Introduction
Navigating the complex landscape of biologics in Australia demands a thorough understanding of risk management plans (RMPs) and the essential templates that guide their submission. These templates not only ensure compliance with stringent regulatory standards but also significantly enhance the efficiency of the approval process, which has seen remarkable improvements in recent years.
With the rapid evolution of guidelines and the intricacies involved in RMP submissions, how can sponsors effectively leverage these tools to secure timely market access for their innovative products?
This article explores ten essential RMP submission templates that are pivotal for biologics in Australia, illuminating their benefits and the strategic approaches necessary for successful implementation.
bioaccess® RMP Submission Template: Streamlined Process for Biologics in Australia
The bioaccess® are expertly crafted to (RMPs). This template not only integrates best practices but also aligns with the latest , ensuring that all essential information is presented in a clear and concise manner. By leveraging this template, sponsors can anticipate a more efficient review process. This is particularly significant given that have improved dramatically, with many achieving market access within just 4 to 6 weeks in 2025.
The template features comprehensive sections dedicated to:
- Threat identification
- Evaluation
- Mitigation strategies
These sections are specifically tailored for the Australian regulatory landscape. This strategic approach enhances compliance and operational efficiency, empowering sponsors to templates for biologics in Australia with greater confidence. Successful implementations of this template have proven its