Introduction
Navigating the complex landscape of medical device regulations in India is essential for manufacturers aiming to establish a foothold in this rapidly growing market. The framework established by the Central Drugs Standard Control Authority provides a foundation, but understanding the classification and compliance requirements is crucial for enhancing the chances of successful market entry. The challenge, however, lies in deciphering the approval process, which can often feel daunting and time-consuming.
How can manufacturers effectively navigate these regulatory hurdles to ensure their products not only meet compliance standards but also thrive in a competitive environment? By grasping the intricacies of the regulatory landscape, manufacturers can position themselves for success and capitalize on the opportunities within this dynamic sector.
Understand the Indian Medical Device Regulatory Framework
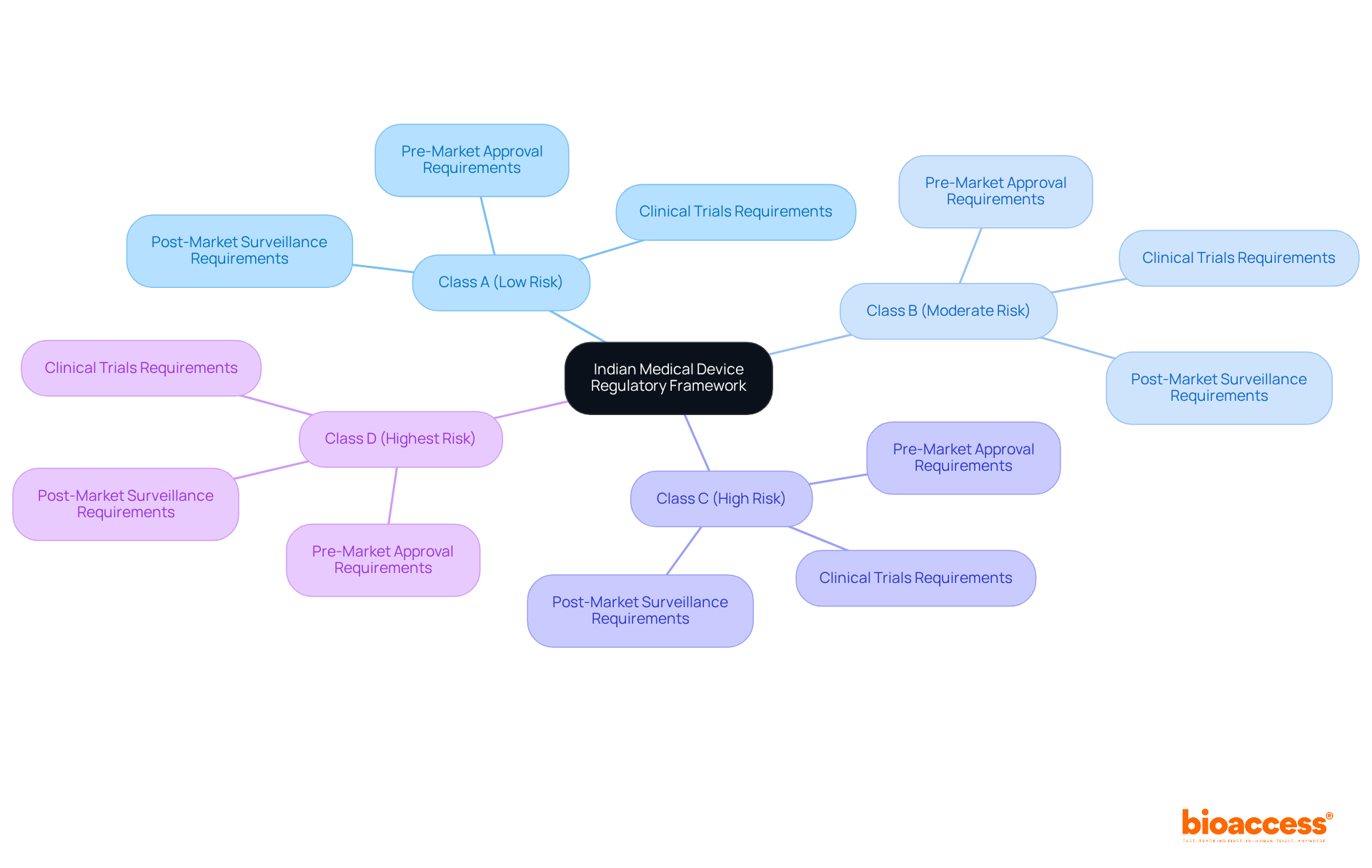
Navigating the for an Indian is essential for anyone looking to enter this complex market. Understanding the framework established by the is crucial. Regulated by the and the s Rules of 2017, this framework categorizes healthcare instruments into four :
- Class A (low risk)
- Class B (moderate risk)
- Class C (high risk)
- Class D (highest risk)
Each category has specific requirements for , , and , all vital for compliance.
