Introduction
In the intricate landscape of clinical research, the success of a study hinges on a well-structured design that encompasses a multitude of critical elements. From defining the right target population to ensuring robust statistical analysis, each component plays a pivotal role in shaping the outcomes of clinical trials. This article delves into ten key elements of clinical study design, offering insights into best practices that not only enhance research efficiency but also uphold ethical standards. As the demand for innovative therapies grows, researchers must effectively navigate these complexities to ensure meaningful results and regulatory compliance.
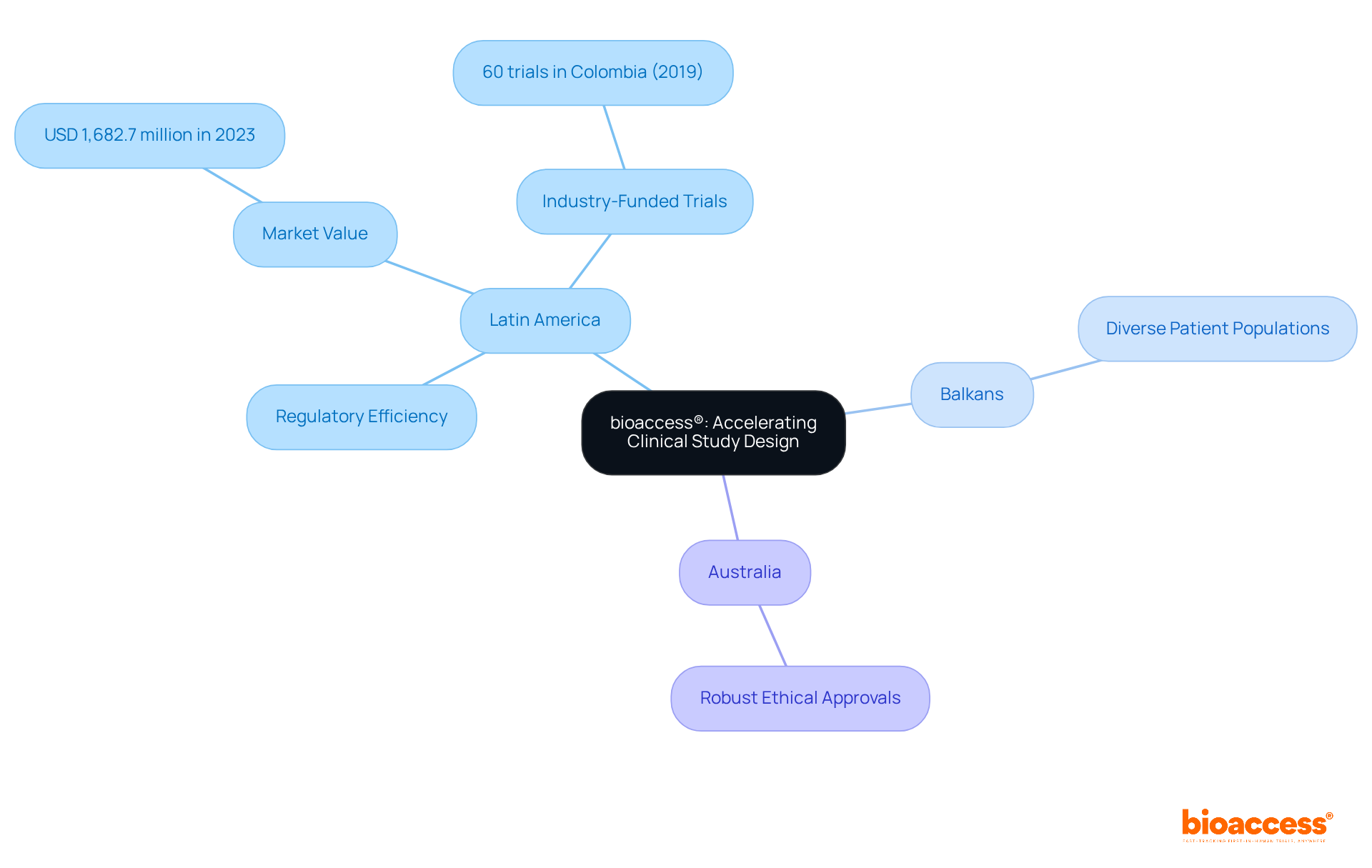
bioaccess®: Accelerating Clinical Study Design with Global Agility
bioaccess® strategically positions itself across Latin America, the Balkans, and Australia, offering unparalleled flexibility in . By capitalizing on , the diverse patient populations in the Balkans, and Australia’s robust ethical approval systems, bioaccess® significantly reduces the time required to initiate and complete . This global-first approach not only but also enhances the overall quality of .
Notably, research conducted in Colombia has successfully attracted , with the country hosting 60 industry-funded trials in 2019 alone. Additionally, the Latin American generated USD 1,682.7 million in 2023, underscoring its increasing significance in the global arena.
These advancements underscore the critical role of in achieving successful s, positioning bioaccess® as an essential partner for Medtech and Biopharma innovators in pursuit of timely breakthroughs.
