Introduction
The initial phases of clinical trials are crucial, laying the groundwork for transformative medical advancements in Chile’s dynamic landscape. By capitalizing on a supportive regulatory environment and a diverse patient base, sponsors can unlock significant advantages in speed and cost efficiency when conducting early phase clinical trials. Navigating the complexities of compliance and patient recruitment poses significant challenges for sponsors. This article explores essential strategies that streamline the trial process and position Chile as a strategic hub for early phase clinical research in Latin America.
Define Early Phase Clinical Trials and Their Importance in Chile
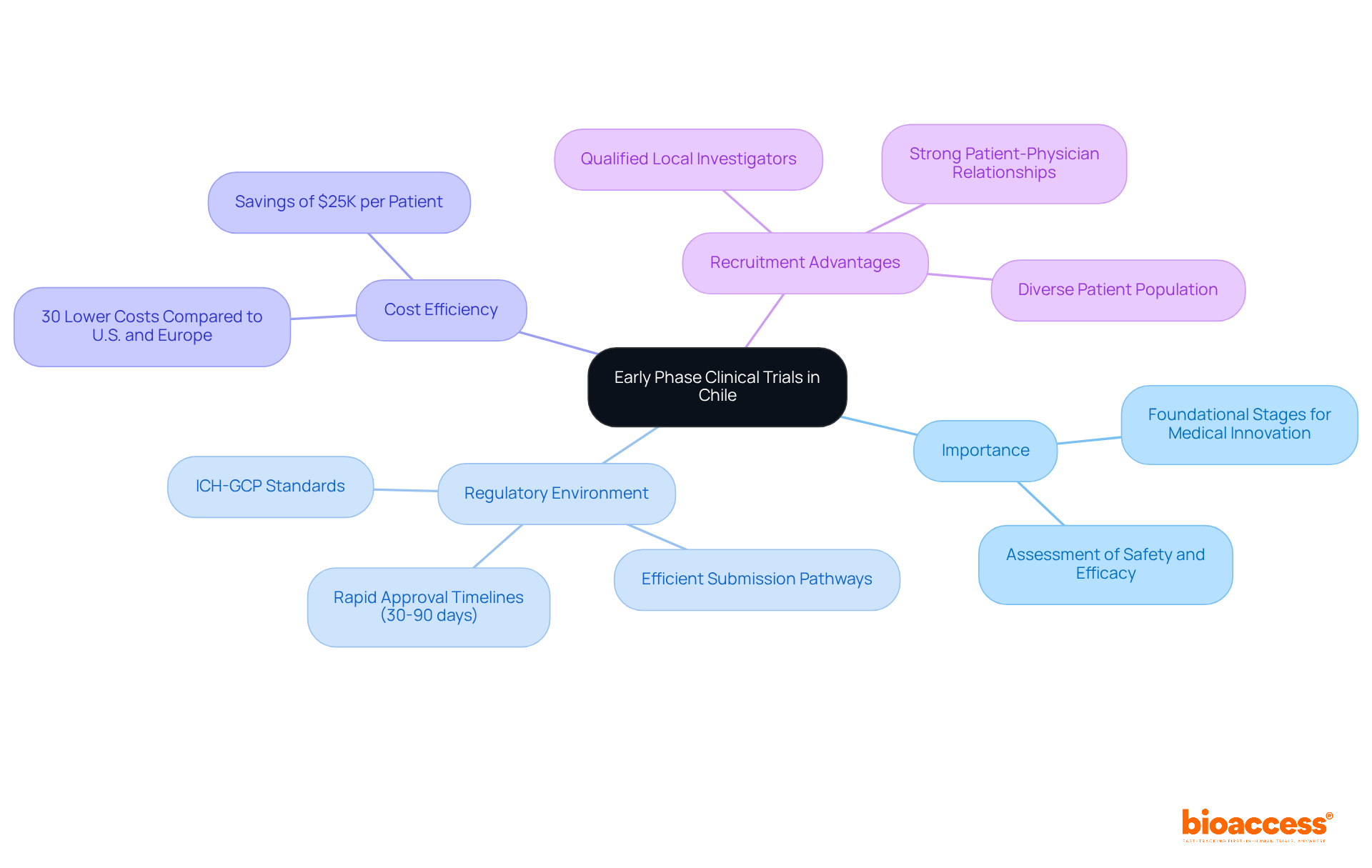
In the realm of clinical research, the initial phases of studies are not just steps; they are critical milestones that determine the future of medical innovation. Initial phase clinical studies, specifically Phase I and II, represent the foundational stages for assessing new medical treatments, drugs, or devices in human subjects. These assessments are crucial for establishing safety, tolerability, pharmacokinetics, and preliminary efficacy. In this nation, the importance of early phase studies is heightened by the robust oversight framework, which includes adherence to ICH-GCP standards and efficient pathways for submissions through agencies like the Instituto de Salud Pública (ISP). This framework not only facilitates faster patient recruitment but also lowers operational costs when compared to the U.S. and Europe.
