Introduction
In the fast-evolving landscape of clinical research, the right medical equipment can be the difference between success and failure. Each type of equipment, from diagnostic tools to life support systems, plays a crucial role in ensuring accurate data collection, patient safety, and effective treatment outcomes. As researchers navigate complex trials, the challenge lies not only in identifying essential equipment but also in understanding how each piece contributes to the overall integrity of the study.
What are the ten essential types of medical equipment that can empower clinical research and enhance healthcare delivery?
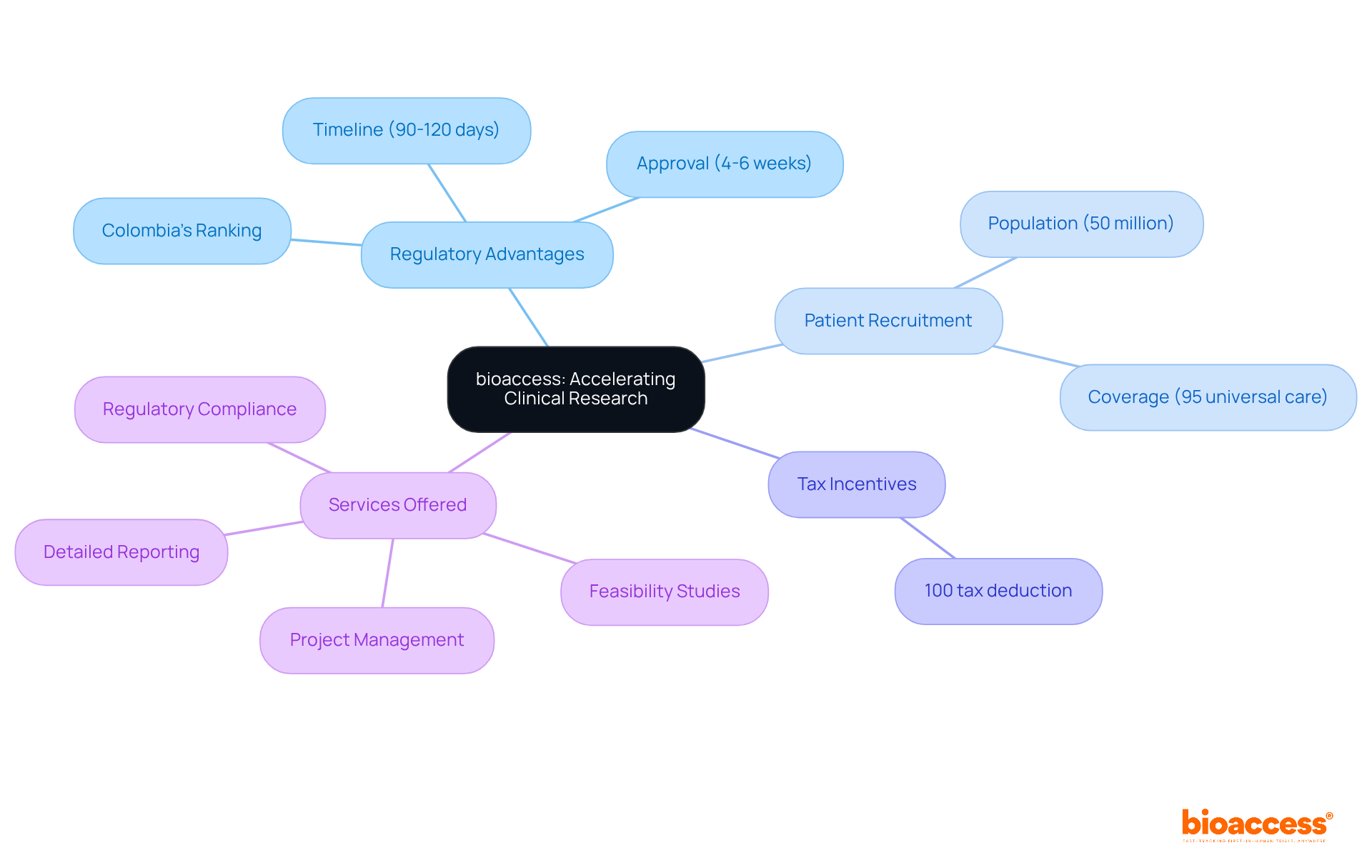
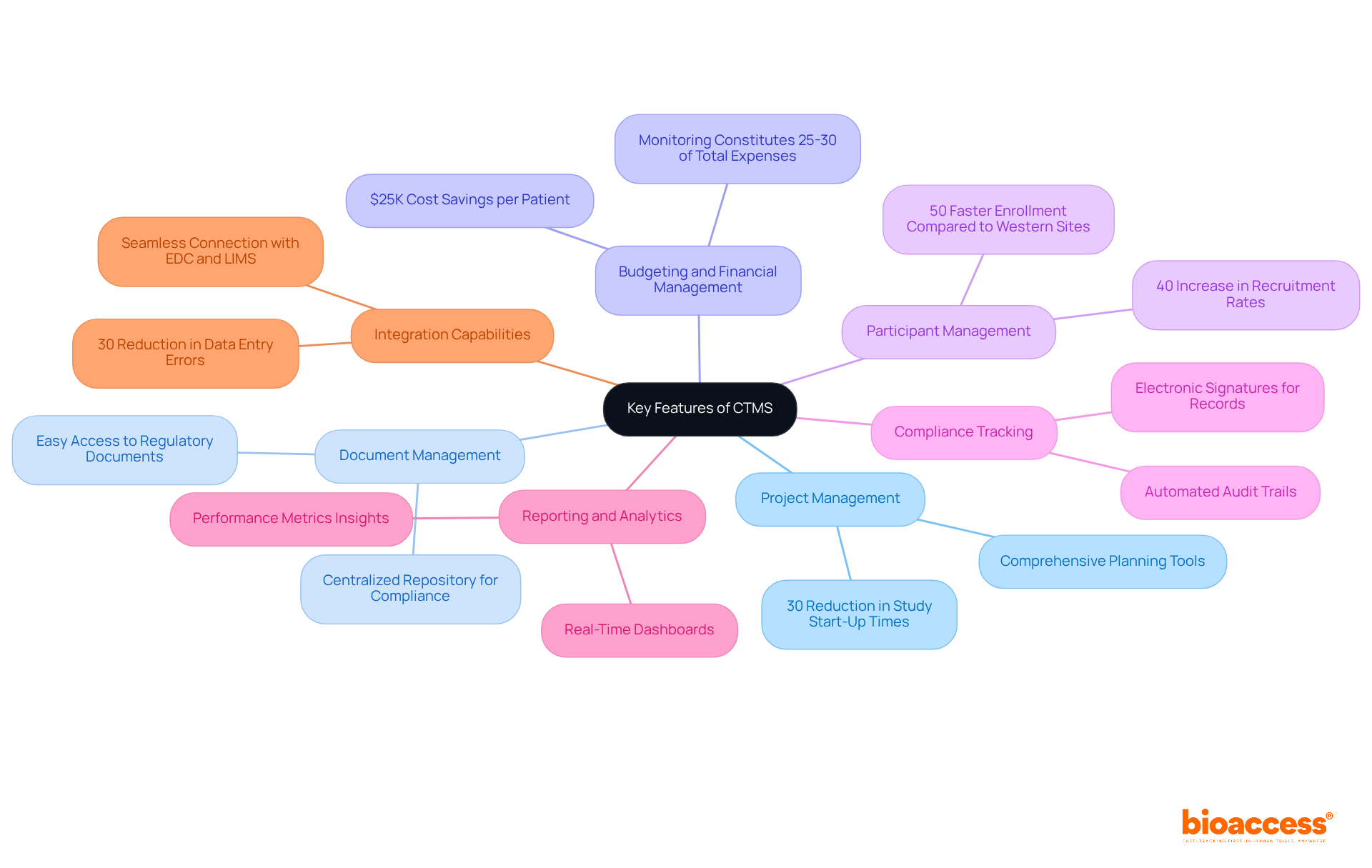
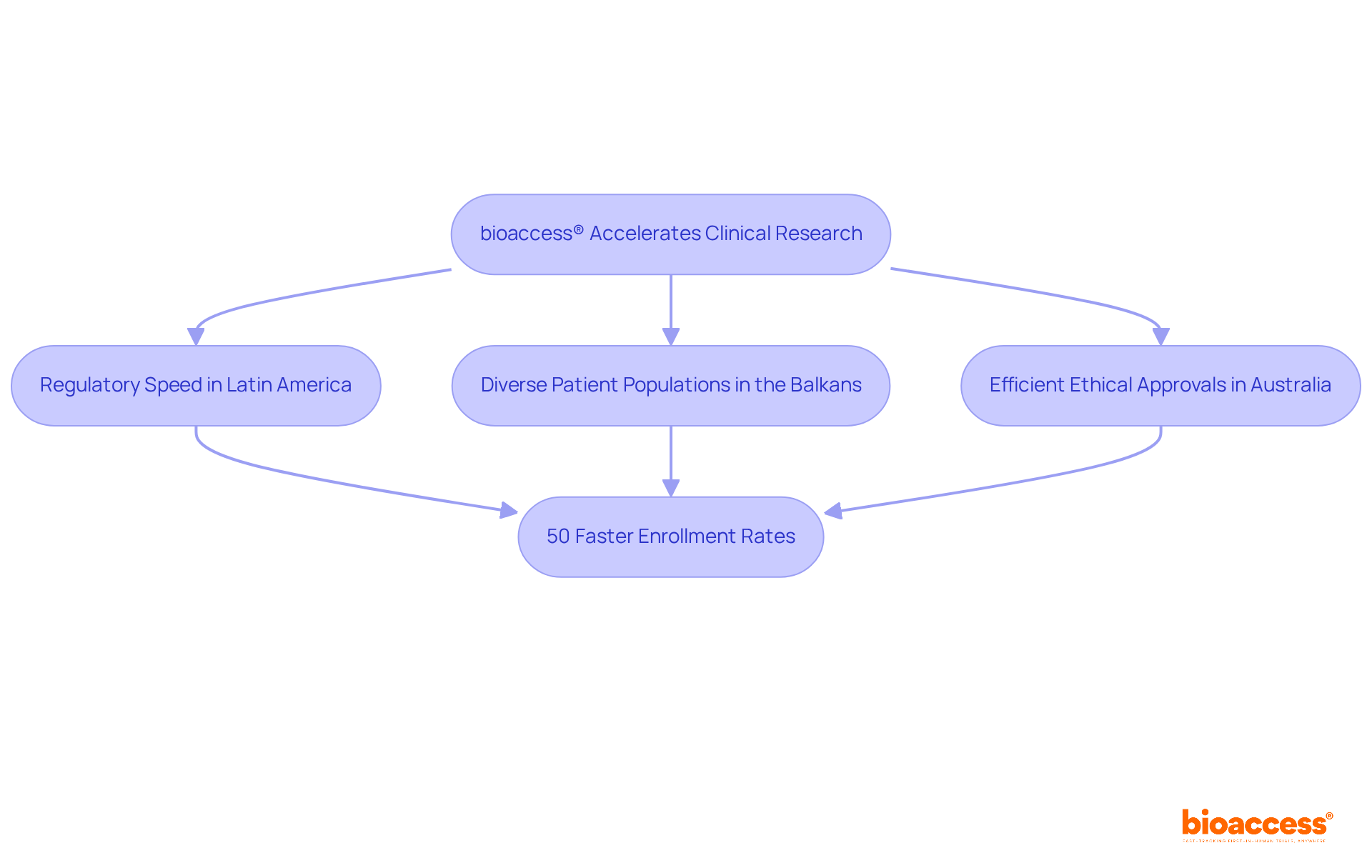
bioaccess®: Accelerating Clinical Research for Medical Devices
bioaccess® strategically positions itself in , the Balkans, and Australia to deliver . With the ability to secure and achieve enrollment rates that are 50% faster than traditional markets, bioaccess® empowers to accelerate their products’ journey to market. This agility is crucial in an environment where timely access to various can significantly impact outcomes and healthcare delivery.
With over 15 years of experience, bioaccess® possesses a deep understanding of the and diverse participant demographics that greatly influence . Their steadfast commitment to ethical practices and high-quality research not only establishes them as a leader in the field but also makes them an invaluable partner for companies navigating the complexities of . As the industry evolves, the focus on remains critical, underscoring the essential role bioaccess® plays in advancing healthcare solutions.