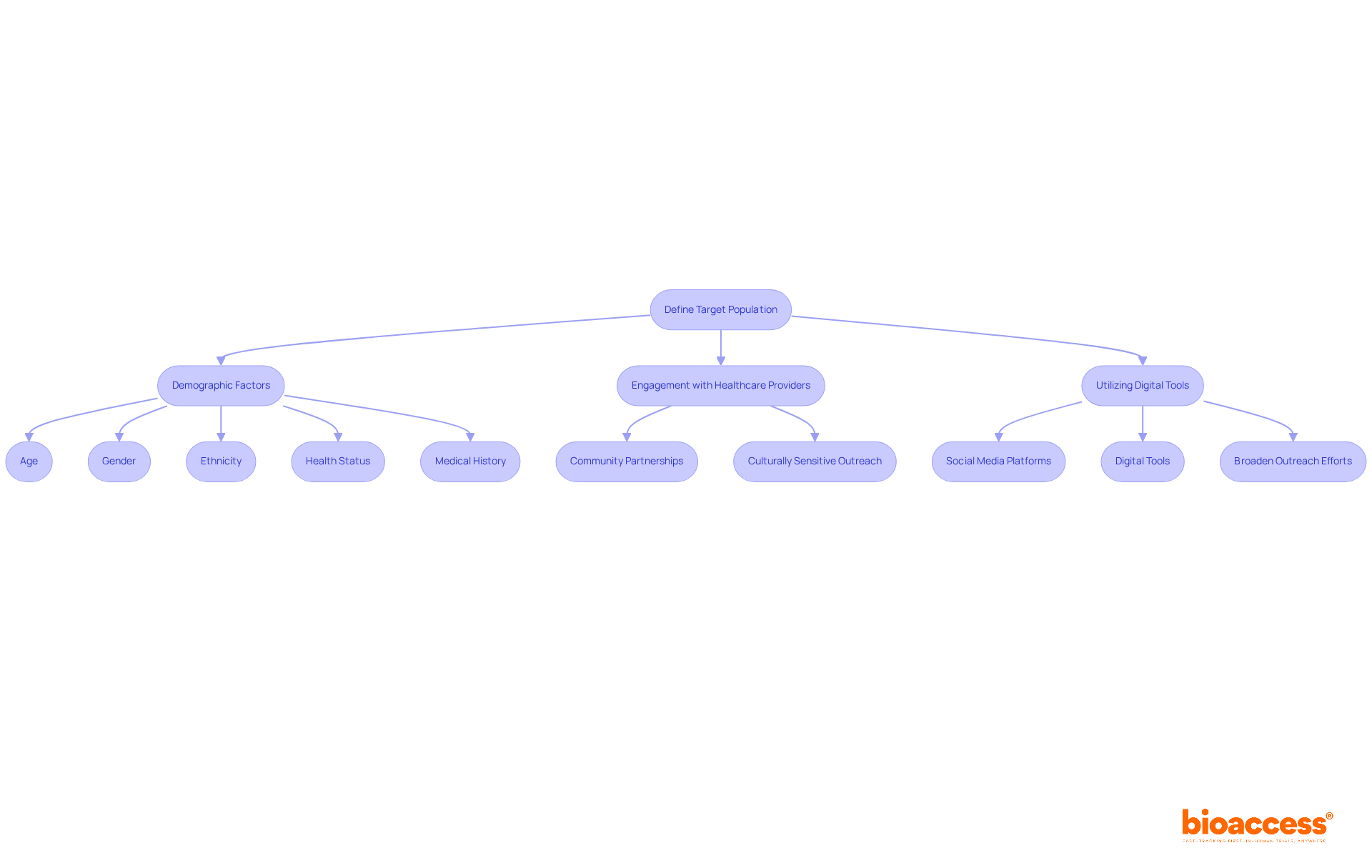
Target Population: Defining the Right Participants for Your Study
Defining the is crucial for aligning participant traits with the research’s objectives. This process includes such as age, gender, ethnicity, health status, and relevant medical history. In , a precisely defined not only enhances the relevance of findings but also ensures that results can be generalized to a broader patient demographic.
Engaging local healthcare providers is vital for implementing effective , particularly in areas with diverse patient populations. For instance, utilizing can significantly improve recruitment rates. Furthermore, employing can broaden outreach efforts, facilitating connections with potential participants.
By concentrating on these strategies, researchers can more effectively manage the within their , ultimately resulting in more .
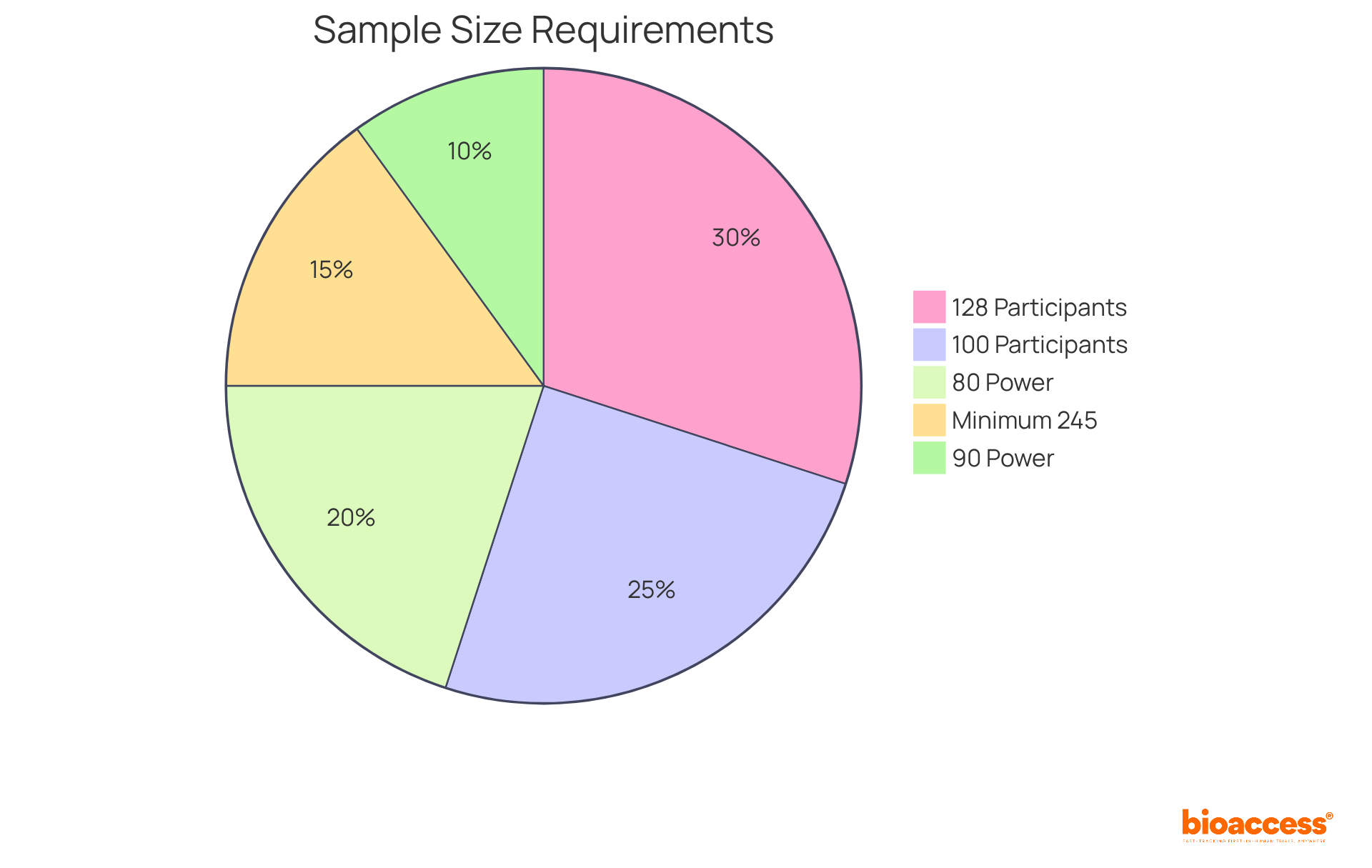
Sample Size: Calculating the Right Number for Statistical Power
Establishing a suitable is vital in medical research, as it directly affects the research’s power—the likelihood of accurately identifying a genuine effect. A larger significantly enhances the likelihood of identifying meaningful differences, while a smaller sample may yield inconclusive results, leading to potential misinterpretations of the data. Research suggests that a is frequently advised to guarantee sufficient power, especially in where the occurrence of outcomes can differ significantly. Additionally, for a medium effect size, a total of 128 participants is required to achieve , which is a common standard among regulators.
is typically set at 80%, meaning there is an 80% chance of detecting an effect if it exists. This threshold is essential for ensuring that the research can reliably support its hypotheses. The most common alpha level chosen is 0.05, indicating a 5% risk of a , where a true null hypothesis is incorrectly rejected. In practical terms, researchers must carefully balance the against the expected effect size and the desired power to avoid underpowered analyses that fail to detect significant treatment effects. Alaa Althubaiti emphasizes that determining the necessary should not be viewed as a solution to an inquiry, highlighting the complexities involved in this process.
