Introduction
The Tanzania Medicines and Medical Devices Authority (TMDA) stands as a pivotal guardian of public health, ensuring that the medicines and healthcare devices accessible to Tanzanians adhere to stringent safety and quality standards.
In a rapidly evolving healthcare landscape, comprehending the multifaceted role of TMDA is crucial for stakeholders, including pharmaceutical companies and healthcare providers.
With the alarming rise of counterfeit products and an urgent demand for innovation, how adeptly can TMDA navigate these challenges to fulfill its mission?
This article explores the authority’s historical evolution, regulatory framework, and significant influence on healthcare in Tanzania, illuminating the essential balance between safety and advancement in medical regulation.
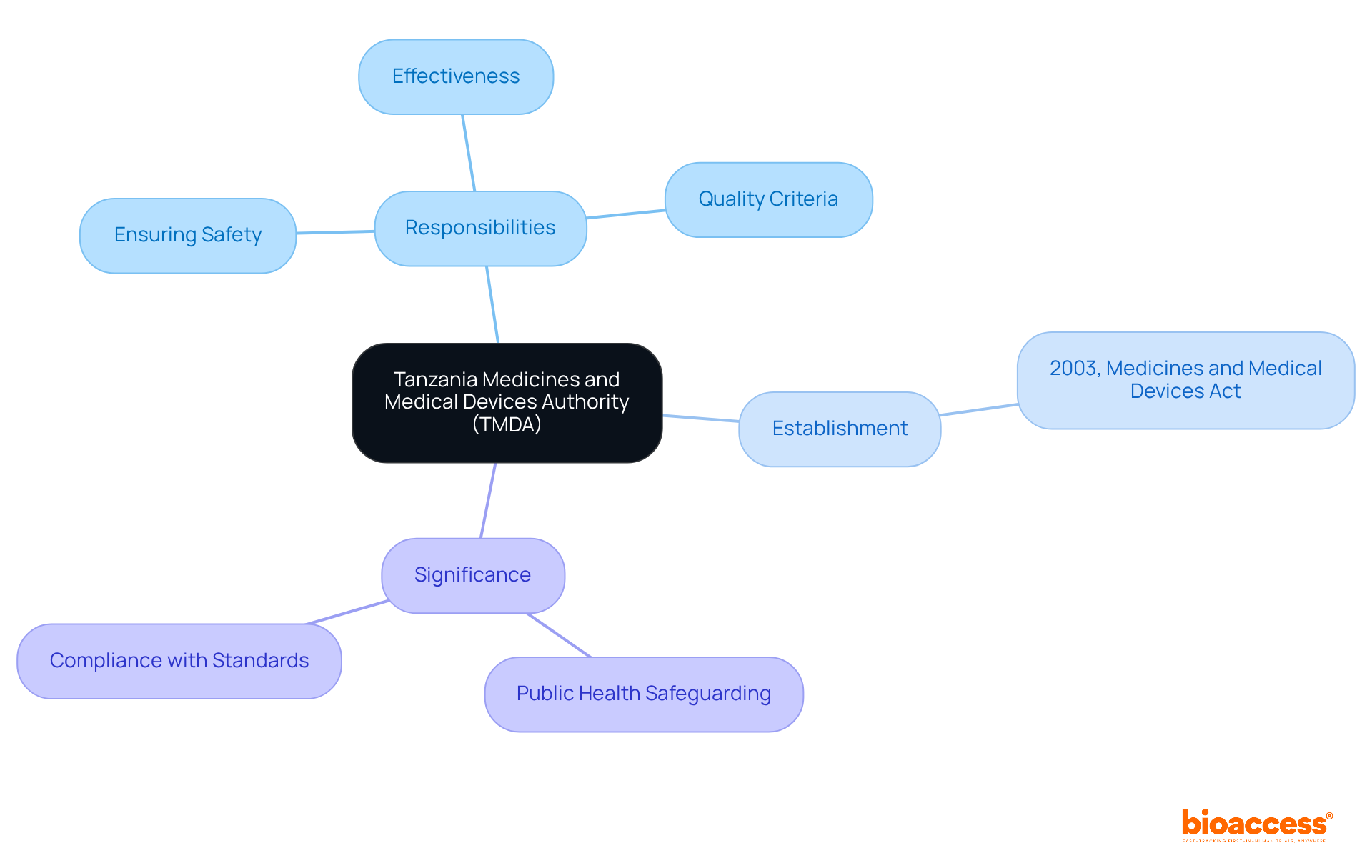
Define TMDA: The Tanzania Medicines and Medical Devices Authority
The tmda is the governing entity responsible for ensuring the safety, effectiveness, and quality of medicines and in Tanzania. Established under the Medicines and Medical Devices Act of 2003, this agency is dedicated to by ensuring that all healthcare products meet before they become available to the public. This authority is pivotal in the oversight environment, functioning as a gatekeeper for and ensuring compliance with both national and international standards.