How can sponsors leverage the regulatory environment designed for rapid approvals, with timelines often ranging from 30 to 90 days for initial submissions? Conducting these trials in Chile allows sponsors to tap into a diverse patient population, enhancing recruitment potential. Local investigators are not just cooperative; they are well-qualified and deeply familiar with the nuances of clinical research, making collaboration smoother.
Additionally, the cost-efficiency of performing studies in this region is a notable benefit. By leveraging local resources and expertise, sponsors can achieve first-in-human milestones more efficiently, preserving capital and extending their operational runway. With bioaccess®, companies can save around $25K per patient through pre-negotiated site contracts and experience overall study costs that are 30% lower than in other regions. The blend of a supportive policy environment and a robust patient-physician rapport boosts adherence and speeds up the research development process, positioning the country as an appealing choice for early phase studies in Latin America. As the landscape of clinical research evolves, embracing the advantages of early phase clinical trial Chile could redefine success for many sponsors.
Explore Regulatory Requirements for Early Phase Trials in Chile
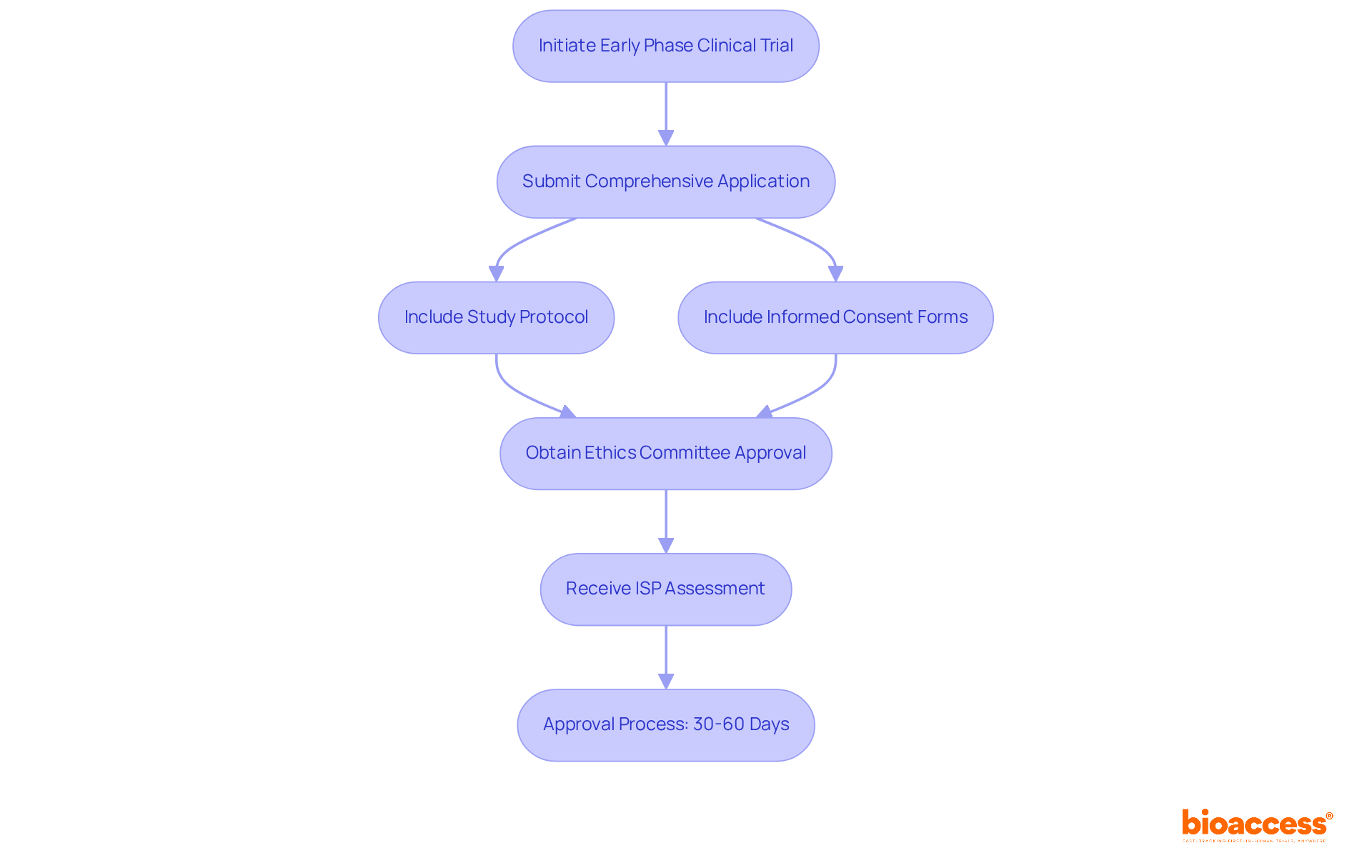
Navigating the regulatory landscape in Chile is crucial for successful clinical research. Sponsors must submit a comprehensive application to initiate an early phase clinical trial in Chile. This application includes:
- The study protocol
- Informed consent forms
- Approvals from an accredited ethics committee
Typically, the approval process lasts 30 to 60 days, depending on the study’s complexity. The ISP usually assesses applications within 30 business days. Compliance with ICH-GCP guidelines is mandatory, and all documentation must be translated into Spanish to meet local requirements. Additionally, securing an Import License for any investigational products is essential, with the process for obtaining this license taking approximately 2 to 4 weeks. Other pertinent oversight bodies, such as INVIMA, ANVISA, and COFEPRIS, may also have a role depending on the nature of the study and the products involved.
How can sponsors navigate these compliance pathways more efficiently? By leveraging bioaccess®’s expertise, they can reduce delays and ensure a smooth study initiation. The expedited ethics approvals and early feasibility studies provided by bioaccess® enhance the efficiency of research in the region, positioning Latin America, particularly Chile, as a strategic advantage for MedTech and Biopharma companies conducting early phase clinical trials in Chile. Understanding these processes not only streamlines research but also positions companies for success in a competitive market.
