Introduction
The intricate world of clinical trials hinges on the expertise of statisticians who navigate complex data to ensure the validity of medical research outcomes. Mastering essential skills in statistical analysis not only enhances the reliability of findings but also significantly influences patient care and treatment protocols. However, with the rapid evolution of methodologies and the growing demands of healthcare, how can clinical trials statisticians stay ahead of the curve and effectively mitigate challenges such as bias and misinterpretation? This article explores ten critical skills every clinical trials statistician must master to thrive in this dynamic field and contribute meaningfully to medical advancements.
bioaccess®: Mastering Statistical Analysis for Clinical Trials
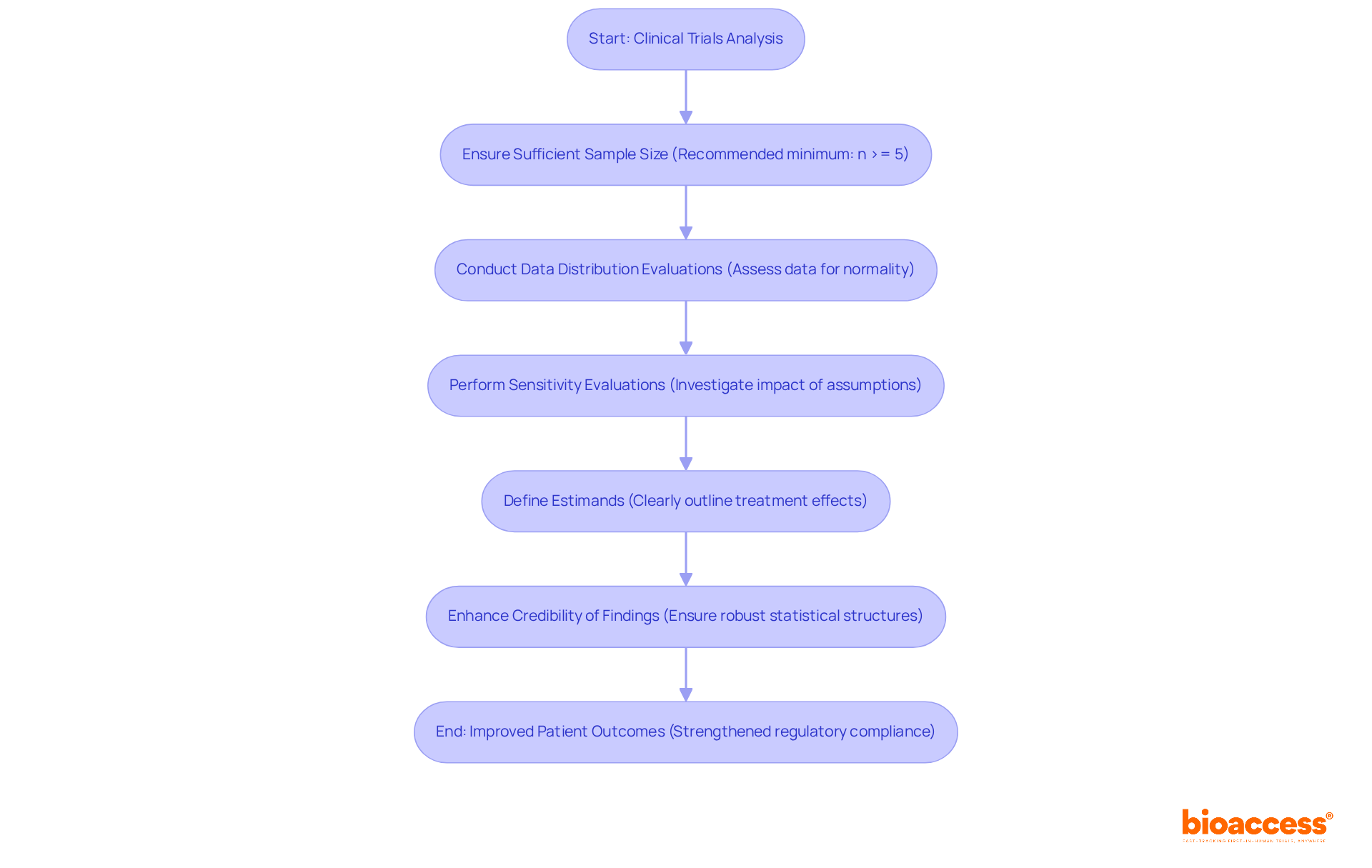
At bioaccess®, the role of a is paramount for mastering to uphold the integrity and reliability of . The organization employs advanced analytical methodologies that are essential for a to assess the efficacy and safety of .
By adhering to —such as ensuring and conducting appropriate data distribution evaluations—bioaccess® aids the in that can compromise study validity. This expertise not only accelerates the research process but also enhances the credibility of findings, ultimately leading to and strengthened .