Utilizing can greatly facilitate this process, allowing researchers to perform complex calculations efficiently. Tools such as G-Power, OpenEpi, PASS, and R offer valuable resources for estimating s based on different statistical analyses, ensuring that research is sufficiently powered to identify significant effects. Ultimately, a well-calculated not only enhances the validity of findings but also upholds by minimizing unnecessary risks to participants. Ethical committees view appropriate calculation as a prerequisite for the approval of , emphasizing its significance in safeguarding participants and maintaining the integrity of the research.
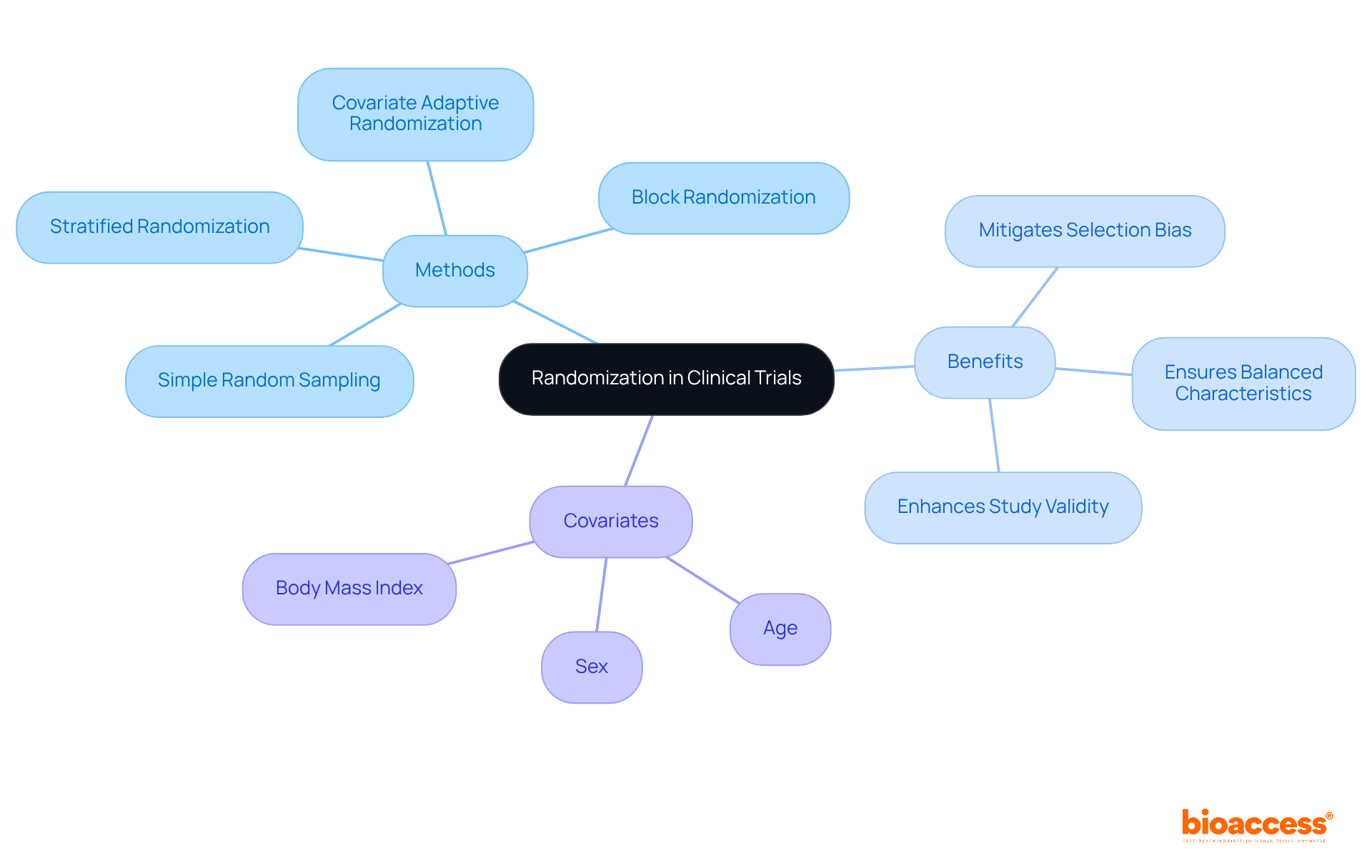
Randomization: Ensuring Fairness in Clinical Trials
is a cornerstone in , as it crucially ensures that participants are assigned to different groups without bias. This objective can be achieved through a variety of methods, including:
- Simple
By providing every participant with an equal opportunity for group assignment, researchers effectively mitigate , thereby enhancing the validity of the results. For example, stratified facilitates the control of specific covariates, such as:
- Age
- Sex
- Body mass index
This ensures balanced characteristics across . This approach proves particularly beneficial in larger studies, where the complexity of can lead to imbalances if not properly managed. Evidence indicates that inadequate can result in an overestimation of treatment effects by as much as 40% compared to studies employing proper , underscoring the critical need for robust .
Furthermore, methods like covariate adaptive , which adjusts group assignments based on prior , have demonstrated a capacity to produce less imbalance than traditional methods. Additionally, guarantees equal sample sizes among , further reinforcing the validity of study results. Ultimately, the application of within is indispensable for achieving reliable and interpretable outcomes in health research.