Recent updates to the Drugs and Cosmetics Act reflect ongoing efforts to streamline these processes and align with global standards. Comprehending these regulations is not just beneficial; it’s necessary for effectively navigating the authority’s requirements. Are you prepared to ensure that your product meets all necessary criteria for in India? Understanding these key elements can significantly impact your .
Classify Medical Devices According to Indian Standards
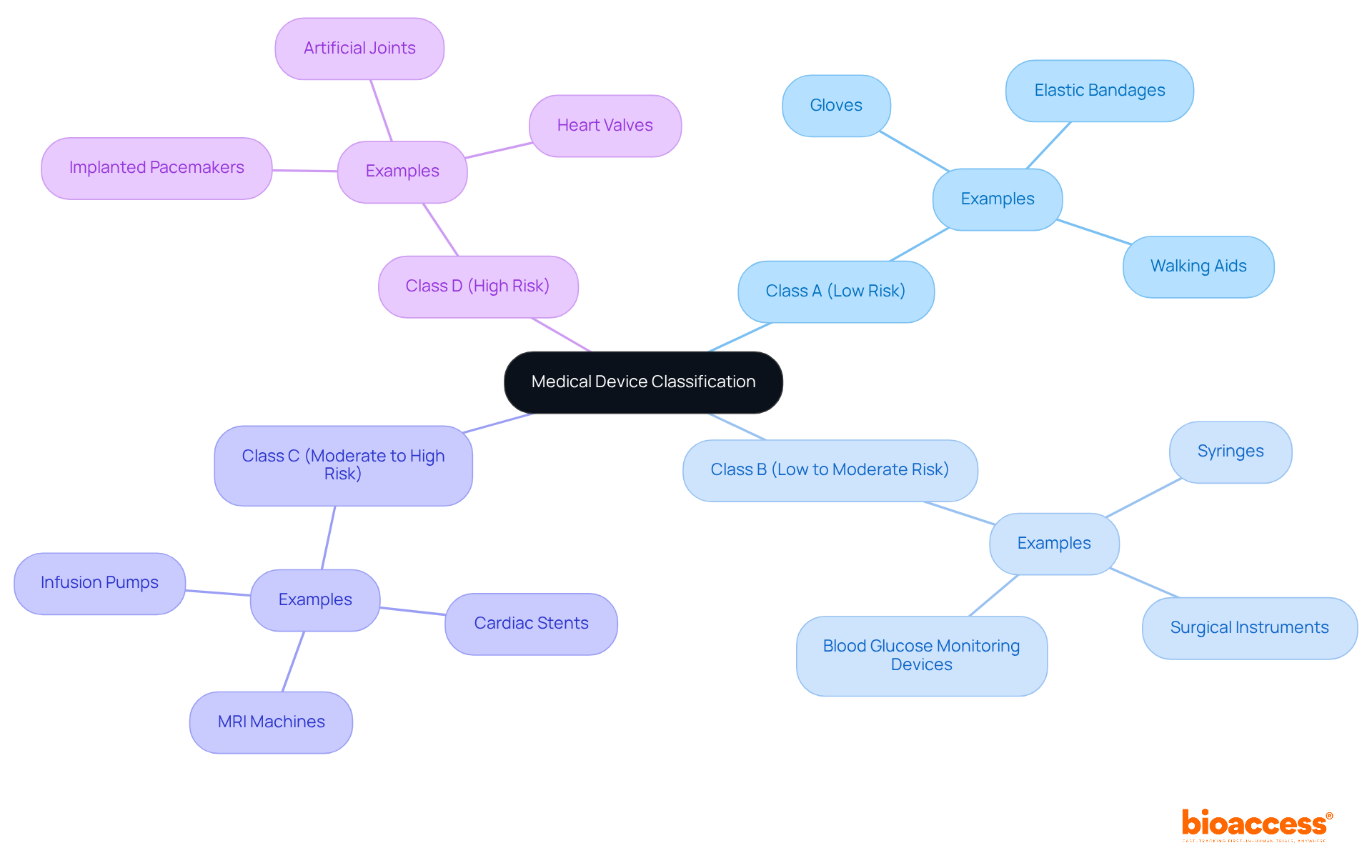
To classify your medical instrument effectively, it’s essential to consult the from the . These guidelines categorize products into four distinct classes based on their intended use and associated risks. Start by evaluating the primary purpose of your apparatus and its potential impact on patient health. The serves as a valuable tool for identifying the appropriate class.