The emphasis on , including sensitivity evaluations and clearly defined estimands, further bolsters the dependability of medical research, ensuring that the results are both significant and actionable.
Understanding Statistical Analysis Methods in Clinical Trials
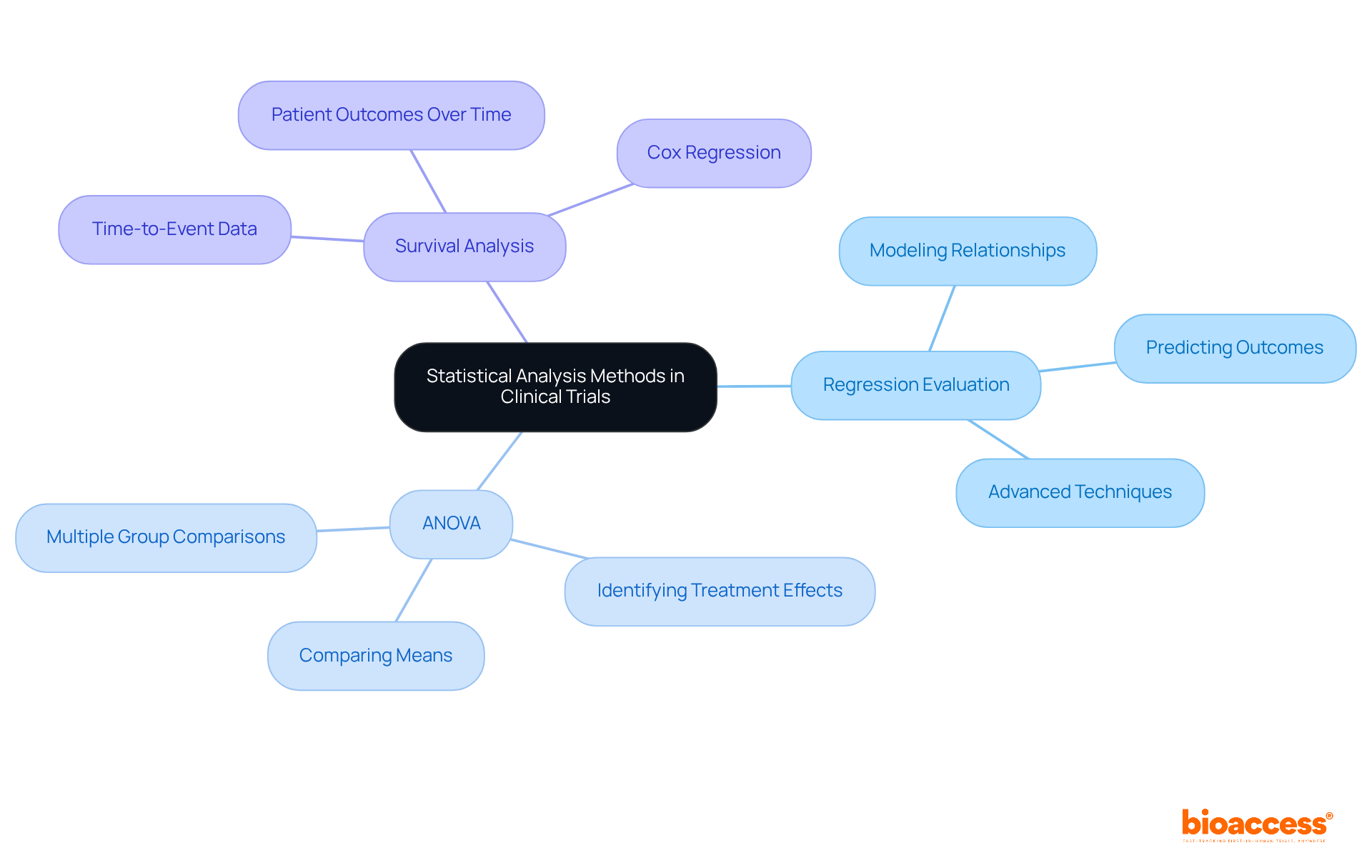
Statistical evaluation methods in are integral to effective . Among these, stands out as pivotal for elucidating relationships between variables, allowing researchers to model and predict outcomes based on diverse factors.
Concurrently, is essential for comparing means across multiple groups, thus facilitating the identification of significant differences in .
Furthermore, plays a crucial role in studies focused on time-to-event data, offering valuable insights into patient outcomes over time.
Mastery of these methodologies is indispensable for a statistician, as they underpin robust and ensure precise interpretations of results.
Current trends reveal an increasing emphasis on and ANOVA applications, reflecting the evolving landscape of medical research and underscoring the necessity for a to adapt to these advancements.