effectiveness in accelerating the approval process, thereby facilitating quicker access to innovative biologics for patients in need. As the continues to refine its processes, the adoption of becomes increasingly essential for Medtech innovators striving for timely market entry.
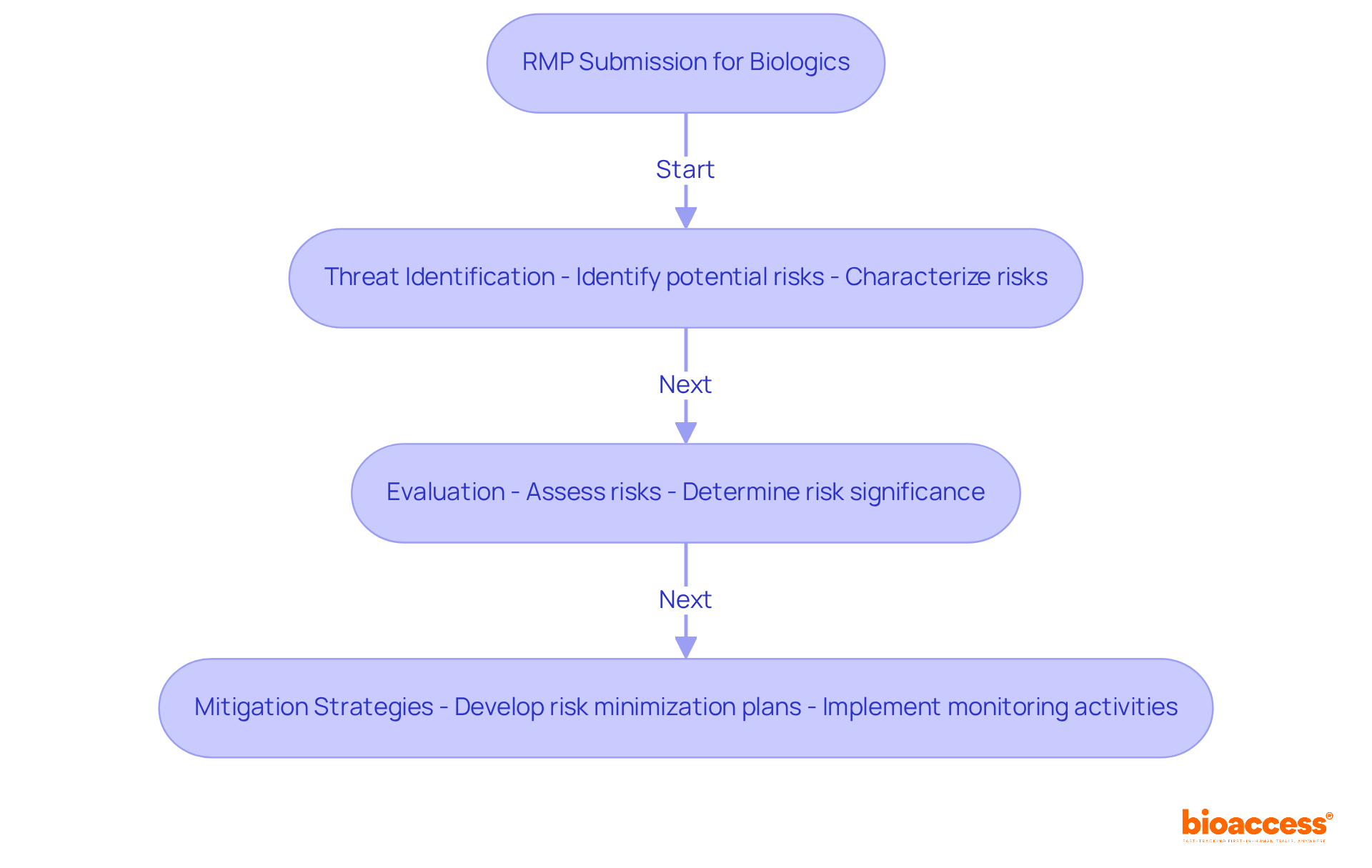
Australian Therapeutic Goods Administration (TGA) RMP Template: Compliance-Focused Framework
The TGA RMP Template serves as a crucial compliance framework that delineates the specific requirements for . This template is vital for sponsors of prescription medicines and biologicals, as it serves as one of the for biologics in Australia, ensuring that all essential components are incorporated into their submissions. Key elements of the TGA RMP Template encompass:
- A thorough
- A robust
- A comprehensive
By adhering strictly to this template, sponsors not only demonstrate their commitment to but also enhance their , significantly streamlining the .
Notably, the average review time for is around 4 to 6 weeks, highlighting the efficiency of the process when all requirements are met. Specialist insights reveal that a well-organized RMP can lead to successful submissions, as evidenced by numerous management plans positively assessed by the TGA. Furthermore, staying abreast of the latest and integrating any necessary changes into the RMP is essential for maintaining compliance and ensuring the safety of biological products throughout their lifecycle.
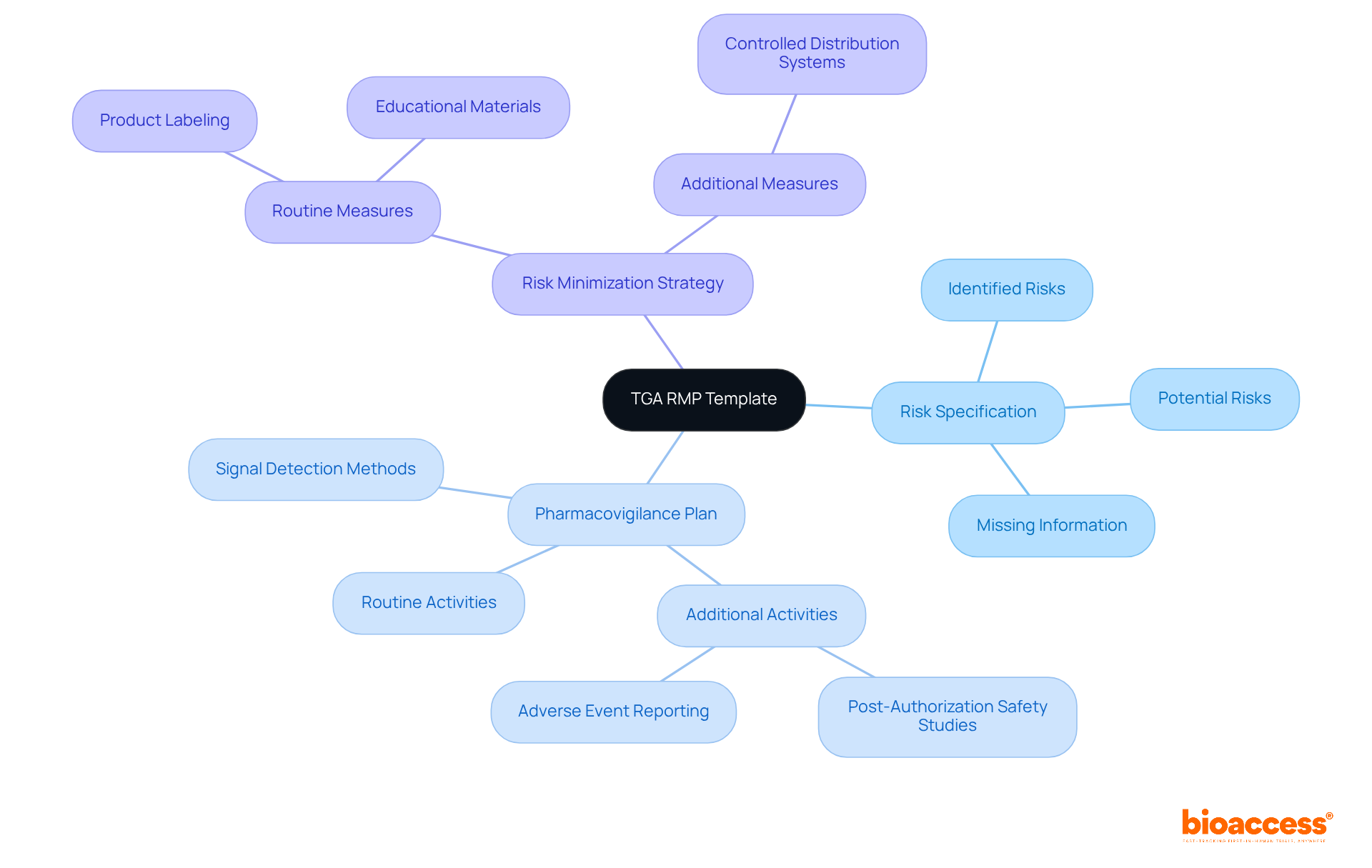
European Medicines Agency (EMA) RMP Template: International Standards for Biologics