Implement Strategies for Successful Execution of Early Phase Trials
Navigating the regulatory landscape for early phase clinical trial Chile can be daunting, but strategic partnerships can pave the way for success. To ensure effective implementation, sponsors should consider the following strategies:
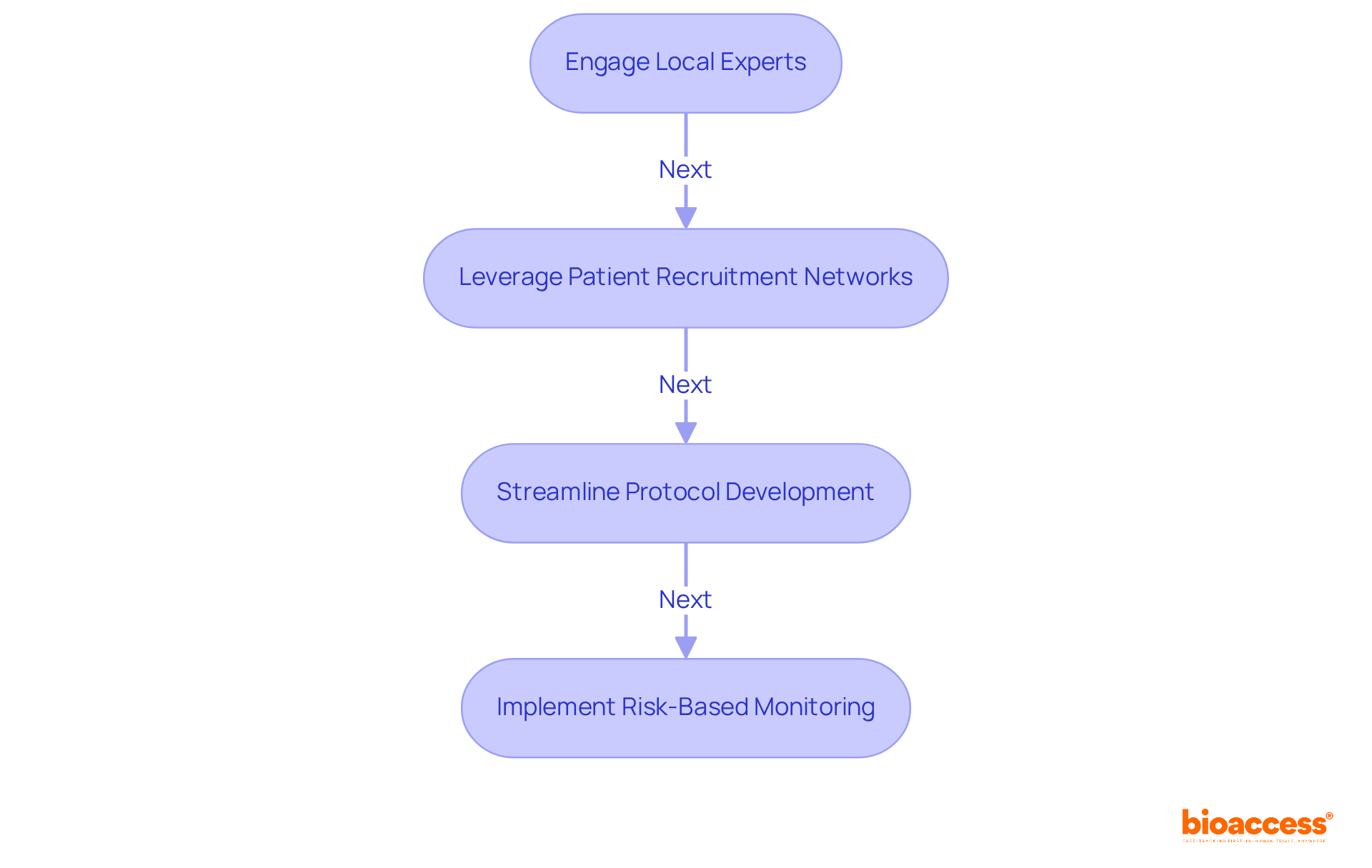
- Engage Local Experts: Partner with specialized CROs like bioaccess®, the only CRO built exclusively for first-in-human trials. This collaboration simplifies the approval process, allowing for quicker navigation of compliance complexities. With the Public Health Institute of Chile (ISP) known for its efficient review processes, approvals for early phase clinical trial Chile can be secured in as little as 4-8 weeks, compared to the 6+ months often seen in the US/EU.
- Leverage Patient Recruitment Networks: Utilize bioaccess®’s established networks to enhance patient recruitment efforts. This ensures a diverse participant pool that accurately reflects the target population. With 80% treatment-naïve patient populations and 50% faster enrollment, bioaccess® can help you efficiently meet your recruitment goals.