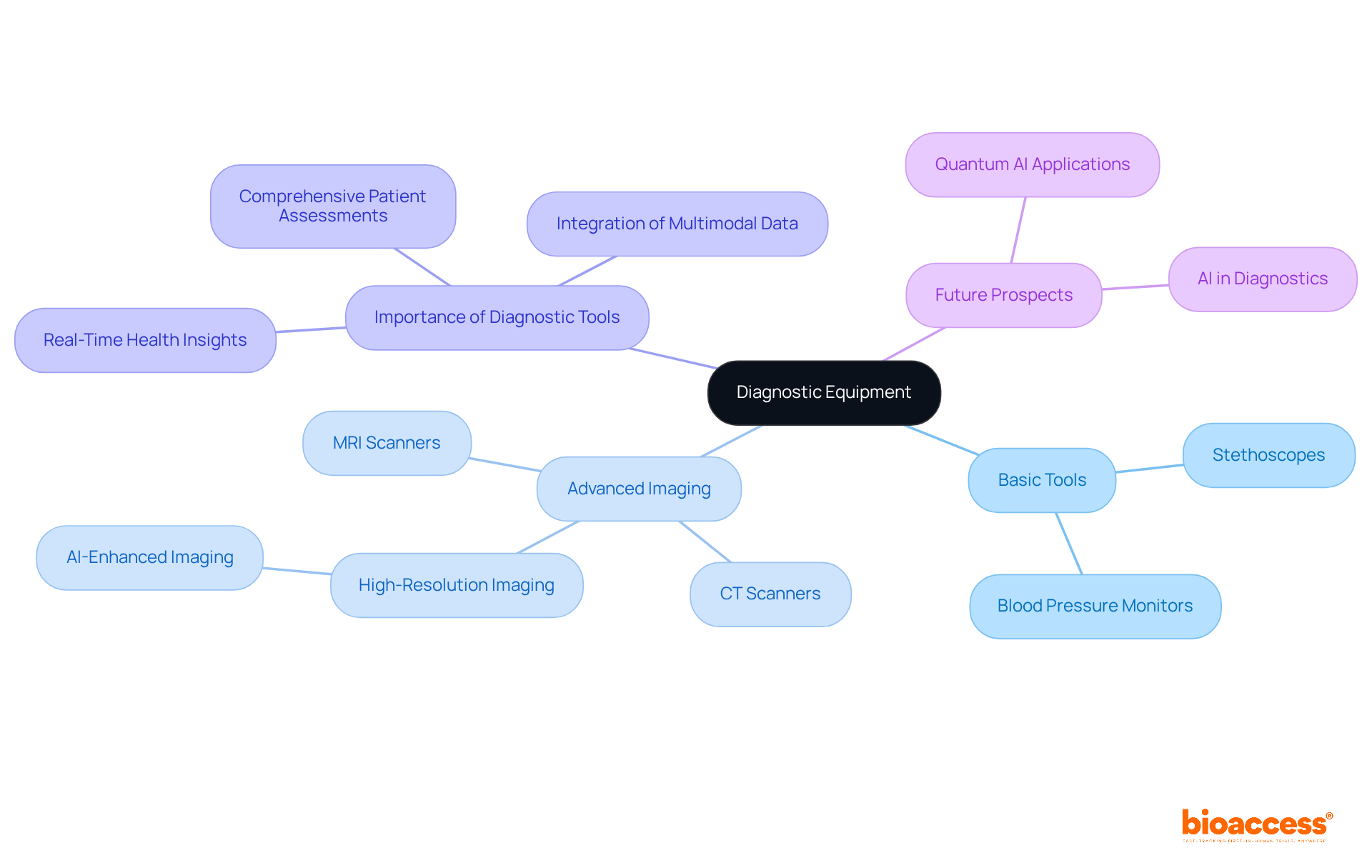
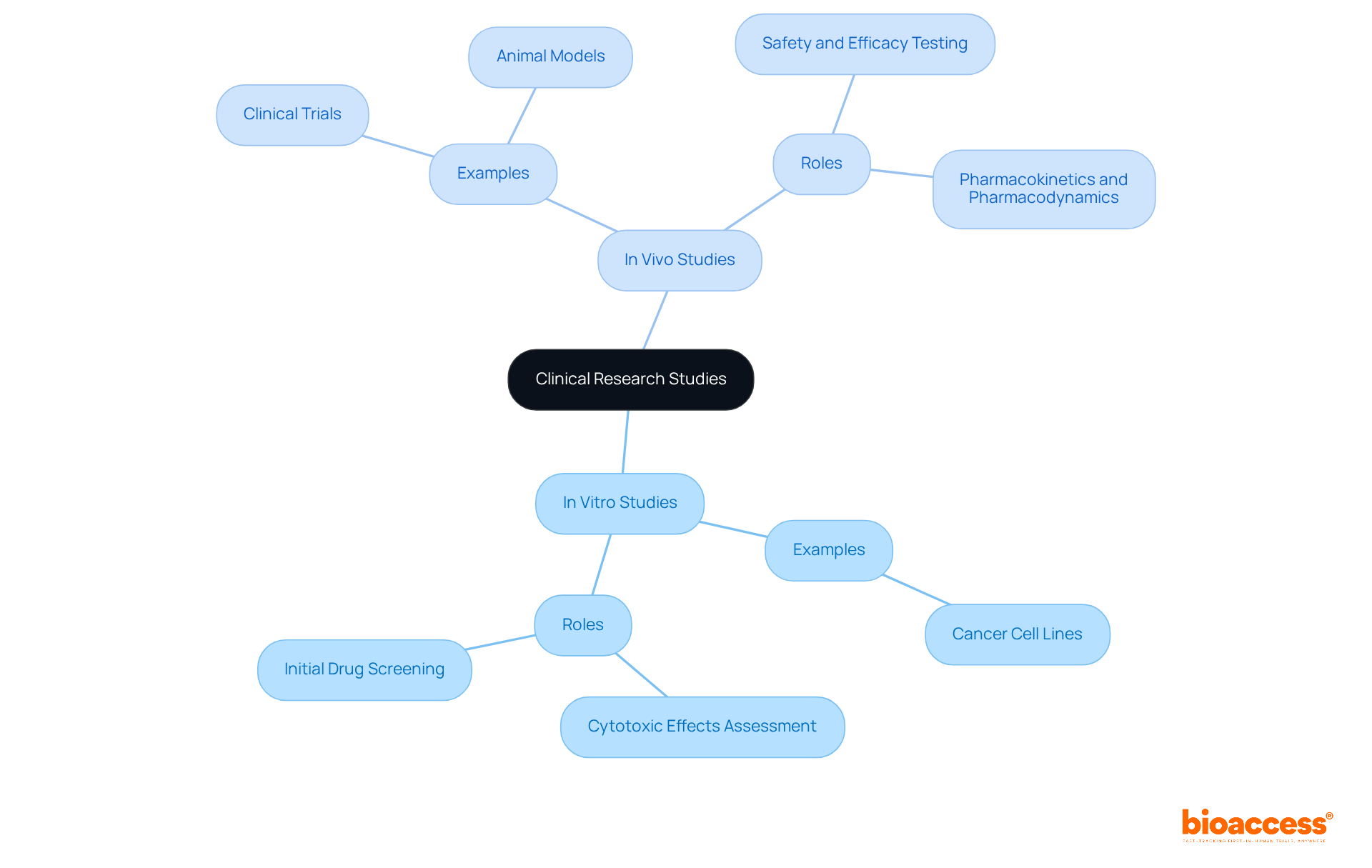
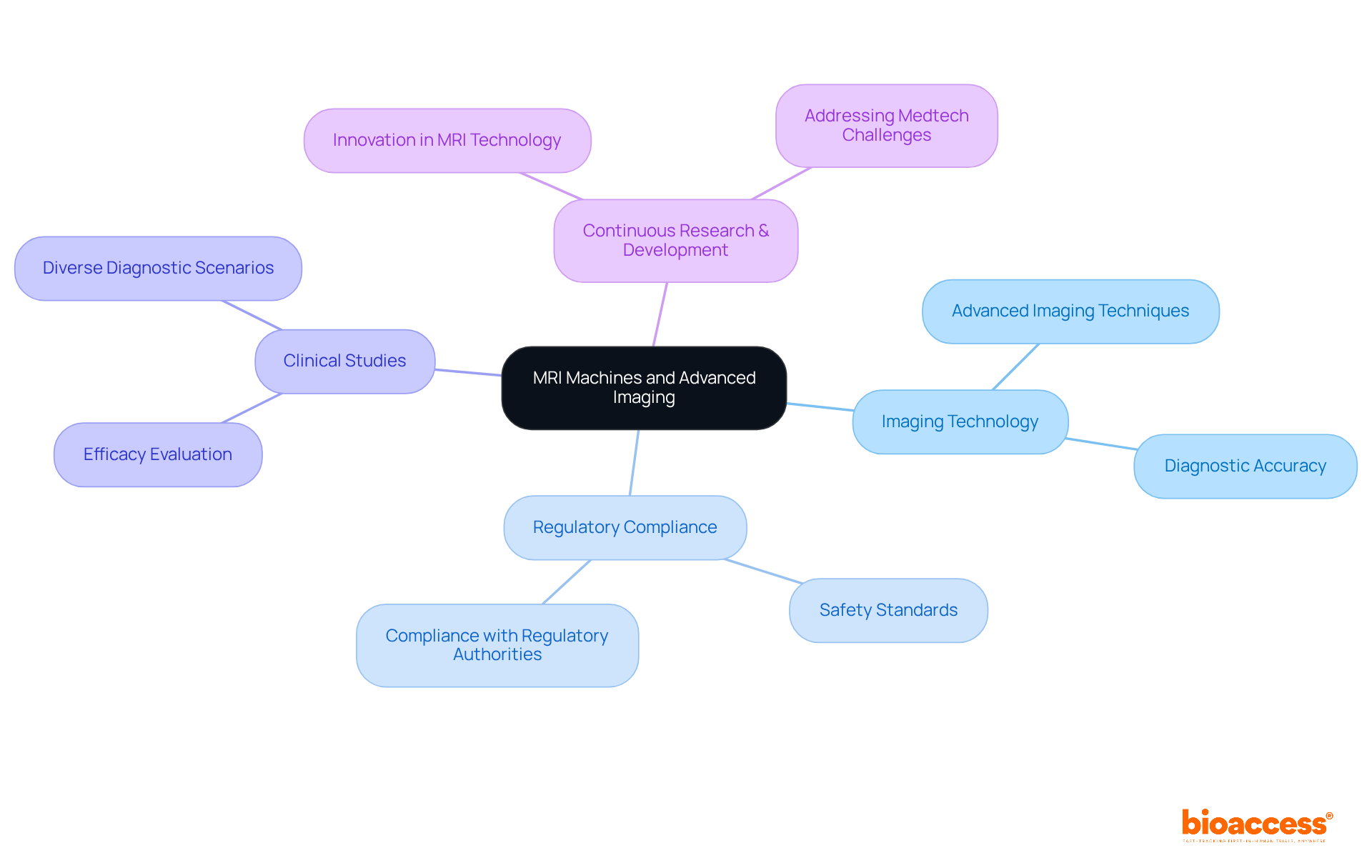
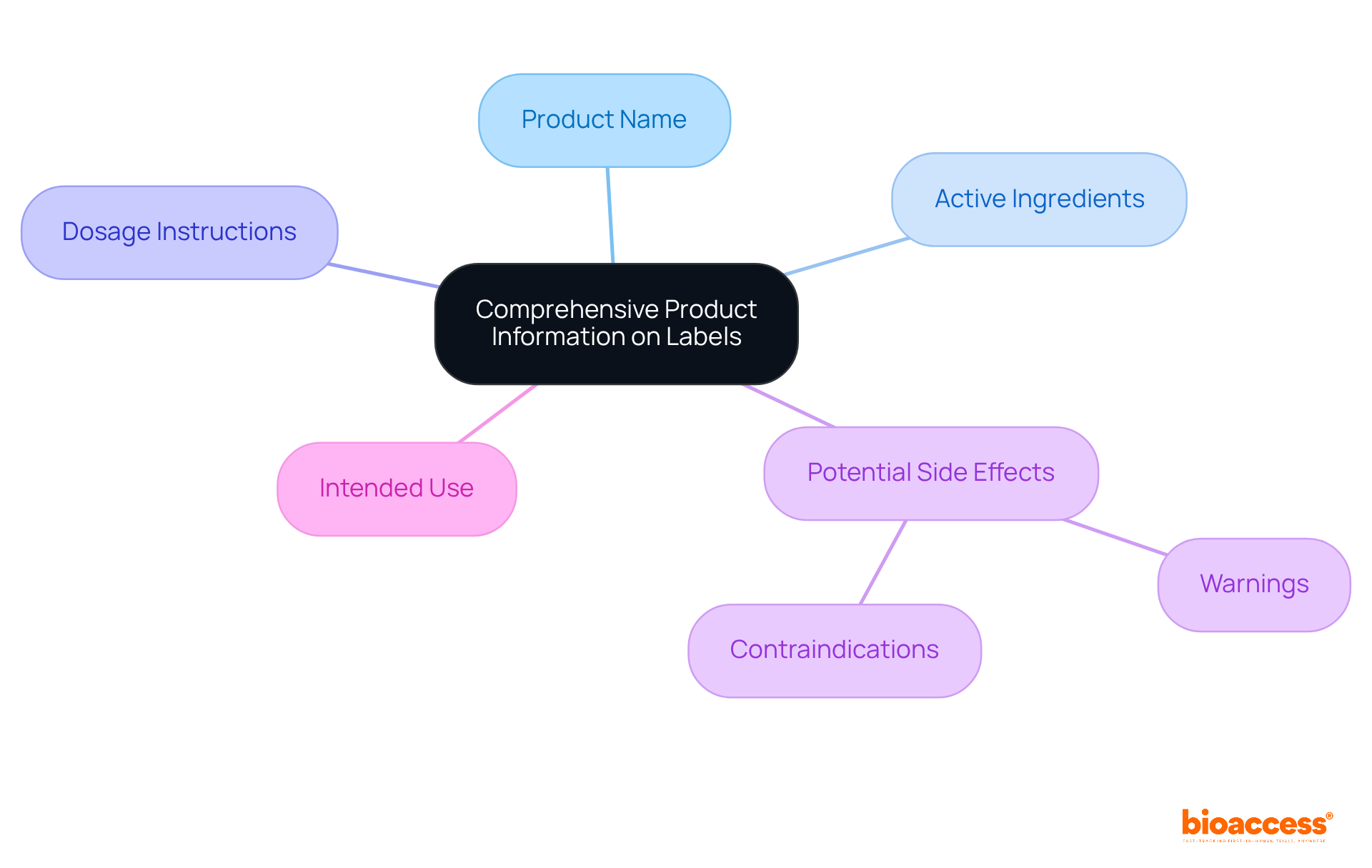
is pivotal in assessing individual health, incorporating a diverse array of tools such as stethoscopes, , and like MRI and CT scanners. These instruments are critical for gathering essential information on individual conditions, which is vital for .