Contextualize TMDA: Historical Development and Regulatory Framework
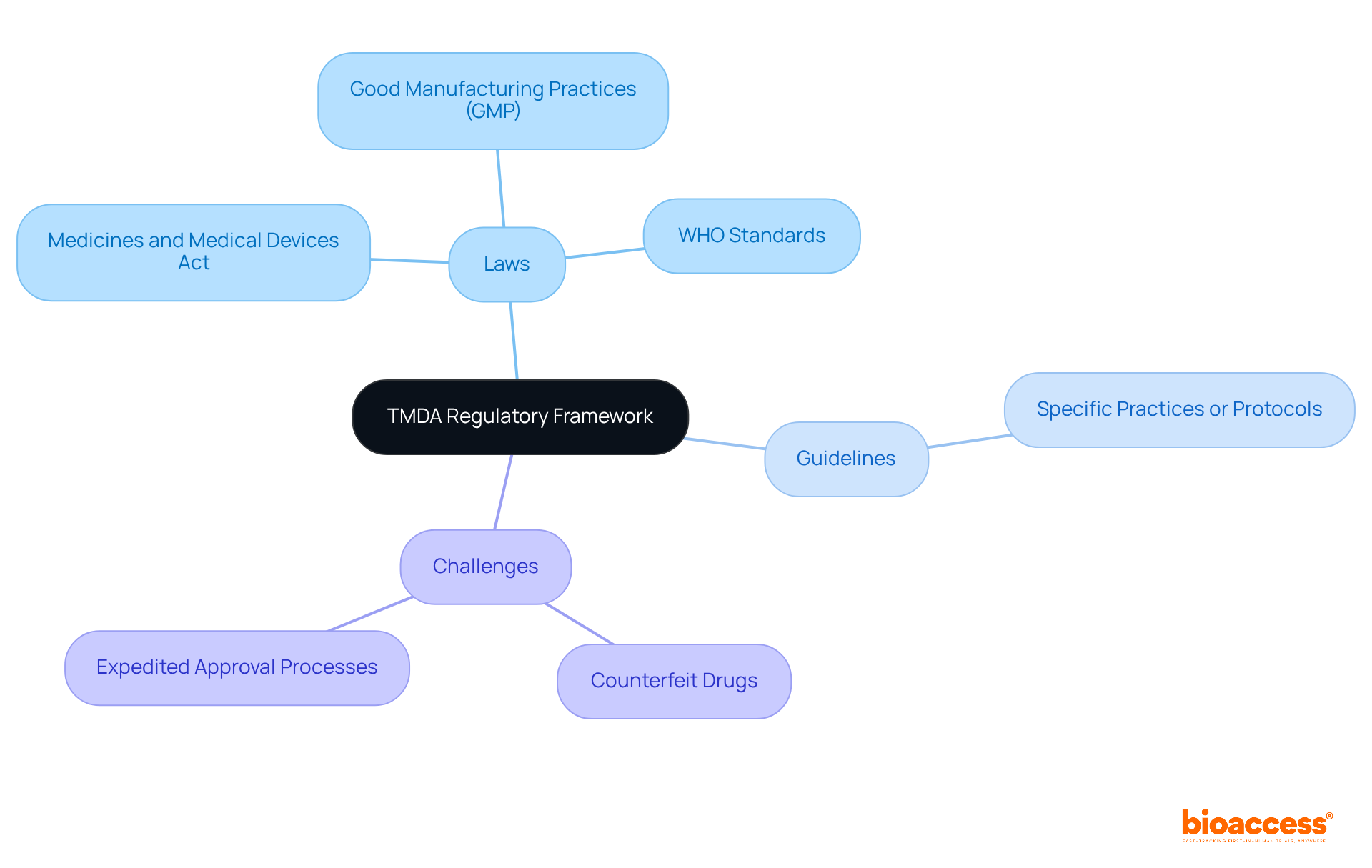
The organization was established to meet the growing demand for a robust oversight structure for the introduction of healthcare products in Tanzania. Operating under the Ministry of Health, Community Development, Gender, Elderly and Children, it adheres to a shaped by various laws and guidelines, including:
Over the years, the organization has adapted its regulations to address , such as the rise of counterfeit drugs and the urgent need for expedited approval processes for innovative health technologies.