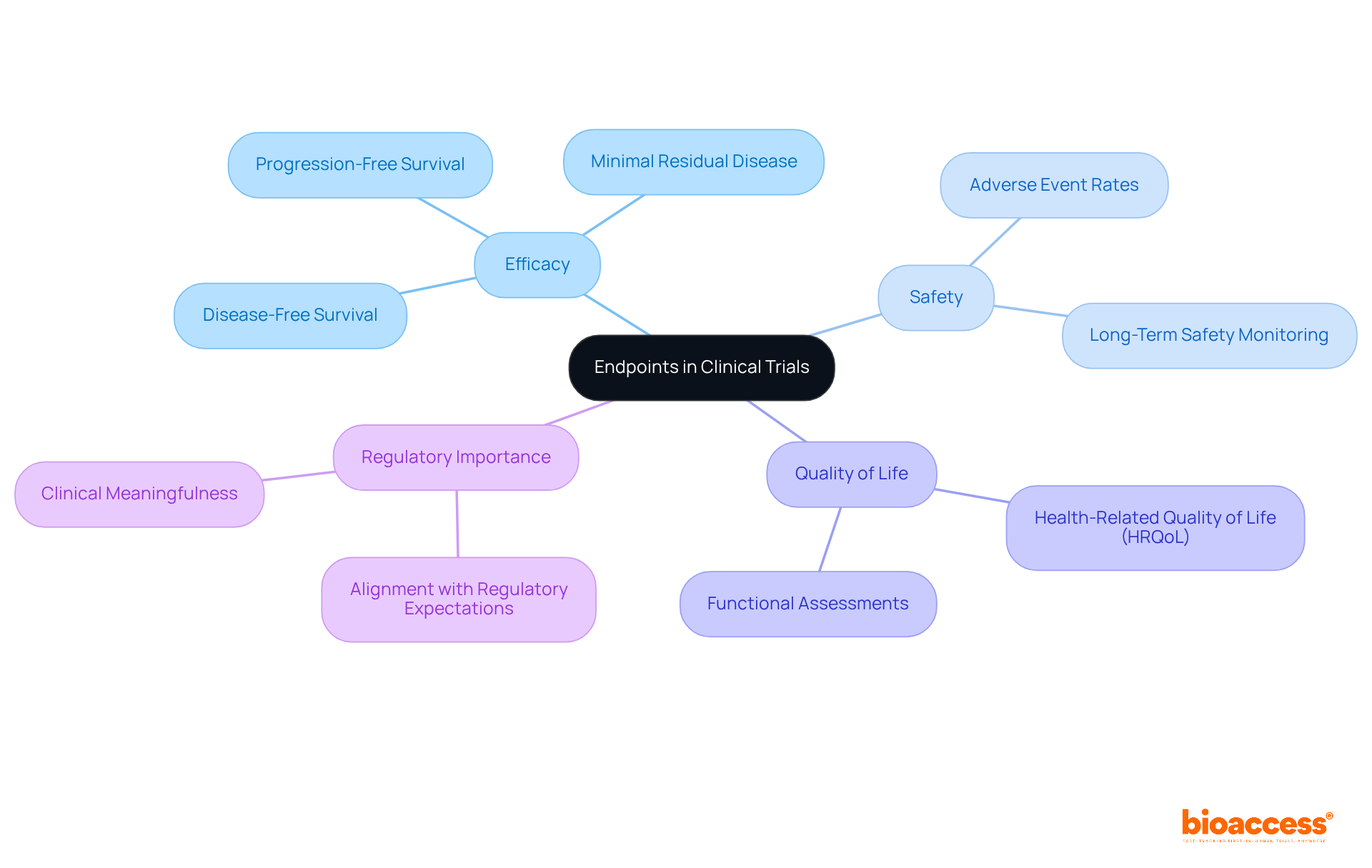
Endpoints: Defining Success in Clinical Trials
Endpoints represent the specific results that a research study aims to assess, encompassing efficacy, safety, and quality of life. Clearly defining at the outset is crucial for steering the study’s focus and analysis. For instance, successful often demonstrate clear endpoints that align with regulatory expectations, thereby enhancing their likelihood of approval.
Recent trends indicate a shift toward the development of endpoints, such as and minimal residual disease, which more effectively reflect therapeutic value, especially in complex conditions like Alzheimer’s disease, where conventional measures may fall short. As emphasized in various studies, the significance of cannot be overstated; they not only direct the inquiry process but also inform payer reimbursement decisions.
The FDA underscores that the hinges on the relevance of the domains assessed and the magnitude of treatment effects. Consequently, ensuring that endpoints are clinically relevant and measurable is vital, as they ultimately determine the being tested.
With completion rates for reported at 84.9%, the emphasis on is more crucial than ever in navigating the complexities of .
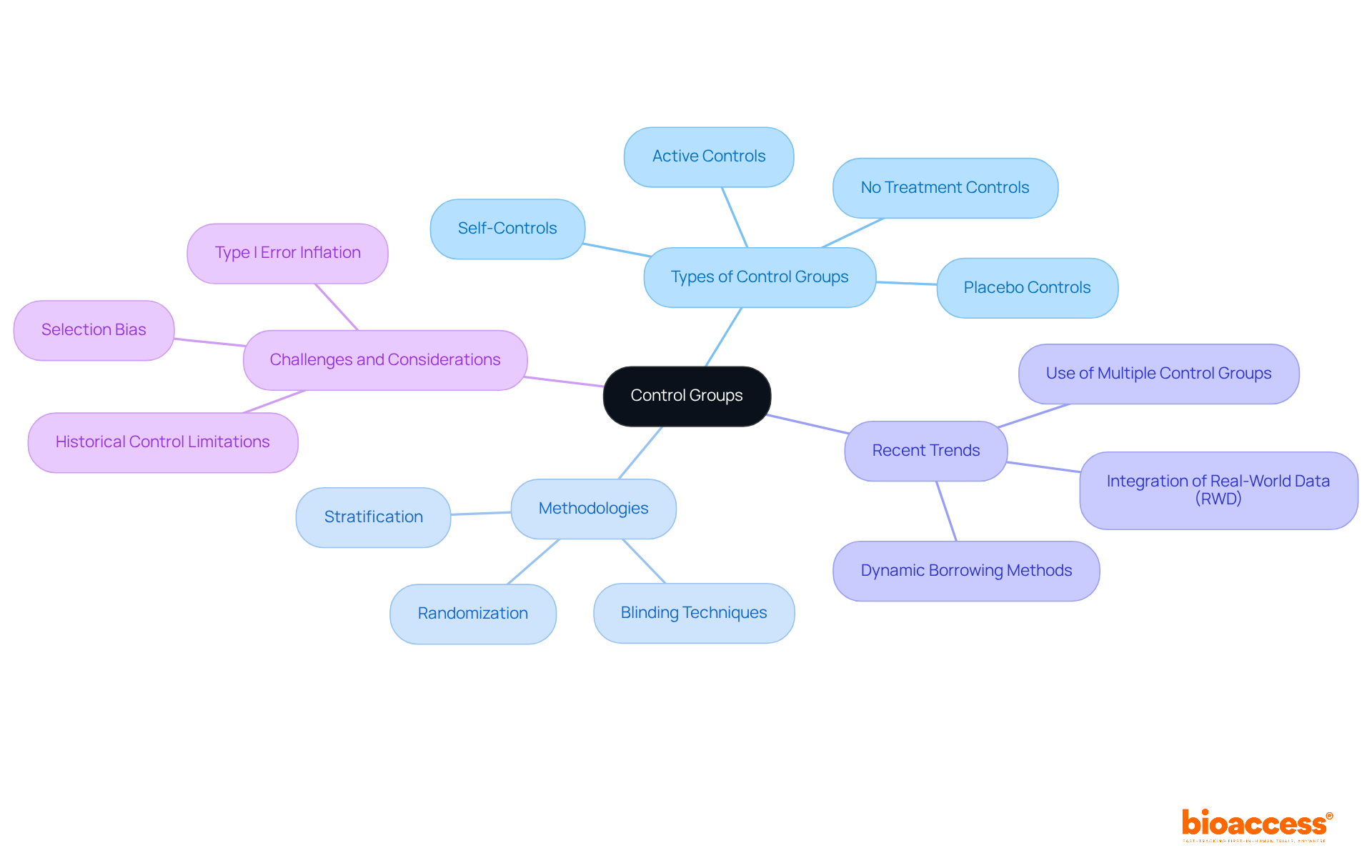
Control Groups: Establishing Baselines for Comparison
, enabling researchers to evaluate the effects of an intervention through comparison with a group that does not receive treatment. This comparison can be established via various methodologies, such as —where participants receive an inert substance—or , where they are given a standard treatment known to be effective. The selection of an appropriate control group must align with the research objectives and the specific nature of the intervention, ensuring that results are interpretable and meaningful.