For instance, provide real-time insights into cardiovascular health, allowing researchers to monitor changes that may influence trial outcomes. Imaging technologies, particularly MRI and CT scanners, deliver detailed visualization of internal structures, facilitating the identification of underlying conditions that could impact evaluations.
Recent advancements in diagnostic tools, including , have markedly enhanced the accuracy and efficiency of evaluations. By 2025, innovations such as are expected to further elevate diagnostic capabilities, leading to more precise assessments and improved outcomes for individuals.
Healthcare experts emphasize the importance of , noting that their dependability directly affects the quality of . The integration of multimodal data—merging imaging, bio-signals, and patient history—has been demonstrated to improve diagnostic accuracy, diminishing the risk of misdiagnosis and enhancing treatment management for .
In summary, the effectiveness of medical research hinges on the quality of , underscoring the essential role these tools play in the research landscape.
Treatment Equipment: Key Instruments for Patient Care
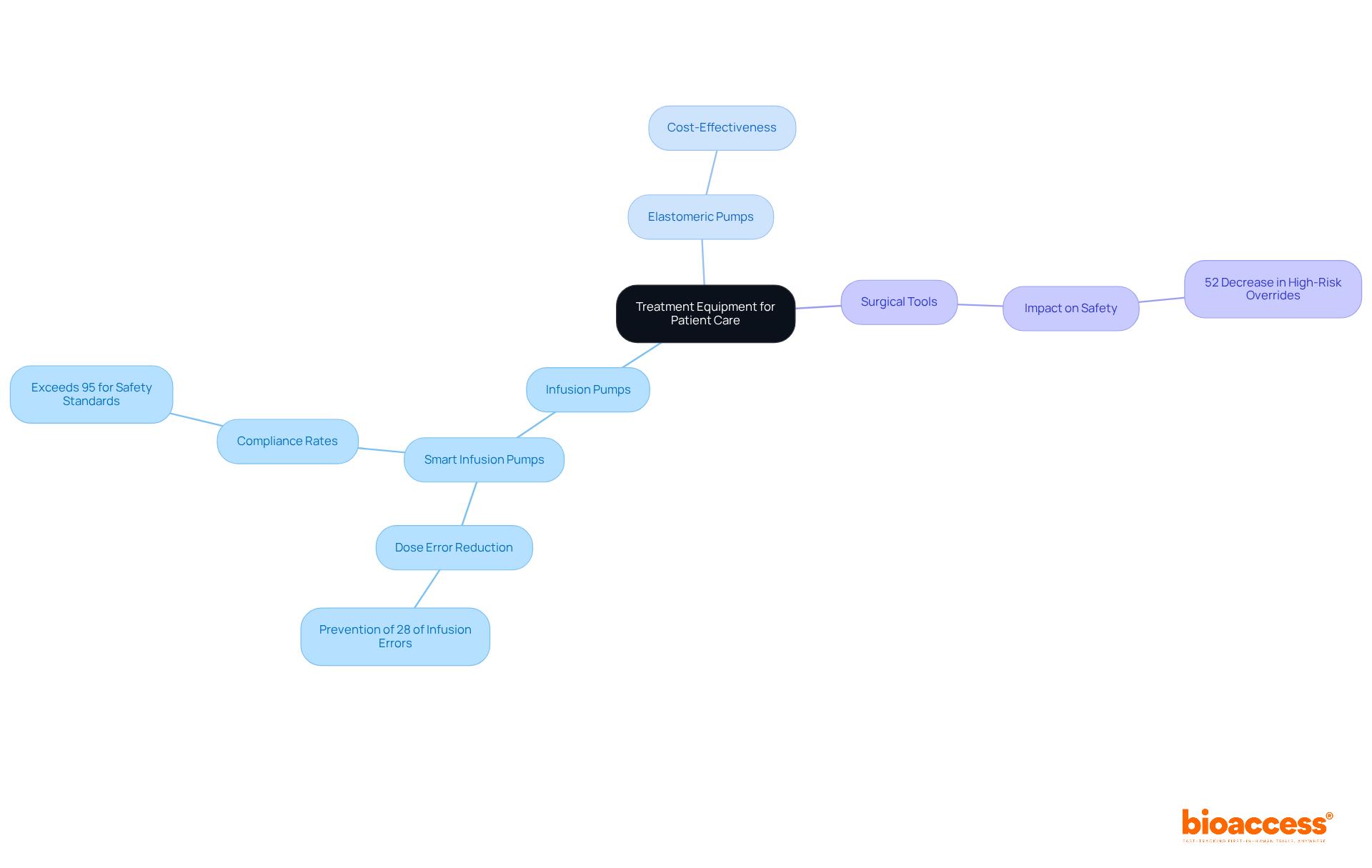
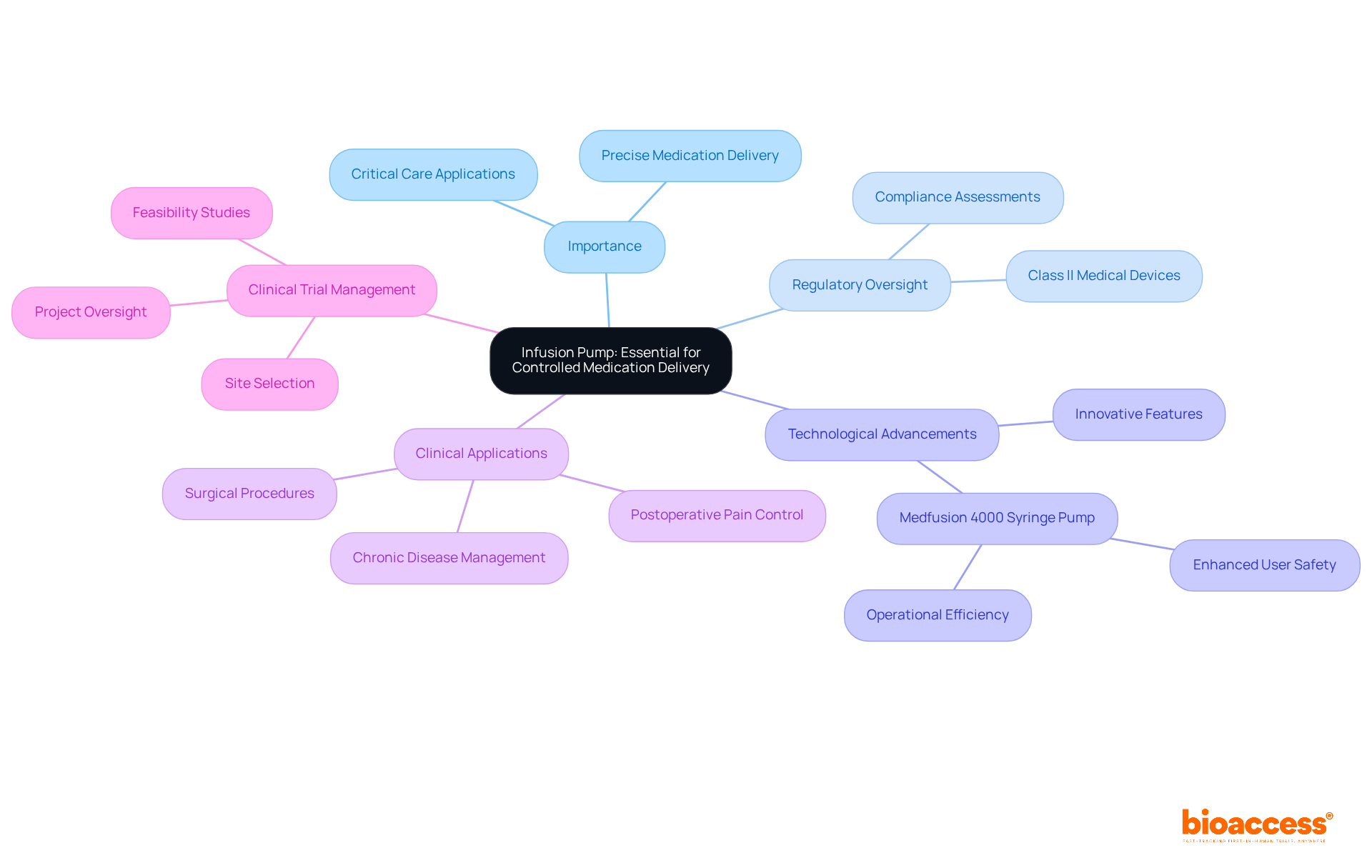
encompasses a diverse array of instruments essential for delivering care during research trials. Among these, emerge as critical devices for administering medications and fluids with precision, guaranteeing that patients receive the correct dosages at the appropriate intervals. A recent study underscores this significance, revealing that smart equipped with dose error reduction software can substantially enhance , potentially preventing 28% of infusion errors by alerting clinicians to dosing discrepancies. Furthermore, these smart maintain compliance rates exceeding 95% for safety standards, underscoring their reliability within healthcare environments.
Equally important are , which, being typically less expensive than more complex infusion systems, provide cost-effective solutions for medication delivery.
also hold paramount importance in medical studies, facilitating procedures that are integral to the investigative process. The effectiveness of these instruments is evidenced by hospitals that have adopted standardized procedures, reporting a remarkable 52% decrease in high-risk overrides. This statistic illustrates the impact of proper equipment usage on individual safety.
As we approach 2025, the role of in is increasingly vital, particularly in managing complex treatment regimens associated with chronic and acute diseases. Their ability to sustain steady flow rates is crucial in critical situations, thereby . Additionally, the integration of with healthcare informatics systems improves tracking and monitoring, significantly reducing medication errors and enhancing safety for patients receiving care.
Training programs are indispensable for , effectively minimizing programming errors and bolstering safety for individuals. In summary, the proper use and maintenance of , including and surgical instruments, are essential for ensuring safety and achieving reliable results in medical studies.
