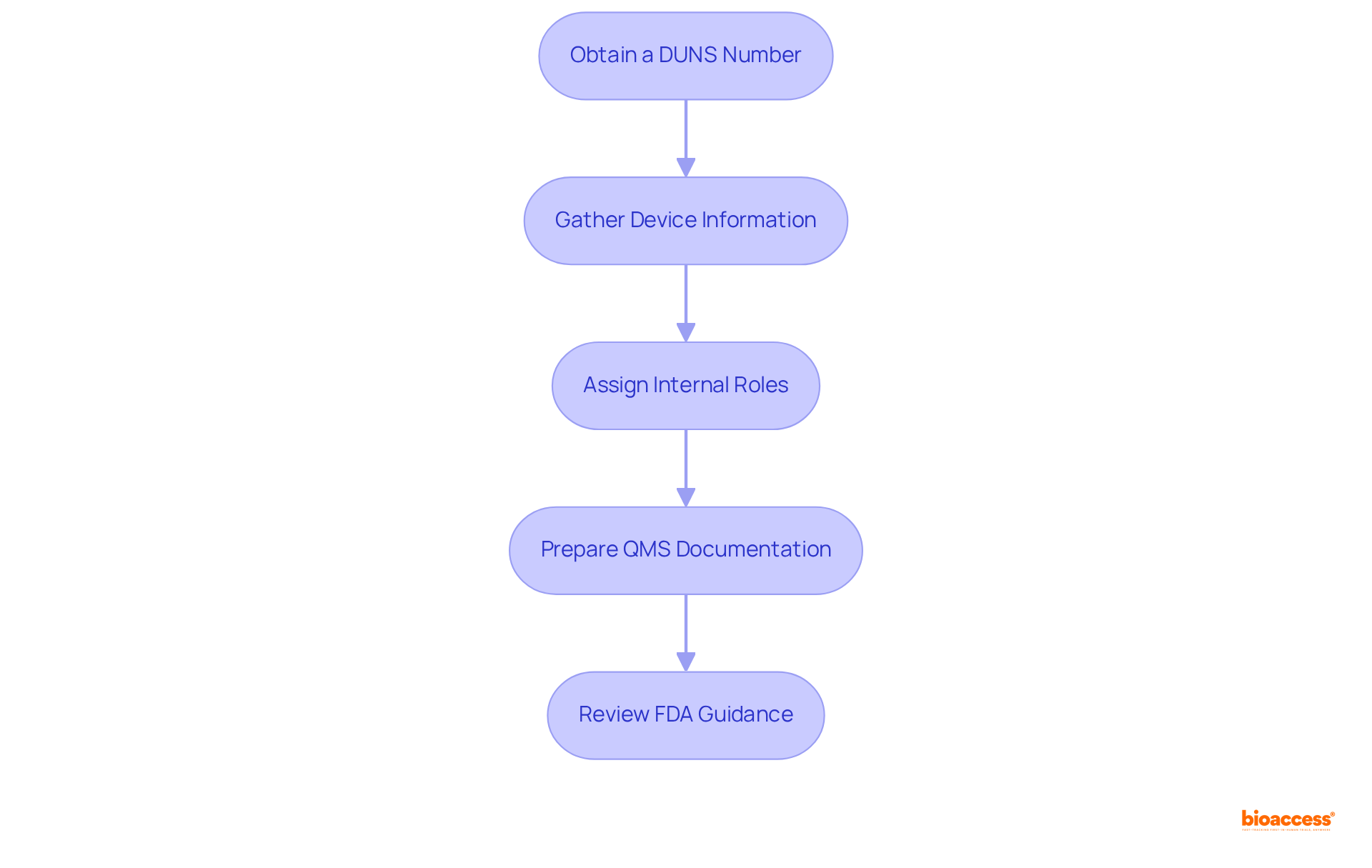
To gain gudid access to the Global Unique Device Identification Database, manufacturers must prepare specific documentation and credentials. Here’s a streamlined guide to facilitate the process:
- Obtain a : This unique identifier is crucial for validating your organization’s identity. Request a via Dun & Bradstreet, as it is a requirement for entries.