Understanding the regulations set forth by the relevant authority is crucial for firms like bioaccess, which provide . Compliance with these guidelines ensures that clinical trials conducted in Tanzania meet essential standards, thereby facilitating smoother approval processes and enhancing the overall success of device studies. As the Medtech landscape evolves, collaboration with regulatory bodies becomes increasingly vital, positioning firms to effectively navigate the complexities of clinical research and drive advancements in healthcare.
Explore TMDA’s Functions: Roles in Medical Product Regulation
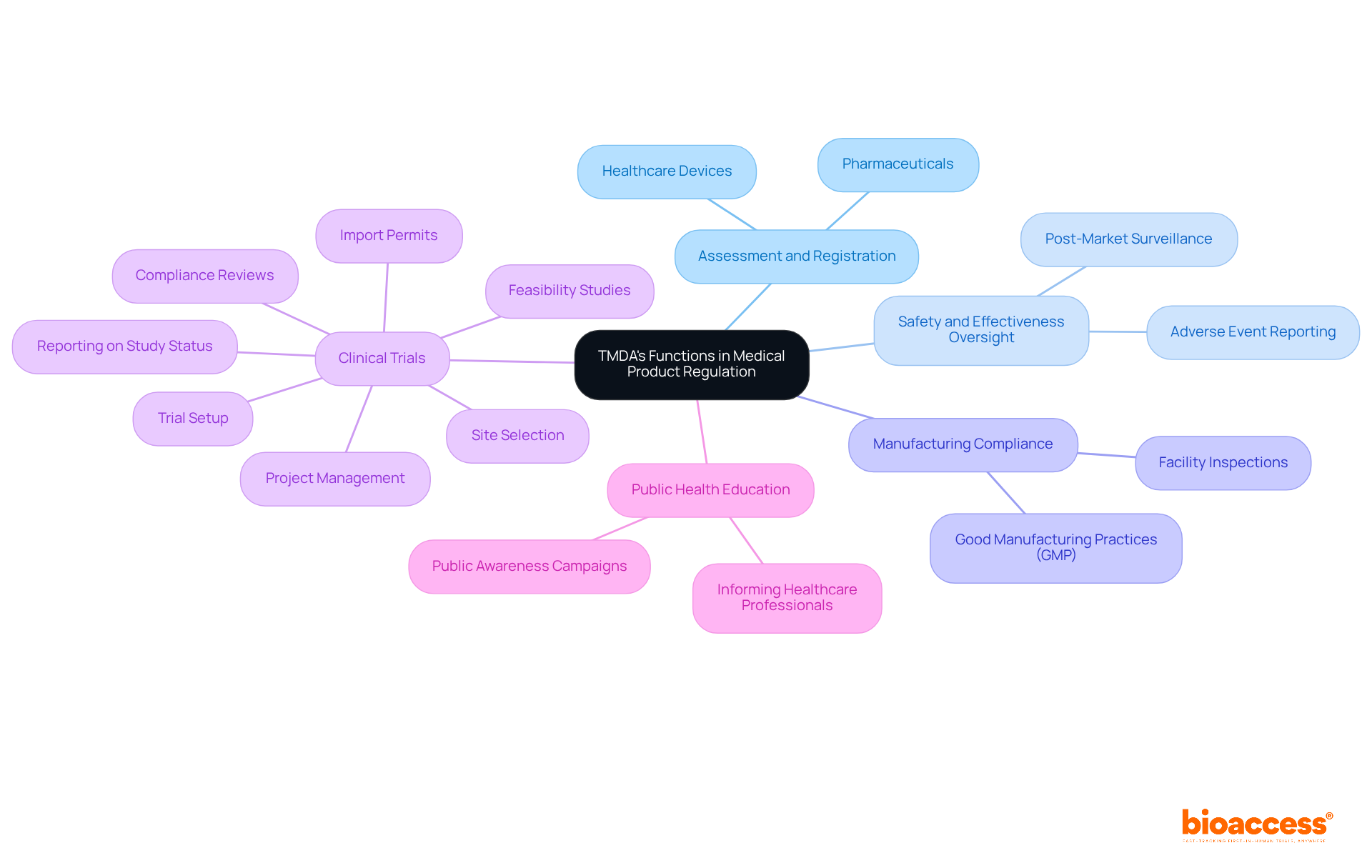
The organization’s primary functions encompass the assessment and registration of pharmaceuticals and healthcare devices, overseeing their safety and effectiveness post-market introduction, and ensuring adherence to regulatory standards. The authority conducts inspections of manufacturing facilities to guarantee compliance with (GMP) and oversees to protect the rights and welfare of participants. Furthermore, the organization plays a crucial role in , informing healthcare professionals and the public about the safe use of medical products. By fulfilling these functions, tmda ensures that only safe and effective products reach the Tanzanian market.
With bioaccess®’s comprehensive , the process is streamlined, allowing for . These services encompass:
- Feasibility studies
- Site selection
- Compliance reviews
- Trial setup
- Import permits
- Project management
- Reporting on study status, inventory, and adverse events