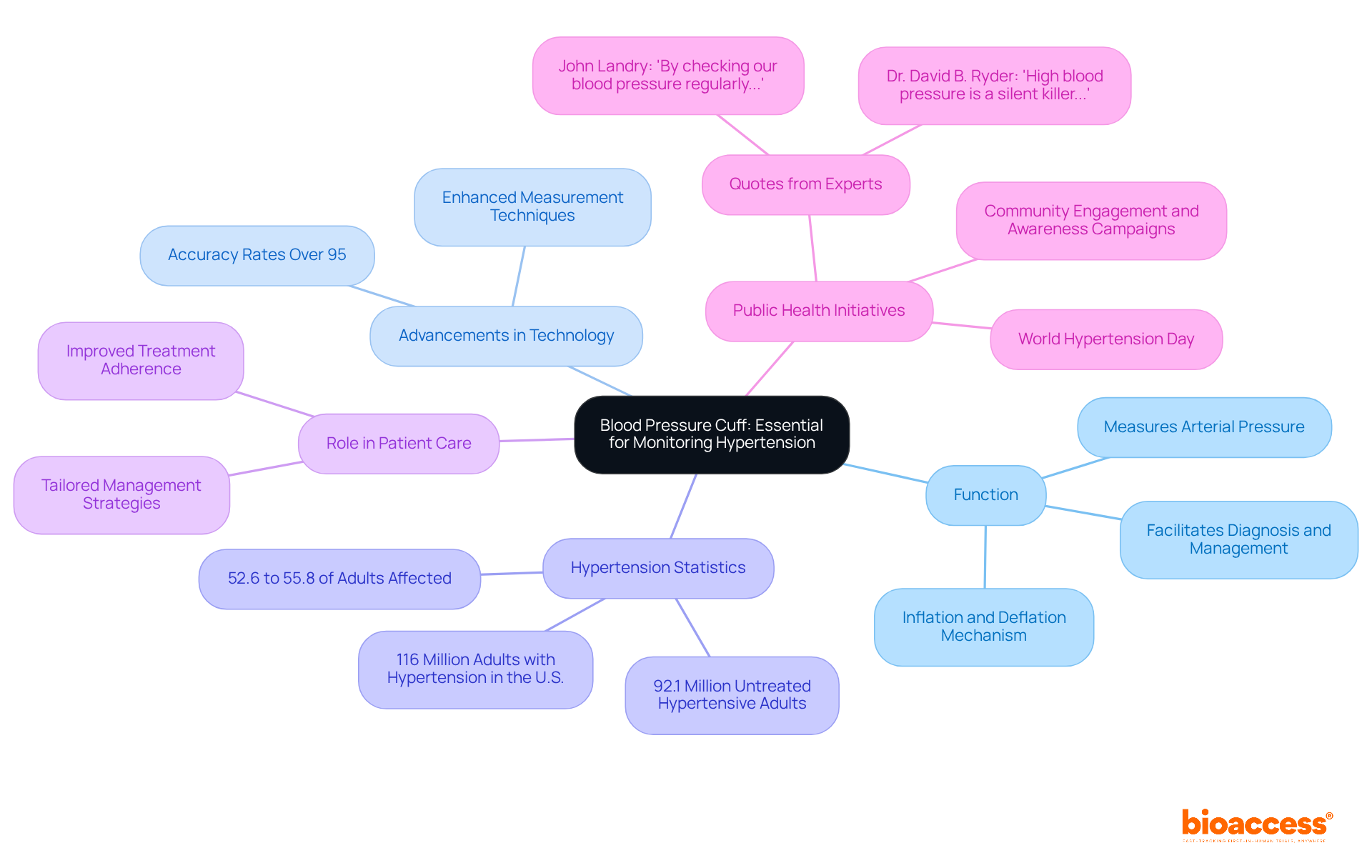
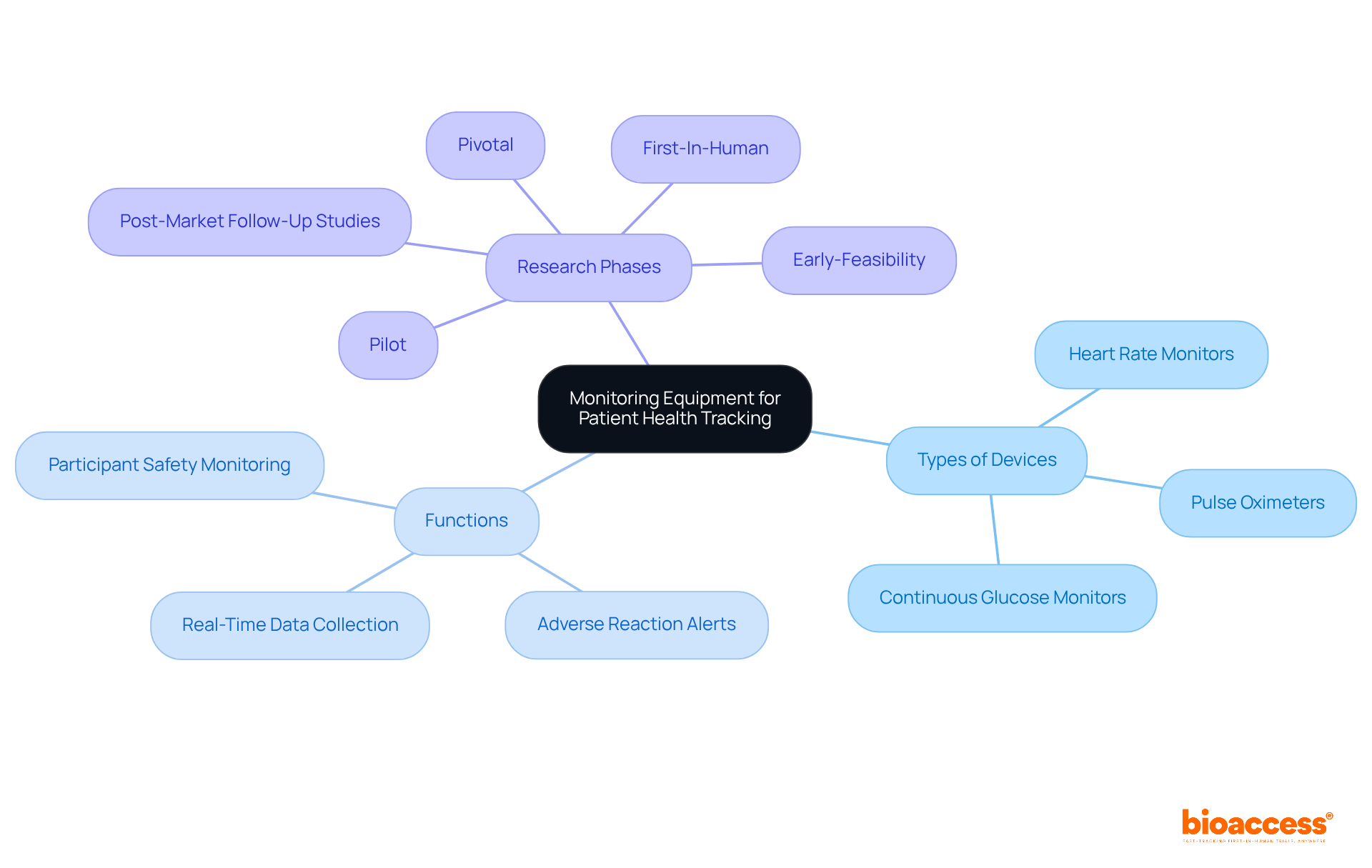
Monitoring Equipment: Vital for Patient Health Tracking
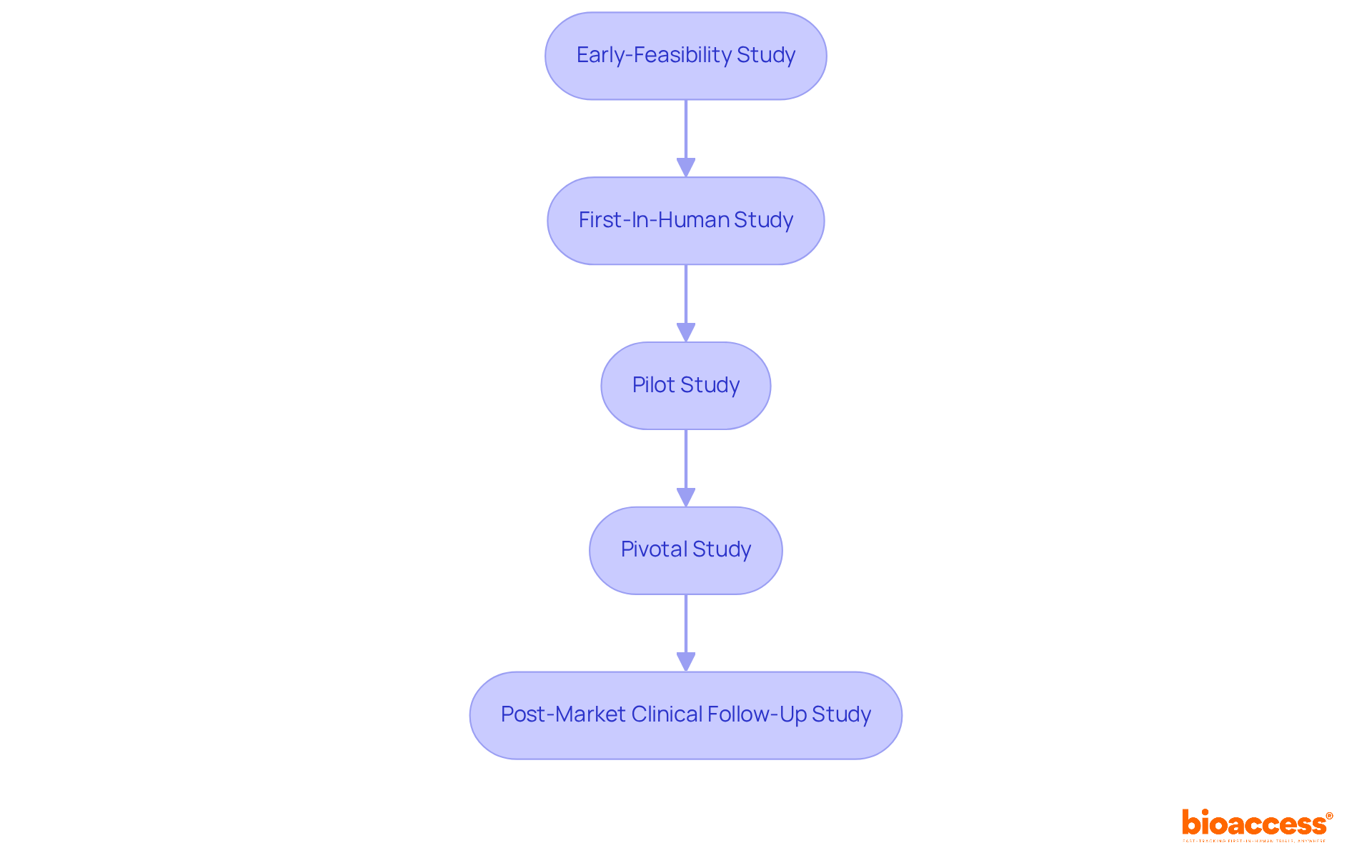
are pivotal in during , particularly in research conducted by bioaccess®, a leading in Latin America. Devices such as:
deliver that reflect an individual’s response to treatment. For example, can alert researchers to any adverse reactions during a trial, facilitating immediate intervention. The ability to consistently track vital signs ensures that individual safety is prioritized and that potential issues are swiftly addressed, thereby enhancing the overall integrity of the research. With bioaccess®’s extensive expertise in managing:
- Early-Feasibility
- Pilot
- Pivotal
- Post-Market Follow-Up Studies
the integration of is indispensable for achieving successful research outcomes.
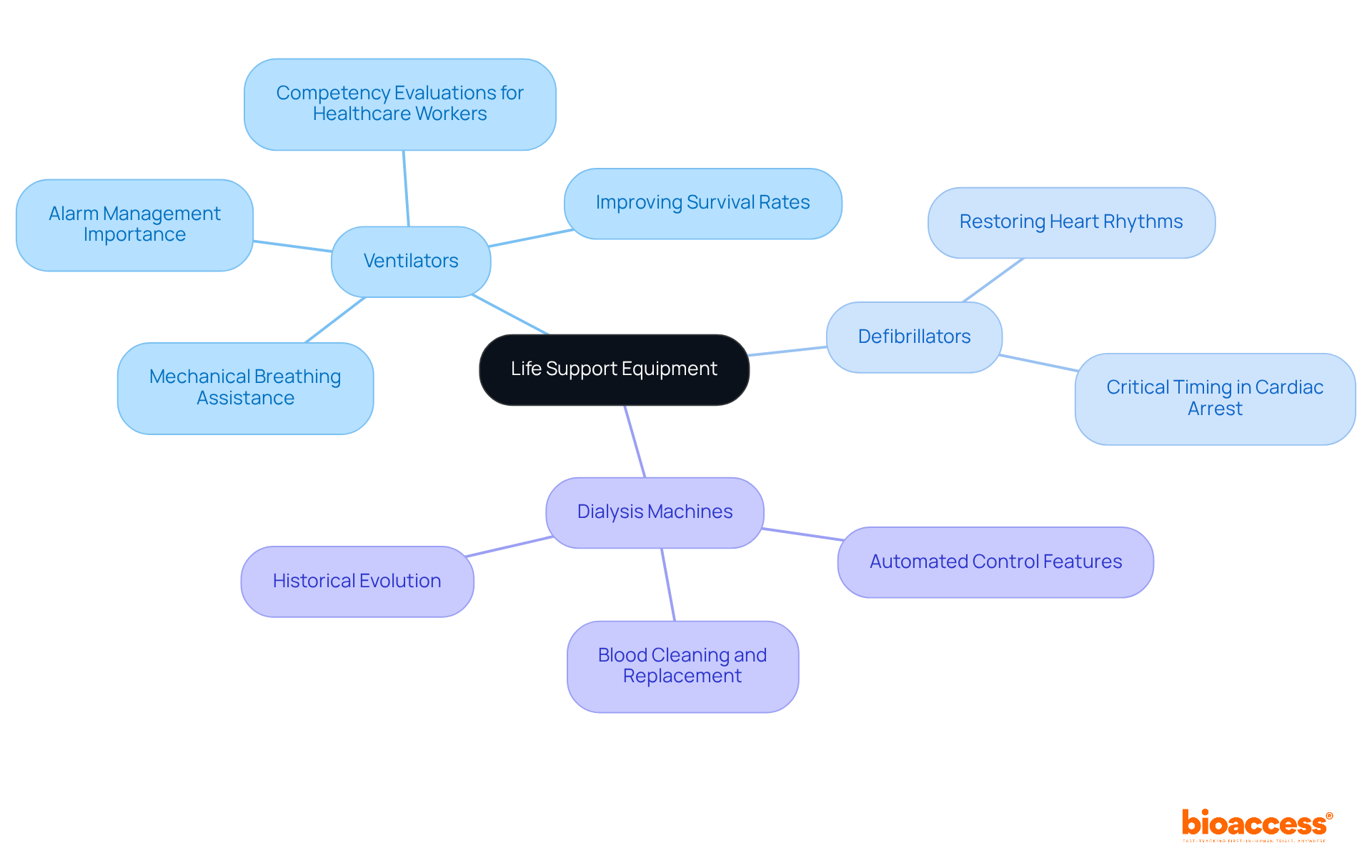
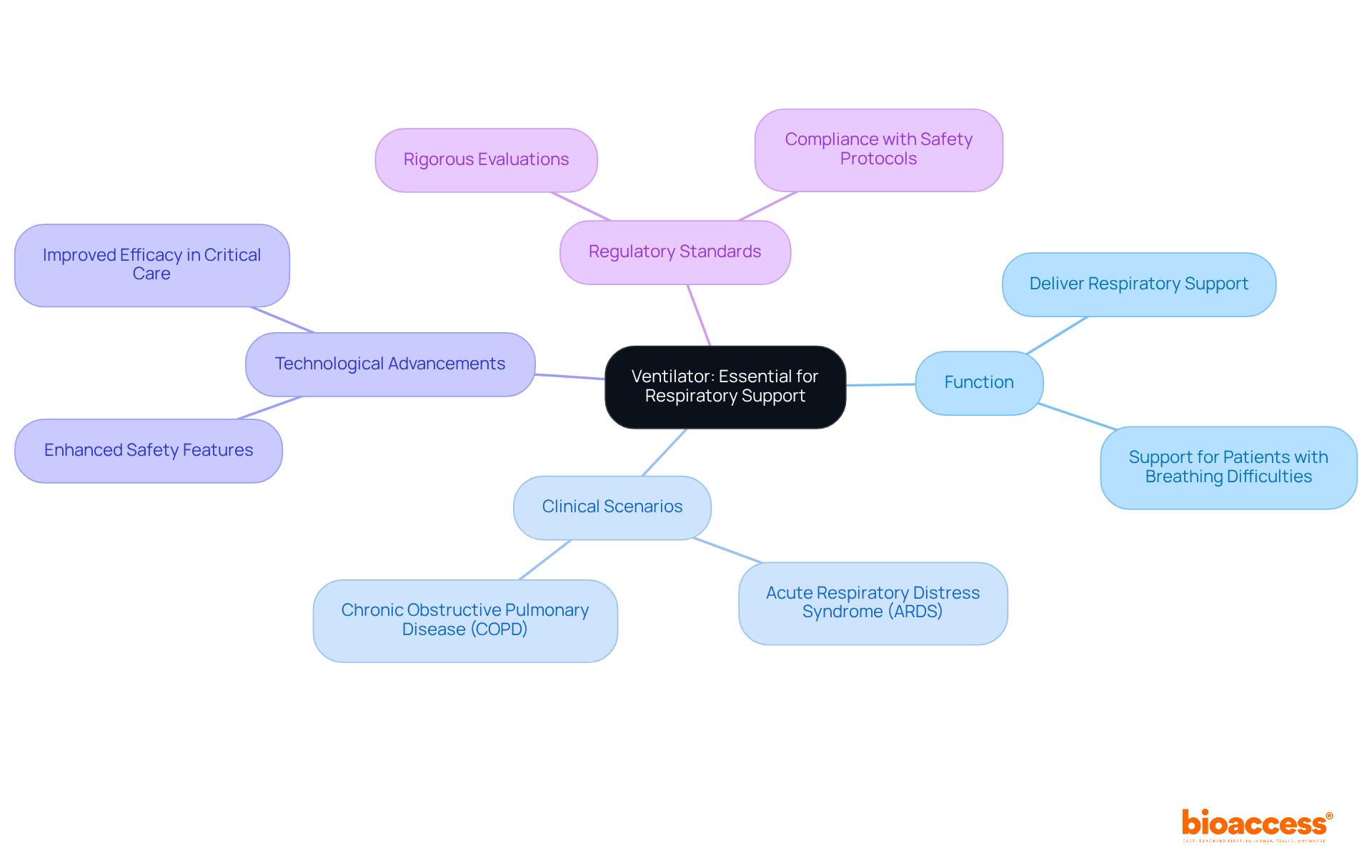
Life Support Equipment: Crucial for Critical Care
, which encompasses several including ventilators, defibrillators, and dialysis machines, is vital for individuals in critical condition or those participating in complex . These devices are engineered to sustain life and support .