Recent trends indicate a growing preference for employing , which enhances the reliability of results by minimizing potential biases associated with reliance on a single control group. For instance, research has shown that utilizing both placebo and active controls can provide a more comprehensive understanding of . Moreover, the integration of is becoming increasingly prevalent, facilitating more accurate baseline comparisons and improved patient recruitment strategies.
Incorporating effective control group methodologies is paramount for preserving the integrity of . Researchers are urged to implement and ensure comparability between treatment and control groups. As the landscape of medical research evolves, mastering the nuances of and execution will remain vital for generating trustworthy and valid results.
Statistical Analysis Plan: Structuring Your Data Analysis
A (SAP) serves as a critical blueprint for the methods and procedures employed in the to analyze data collected during a . It meticulously outlines the , strategies for addressing missing data, and the framework for conducting . By establishing a prior to the commencement of data collection, researchers can ensure a systematic approach that aligns directly with the study’s objectives. This proactive strategy not only bolsters the integrity of the research but also promotes reproducibility, a fundamental aspect of that is underscored by experts in the field.
Carrol Gamble, PhD, notably emphasizes that a clear and is essential for enhancing . Alarmingly, fewer than 1% of researchers publish their SAPs in peer-reviewed journals, underscoring the critical need for transparency and the necessity for thorough SAPs in medical studies. The prevalence of common statistical tests, such as t-tests and ANOVA, in medical research further highlights the importance of a well-structured to effectively guide these methodologies.
Furthermore, conducting multiple analyses without appropriate adjustments can inflate the alpha spend, thereby increasing the likelihood of Type I error. This reality accentuates the urgent need for a . By prioritizing the development of a robust SAP, researchers can significantly enhance the quality and reliability of their studies.
, with branches showing different essential components like statistical tests and strategies for missing data. Each branch provides a visual way to see how these components contribute to the overall integrity of clinical research. At the center is the main concept of the Statistical Analysis Plan, with branches showing different essential components like statistical tests and strategies for missing data. Each branch provides a visual way to see how these components contribute to the overall integrity of clinical research.](https://images.tely.ai/telyai/gpietquo-at-the-center-is-the-main-concept-of-the-statistical-analysis-plan-with-branches-showing-different-essential-components-like-statistical-tests-and-strategies-for-missing-data-each-branch-provides-a-visual-way-to-see-how-these-components-contribute-to-the-overall-integrity-of-clinical-research.webp)
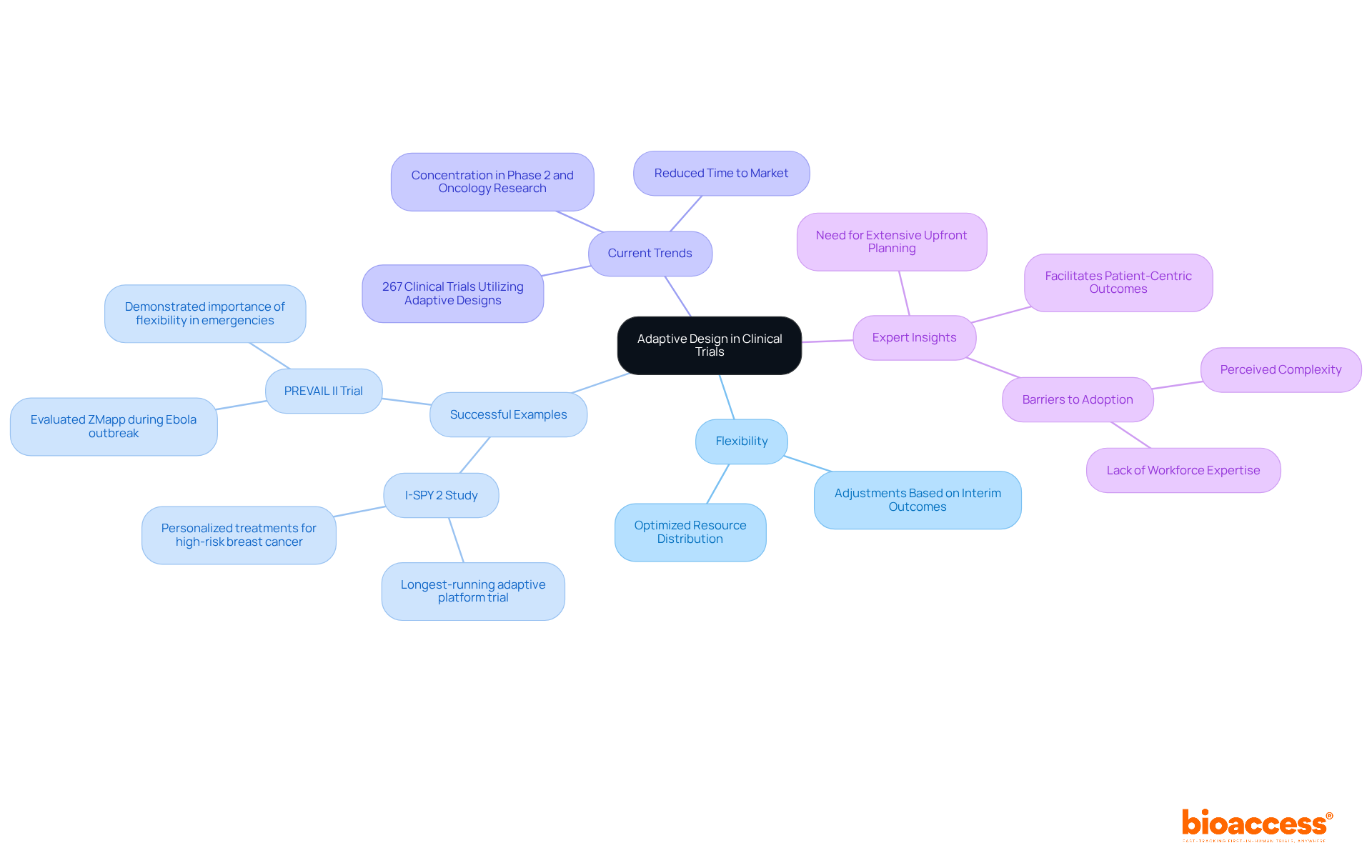
Adaptive Design: Flexibility in Clinical Trial Methodology
empower researchers to implement predetermined modifications during studies based on interim outcomes. This inherent flexibility allows for adjustments in sample size, treatment regimens, and even endpoints, facilitating a more adaptable management strategy. By leveraging adaptive designs in , researchers can and , ultimately leading to more effective clinical studies.