Interpreting Statistical Significance in Clinical Research
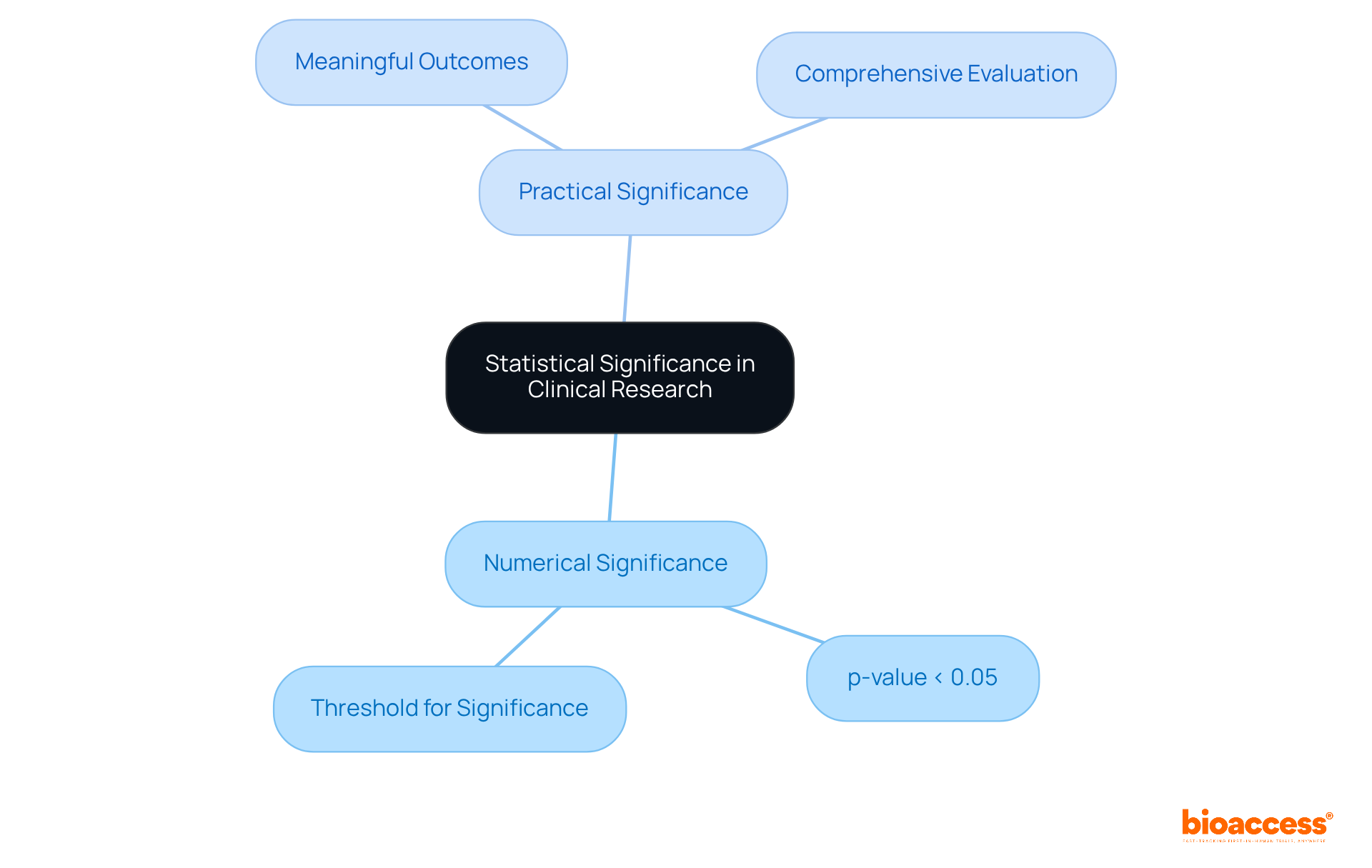
Understanding the is essential in evaluating whether the outcomes of a are likely a result of randomness. A common threshold for significance is a of less than 0.05, which indicates that there is less than a 5% probability that the observed results occurred by random chance.
However, it is crucial to consider the alongside . A numerically significant result may not always translate into meaningful practical outcomes, underscoring the need for a .