The stands as the gold standard for across Europe, serving as a crucial reference for international submissions. When companies apply for , they must submit a (RMP) to the European Medicines Agency (EMA), underscoring the necessity of utilizing the . This comprehensive template encompasses vital sections for:
All adhering to the modular format outlined in GVP Module V. By aligning their submissions with the EMA’s RMP Template, sponsors not only fulfill European regulatory requirements but also bolster their credibility in the global market. This strategic alignment proves especially advantageous for companies aiming to penetrate European markets while ensuring compliance with Australian regulations, ultimately facilitating smoother international submissions and the effective use of to enhance the overall success of their products.
Moreover, it is essential to recognize that RMPs are that require revisions as new safety information emerges, reflecting the evolving nature of risk management. As Safwan Azeem aptly noted, ‘A well-crafted Risk Management Plan (RMP) is no longer a – it’s a foundational requirement for any medicinal product seeking approval in today’s global market.’ This statement reinforces the critical role of RMPs in navigating the complexities of regulatory landscapes.
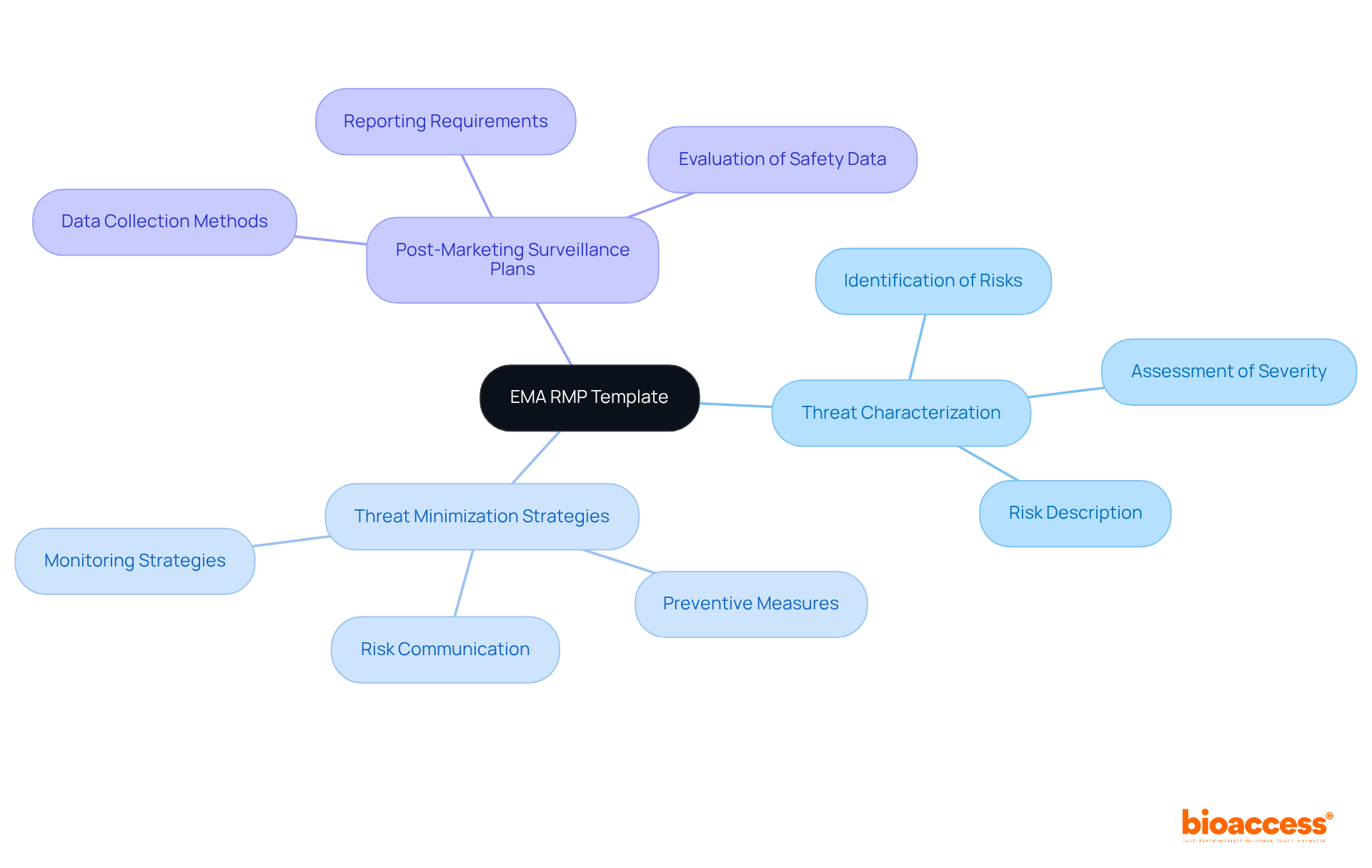
World Health Organization (WHO) RMP Template: Global Guidelines for Biologics
The provides . It underscores the importance of a , which includes:
- Identification
- Assessment
By following the WHO’s recommendations, sponsors can ensure their products comply with and meet . This compliance is crucial for , ultimately leading to improved .
Risk Evaluation and Mitigation Strategy (REMS) Template: Essential for Biologics Safety