- Streamline Protocol Development: Develop clear and concise study protocols that align with compliance expectations, such as ICH-GCP adherence. bioaccess®’s expertise guarantees that your protocols meet the required standards for FDA acceptance, utilizing the Innovation Runway to expedite milestones in the trial process.
- Implement Risk-Based Monitoring: Adopt risk-based monitoring strategies to ensure compliance and data integrity while minimizing costs. By employing these strategies, sponsors can enhance their operational efficiency and reduce time to market, ultimately preserving equity and extending their runway for further R&D investments.
By embracing these strategies, sponsors not only enhance their operational efficiency but also position themselves for sustainable growth in the competitive Medtech landscape.
Identify and Overcome Challenges in Early Phase Clinical Trials
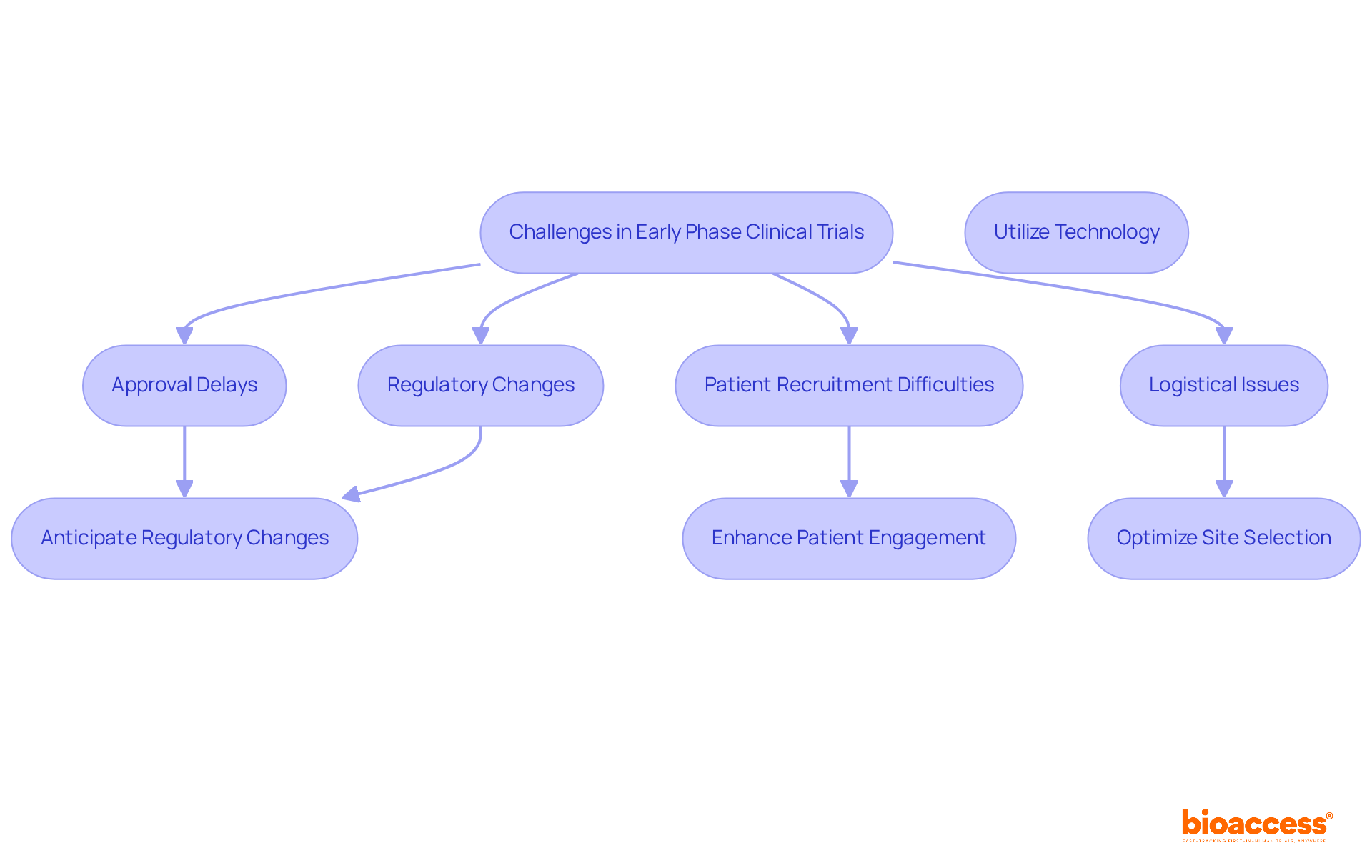
Navigating the complexities of early phase clinical trial Chile presents significant challenges that demand strategic foresight. Approval delays, patient recruitment difficulties, and logistical issues can significantly hinder the progress of early phase clinical trial Chile. To tackle these obstacles effectively, sponsors need to consider several key strategies:
- Anticipate Regulatory Changes: Stay informed about evolving regulations from the Instituto de Salud Pública (ISP) and engage with them early in the process to mitigate potential compliance issues. Recent regulatory modifications, including Law 14.874/24, which was approved in May 2024, aim to simplify the assessment process, minimizing bureaucratic hurdles and improving predictability in approval processes. With Bioaccess® on your side, you’ll be well-prepared for these changes.
- Enhance Patient Engagement: Develop targeted outreach programs to educate potential participants about the benefits of the study, which can significantly improve recruitment rates. Given that dropout rates in Latin America are one-third of those in the U.S. and EU, fostering strong relationships between patients and physicians can enhance retention. Bioaccess® has successfully navigated these challenges, helping clients engage effectively with local populations.
- Optimize Site Selection: Choose research sites with demonstrated success in early phase studies to ensure efficient operations and effective patient management. Chile’s urban centers provide a concentrated subject pool, making it easier to recruit participants who are often eager to engage in early phase clinical trial Chile. Bioaccess®’s extensive network can assist in identifying the most suitable sites.