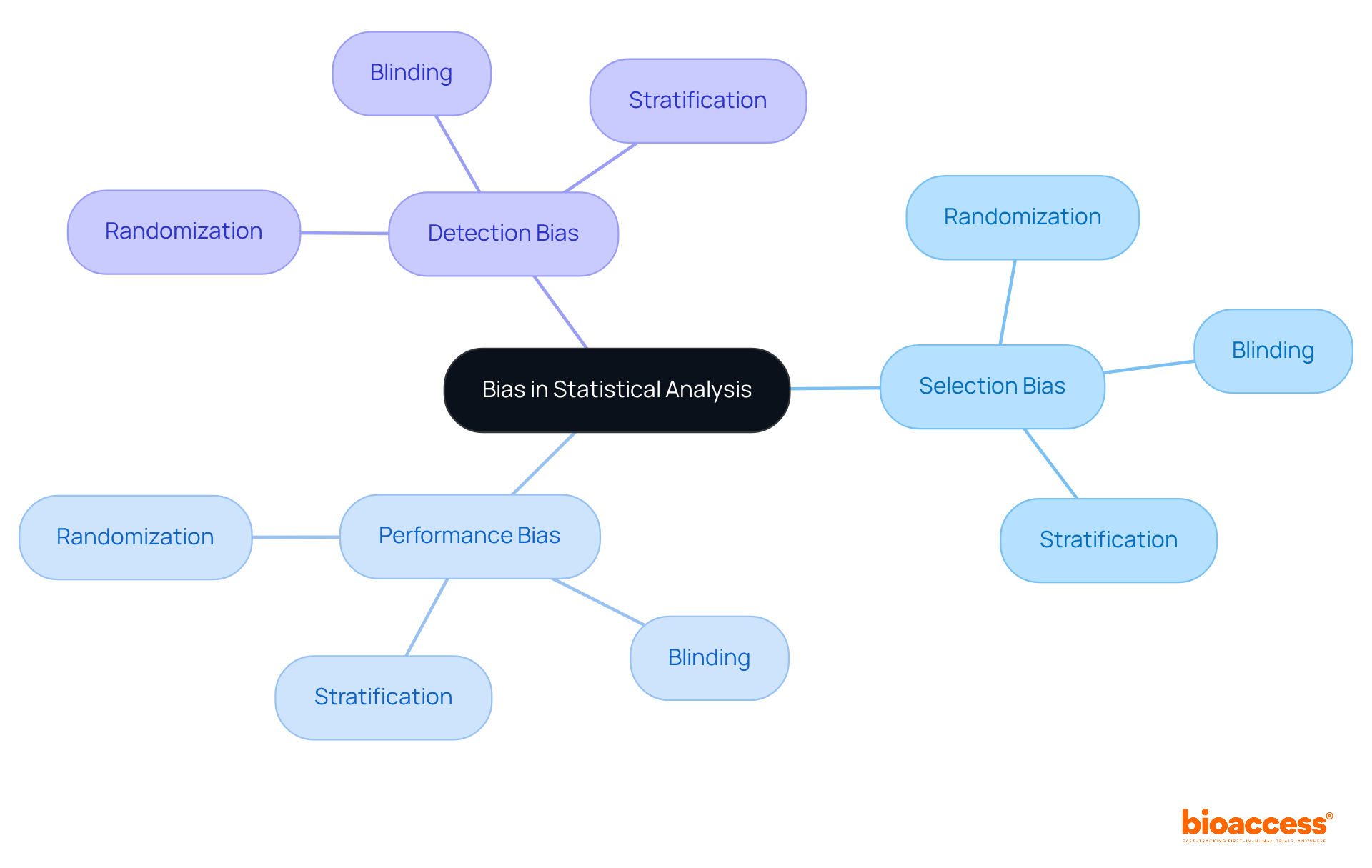
Avoiding Bias in Statistical Analysis for Clinical Trials
is essential for the reliability of . from various sources, including:
- Performance bias
- Detection bias
To mitigate these biases, implementing strategies such as:
- Randomization
- Blinding
- Stratification
is crucial. By ensuring that the , researchers can enhance the reliability of their findings, thereby supporting more accurate conclusions about the efficacy of treatments.
Utilizing Descriptive Statistics in Clinical Trials
play a pivotal role in summarizing the characteristics of participants in clinical research studies conducted by a . Metrics such as means, medians, modes, and standard deviations provide a comprehensive overview of the data, which enables a to discern the distribution and central tendencies within their study populations. Furthermore, like histograms and box plots significantly enhance the communication of findings. These tools facilitate a , allowing them to appreciate the implications of the data more readily.
Applying Inferential Statistics in Clinical Trials
Inferential statistics empower researchers to make predictions and generalizations about broader populations based on sample data. This method is crucial in for a , where common techniques such as:
- Confidence intervals
serve as foundational tools for drawing conclusions from study results. For instance, that likely include the true population parameter, offering insights into the precision of estimates derived from samples. Furthermore, the of observed treatment effects, ensuring that findings are not merely coincidental. Continuous or adverse occurrences during studies, which may necessitate adjustments to research protocols. This not only enhances the reliability of results but also aids a in making critical decisions in medical research, ensuring that conclusions are firmly grounded in rigorous data analysis.
Understanding P-values and Confidence Intervals in Clinical Trials
serve as fundamental components of in . A under the assumption that the null hypothesis is true, while a . For instance, a , 95% of the intervals would capture the true population parameter. Grasping these concepts is crucial for a statistician, as it enables them to assess the of their findings, ultimately enhancing the .