- Gather Device Information: Compile all necessary details about your medical device, including the , brand name, model number, and a comprehensive description.
- Assign Internal Roles: Designate team members responsible for overseeing entries, ensuring they receive the necessary training and access to relevant systems.
- Prepare : Ensure your QMS is current and includes procedures for , as this is vital for regulatory adherence.
- : Familiarize yourself with the FDA’s guidelines on the Global Unique Device Identifier entries to ensure all requirements are met, including the need for precise and validated data.
By preparing these documents in advance, you can streamline the application process and minimize potential delays. Notably, the has seen a significant increase in s issued, reflecting the growing compliance landscape. According to recent reports, the market size of in the U.S. is anticipated to reach $56.4 billion by 2025, highlighting the significance of prompt and precise entries for market access. Furthermore, the system oversees 57 attribute fields via organized entries, emphasizing the intricacy involved in the process. The was finalized in 2020, highlighting the urgency for adherence in this changing regulatory environment.
Follow the Application Process for GUDID Access
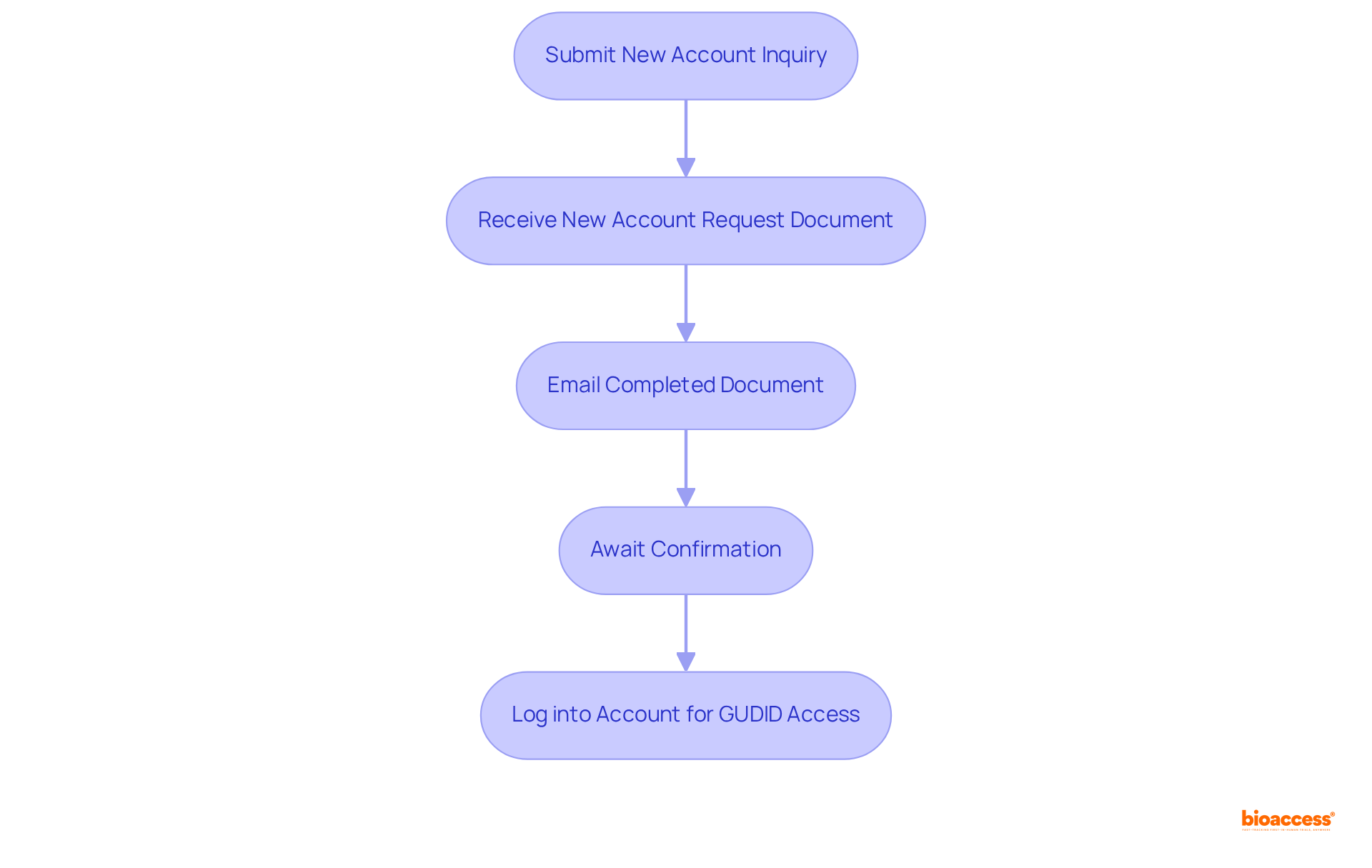
To apply for , adhere to the following essential steps:
- Submit a : Navigate to the ’s device identification website and complete the form, ensuring that all details are accurate and comprehensive. It is crucial to note that is mandatory for all devices intended for commercial distribution in the U.S.
- Receive the : Upon submission, the will send you a fillable PDF document via email. Complete this document with the necessary information.