For example:
- like gloves and elastic bandages.
- Class D encompasses and artificial joints.
Thorough documentation of your classification rationale is crucial, as it will be vital for your compliance submissions. Understanding these classifications not only aids in adherence but also for the Indian in India’s rapidly evolving healthcare equipment landscape. By grasping these essential classifications, you position yourself for success in navigating the complexities of .
Follow the Market Pathway for Regulatory Submissions
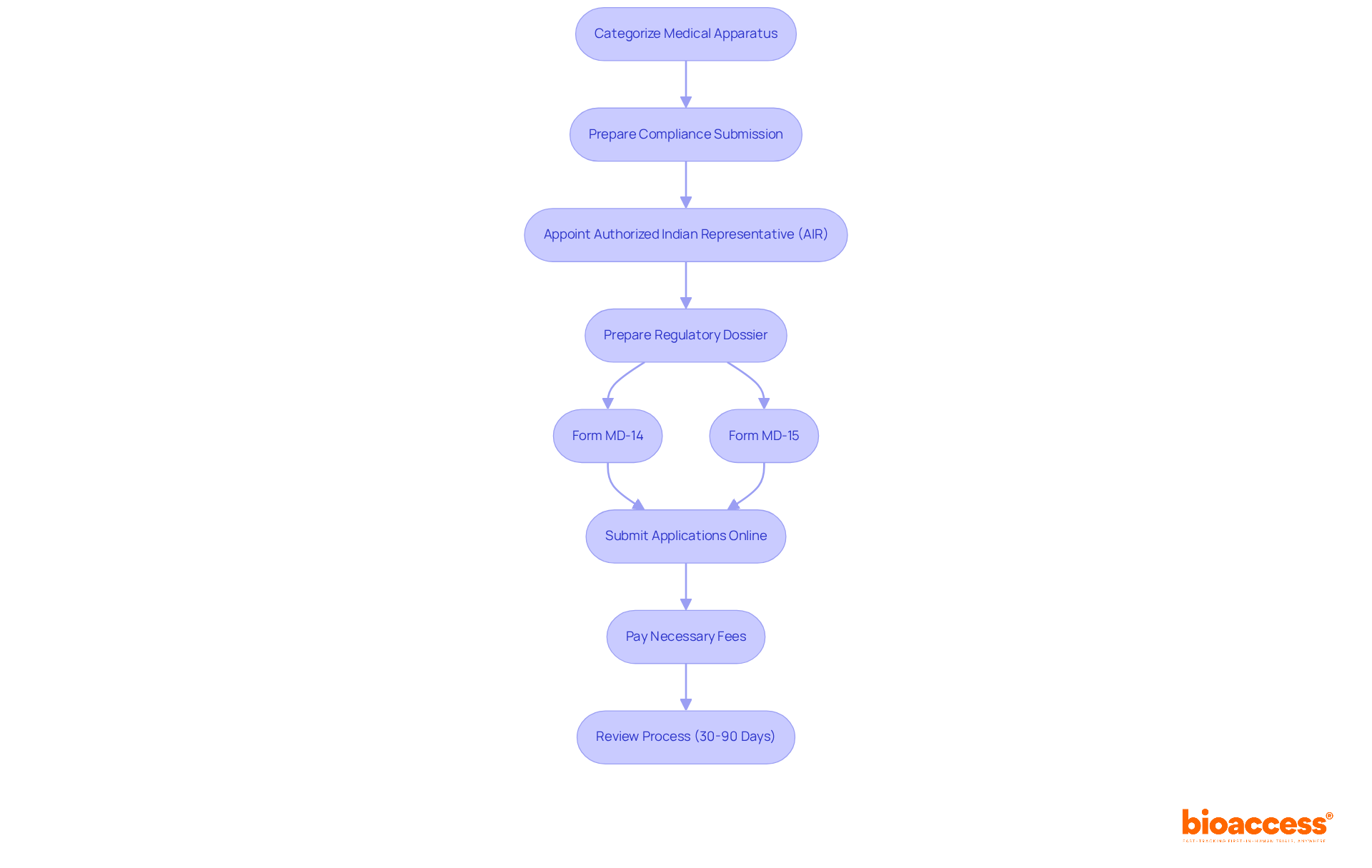
After categorizing your medical apparatus, the next essential step is preparing for compliance submission. This process is particularly crucial for foreign manufacturers, who must appoint an . This representative will facilitate communication with the , ensuring that your submission is both .
The must be meticulously prepared, typically including:
- Form MD-14 for
- Form MD-15 for
Applications should be submitted through the online portal, with all necessary fees paid in advance. Keep in mind that the review process can extend over several months, making it vital to plan accordingly. Are you ready to address any queries from the CDSCO during their evaluation? Timely responses can significantly impact your .
Recent statistics indicate that the approval process can take anywhere from:
- 30 working days for domestic trials
- 90 working days for global trials
This underscores the importance of thorough preparation and compliance with all . Leveraging the expertise of professionals like Ana Criado, Director of Regulatory Affairs at bioaccess, and Katherine Ruiz, an expert in Regulatory Affairs for Medical Devices and In Vitro Diagnostics, can provide invaluable guidance throughout this process.