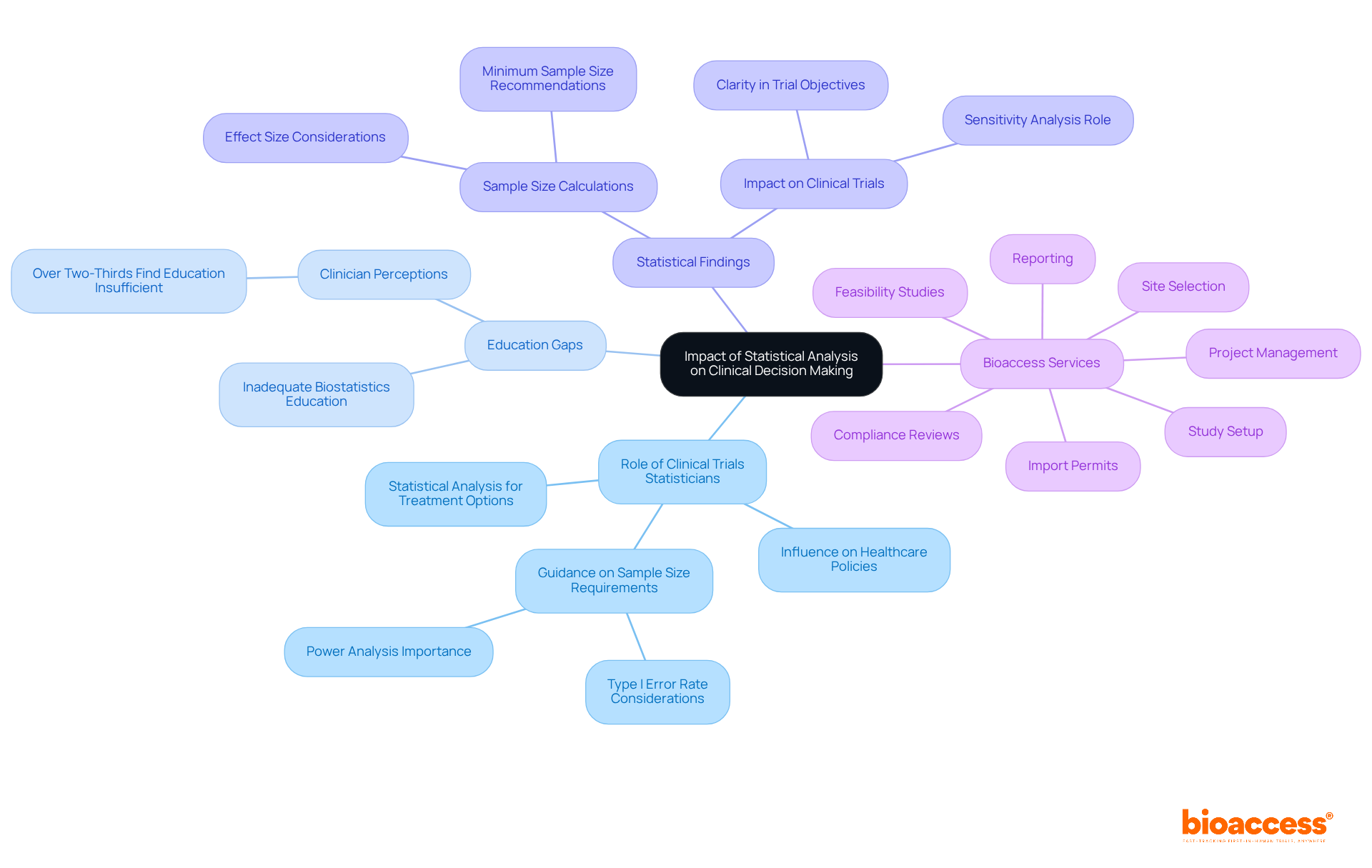
Impact of Statistical Analysis on Clinical Decision Making
The role of a is indispensable in medical decision-making, as they provide statistical analysis and evidence-based insights that shape treatment options and patient care strategies. For instance, a can help ensure that directly inform healthcare providers regarding the efficacy and safety of new therapies, empowering them to make well-informed recommendations to patients. Notably, over two-thirds of clinicians perceive the level of offered to medical students as inadequate, underscoring a gap that could impact future healthcare decisions.
Furthermore, the impact of statistical findings extends to healthcare policy decisions and guidelines. A can use rigorous to pave the way for the establishment of new protocols that enhance patient outcomes. Recent data indicate that maintaining a often necessitates a , which a would advise, to achieve a power of 0.8, ensuring that studies yield reliable results. This underscores the critical importance of sufficient sample size considerations in medical research, which a must take into account, as smaller differences require larger sample sizes to achieve significant results.
Within the realm of , bioaccess offers capabilities such as:
- feasibility studies
- site selection
- compliance reviews
- study setup
- import permits
- project management
- reporting
This thorough management not only guarantees adherence to regulatory standards but also promotes international collaboration, advancing global health improvements through innovative Medtech solutions.