Bioaccess®’s innovative 6-8 week sprint approach enables faster enrollment of , significantly impacting local economies through job creation and healthcare improvement. This collaboration not only enhances the efficiency of but also promotes international partnerships, ultimately aiding health on a broader scale.
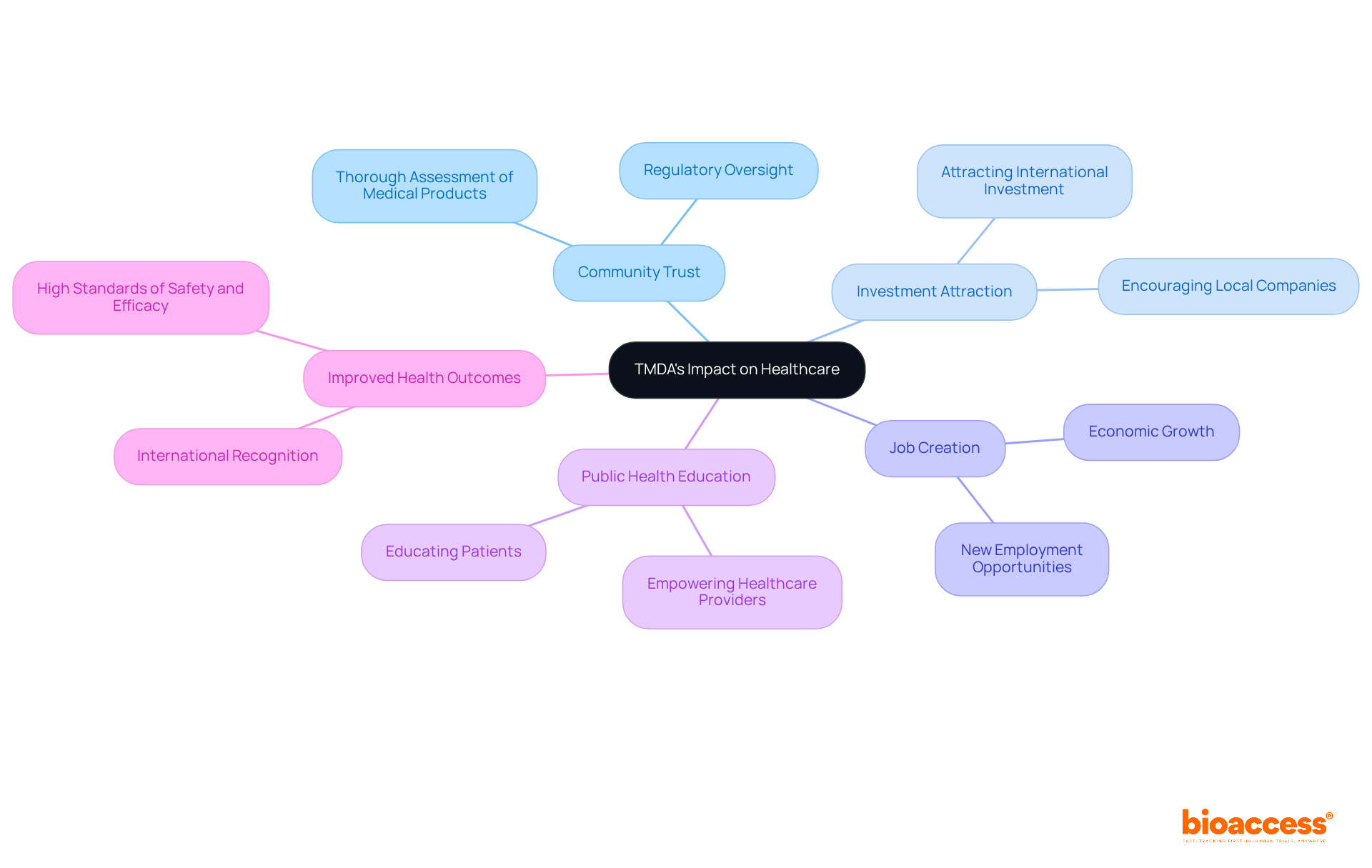
Assess TMDA’s Impact: Implications for Healthcare and Stakeholders
The influence of the organization on healthcare in Tanzania is profound. By ensuring that medical products are thoroughly assessed and overseen, the agency enhances community trust in healthcare systems and promotes an atmosphere favorable to innovation. This to invest in the Tanzanian market, creating jobs and promoting economic growth.
Furthermore, the organization’s dedication to public health education empowers healthcare providers and patients, resulting in . Stakeholders, including pharmaceutical companies, healthcare professionals, and patients, benefit from , which also contributes to TMDA’s international recognition.
Ultimately, these initiatives contribute to the overall improvement of healthcare delivery in Tanzania while driving through international collaboration and innovation in Medtech.
Conclusion
The Tanzania Medicines and Medical Devices Authority (TMDA) serves as a vital cornerstone in the nation’s healthcare framework, committed to ensuring that medicines and medical devices are not only safe and effective but also of the highest quality. Established under the Medicines and Medical Devices Act of 2003, TMDA is instrumental in safeguarding public health while fostering innovation within the healthcare sector.