The serves as a crucial framework for sponsors of high-risk biologics, delineating . This comprehensive template encompasses , monitoring, and evaluation, ensuring that all stakeholders remain well-informed and actively engaged in the protection process. By implementing a robust REMS, sponsors not only bolster but also ensure compliance with , thereby fostering trust in their products.
Effective within the REMS framework are vital, as they promote transparency and instill confidence among healthcare providers and patients alike. The timeline for implementing REMS for can vary; however, early planning during the is recommended to streamline the process and avert delays. Recent updates in for biologics underscore the evolving landscape of regulatory expectations, highlighting the necessity for .
Australian Clinical Trials Notification (CTN) RMP Template: Localized Compliance for Biologics
The in Australia serve a crucial purpose for sponsors conducting . It outlines the necessary that must be submitted as part of the process. Essential elements include:
- Protection monitoring strategies
- Reporting responsibilities
By utilizing the for , sponsors can ensure their comply with Australian regulations, which facilitates smoother approvals and enhances the well-being of trial participants.
Stakeholder Engagement RMP Template: Enhancing Communication for Biologics
The for highlight the critical role of communication in managing biologics. This template delineates strategies for engaging healthcare professionals, regulatory bodies, and patients, ensuring that all stakeholders remain well-informed and actively participate in . Effective communication fosters clarity and trust-elements essential for successfully navigating uncertainties. By adopting robust , sponsors can significantly boost the effectiveness of their , ultimately leading to improved .