In conclusion, the integration of robust data evaluation by a in medical studies not only directs treatment choices but also profoundly influences healthcare policies and provider recommendations, ultimately enhancing patient care.
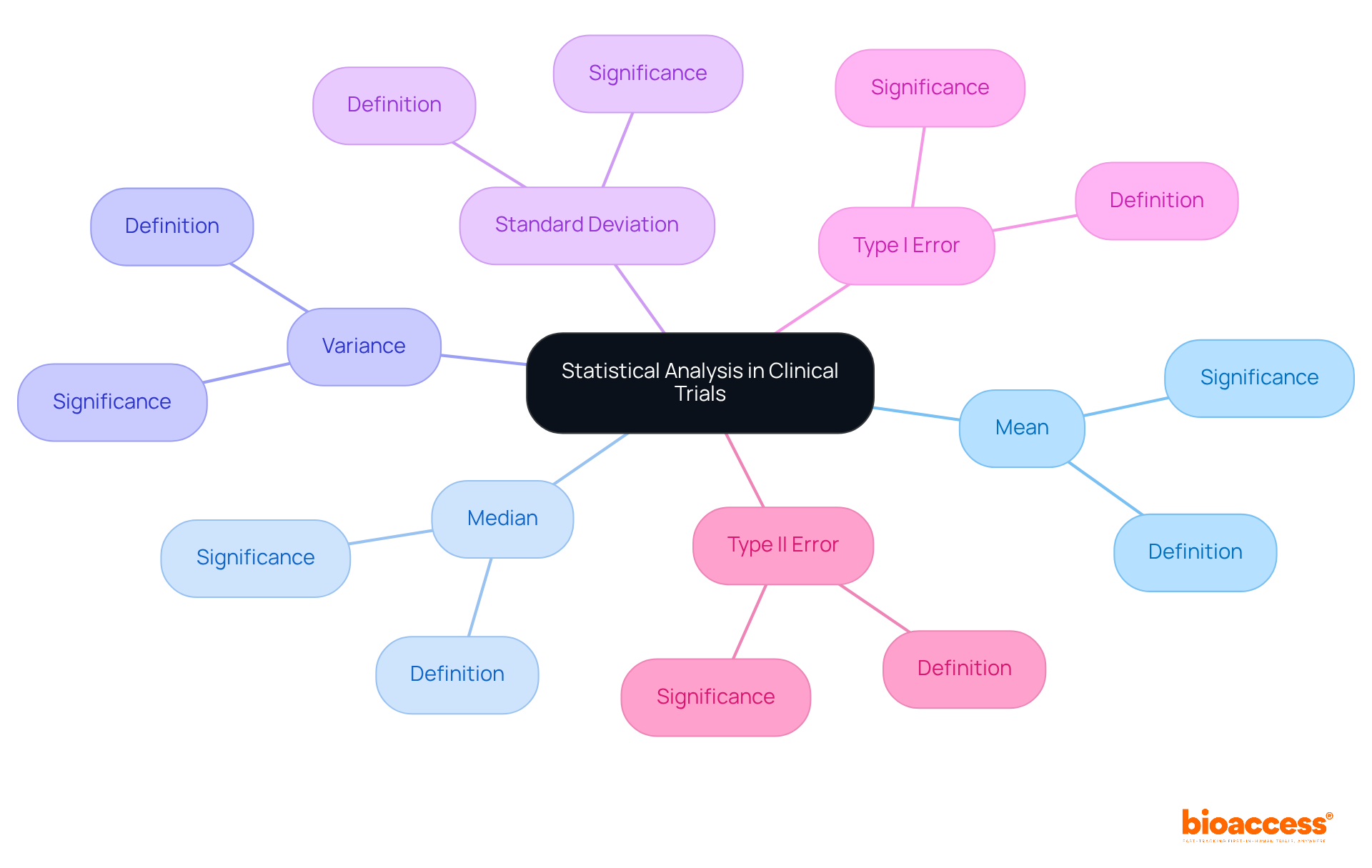
Mastering Statistical Analysis Terms in Clinical Trials
Proficiency in is essential for effective communication in medical studies. Fundamental terms such as ‘mean,’ ‘median,’ ‘variance,’ and ‘standard deviation’ serve as the backbone of , enabling researchers to summarize and interpret information accurately.
Understanding ‘—is crucial for grasping the implications of . For instance, a commonly accepted significance level is 0.05, indicating a 5% chance of a type I error in a study. This knowledge empowers researchers and clinicians to engage in informed discussions about , interpret results effectively, and understand their implications for patient care.
Furthermore, s can uncover negative occurrences or patterns during , underscoring the importance of thorough . , emphasizing the vital role of continuous numerical assessment.
In case studies involving inferential statistics, researchers employ and regression analyses to draw conclusions about broader populations based on sample data. Descriptive statistics are also essential for summarizing participant characteristics and treatment outcomes, aiding in effective data interpretation.