bioaccess offers services such as and trial data delivery, ensuring that your submission is both . This ultimately facilitates a smoother path to market, allowing you to navigate the complexities of with confidence.
Comply with Post-Market Surveillance and Reporting Requirements
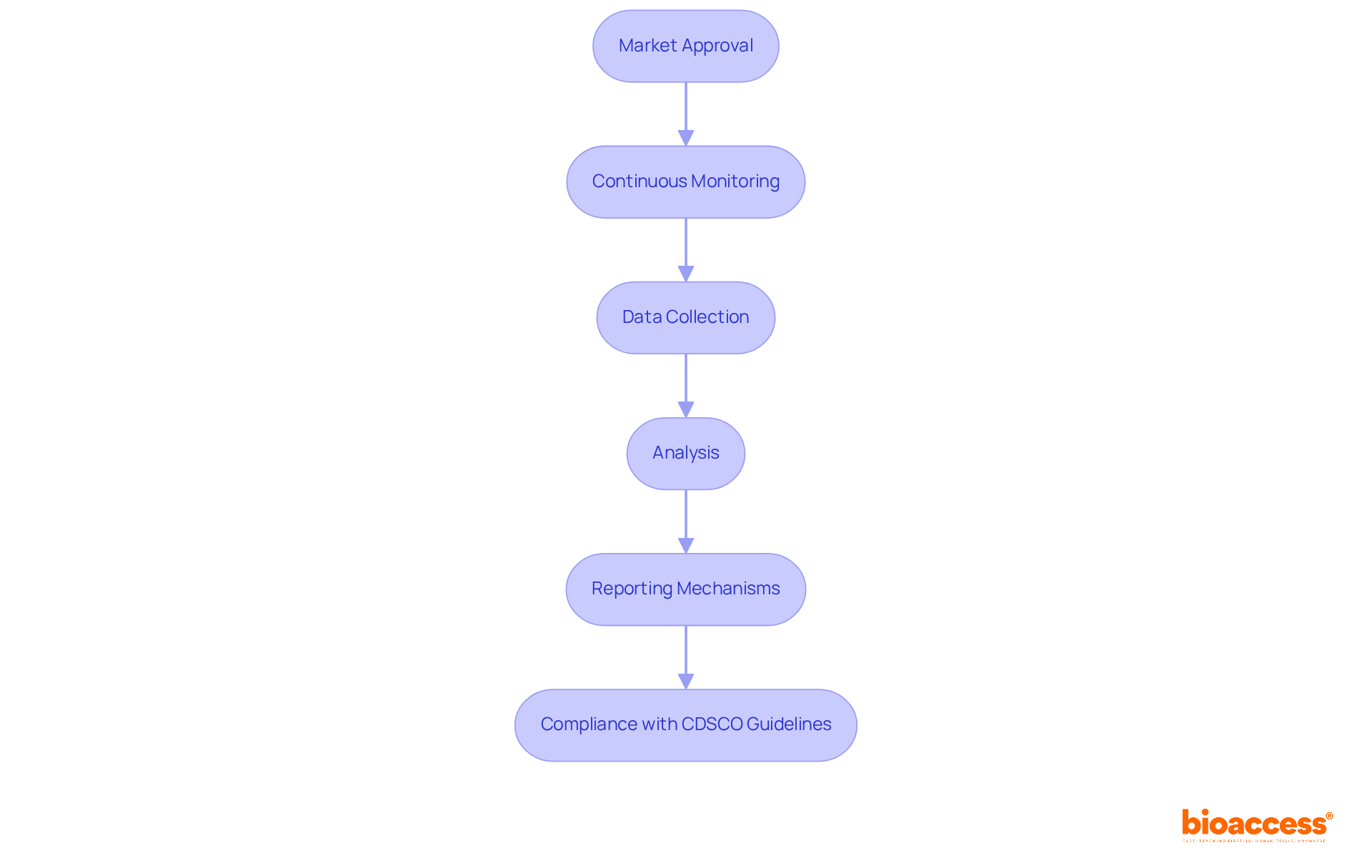
Once your medical instrument secures market approval, adhering to requirements is not just important; it’s crucial. This process involves in real-world applications. Manufacturers are obligated to promptly report any or equipment malfunctions to the Central Drugs Standard Control Authority (CDSCO). Statistics reveal that from July 2015 to October 2019, the CDSCO received 1,931 reports of , with 1,277 classified as serious. This underscores the necessity for vigilant monitoring. Notably, the reporting of surged after 2017, reflecting an increasing awareness of the need for thorough oversight.
To ensure compliance, it’s essential to develop a that includes:
- Regular data collection
- Analysis
This plan should provide clear guidelines for identifying and documenting , along with a structured approach for assessing equipment performance over time. Maintaining detailed records of all post-market activities is vital, as these may be subject to audits by regulatory authorities.