Research shows that companies with strong are 30% more likely to succeed with new products. A well-structured communication strategy not only keeps stakeholders updated on RMP progress but also encourages their involvement by utilizing for , enhancing project results and mitigating challenges associated with biological products. Furthermore, effective necessitates clear, measurable to assess the success of . Understanding the , including the oversight from authorities like INVIMA in Colombia-recognized as a Level 4 health authority by PAHO/WHO-can further amplify the effectiveness of these engagement strategies within the Australian context.
Post-Marketing Surveillance RMP Template: Monitoring Biologics After Approval
The are essential for tracking the safety and efficacy of biologics after approval. This template, which is one of the , outlines , prompt adverse event reporting, and regular assessment updates. Given that the average time for is critical for patient well-being, a robust enables sponsors to swiftly identify potential issues and implement necessary risk mitigation measures.
Continuous oversight is vital for ensuring the . It allows for the adjustment of strategies based on real-world data, reinforcing the importance of effective monitoring. Not only do these strategies enhance product safety, but they also foster trust with regulatory bodies, ultimately leading to improved patient outcomes. Furthermore, bioaccess provides comprehensive that are crucial in supporting these efforts. These services encompass:
- Site selection
- Compliance reviews
- Trial setup
- Import permits
- Project management
- Reporting
Ensuring meticulous management of all aspects of the clinical trial process.
Risk Identification and Assessment RMP Template: Key for Biologics Development
The , including the , serve as a vital resource for sponsors involved in the development of . This template not only facilitates a , such as security concerns and efficacy issues, but also outlines effective strategies for evaluating these uncertainties. By utilizing , sponsors are equipped to proactively address the , ultimately enhancing the security and effectiveness of their .
Moreover, are essential. These revisions guide and align with evolving regulatory standards. This structured approach not only ensures compliance but also reinforces a commitment to . In a landscape where collaboration is key, utilizing the positions sponsors to navigate the complexities of with confidence.
Continuous Improvement RMP Template: Evolving Strategies for Biologics Safety
The RMP Submission Templates for emphasize the critical need for ongoing assessment and enhancement of management strategies for biological products. This template delineates mechanisms for the , ensuring that new data and emerging information are seamlessly integrated. As W. Edwards Deming aptly noted, ” Improvement is not compulsory; it’s voluntary. But to survive, we must learn.” By , sponsors can adapt their strategies to meet evolving challenges, thereby bolstering the reliability and efficacy of their biologics, which is essential for adhering to in Australia.
Frequent updates to are not just beneficial; they are essential. Such updates facilitate swift responses to new insights and regulatory demands, ultimately promoting a proactive management strategy. For example, the case study on the “Human Side of Change Management” illustrates how organizations that prioritize can adeptly . Furthermore, it is advisable that , particularly those involving RMP submission templates for , be updated at least biannually to ensure alignment with the latest regulatory expectations and safety data.
Conclusion
The significance of effective RMP submission templates for biologics in Australia is paramount. These templates not only ensure compliance with regulatory standards but also streamline the submission process, ultimately granting patients quicker access to innovative treatments. By implementing structured frameworks like the bioaccess® RMP submission template and the TGA RMP Template, sponsors can navigate the complexities of regulatory requirements with enhanced ease and confidence.
This article has presented key insights into various RMP templates, each designed to address specific regulatory needs and challenges. From the comprehensive guidelines provided by the WHO and EMA to the localized compliance frameworks essential for Australian clinical trials, these templates are indispensable tools for sponsors. They encompass critical elements such as:
- Risk identification
- Stakeholder engagement
- Post-marketing surveillance
All vital for ensuring the safety and efficacy of biologics.
As the biologics landscape evolves, embracing these RMP submission templates is crucial for upholding high standards of patient safety and regulatory compliance. Companies must prioritize the integration of these templates into their processes, fostering a culture of continuous improvement and proactive risk management. By doing so, they not only enhance their chances of successful market entry but also contribute to the overall advancement of healthcare innovation in Australia and beyond.
Frequently Asked Questions
What is the purpose of the bioaccess® RMP submission template for biologics in Australia?
The bioaccess® RMP submission template is designed to streamline the submission of risk management plans (RMPs) for biologics in Australia, integrating best practices and aligning with the latest regulatory requirements to enhance the efficiency of the review process.