For example, for individuals facing respiratory failure, significantly improving survival rates in critical care settings. Research indicates that effective ventilator management can reduce complications and enhance outcomes for individuals, underscoring their importance in medical studies. Moreover, healthcare professionals caring for individuals on ventilators must undergo annual competency evaluations, ensuring they are adequately trained to operate these essential devices.
during cardiac arrest, where every second is critical. Their prompt application can substantially elevate survival rates, rendering them indispensable in emergency situations. The integration of reliable for life support is essential not only for safety but also for the ethical conduct of involving high-risk populations.
Additionally, have revolutionized healthcare since their inception in the early 1940s. and distribution, along with alert systems for unforeseen issues, ensuring that individuals with kidney failure remain healthy and toxin-free while awaiting replacement therapies. This evolution highlights the ongoing commitment to through innovative medical technology.
In summary, the availability of robust , particularly life support equipment, is crucial for ensuring and facilitating the ethical conduct of research studies, ultimately leading to enhanced health outcomes for patients in critical care.
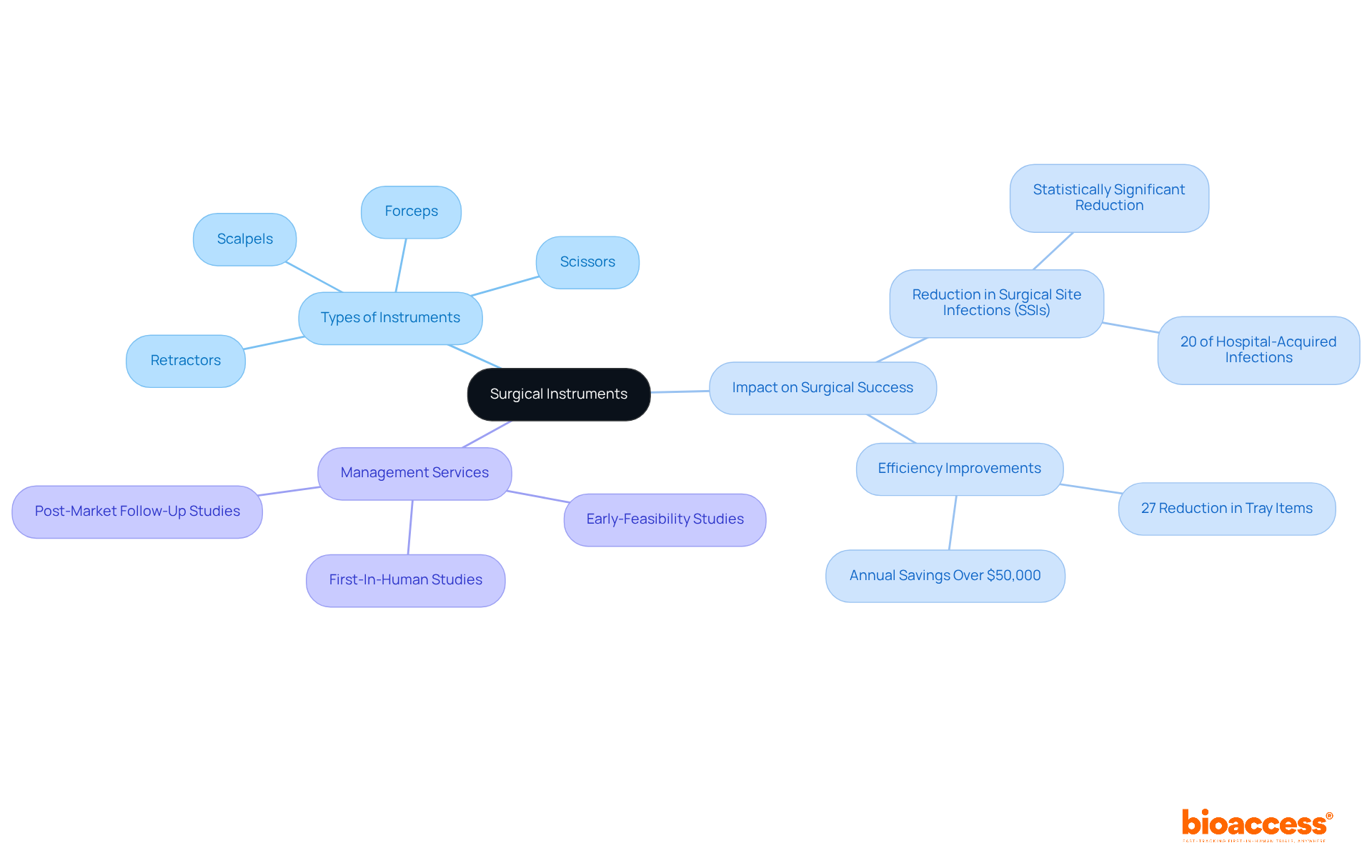
Surgical Instruments: Fundamental for Medical Procedures
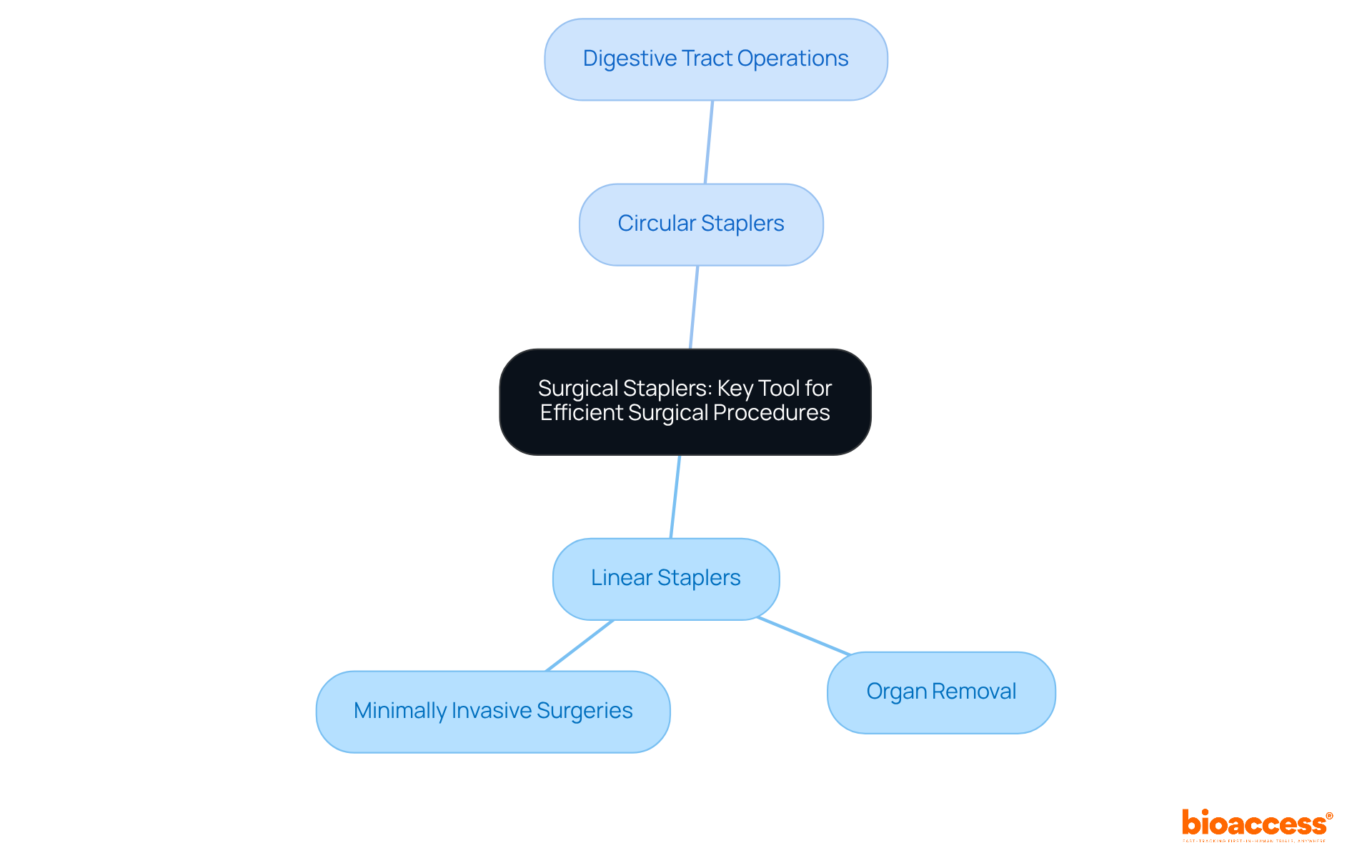
are essential components of different medical equipment types used in a variety of medical processes, particularly in . Key instruments such as scalpels, forceps, scissors, and retractors each serve distinct purposes that are critical for achieving successful outcomes. For instance, scalpels are primarily employed for making precise incisions, while forceps play a vital role in grasping and manipulating tissues during .
The impact of high-quality on success rates is significant. A study involving 233 patients revealed a statistically significant reduction in through the implementation of a . Additionally, the efficiency of is markedly improved when instruments are , which led to a 27% reduction in tray items and annual savings exceeding $50,000.
Innovations in surgical instruments continue to emerge, with advancements anticipated in 2025 that are expected to enhance research outcomes. The importance of utilizing , which are among essential medical equipment types, is underscored by the fact that nearly 20% of all hospital-acquired infections in the United States are associated with SSIs, highlighting the necessity for meticulous instrument selection and management within medical environments. As the healthcare landscape evolves, the focus on reducing complications through advanced remains essential for the success of medical studies.
In this context, partnering with bioaccess® ensures that research trials are supported by comprehensive management services, including:
- Post-Market Follow-Up Studies
These services are crucial for navigating the complexities of , reinforcing the importance of collaboration in achieving favorable research outcomes.