Successful examples of adaptive studies underscore their potential impact. The I-SPY 2 Study, launched in 2010, stands as the , focusing on . It has effectively identified therapies tailored to various tumor subtypes, exemplifying the efficacy of adaptive methodologies in personalizing cancer treatment. Similarly, the PREVAIL II Trial, conducted during the Ebola outbreak, employed an adaptive design to swiftly evaluate the efficacy of ZMapp, highlighting the critical importance of .
Current trends reveal a growing acceptance of within the Medtech and Biopharma sectors. A 2023 study identified 267 s utilizing adaptive designs, with a significant concentration in Phase 2 and oncology research. This shift signifies a sector-wide acknowledgment of the advantages adaptive studies offer, including and improved patient safety through continuous observation and modifications based on real-time data.
Experts note that while adaptive trials may necessitate more extensive upfront planning, their capacity to facilitate seamless transitions between trial phases and enhance patient-centric outcomes renders them invaluable. As the sector evolves, the integration of flexible approaches is expected to foster sustainable growth and innovation in healthcare studies.
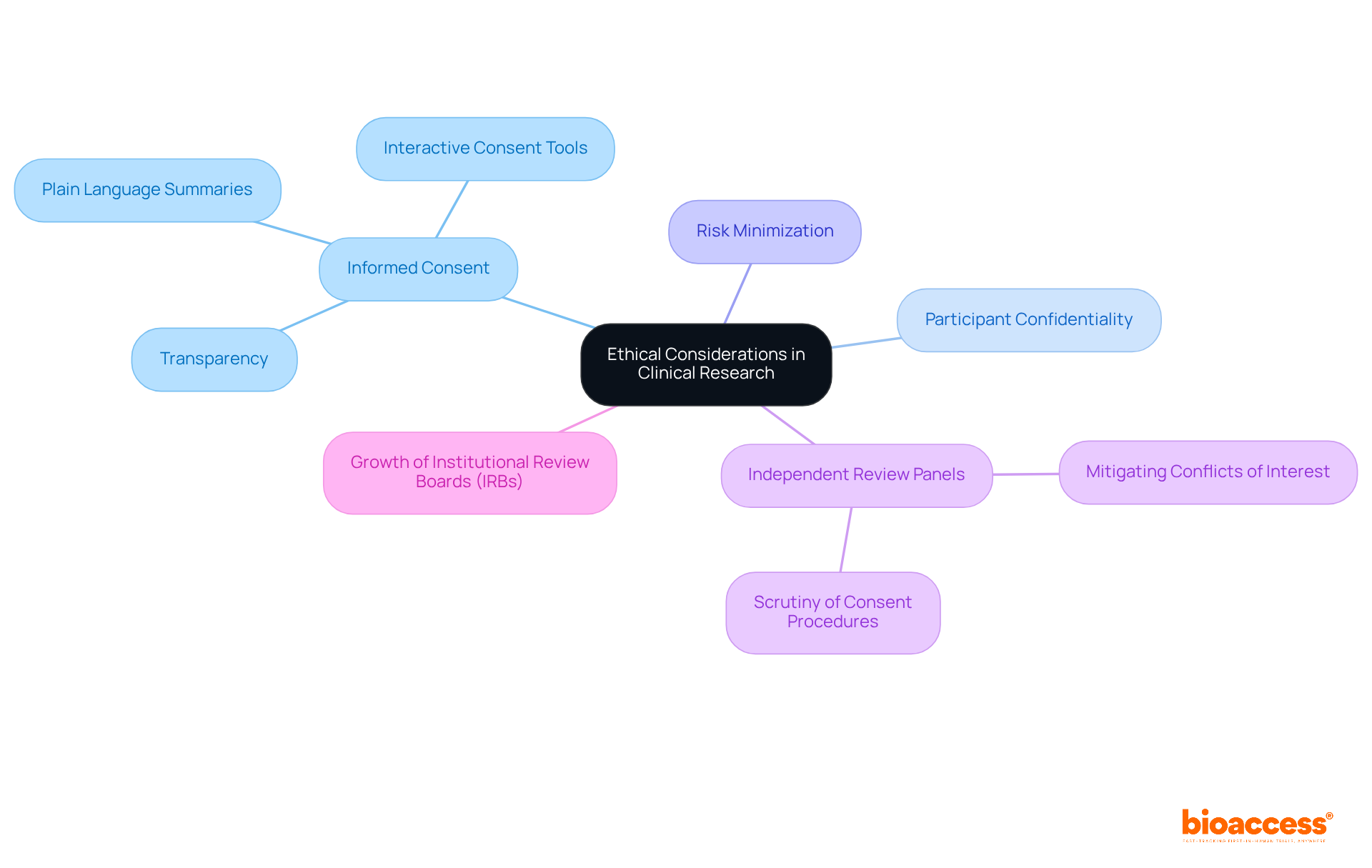
Ethical Considerations: Safeguarding Participants in Clinical Research
are paramount, encompassing responsibilities such as obtaining , safeguarding participant confidentiality, and minimizing risks. is not merely a formality; it is a fundamental ethical requirement that ensures participants are fully aware of the study’s purpose, methods, risks, and potential benefits. Recent trends highlight a shift towards more transparent and comprehensive processes, which are essential for fostering trust between researchers and participants.
For instance, the Belmont Report emphasizes the , mandating that participants understand the uncertainties surrounding treatment efficacy. This is especially significant in early-phase studies, where the risk-benefit ratio is often unclear. Moreover, independent review panels are increasingly scrutinizing procedures to ensure . The significant growth in the number of (IRBs), alongside the increase in , further underscores the importance of this scrutiny.
Examples of effective processes include the use of plain language summaries and interactive consent tools that enhance participant understanding. Such practices not only adhere to ethical standards but also enable participants to make informed choices about their involvement in studies. Notably, only 63% of , highlighting the need for .
Statements from specialists highlight the significance of :
“Has everything been done to minimize the risks and inconvenience to participants, to maximize the potential benefits, and to determine that the potential benefits to individuals and society are proportionate to, or outweigh, the risks?”
This underscores the ethical obligation to prioritize . The ethical evaluation of studies involving human participants has advanced considerably over the last two decades, emphasizing the importance of justifying eligibility standards in trials. By following these , researchers can improve the integrity of their research and ensure the well-being of all participants.