This integration of not only enhances the reliability of medical studies but also fosters collaboration among researchers, ensuring that results are conveyed clearly and precisely.
Embracing Continuous Learning in Clinical Trial Statistics
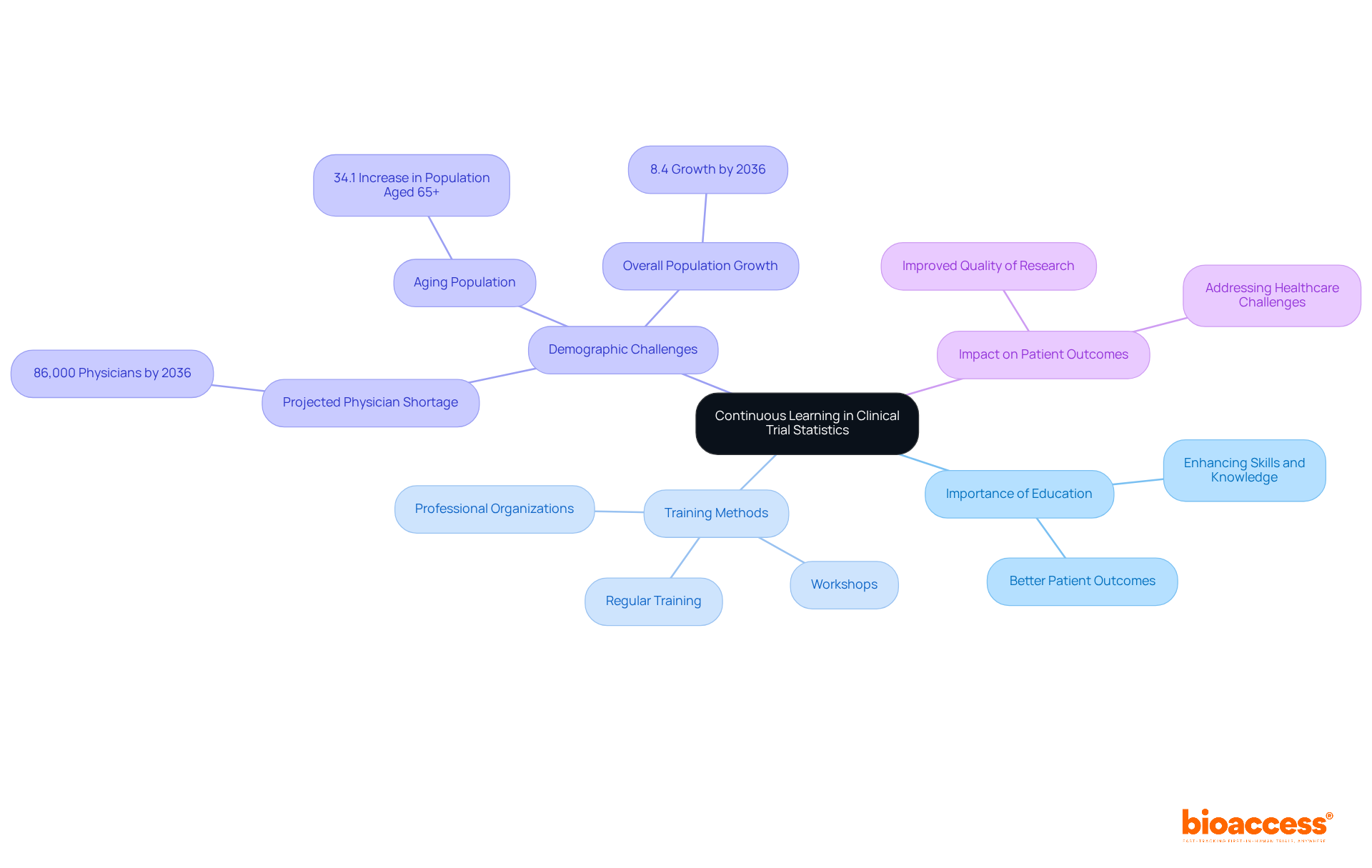
Embracing in statistics to remain abreast of advancements in statistical methods and technologies. Regular training, attending workshops, and engaging with professional organizations significantly enhance skills and knowledge. Given the , along with a projected 34.1% increase in the population aged 65 and above, the need for a in is more urgent than ever. Staying informed about , including machine learning and big data analytics, can greatly improve the quality and efficiency of medical research.
As David J. Skorton, President and CEO of AAMC, emphasizes, “The medical education community and policymakers are making real progress in our efforts to meet the of our communities, but we must not be complacent.”
This unwavering ultimately contributes to and more effective , highlighting the critical role of a in addressing the evolving challenges in healthcare through continuous professional development.
Conclusion
Mastering the essential skills of a clinical trials statistician is crucial for ensuring the integrity and reliability of medical research outcomes. This underscores the importance of various statistical methodologies, from descriptive and inferential statistics to understanding significance and avoiding bias. Such competencies not only enhance the validity of clinical trials but also significantly contribute to informed decision-making in healthcare.