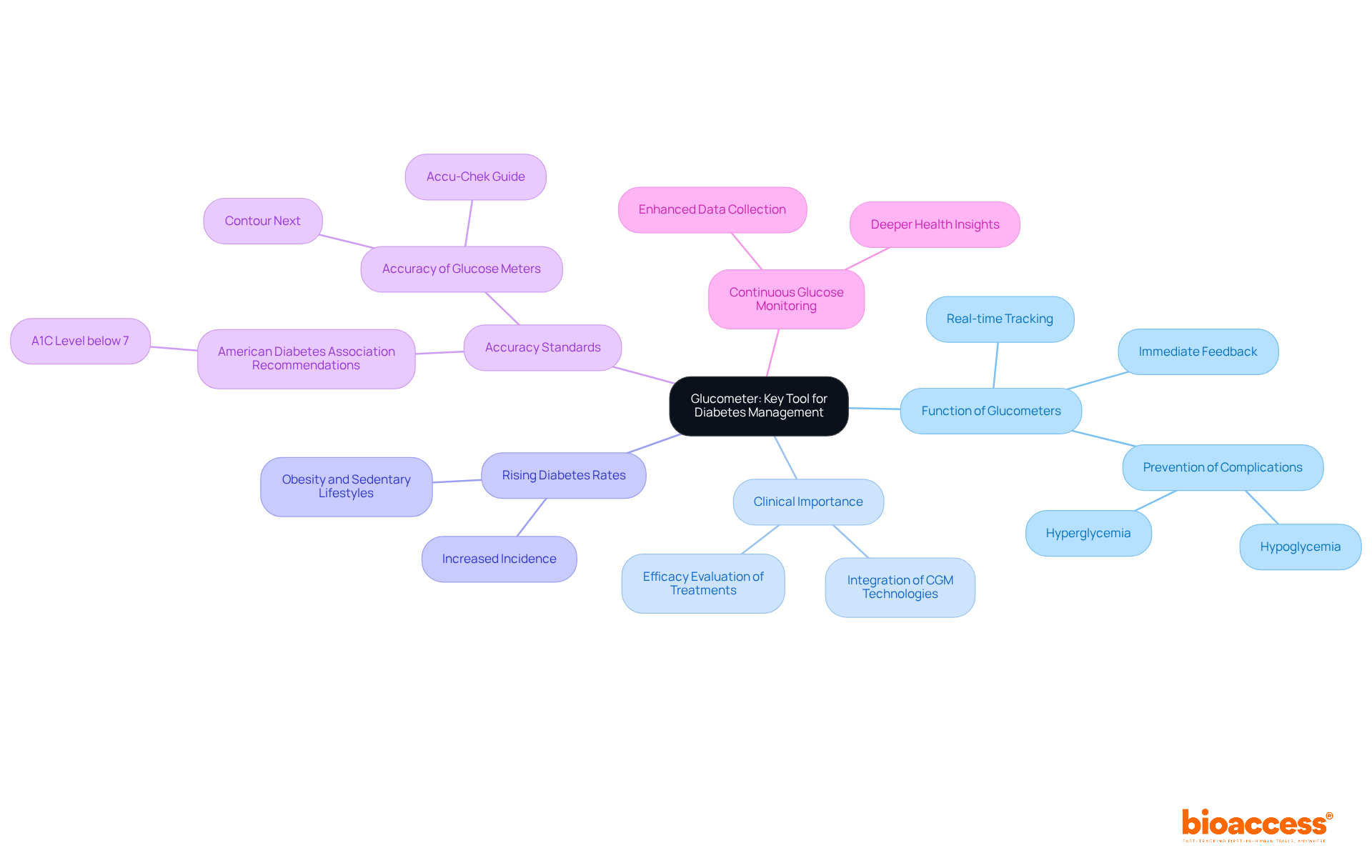
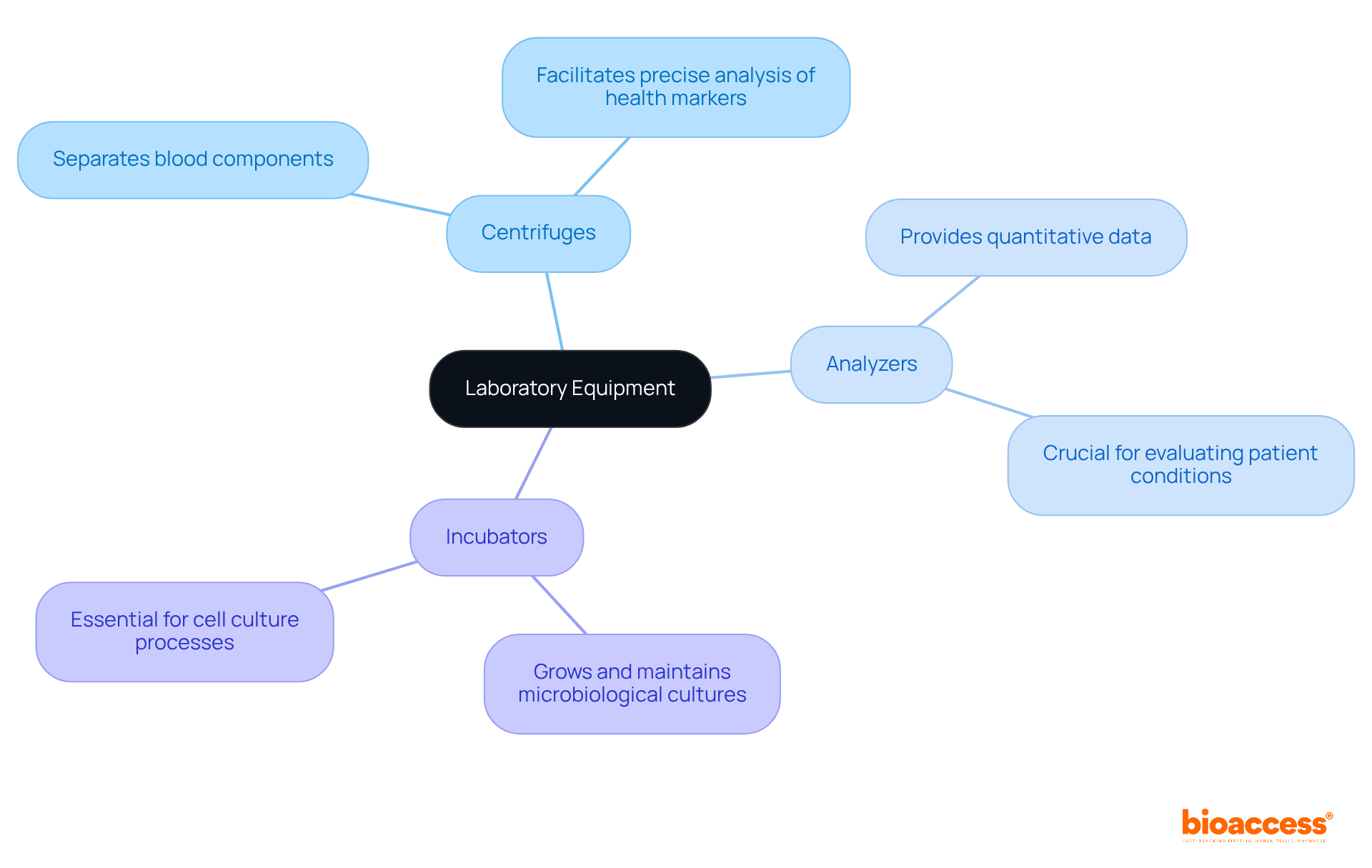
Laboratory Equipment: Supporting Diagnostics and Research
Various are essential for conducting that are vital to medical research. Key , such as centrifuges, analyzers, and incubators, play an integral role in processing samples and generating reliable results. For instance, centrifuges effectively separate blood components, facilitating precise analysis of various health markers. Analyzers, conversely, provide quantitative data that is crucial for evaluating patient conditions.
The precision and dependability of these significantly impact the quality of data collected during . Research shows that hospitals equipped with have experienced a 30% increase in specialist referrals, highlighting the necessity of utilizing . Furthermore, has been proven to decrease test turnaround times, thereby enhancing overall efficiency in experimental environments.
As we approach 2025, the significance of centrifuges and analyzers in trials cannot be overstated. Their ability to deliver is essential for advancing medical studies and improving outcomes for patients. Laboratory professionals assert that types are fundamental to achieving accurate testing and streamlined workflows, which ultimately drive progress in healthcare. Regular calibration and are also critical for maintaining accuracy and ensuring safety in laboratory settings.
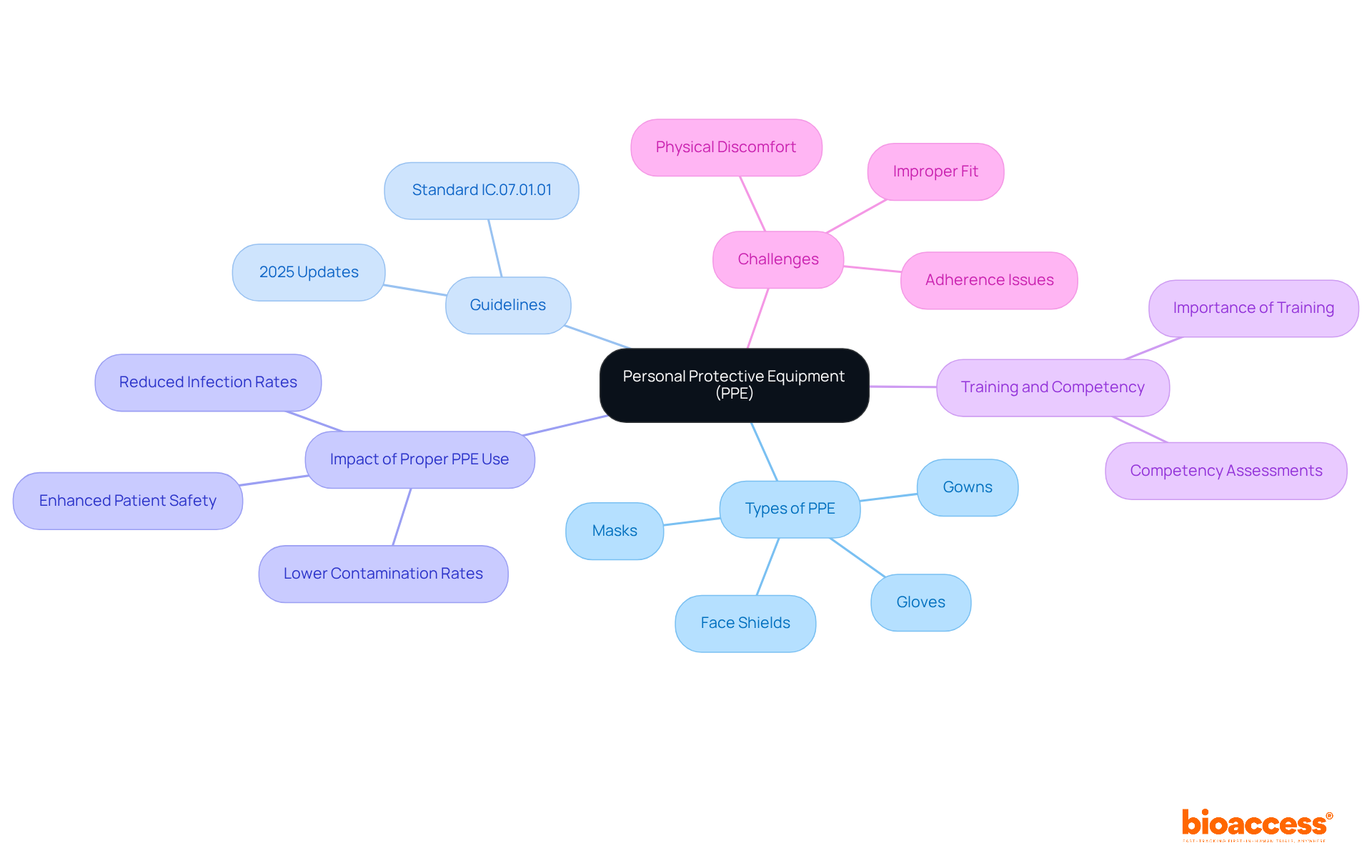
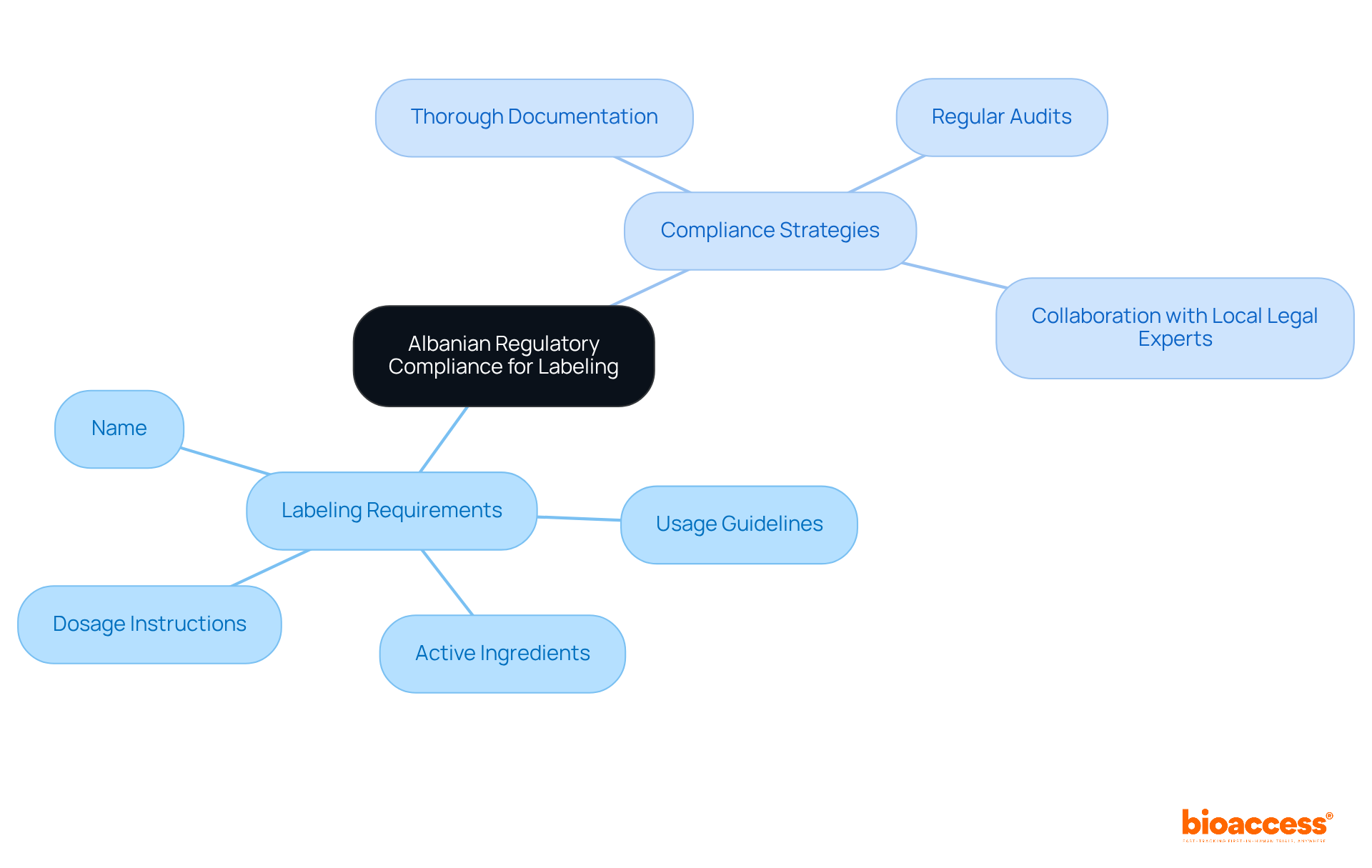
Personal Protective Equipment (PPE): Ensuring Safety in Healthcare
is essential in research settings, as it protects healthcare workers and patients from infectious agents and hazardous substances. Key types of PPE include:
- Gloves
- Masks
- Gowns
- Face shields
Each is designed to effectively mitigate exposure risks.