- Utilize Technology: Implement digital tools for data collection and monitoring to streamline processes and enhance communication among stakeholders. Utilizing technology can assist in overcoming logistical difficulties and enhancing overall study efficiency. Bioaccess® utilizes cutting-edge technological solutions to enable smoother study operations.
By leveraging local expertise and innovative strategies, sponsors can turn these challenges into opportunities for success in clinical research.
Conclusion
Navigating the complexities of early phase clinical trials in Chile presents both challenges and opportunities for sponsors in the MedTech and Biopharma sectors. These initial studies, which include Phase I and II, are crucial for laying the groundwork for future medical advancements. The unique regulatory environment in Chile, with its efficient approval timelines and supportive infrastructure, makes the country an attractive destination for conducting these trials. This enables sponsors to achieve significant milestones swiftly and cost-effectively.
What strategies can enhance the success of early phase trials in Chile? Here are several key tactics to consider:
- Engaging local experts
- Leveraging patient recruitment networks
- Streamlining protocol development
- Implementing risk-based monitoring
These critical tactics can mitigate challenges and optimize operational efficiency. Moreover, understanding the regulatory landscape, including compliance with ICH-GCP guidelines and navigating the approval process with the ISP, is fundamental for ensuring a smooth trial initiation and execution.
As the clinical research landscape continues to evolve, embracing the advantages offered by early phase clinical trials in Chile is essential for sponsors aiming to achieve a competitive edge. Reduced operational costs, faster patient recruitment, and a robust regulatory framework accelerate the path to market and foster innovation in medical treatments. By capitalizing on these insights and strategies, stakeholders can effectively navigate the complexities of early phase trials, ultimately contributing to the advancement of healthcare solutions in Latin America and beyond. By not seizing these opportunities, sponsors risk falling behind in the rapidly evolving landscape of medical innovation.
Frequently Asked Questions
What are early phase clinical trials and why are they important in Chile?