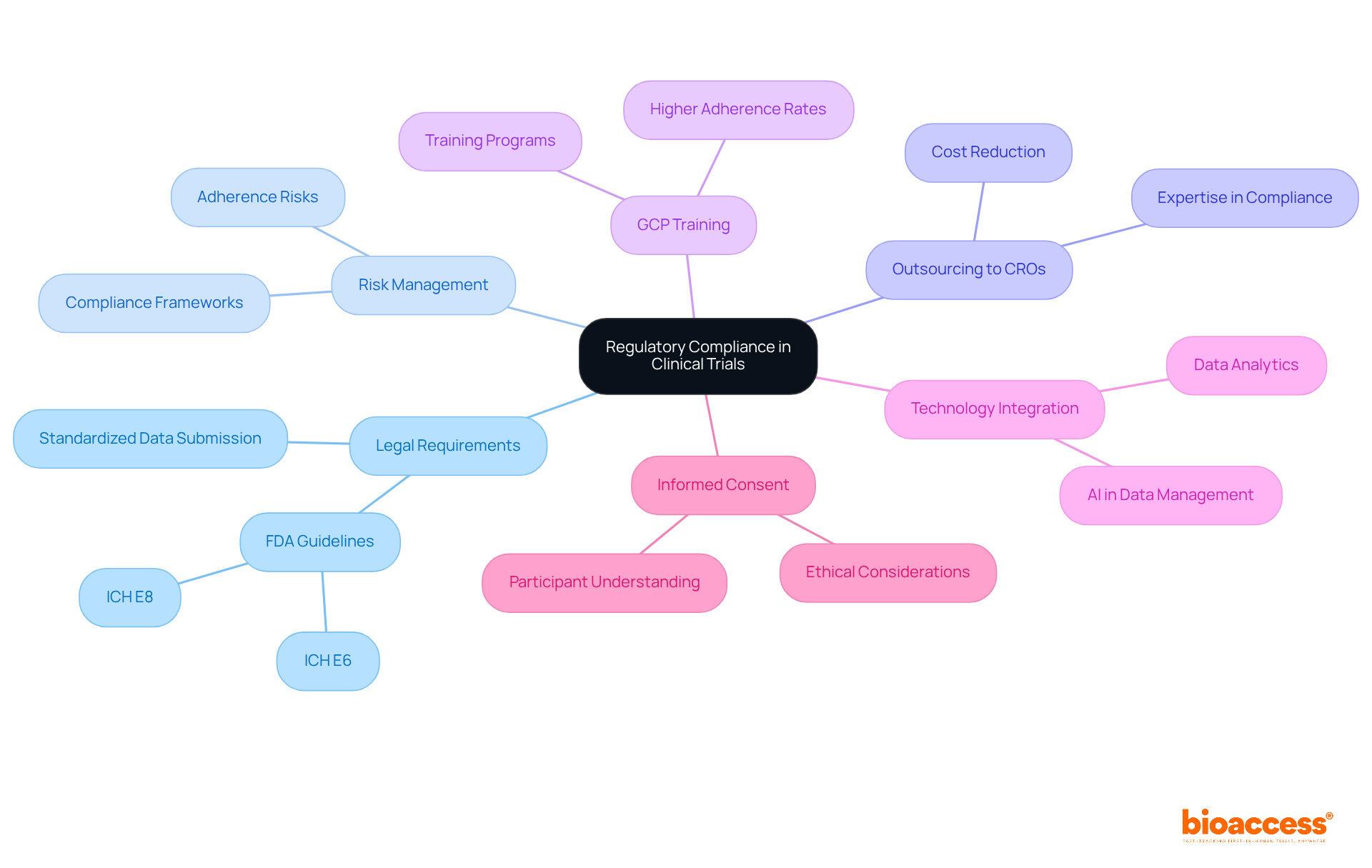
Regulatory Compliance: Navigating Legal Requirements in Clinical Trials
stands as a cornerstone in medical research, necessitating unwavering adherence to laws and guidelines that vary across regions. This intricate process involves securing and meticulously following . For instance, the FDA’s guidelines, particularly ICH E6 and ICH E8, delineate comprehensive steps for integrating risk management into , enabling researchers to identify adherence risks early in the phase. Navigating these complex regulatory landscapes not only ensures that investigations are conducted ethically but also safeguards participant welfare and upholds the integrity of the work.
Recent trends indicate that to contract organizations (CROs) to leverage their expertise in navigating these legal requirements. This shift is propelled by the imperative for compliance with evolving regulations and the aspiration to enhance operational efficiencies. Moreover, organizations that implement robust training programs experience markedly higher adherence rates, underscoring the significance of .
As the landscape of medical studies grows more intricate, with only 20% of research meeting deadlines due to inefficiencies, . These tools not only streamline data management but also enhance adherence tracking, facilitating prompt corrective actions when necessary.
Informed consent remains a pivotal area, as over 50% of research participants find it challenging to fully comprehend key components, which can lead to ethical violations. Addressing these concerns is vital for safeguarding participant rights and ensuring the ethical practice of research studies. By prioritizing compliance and ethical standards, researchers can adeptly navigate the legal complexities of , ultimately contributing to the success of their trials.
Conclusion
The success of clinical study design is fundamentally rooted in a meticulous understanding of its key elements, which collectively enhance the reliability and efficiency of research outcomes. By addressing critical components such as:
- Defining the target population
- Calculating appropriate sample sizes
- Implementing robust randomization techniques
- Establishing clear endpoints
Researchers can significantly improve the validity of their findings. Furthermore, ethical considerations and regulatory compliance are pivotal in safeguarding participants and ensuring the integrity of the research process.
This article has highlighted various strategies, from leveraging global agility in clinical study design to utilizing adaptive methodologies that allow for flexibility in trial execution. The importance of engaging local healthcare providers for effective recruitment, the necessity of well-structured statistical analysis plans, and the critical role of control groups in establishing baselines have been underscored as essential practices. These insights not only aid in navigating the complexities of clinical research but also serve to foster innovation within the Medtech and Biopharma sectors.
As the landscape of clinical trials continues to evolve, embracing these best practices becomes increasingly vital. Researchers are urged to prioritize ethical standards, maintain regulatory compliance, and leverage advanced methodologies to enhance the quality and impact of their studies. By doing so, the clinical research community can drive meaningful advancements in healthcare, ultimately benefiting patients and society at large.
Frequently Asked Questions
What is bioaccess® and what regions does it operate in?
bioaccess® is a company that strategically positions itself across Latin America, the Balkans, and Australia, providing flexibility in research design and accelerating clinical study timelines.
How does bioaccess® enhance clinical study design?
bioaccess® enhances clinical study design by leveraging rapid regulatory processes in Latin America, diverse patient populations in the Balkans, and robust ethical approval systems in Australia, significantly reducing the time to initiate and complete research projects.
What is the significance of the Latin American clinical trials market?
The Latin American clinical trials market generated USD 1,682.7 million in 2023, highlighting its increasing importance in the global clinical research landscape.
Why is defining the target population important in clinical studies?
Defining the target population is crucial for aligning participant traits with research objectives, enhancing the relevance of findings, and ensuring that results can be generalized to a broader patient demographic.
What strategies can improve participant recruitment in clinical studies?
Effective recruitment strategies include engaging local healthcare providers, utilizing community partnerships, employing culturally sensitive outreach, and leveraging digital tools and social media platforms to broaden outreach efforts.