The latest guidelines for PPE in healthcare, set to take effect in 2025, underscore the necessity of to enhance protection. The updated standard IC.07.01.01 emphasizes , detailing protocols for screening, isolation, and waste management. Experts in assert that significantly reduces infection rates in medical environments. For example, studies indicate that healthcare personnel (HCP) wearing short sleeves experienced less contamination than those in long sleeves, highlighting the critical role of appropriate attire in minimizing risks.
Real-world instances illustrate the impact of stringent in healthcare studies. Facilities that implement report , showcasing the effectiveness of well-structured protocols. In these high-stakes environments, a commitment to PPE not only protects individuals but also upholds the ethical standards of medical research, ensuring that remains a top priority.
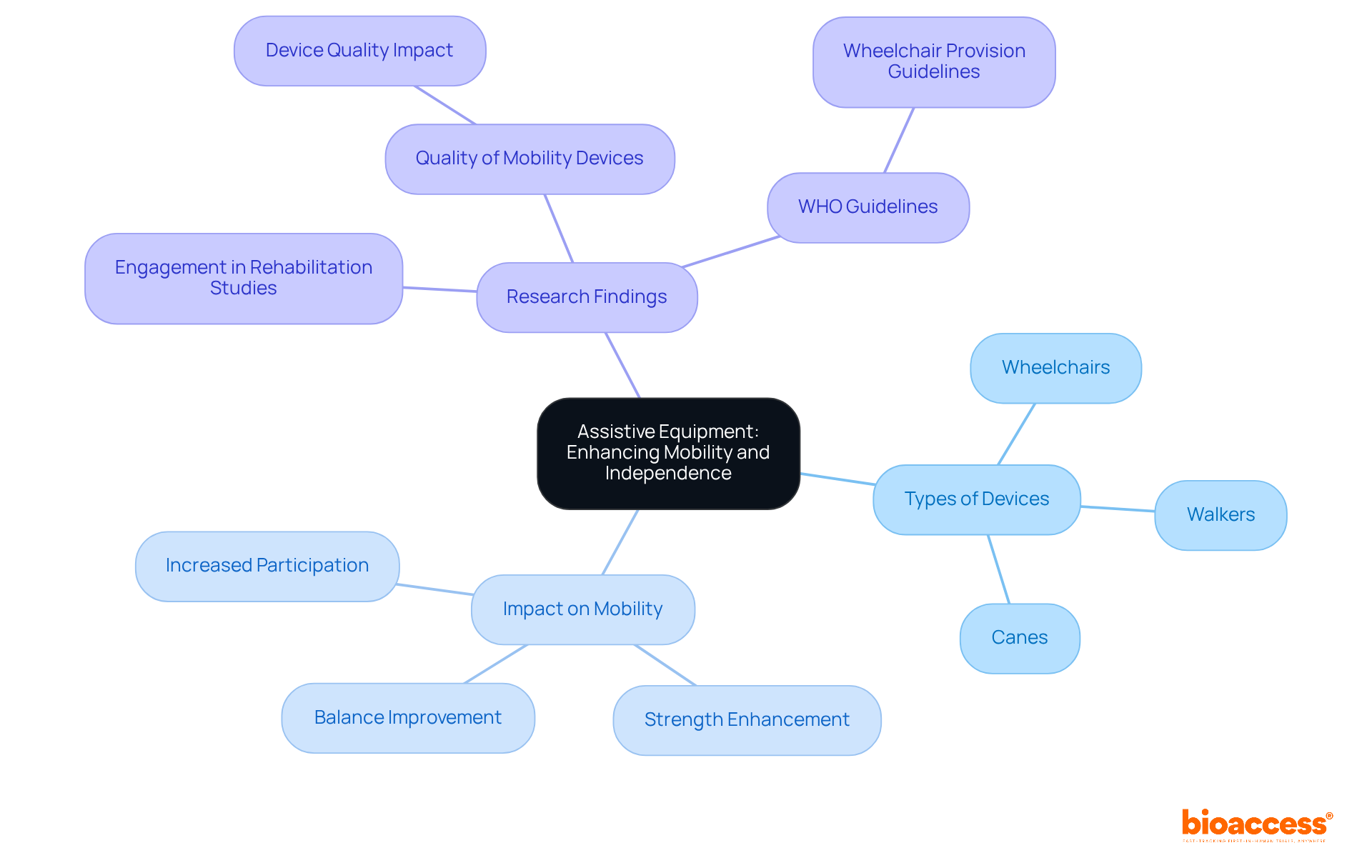
Assistive Equipment: Enhancing Patient Mobility and Independence
Assistive devices, including wheelchairs, walkers, and canes, are pivotal in enhancing mobility and independence, particularly within centered on . These devices empower individuals to participate more actively in their care, facilitating their contributions to during trials.
For example, walkers are essential for helping individuals regain balance and strength, while wheelchairs offer crucial mobility for those with limited physical abilities. The integration of assistive devices into not only leads to improved outcomes for individuals but also enriches the understanding of the effectiveness of various interventions.
Research demonstrates that the timely provision of significantly boosts participant engagement in , ultimately promoting greater independence. Rehabilitation specialists underscore the necessity of for recovery, as they allow individuals to navigate their environments more effectively, thereby encouraging involvement in therapeutic activities.
The importance of in recovery studies is underscored by findings that show . By addressing mobility challenges, these aids not only facilitate physical recovery but also enhance the overall , enabling them to maintain greater autonomy in their daily activities. Furthermore, research indicates that factors related to device quality account for 20% of the variance in participation results, highlighting the critical role of reliable mobility devices in enhancing participant engagement and outcomes in . Additionally, the WHO’s new wheelchair provision guidelines released in June 2023 reinforce existing standards and practices in assistive technology provision, emphasizing the significance of quality and accessibility in .
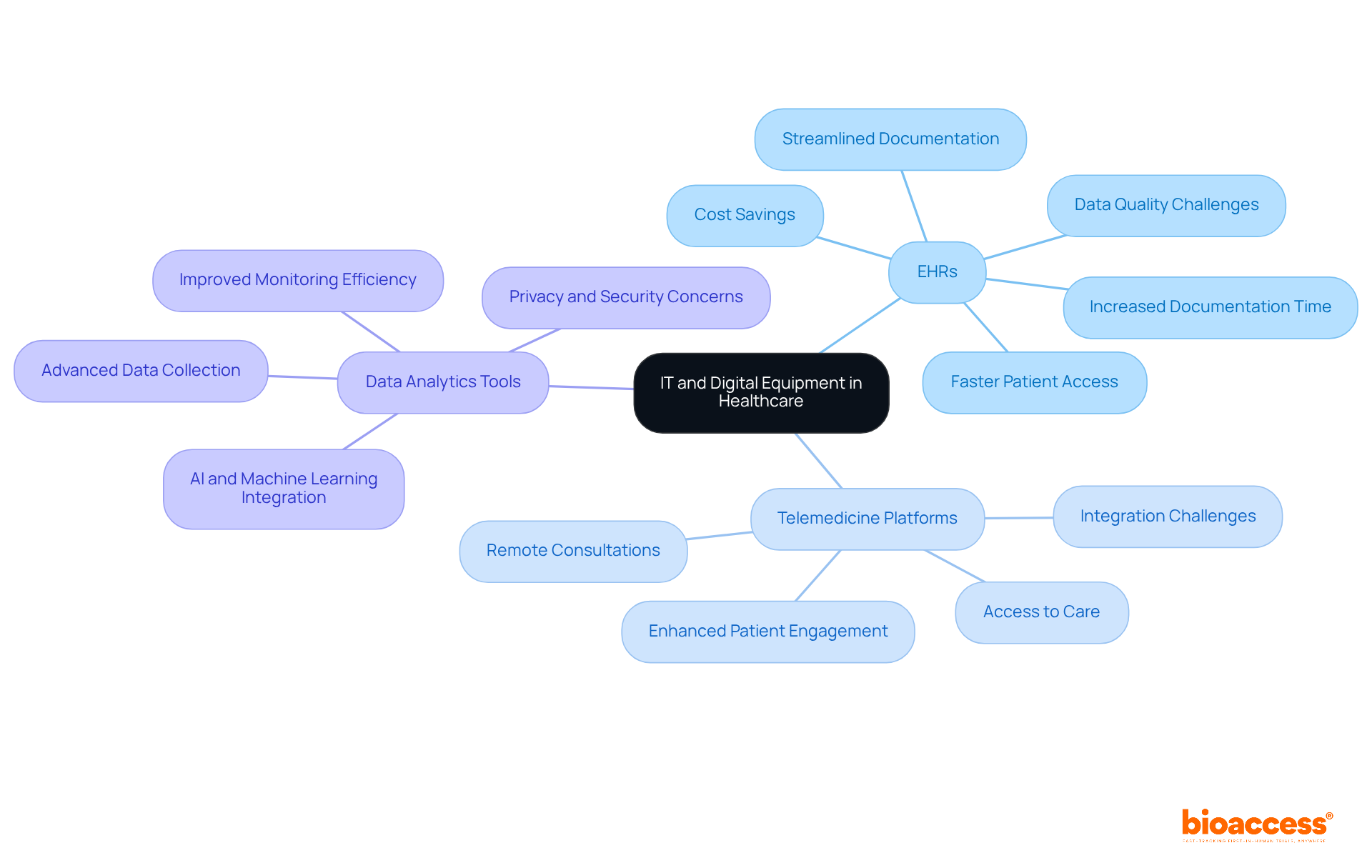
IT and digital medical equipment types are fundamentally transforming healthcare management, particularly within . Technologies such as , , and advanced data analytics tools are crucial for enhancing .
EHRs significantly streamline the documentation process, allowing researchers to access patient information swiftly and securely. This rapid access not only reduces the time spent on administrative duties but also minimizes documentation errors that could affect patient care. Research indicates that EHR integration can in recruitment, screening, and . With bioaccess’s , studies can achieve , resulting in substantial cost savings of $25K per individual, thus addressing common recruitment challenges faced by Medtech and biopharma startups.