Early phase clinical trials, specifically Phase I and II, are critical milestones in clinical research that assess new medical treatments, drugs, or devices in human subjects. They are essential for establishing safety, tolerability, pharmacokinetics, and preliminary efficacy, making them foundational for future medical innovation.
How does the regulatory environment in Chile support early phase clinical trials?
Chile has a robust oversight framework that adheres to ICH-GCP standards and provides efficient submission pathways through agencies like the Instituto de Salud Pública (ISP). This environment facilitates faster patient recruitment and reduces operational costs compared to the U.S. and Europe.
What are the benefits of conducting early phase clinical trials in Chile?
Conducting trials in Chile allows sponsors to access a diverse patient population, enhancing recruitment potential. The cost-efficiency of studies in this region is notable, with companies able to save around $25K per patient and experience overall study costs that are 30% lower than in other regions.
What is the typical timeline for initial submissions for early phase trials in Chile?
The typical timeline for initial submissions in Chile ranges from 30 to 90 days, allowing for rapid approvals and facilitating quicker progression of clinical studies.
How do local investigators contribute to the success of early phase trials in Chile?
Local investigators in Chile are well-qualified and familiar with the nuances of clinical research, which makes collaboration smoother and enhances the overall efficiency of conducting trials.
What financial advantages do sponsors gain from conducting early phase trials in Chile?
By leveraging local resources and expertise, sponsors can achieve first-in-human milestones more efficiently, preserving capital and extending their operational runway, thereby enhancing the overall financial viability of their clinical studies.
How does the relationship between patients and physicians in Chile impact clinical research?
The strong rapport between patients and physicians in Chile boosts adherence to clinical trials and speeds up the research development process, making the country an appealing choice for early phase studies in Latin America.
List of Sources
- Define Early Phase Clinical Trials and Their Importance in Chile
- Latin America Clinical Trials Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trials-market/latin-america)
- Early Feasibility Studies in Latin America (https://greenlight.guru/blog/early-feasibility-studies-in-latin-america)
- Clinical Trials – Considerations for Latin America (https://clinicaltrialsarena.com/news/clinical-trials-considerations-for-latin-america-5813786-2)
- Explore Regulatory Requirements for Early Phase Trials in Chile
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Chile (ISP – Instituto de Salud Pública) – PharmaRegulatory.in – India’s Regulatory Knowledge Hub (https://pharmaregulatory.in/country-specific-regulatory-affairs/chile-isp-instituto-de-salud-publica)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Chile Clinical Trial Regulations | ISP Guide | bioaccess® (https://bioaccessla.com/regulatory-guide/chile)
- Implement Strategies for Successful Execution of Early Phase Trials
- Chile First-in-Human Clinical Trial Market Size, Share, Growth & Trends 2025–2033 (https://reedintelligence.com/market-analysis/chile-first-in-human-clinical-trial-market)
- Clinical Trials Patient Recruitment in Latin America | H Clinical (https://hclinical.com/patient-recruitment)
- The Importance of Patient Recruitment in Clinical Trials | Novotech CRO (https://novotech-cro.com/blog/importance-patient-recruitment-clinical-trials)
- FDA Issues Guidance on Modernizing Statistical Methods for Clinical Trials (https://fda.gov/news-events/press-announcements/fda-issues-guidance-modernizing-statistical-methods-clinical-trials)
- Optimizing Early Phase Clinical Trials to Minimize Risk and Improve Decision-Making | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/optimizing-early-phase-clinical-trials-to-minimize-risk-and-improve-decision-making)
- Identify and Overcome Challenges in Early Phase Clinical Trials
- 7 quotes from 2023 to guide you on the medtech market (https://tiinatyni.com/blogi/7-quotes-from-2023-to-guide-you-on-the-medtech-market)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- Clinical Trials Quotes (2 quotes) (https://goodreads.com/quotes/tag/clinical-trials)
- Clinical Trials Patient Recruitment in Latin America | H Clinical (https://hclinical.com/patient-recruitment)

Leave a Reply