- Email the Completed Document: Return the filled PDF to the , including any additional documentation they may have requested.
- Await Confirmation: The will assess your application and inform you of your account status. This review process can take several weeks; therefore, it is advisable to plan accordingly. Manufacturers should develop an and schedule for compliance, as the process may extend over time.
- Log into your account to gain and begin submitting your product information once your application is approved.
In recent years, the has observed a notable rise in applications, reflecting an increasing focus on compliance and responsibility within the . Common mistakes during the inquiry process include incomplete forms and missing documentation, which can significantly delay approval. As Marco Theobold, a specialist in medical equipment and drug regulations, emphasizes, careful preparation is essential to avoid these pitfalls. To ensure a seamless application process and , verify that all information is meticulously prepared before submission. Furthermore, labelers are responsible for preserving data accuracy over time and must promptly update records if device characteristics change.
Troubleshoot Common Issues in GUDID Access
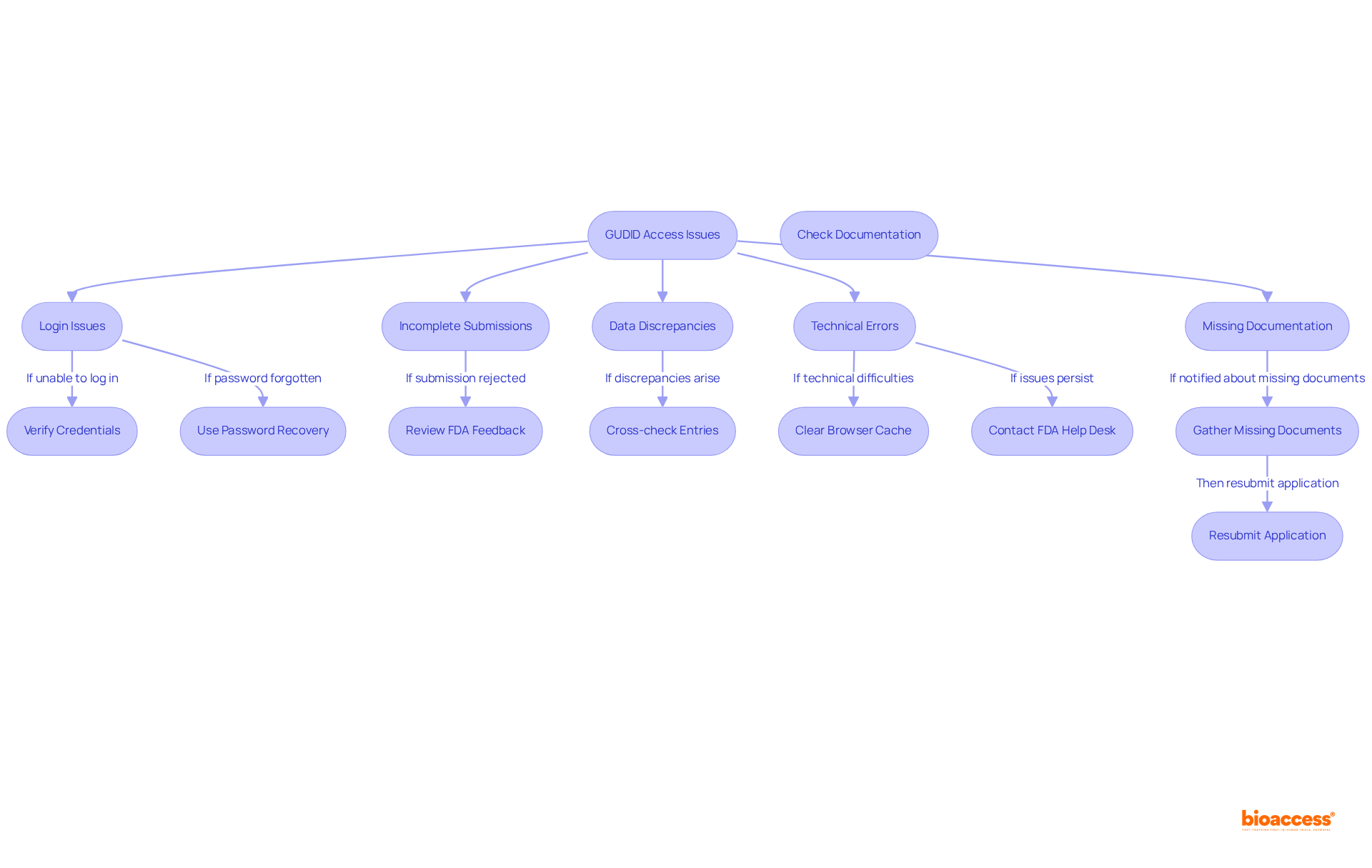
When requesting access, you may encounter several typical issues that require effective troubleshooting. Consider the following strategies:
- If you are unable to log into your , verify that you are using the correct credentials to resolve your . For forgotten passwords, utilize the to regain access.
- Incomplete Submissions: In the event of a , carefully review the feedback provided by the FDA. Ensure that all required fields are accurately and completely filled out to avoid further complications.