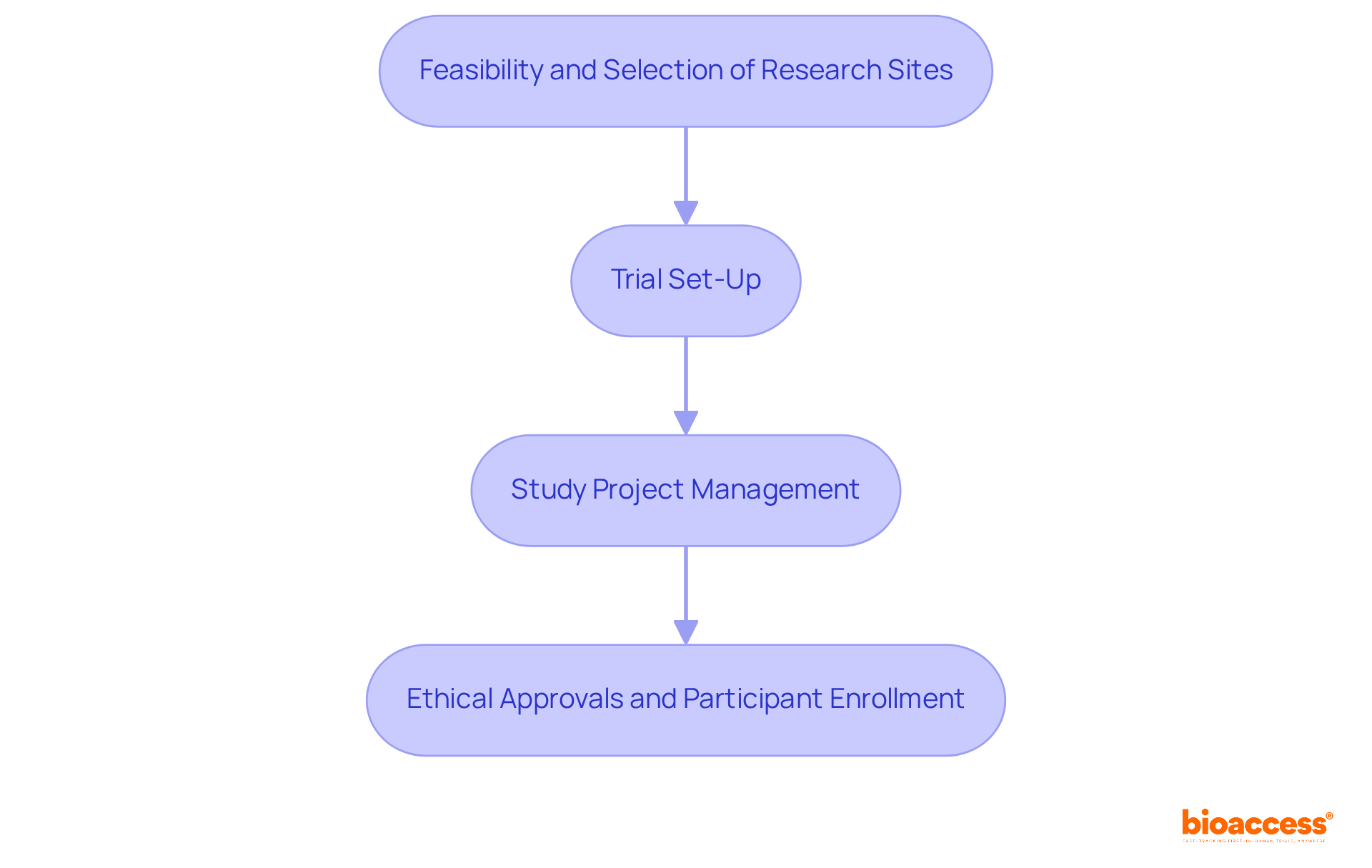
revolutionize patient engagement by enabling remote consultations, thereby expanding access to care. This capability is particularly vital in research studies, where participant involvement may be distributed across various locations. By integrating these digital solutions into medical studies, organizations can improve operational efficiency and elevate the overall quality of patient care, ensuring that innovative treatments reach patients more effectively and swiftly. Furthermore, —including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting—play a critical role in enhancing the success of medical initiatives.
Despite these advancements, such as data quality and system diversity persist, complicating the integration of EMRs into healthcare studies. Addressing these challenges through policy reform and technological innovation is essential for maximizing the impact of EMRs on and healthcare delivery, ultimately contributing to job creation, economic growth, and improved healthcare outcomes.
Conclusion
The significance of medical equipment types in clinical research cannot be overstated; they serve as the backbone for successful trials and patient safety. Each category of equipment, from diagnostic tools to life support apparatus, plays a crucial role in ensuring that research outcomes are reliable and that patient care standards are upheld. The integration of advanced technologies and reliable instruments is essential for facilitating the ethical conduct of clinical studies, ultimately leading to improved healthcare solutions.
Throughout this article, various types of medical equipment have been highlighted, emphasizing their importance in different aspects of clinical research.
- Diagnostic equipment aids in accurate patient assessments
- Treatment devices ensure precise medication delivery
- Monitoring tools track vital signs
- Life support machines sustain critical functions
- Surgical instruments
- Laboratory tools
- Personal protective equipment
- Assistive devices
- IT solutions
All contribute to a comprehensive approach to healthcare management and research efficacy.
As the landscape of clinical research continues to evolve, the call to action is clear: investing in high-quality medical equipment and embracing technological advancements is vital for enhancing patient outcomes and accelerating the development of innovative treatments. Stakeholders in the healthcare sector must prioritize the integration of reliable equipment and robust systems to navigate the complexities of clinical trials effectively, ensuring that the future of healthcare is not only innovative but also safe and effective for all patients involved.
Frequently Asked Questions
What is bioaccess® and what services does it provide?
bioaccess® is a research service provider that operates in Latin America, the Balkans, and Australia, specializing in accelerating clinical research for medical devices and biopharma innovators.
How quickly can bioaccess® secure ethical approvals?
bioaccess® can secure ethical approvals in just 4-6 weeks.
How does bioaccess®’s enrollment rate compare to traditional markets?
bioaccess® achieves enrollment rates that are 50% faster than those in traditional markets.
Why is the agility of bioaccess® important for healthcare delivery?
The agility of bioaccess® is crucial because timely access to various medical equipment types can significantly impact patient outcomes and healthcare delivery.
What experience does bioaccess® have in the field?
bioaccess® has over 15 years of experience and a deep understanding of regulatory nuances and diverse participant demographics that influence research success.
What commitment does bioaccess® have regarding its research practices?
bioaccess® is committed to ethical practices and high-quality research, establishing itself as a leader in the field.
What role do diagnostic equipment play in patient assessment?
Diagnostic equipment, such as stethoscopes and imaging devices, is essential for assessing individual health and gathering information critical for evaluating new medical devices.
How do advancements in diagnostic tools affect medical research?
Recent advancements, including high-resolution imaging technologies, enhance the accuracy and efficiency of evaluations, leading to improved outcomes.
What are the key types of treatment equipment mentioned?
Key treatment equipment includes infusion pumps, elastomeric pumps, and surgical tools, all of which are essential for delivering care during research trials.
How do smart infusion pumps improve medication accuracy?
Smart infusion pumps equipped with dose error reduction software can prevent 28% of infusion errors by alerting clinicians to dosing discrepancies.
What is the significance of surgical tools in medical studies?
Surgical tools are crucial for facilitating procedures integral to the investigative process, and their proper usage can significantly enhance patient safety.
Why is training important for infusion practices?
Training programs are essential for ensuring safe infusion practices, minimizing programming errors, and bolstering safety for patients receiving care.
How does the integration of infusion pumps with healthcare informatics systems benefit clinical studies?
This integration improves tracking and monitoring, significantly reducing medication errors and enhancing patient safety.
List of Sources
- bioaccess®: Accelerating Clinical Research for Medical Devices
- Latin America’s Landscape For Medtech Clinical Trials (https://clinicalleader.com/doc/latin-america-s-landscape-for-medtech-clinical-trials-0001)
- bioaccessla.com (https://bioaccessla.com/blog/understanding-the-cra-position-key-roles-and-impact-in-research)
- bioaccessla.com (https://bioaccessla.com/blog/10-essential-insights-into-pharmacy-regulatory-affairs-for-leaders)
- bioaccessla.com (https://bioaccessla.com/news)
- linkedin.com (https://linkedin.com/posts/bioaccess_understanding-the-invima-approval-process-activity-7277735942495301632-9hnL)
- Diagnostic Equipment: Essential Tools for Patient Assessment
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC9955430)
- icrp.org (https://icrp.org/publication.asp?id=icrp%20publication%20135)
- The Role of AI in Hospitals and Clinics: Transforming Healthcare in the 21st Century – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11047988)
- Medical devices | European Medicines Agency (EMA) (https://ema.europa.eu/en/human-regulatory-overview/medical-devices)
- grundium.com (https://grundium.com/blog/why-high-resolution-imaging-is-critical-for-accurate-diagnoses)
- Treatment Equipment: Key Instruments for Patient Care
- pharmko.com (https://pharmko.com/blog/how-do-infusion-pumps-improve-patient-care)
- Monitoring Equipment: Vital for Patient Health Tracking
- Clinical Trial Monitoring – Biostatistics.ca (https://biostatistics.ca/clinical-trial-monitoring)
- infiniummedical.com (https://infiniummedical.com/vital-signs-monitoring)
- lindushealth.com (https://lindushealth.com/blog/real-time-monitoring-in-clinical-trials-enhancing-accuracy-and-efficiency)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC11258279)
- credevo.com (https://credevo.com/articles/2024/06/25/clinical-trial-monitoring-key-strategies)
- Life Support Equipment: Crucial for Critical Care
- cidrap.umn.edu (https://cidrap.umn.edu/covid-19/life-or-death-experts-guide-pandemic-medical-equipment-decisions)
- qualitymedicalgroup.com (https://qualitymedicalgroup.com/life-support-equipment-vital-importance-healthcare)
- med.umn.edu (https://med.umn.edu/news/worlds-first-ever-ecmo-based-clinical-trial-shows-six-times-higher-survival-rates-among-cardiac-arrest-patients)
- ncbi.nlm.nih.gov (https://ncbi.nlm.nih.gov/books/NBK526044)
- quercusfoundation.org (https://quercusfoundation.org/news/the-importance-of-clinical-technology-and-vital-medical-equipment)
- Surgical Instruments: Fundamental for Medical Procedures
- vizientinc.com (https://vizientinc.com/what-we-do/supply-chain/vizient-viewpoints/surgical-tray-optimization)
- frontiersin.org (https://frontiersin.org/journals/surgery/articles/10.3389/fsurg.2023.1183950/full)
- Laboratory Equipment: Supporting Diagnostics and Research
- bdhme.com (https://bdhme.com/choosing-lab-equipment-manufacturers)
- kanboapp.com (https://kanboapp.com/en/industries/healthcare/revolutionizing-healthcare-the-essential-impact-of-laboratory-equipment-on-precision-and-innovation)
- mms.mckesson.com (https://mms.mckesson.com/content/insights/laboratory-equipment-and-its-impact-on-healthcare)
- des3tech.com (https://des3tech.com/blog/essential-laboratory-equipment-every-scientist-needs-for-accurate-research)
- Personal Protective Equipment (PPE): Ensuring Safety in Healthcare
- Department of Labor finalizes rule on proper fit requirements for personal protective equipment in construction (https://dol.gov/newsroom/releases/osha/osha20241211-0)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC12067588)
- The Joint Commission’s 2024 Infection Control Standards: A Complete Guide for Healthcare Facilities (https://netec.org/2025/01/28/the-joint-commissions-2024-infection-control-standards-a-complete-guide-for-healthcare-facilities)
- academic.oup.com (https://academic.oup.com/cid/article/69/Supplement_3/S165/5568516)
- uab.edu (https://uab.edu/news/research-innovation/study-shows-ppe-was-highly-effective-against-covid-19-in-emergency-department-workers)
- Assistive Equipment: Enhancing Patient Mobility and Independence
- tandfonline.com (https://tandfonline.com/doi/abs/10.3109/17483107.2015.1027295?journalCode=iidt20)
- researchgate.net (https://researchgate.net/publication/274142473_The_impact_of_mobility_assistive_technology_devices_on_participation_for_individuals_with_disabilities)
- who.int (https://who.int/news-room/fact-sheets/detail/assistive-technology)
- sciencedirect.com (https://sciencedirect.com/science/article/abs/pii/S1934148215000581)
- Incidence and dynamics of mobility device use among community-dwelling older adults in the United States – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC12288708)
- IT and Digital Equipment: Transforming Healthcare Management
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC10946197)
- sciencedirect.com (https://sciencedirect.com/science/article/abs/pii/S0168851018301635)
- jamanetwork.com (https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2836552)
- mdpi.com (https://mdpi.com/2072-6694/17/9/1552)
- The Impact of Electronic Health Records on Time Efficiency of Physicians and Nurses: A Systematic Review – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC1205599)
















 influences [long-term complications](https://bioaccessla.com/blog/10-benefits-of-clinical-trial-remote-monitoring-for-research-directors), helping us see the connections that led to improved diabetes management. The center represents the DCCT trial. The branches show how intensive insulin therapy influences long-term complications, helping us see the connections that led to improved diabetes management.](https://images.tely.ai/telyai/znpozpjk-the-center-represents-the-dcct-trial-the-branches-show-how-intensive-insulin-therapy-influences-long-term-complications-helping-us-see-the-connections-that-led-to-improved-diabetes-management.webp)