What sections are included in the bioaccess® RMP submission template?
The template includes comprehensive sections dedicated to threat identification, evaluation, and mitigation strategies, specifically tailored for the Australian regulatory landscape.
How has the approval time for biological products in Australia changed recently?
Average approval times for biological products in Australia have improved significantly, with many achieving market access within just 4 to 6 weeks in 2025.
What is the significance of the TGA RMP Template?
The TGA RMP Template serves as a compliance framework that outlines the specific requirements for management plans in Australia, ensuring all essential components are included in submissions for prescription medicines and biologicals.
What are the key elements of the TGA RMP Template?
Key elements of the TGA RMP Template include a thorough risk specification, a robust pharmacovigilance plan, and a comprehensive risk minimization strategy.
What is the average review time for RMP submission templates for biologics in Australia?
The average review time for RMP submission templates for biologics in Australia is around 4 to 6 weeks.
How does the EMA RMP Template relate to international submissions?
The EMA RMP Template is considered the gold standard for risk management plans in Europe and is essential for companies applying for marketing authorization, ensuring compliance with European regulatory requirements.
What sections are included in the EMA RMP Template?
The EMA RMP Template includes sections for threat characterization, threat minimization strategies, and post-marketing surveillance plans, adhering to the modular format outlined in GVP Module V.
Why is it important for RMPs to be dynamic documents?
RMPs are dynamic documents that require revisions as new safety information emerges, reflecting the evolving nature of risk management and ensuring ongoing compliance and safety for biological products.
What is the foundational requirement for any medicinal product seeking approval in today’s global market?
A well-crafted Risk Management Plan (RMP) is considered a foundational requirement for any medicinal product seeking approval in today’s global market.
List of Sources
- bioaccess® RMP Submission Template: Streamlined Process for Biologics in Australia
- Submitting risk management plans for medicines and biologicals (https://tga.gov.au/resources/guidance/submitting-risk-management-plans-medicines-and-biologicals)
- Product Information safety updates – July 2025 (https://tga.gov.au/news/safety-updates/product-information-safety-updates-july-2025)
- Australia’s TGA updates risk management plan requirements for new drugs and biologics (https://bioworld.com/articles/349899-australia-s-tga-updates-risk-management-plan-requirements-for-new-drugs-and-biologics)
- Australian Therapeutic Goods Administration (TGA) RMP Template: Compliance-Focused Framework
- Submitting risk management plans for medicines and biologicals (https://tga.gov.au/resources/guidance/submitting-risk-management-plans-medicines-and-biologicals)
- What is Risk Management Plan in Pharma, RMP Guide (https://masuuglobal.com/what-is-a-risk-management-plan-rmp)
- European Medicines Agency (EMA) RMP Template: International Standards for Biologics
- lsacademy.com (https://lsacademy.com/en/fda-vs-ema-key-regulatory-differences-for-pharmaceuticals)
- Risk Management Plans (RMPs): Comprehensive Pharmacovigilance Guide (https://ccrps.org/clinical-research-blog/risk-management-plans-rmps-comprehensive-pharmacovigilance-guide)
- Risk management plans | European Medicines Agency (EMA) (https://ema.europa.eu/en/human-regulatory-overview/marketing-authorisation/pharmacovigilance-marketing-authorisation/risk-management/risk-management-plans)
- World Health Organization (WHO) RMP Template: Global Guidelines for Biologics
- Submitting risk management plans for medicines and biologicals (https://tga.gov.au/resources/guidance/submitting-risk-management-plans-medicines-and-biologicals)
- Risk Management Plans (RMPs): Comprehensive Pharmacovigilance Guide (https://ccrps.org/clinical-research-blog/risk-management-plans-rmps-comprehensive-pharmacovigilance-guide)
- Risk management plans | European Medicines Agency (EMA) (https://ema.europa.eu/en/human-regulatory-overview/marketing-authorisation/pharmacovigilance-marketing-authorisation/risk-management/risk-management-plans)