- Data Discrepancies: Should discrepancies arise in your device information, cross-check your entries against your documentation. Consistency is essential for adherence, particularly as the in the database.
- Technical Errors: If you encounter with the web application, clear your browser cache or consider switching to another browser. Should issues persist, do not hesitate to reach out to the for support.
- Missing Documentation: If notified about , promptly gather the necessary information and resubmit your application to expedite the process.
By proactively addressing these common issues, you can facilitate a smoother experience with GUDID access, ultimately enhancing your compliance and operational efficiency. Remember, resolving these issues quickly is essential, as than traditional markets when processes are streamlined.
Conclusion
Understanding and securing access to the Global Unique Device Identification Database (GUDID) is paramount for manufacturers of medical devices. This system not only enhances traceability and compliance but also significantly contributes to patient safety and effective post-market surveillance. By familiarizing themselves with GUDID, manufacturers can ensure their products are accurately represented and monitored, ultimately leading to improved health outcomes.
The article outlines essential steps to gain GUDID access, including:
- Preparing the necessary documentation
- Understanding the application process
- Troubleshooting common issues
Key actions such as obtaining a DUNS number, compiling device information, and adhering to FDA guidelines are vital for a smooth application experience. Additionally, addressing potential problems proactively can prevent delays and enhance operational efficiency within the regulatory framework.