With a robust regulatory framework, TMDA diligently assesses and registers pharmaceuticals and healthcare devices, overseeing their safety and efficacy even after they reach the market. Its unwavering dedication to Good Manufacturing Practices and adherence to international standards cultivates community trust and stimulates investment in Tanzanian healthcare. By actively engaging with stakeholders, including pharmaceutical firms and healthcare professionals, TMDA enhances the overall quality of healthcare delivery and propels economic growth.
Given TMDA’s significant contributions, it is essential for stakeholders to engage proactively with this regulatory authority. The collaboration between TMDA and healthcare innovators is crucial for effectively navigating the complexities of medical product regulation and advancing public health initiatives. By emphasizing compliance and nurturing a culture of safety, TMDA’s impact will resonate throughout Tanzania, ultimately leading to improved health outcomes and a more resilient healthcare system.
Frequently Asked Questions
What is the TMDA?
The TMDA, or Tanzania Medicines and Medical Devices Authority, is the governing entity responsible for ensuring the safety, effectiveness, and quality of medicines and healthcare devices in Tanzania.
When was the TMDA established?
The TMDA was established under the Medicines and Medical Devices Act of 2003.
What is the primary role of the TMDA?
The primary role of the TMDA is to safeguard public health by ensuring that all healthcare products meet stringent safety and quality criteria before they are made available to the public.
How does the TMDA contribute to public health?
The TMDA contributes to public health by acting as a gatekeeper for healthcare innovations and ensuring compliance with both national and international standards for medicines and medical devices.

Leave a Reply