Efficient in the Indian often require collaboration with healthcare practitioners to gather feedback and insights on the usage of Indian medical devices. Ongoing training and awareness programs can foster a culture of reporting, enhancing the overall safety and effectiveness of healthcare tools. As highlighted by the Indian Pharmacopoeia Commission, creating a will significantly improve the management of safety concerns, ultimately safeguarding patient health. Furthermore, the implementation of national regulations on medical devices, which began in January 2018, has profoundly impacted PMS practices. Staying updated with the for 2026 is also essential to ensure compliance.
Conclusion
Navigating the Indian medical device regulatory process is a complex journey that demands a thorough understanding of the established framework, classification, and compliance requirements. Grasping the intricacies of the regulations set forth by the Central Drugs Standard Control Authority is essential for stakeholders aiming to position their products for successful market entry. The emphasis on risk classification and the necessity of meticulous regulatory submissions cannot be overstated, as these elements lay the groundwork for compliance and operational success.
Key insights from this discussion underscore the importance of:
- Categorizing medical devices according to their risk levels
- Preparing detailed regulatory submissions
- Adhering to stringent post-market surveillance requirements
Each step in this process is critical-from understanding the classification matrix to ensuring ongoing compliance with post-market reporting. The role of an Authorized Indian Representative is particularly significant for foreign manufacturers, facilitating smoother interactions with regulatory authorities and enhancing the likelihood of timely approvals.