In conclusion, the importance of GUDID access extends beyond mere compliance; it plays a vital role in the broader context of healthcare quality and safety. Manufacturers are encouraged to prioritize their GUDID preparations and stay informed about evolving requirements. By doing so, they not only fulfill regulatory obligations but also contribute to a more reliable and safer healthcare environment for all stakeholders involved.
Frequently Asked Questions
What is the Global Unique Identifier Database (GUDID)?
The GUDID is a comprehensive register established by the FDA that serves as a pivotal system for medical products assigned a Unique Device Identifier (UDI).
Why is understanding GUDID important for manufacturers?
Understanding GUDID is essential for manufacturers as it enhances traceability, bolsters patient safety, and ensures access for regulatory compliance.
What type of information is included in the GUDID registry?
Each entry in the GUDID registry includes crucial information such as the item identifier, manufacturer details, and production specifics.
How does GUDID contribute to post-market surveillance?
GUDID facilitates efficient recall management and supports adverse event reporting, which are critical components of post-market surveillance.
Which classes of products are required to be monitored by manufacturers according to the FDA?
Manufacturers are required to monitor Class II and III products, which present serious health risks, ensuring traceability from production to patient.
What are the benefits of familiarizing oneself with the GUDID?
Familiarizing oneself with GUDID is critical for ensuring that medical products have access to the database, ultimately improving patient outcomes and safety.
What do case studies indicate about the implementation of GUDID?
Case studies indicate that successful implementation of GUDID results in enhanced inventory management and quicker identification of defective devices, highlighting its importance in the healthcare sector.
List of Sources
- Understand GUDID and Its Importance for Medical Devices
- jnjmedtech.com (https://jnjmedtech.com/en-NZ/eu-mdr/resources/blogs/improving-traceability-transparency)
- airistaflow.com (https://airistaflow.com/resources/understanding-medical-device-tracking-and-traceability)
- Advancing Patient Safety Surrounding Medical Devices: Barriers, Strategies, and Next Steps in Health System Implementation of Unique Device Identifiers – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC9233486)
- registrarcorp.com (https://registrarcorp.com/blog/medical-devices/medical-device-registration/gudid)
- Prepare Required Documentation and Credentials for GUDID Access
- ibisworld.com (https://ibisworld.com/united-states/market-size/medical-device-manufacturing/764)
- Medical Device Industry Facts, Trends and Statistics 2025 (https://arterexmedical.com/medical-device-industry-statistics)
- registrarcorp.com (https://registrarcorp.com/blog/medical-devices/medical-device-registration/gudid)
- reedtech.com (https://reedtech.com/knowledge-center/one-step-in-fda-udi-compliance-the-dun-bradstreet-duns-number)
- insider.thefdagroup.com (https://insider.thefdagroup.com/p/fda-gudid-guidance)
- Follow the Application Process for GUDID Access
- fda.gov (https://fda.gov/food/online-registration-food-facilities/fda-industry-systems-user-guide-create-new-account)
- reedtech.com (https://reedtech.com/knowledge-center/gudid-account-creation-five-steps-to-follow)
- registrarcorp.com (https://registrarcorp.com/blog/medical-devices/medical-device-registration/gudid)
- fda.gov (https://fda.gov/medical-devices/global-unique-device-identification-database-gudid/prepare-gudid)
- Troubleshoot Common Issues in GUDID Access
- britest.co.uk (https://britest.co.uk/case_studies)
- emergobyul.com (https://emergobyul.com/news/us-fda-looking-medical-device-registrants-udi-gudid-compliance)
- europe-it-consulting.ch (https://europe-it-consulting.ch/fda-warns-manufacturers-about-gudid-data-discrepancies?lang=en)

Leave a Reply