Key arguments highlight the necessity of advanced statistical techniques, including:
- Regression analysis
- ANOVA
These techniques facilitate accurate data interpretation and help mitigate biases that can skew results. Furthermore, the integration of continuous learning and adaptation to emerging trends in data analysis is vital for clinical trials statisticians to remain effective in their roles, especially in a rapidly evolving medical landscape.
Ultimately, the role of a clinical trials statistician extends beyond mere data analysis; it is integral to shaping treatment protocols and improving patient outcomes. As the demand for skilled statisticians continues to grow, a commitment to ongoing education and proficiency in statistical methods will be essential for addressing the challenges of modern healthcare and ensuring that clinical research continues to drive advancements in patient care.
Frequently Asked Questions
What is the role of a clinical trials statistician at bioaccess®?
The clinical trials statistician at bioaccess® is responsible for mastering data analysis to ensure the integrity and reliability of clinical trial outcomes by employing advanced analytical methodologies.
How does bioaccess® support clinical trials statisticians?
bioaccess® supports clinical trials statisticians by ensuring sufficient sample sizes, conducting appropriate data distribution evaluations, and helping to mitigate common analytical errors that can compromise study validity.
What are the benefits of adhering to best practices in statistical analysis during clinical trials?
Adhering to best practices accelerates the research process, enhances the credibility of findings, improves patient outcomes, and strengthens regulatory compliance.
What statistical methods are commonly used in clinical trials?
Common statistical methods include regression evaluation for modeling relationships between variables, Analysis of Variance (ANOVA) for comparing means across multiple groups, and survival analysis for studying time-to-event data.
Why is it important for a clinical trials statistician to master statistical methodologies?
Mastery of statistical methodologies is essential for ensuring robust study designs and precise interpretations of results, which are critical for the validity of clinical research.
What is the significance of a p-value in clinical research?
A p-value of less than 0.05 is commonly used as a threshold for statistical significance, indicating that there is less than a 5% probability that the observed results occurred by random chance.
How should researchers evaluate the significance of study findings?
Researchers should consider both the numerical significance (such as p-values) and the practical importance of findings, as a numerically significant result may not always lead to meaningful practical outcomes.
List of Sources
- bioaccess®: Mastering Statistical Analysis for Clinical Trials
- ICH E9 statistical principles for clinical trials – Scientific guideline | European Medicines Agency (EMA) (https://ema.europa.eu/en/ich-e9-statistical-principles-clinical-trials-scientific-guideline)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC10328100)
- Understanding Statistical Analysis Methods in Clinical Trials
- Understanding Statistical Analysis in Clinical Trials (https://lindushealth.com/blog/understanding-clinical-trial-statistics-a-comprehensive-guide)
- Statistical Issues and Recommendations for Clinical Trials Conducted During the COVID-19 Pandemic – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC8011486)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC7051800)
- Case Studies (Illustrating the SDC Process) — SDC Practice Guide documentation (https://sdcpractice.readthedocs.io/en/latest/case_studies.html)
- Applying Inferential Statistics in Clinical Trials
- viares.com (https://viares.com/blog/clinical-research-explained/data-analysis)
- Understanding Statistical Analysis in Clinical Trials (https://lindushealth.com/blog/understanding-clinical-trial-statistics-a-comprehensive-guide)
- nature.com (https://nature.com/articles/s41598-025-88400-x)
- Understanding P-values and Confidence Intervals in Clinical Trials
- evidence.nejm.org (https://evidence.nejm.org/doi/full/10.1056/EVIDoa2300003)
- rethinkingclinicaltrials.org (https://rethinkingclinicaltrials.org/news/tag/greg-simon)
- ledidi.com (https://ledidi.com/academy/understanding-p-values-in-medical-research)
- nejm.org (https://nejm.org/doi/full/10.1056/NEJMoa2413258)
- rethinkingclinicaltrials.org (https://rethinkingclinicaltrials.org/news/tag/corita-grudzen)
- Impact of Statistical Analysis on Clinical Decision Making
- ICH E9 statistical principles for clinical trials – Scientific guideline | European Medicines Agency (EMA) (https://ema.europa.eu/en/ich-e9-statistical-principles-clinical-trials-scientific-guideline)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC7745163)
- Mastering Statistical Analysis Terms in Clinical Trials
- Understanding Statistical Analysis in Clinical Trials (https://lindushealth.com/blog/understanding-clinical-trial-statistics-a-comprehensive-guide)
- Fundamental Statistical Concepts in Clinical Trials and Diagnostic Testing – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC8729862)
- Embracing Continuous Learning in Clinical Trial Statistics
- aamc.org (https://aamc.org/news/press-releases/new-aamc-report-shows-continuing-projected-physician-shortage)

Leave a Reply