Why is calculating the right sample size important in medical research?
A suitable sample size is vital as it affects the research’s power, which is the likelihood of accurately identifying a genuine effect. An appropriate sample size enhances the reliability of the findings and supports the research hypotheses.
What is the recommended minimum sample size for clinical trials?
A minimum sample size of 100 participants per group is frequently advised to ensure sufficient power, especially in clinical trials where outcome occurrences can vary significantly.
What is statistical power and why is it important?
Statistical power is typically set at 80%, indicating the chance of detecting an effect if it exists. It is essential for ensuring that research can reliably support its hypotheses and avoid underpowered analyses.
What tools can assist researchers in calculating sample sizes?
Statistical software tools such as G-Power, OpenEpi, PASS, and R can help researchers perform complex calculations to estimate sample sizes based on different statistical analyses.
How does appropriate sample size calculation relate to ethical standards in research?
Appropriate sample size calculation is viewed as a prerequisite for research project approval by ethical committees, as it minimizes unnecessary risks to participants and maintains the integrity of the research.
List of Sources
- bioaccess®: Accelerating Clinical Study Design with Global Agility
- 75 Quotes About AI: Business, Ethics & the Future (https://deliberatedirections.com/quotes-about-artificial-intelligence)
- Latin America Clinical Trials Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trials-market/latin-america)
- Quotes on Medicine and Drug Development – Related Articles – Therapy, Diagnosis, Life Sciences, and Medical Research Discoveries and News – Discovery Medicine (https://discoverymedicine.com/related/2/907)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- statista.com (https://statista.com/statistics/1013599/latin-america-clinical-trials)
- Target Population: Defining the Right Participants for Your Study
- Patient Recruitment and Retention in Clinical Trials: Strategies and Challenges (https://mdgroup.com/blog/patient-recruitment-and-retention-in-clinical-trials-strategies-and-challenges)
- Data Analytics Help Achieve Clinical Trial Diversity (https://globalforum.diaglobal.org/issue/september-2023/data-analytics-help-achieve-clinical-trial-diversity)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC11348161)
- Enrollment in Clinical Trials: Statistics and Patient Recruitment Strategies | Power (https://withpower.com/guides/enrollment-in-clinical-trials-statistics-and-patient-recruitment-strategies)
- wcgclinical.com (https://wcgclinical.com/insights/diverse-recruitment-strategies-and-the-future-of-inclusive-clinical-trials)
- Sample Size: Calculating the Right Number for Statistical Power
- journals.lww.com (https://journals.lww.com/picp/fulltext/2024/15030/sample_size_calculation_in_clinical_research.10.aspx)
- Statistical Sample Size Calculations for Clinical Trials – Quantics Biostatistics (https://quantics.co.uk/blog/statistical-calculations-of-sample-size-for-clinical-trials)
- Sample size determination: A practical guide for health researchers – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10000262)
- Sample size estimation and power analysis for clinical research studies – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC3409926)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC7745163)
- Randomization: Ensuring Fairness in Clinical Trials
- Issues in Outcomes Research: An Overview of Randomization Techniques for Clinical Trials – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC2267325)
- tandfonline.com (https://tandfonline.com/doi/abs/10.1080/19466315.2023.2225451)
- bmcmedresmethodol.biomedcentral.com (https://bmcmedresmethodol.biomedcentral.com/articles/10.1186/s12874-022-01786-4)
- The importance of randomization in clinical research – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC9424468)
- Randomization in Clinical Trials: Everything You Need to Know (https://greenlight.guru/blog/randomization-medical-devices-clinical-trials)
- Endpoints: Defining Success in Clinical Trials
- 10 Trends and Statistics for Clinical Trials in 2023 (https://xtalks.com/10-trends-and-statistics-for-clinical-trials-in-2023-3377)
- iqvia.com (https://iqvia.com/insights/the-iqvia-institute/reports-and-publications/reports/evolving-oncology-endpoints)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC9843702)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC6409418)
- Control Groups: Establishing Baselines for Comparison
- novotech-cro.com (https://novotech-cro.com/faq/importance-control-groups-clinical-trials)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC3951812)
- numberanalytics.com (https://numberanalytics.com/blog/ultimate-guide-control-group-clinical-trials-data-analysis)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC3461178)
- Statistical Analysis Plan: Structuring Your Data Analysis
- How to Understand and Create a Statistical Analysis Plan (SAP) (https://quanticate.com/blog/how-to-understand-and-create-a-statistical-analysis-plan)
- trialsjournal.biomedcentral.com (https://trialsjournal.biomedcentral.com/articles/10.1186/s13063-025-08756-3)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S2451865423000467)
- Guidelines for the Content of Statistical Analysis Plans in Clinical Trials (https://jamanetwork.com/journals/jama/fullarticle/2666509)
- pubmed.ncbi.nlm.nih.gov (https://pubmed.ncbi.nlm.nih.gov/29260229)
- Adaptive Design: Flexibility in Clinical Trial Methodology
- Adaptive trial designs: understanding the potential of statistical innovation (https://iqvia.com/blogs/2021/11/adaptive-trial-designs-understanding-the-potential-of-statistical-innovation)
- Adaptive trial designs will increase clinical trial speed, safety, and effectiveness (https://statnews.com/2024/07/12/adaptive-trial-designs-increase-speed-safety-effectiveness)
- Figure 1. Annual statistics of clinical trial using adaptive design…. (https://researchgate.net/figure/Annual-statistics-of-clinical-trial-using-adaptive-design-Number-of-adaptive-design_fig1_376859898)
- biopharmadive.com (https://biopharmadive.com/spons/drug-development-innovations-that-work-adaptive-trial-design/542907)
- Ethical Considerations: Safeguarding Participants in Clinical Research
- projecteuclid.org (https://projecteuclid.org/journals/statistical-science/volume-6/issue-1/Ethics-and-Statistics-in-Randomized-Clinical-Trials/10.1214/ss/1177011934.full)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/0378375894901988)
- Statistics and ethics: Models for strengthening protection of human subjects in clinical research | PNAS (https://pnas.org/doi/10.1073/pnas.0912882107)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC4465206)
- Ethics Research | Clinical Center (https://cc.nih.gov/recruit/ethics)
- Regulatory Compliance: Navigating Legal Requirements in Clinical Trials
- 10 Trends and Statistics for Clinical Trials in 2023 (https://xtalks.com/10-trends-and-statistics-for-clinical-trials-in-2023-3377)
- 12 Best Quotes on the Future of Healthcare (https://klara.com/blog/12-best-quotes-on-the-future-of-healthcare)
- bioaccessla.com (https://bioaccessla.com/blog/top-7-compliance-risks-in-trial-design-you-need-to-know)
- Regulatory Challenges With Data Collection | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/regulatory-challenges-data-collection)
- numberanalytics.com (https://numberanalytics.com/blog/clinical-data-management-pharma-7-stats)

Leave a Reply