- 50 Risk Management Quotes: Wisdom for Smart Decision-making | ITD World (https://itdworld.com/blog/leadership/risk-management-quotes)
- Statistical Tools for Biologics Stability Testing (https://bioprocessintl.com/qa-qc/comprehensive-stability-assessment-of-biotechnological-products-degradation-mechanisms-statistical-tools-and-regulatory-compliance-part-1)
- Risk Evaluation and Mitigation Strategy (REMS) Template: Essential for Biologics Safety
- Breaking Down Risk Evaluation and Mitigation Strategies (REMS) – Pearl Pathways (https://pearlpathways.com/breaking-down-risk-evaluation-and-mitigation-strategies-rems)
- REMS programs explained: A guide for sponsors | PPD (https://ppd.com/blog/rems-programs-explained-guide-for-sponsors)
- FDA Guidance for Effective Risk Evaluation & Mitigation Strategy | Rho (https://rhoworld.com/risk-evaluation-and-mitigation-strategies-fda-guidances-for-assessing-effectiveness)
- Adaptation for Regulatory Application: A Content Analysis of FDA Risk Evaluation and Mitigation Strategies Assessment Plans (2014–2018) Using RE-AIM – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC7052173)
- Use of Risk Evaluation and Mitigation Strategies by the US Food and Drug Administration, 2008-2019 – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC12543398)
- Stakeholder Engagement RMP Template: Enhancing Communication for Biologics
- Stakeholder Engagement Effectiveness Statistics (https://zoetalentsolutions.com/stakeholder-engagement-effectiveness)
- TOP 25 STAKEHOLDER QUOTES (of 56) | A-Z Quotes (https://azquotes.com/quotes/topics/stakeholder.html)
- 70 Quotes on Risk Taking and Smart Decision-Making (https://deliberatedirections.com/risk-management-quotes)
- 50 Risk Management Quotes: Wisdom for Smart Decision-making | ITD World (https://itdworld.com/blog/leadership/risk-management-quotes)
- Post-Marketing Surveillance RMP Template: Monitoring Biologics After Approval
- Post-marketing surveillance framework of cell and gene therapy products in the European Union, the United States, Japan, South Korea and China: a comparative study – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11438358)
- 7 Key Insights on Post-Market Surveillance under ANVISA | bioaccess® (https://bioaccessla.com/blog/7-key-insights-on-post-market-surveillance-under-anvisa)
- Experts’ Views on FDA Regulatory Standards for Drug and High-Risk Medical Devices: Implications for Patient Care – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC9708961)
- Why Pharmacovigilance Is More Critical Than Ever (https://news-medical.net/life-sciences/Why-Pharmacovigilance-Is-More-Critical-Than-Ever.aspx)
- Postmarketing safety-related regulatory actions for new therapeutic biologics approved in the U.S. 2002 – 2014: Similarities and differences with new molecular entities – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC8159207)
- Risk Identification and Assessment RMP Template: Key for Biologics Development
- What is Risk Management Plan in Pharma, RMP Guide (https://masuuglobal.com/what-is-a-risk-management-plan-rmp)
- 50 Risk Management Quotes: Wisdom for Smart Decision-making | ITD World (https://itdworld.com/blog/leadership/risk-management-quotes)
- Risk Management Plans (RMPs): Comprehensive Pharmacovigilance Guide (https://ccrps.org/clinical-research-blog/risk-management-plans-rmps-comprehensive-pharmacovigilance-guide)
- A Science-Based Methodology Framework for the Assessment of Combination Safety Risks in Clinical Trials – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10205853)
- Submitting risk management plans guidance document: Overview – Canada.ca (https://canada.ca/en/health-canada/services/drugs-health-products/reports-publications/medeffect-canada/guidance-submission-risk-management-plans-policy-overview.html)
- Continuous Improvement RMP Template: Evolving Strategies for Biologics Safety
- 85 Quotes – Operational Excellence and Continuous Improvement | Rever (https://reverscore.com/85-quotes-operational-excellence-and-continuous-improvement)
- A Novel Metric for Continuous Improvement During Stage Three | BioPharm International (https://biopharminternational.com/view/novel-metric-continuous-improvement-during-stage-three)
- 26 Quotes About Improving Processes That Will Inspire Change (https://niagarainstitute.com/blog/quotes-improving-processes)
- goretro.ai (https://goretro.ai/post/continuous-improvement-quotes)
- blog.kainexus.com (https://blog.kainexus.com/continuous-improvement/continuous-improvement-leadership-quotes)

Leave a Reply