Ultimately, navigating the Indian medical device regulatory landscape transcends mere compliance; it fosters a culture of safety and vigilance that benefits both patients and healthcare providers. As the market evolves, staying informed about current regulations and best practices is essential for manufacturers seeking to thrive in this competitive environment. By embracing these guidelines and actively participating in post-market surveillance, manufacturers can significantly enhance the safety and effectiveness of medical devices, ensuring they meet the highest standards of quality and performance.
Frequently Asked Questions
What is the importance of understanding the Indian medical device regulatory framework?
Understanding the Indian medical device regulatory framework is crucial for anyone looking to enter the complex market, as it helps navigate the approval process established by the Central Drugs Standard Control Authority.
What laws govern the regulation of medical devices in India?
The regulation of medical devices in India is governed by the Drugs and Cosmetics Act of 1940 and the Medical Devices Rules of 2017.
How are medical devices categorized in India?
Medical devices in India are categorized into four risk levels: Class A (low risk), Class B (moderate risk), Class C (high risk), and Class D (highest risk).
What are the requirements for each category of medical devices?
Each category has specific requirements for pre-market approval, clinical trials, and post-market surveillance, which are vital for compliance.
Have there been any recent updates to the regulatory framework?
Yes, recent updates to the Drugs and Cosmetics Act reflect ongoing efforts to streamline processes and align with global standards.
Why is it necessary to comprehend these regulations?
Comprehending these regulations is necessary for effectively navigating the authority’s requirements and ensuring that products meet all necessary criteria for market entry in India.
List of Sources
- Understand the Indian Medical Device Regulatory Framework
- Medical Devices Industry in India – Market Share, Growth & Scope (https://ibef.org/industry/medical-devices)
- Statistics & Figures: Medical Device Registration India (https://morulaa.com/statistics-figures-medical-device-registration-india)
- Asia-Pacific Roundup: India’s CDSCO seeks feedback on medical device software regulation (https://raps.org/news-and-articles/news-articles/2025/10/asia-pacific-roundup-india-s-cdsco-seeks-feedback)
- India Medical Devices (https://trade.gov/market-intelligence/india-medical-devices)
- Classify Medical Devices According to Indian Standards
- India’s Medical Device Regulations: A 2025 Update (https://pacificbridgemedical.com/publication/india-medical-device-regulations-2025-update)
- CDSCO New Medical Device Classification List – India Update (https://elexes.com/new-classification-list-for-medical-device)
- Medical Devices Risk Classification in India | CDSCO Guidelines (https://nkgabc.com/medical-devices-risk-based-classification)
- CDSCO Classification for Medical Devices in India | Operon Strategist (https://operonstrategist.com/cdsco-classification-for-medical-devices)
- An Overview of CDSCO Medical Devices Classification (https://pharmadocx.com/an-overview-of-cdsco-medical-devices-classification)
- Follow the Market Pathway for Regulatory Submissions
- Medical Device Import Regulations in India: Step-by-Step (2025) (https://cliniexperts.com/step-by-step-guide-to-medical-device-import-regulations-in-india-2025)
- Indian Regulatory Dossier preparation – Things to remember (https://morulaa.com/things-to-remember-while-preparing-your-regulatory-dossier)
- The Evolving Regulatory Framework in India: Impact on the Pharmaceutical Industry (https://globalforum.diaglobal.org/issue/december-2024/the-evolving-regulatory-framework-in-india-impact-on-the-pharmaceutical-industry)
- CDSCO Registration Timeline for Medical Devices in India (https://cliniexperts.com/cdsco-registration-timeline-how-long-does-it-take-for-medical-devices)
- Comprehensive Guide To Registering Medical Devices In India – Tecnova (https://tecnovaglobal.com/blog/comprehensive-guide-to-registering-medical-devices-in-india)
- Comply with Post-Market Surveillance and Reporting Requirements
- Reporting of adverse events related to medical devices: A… : Indian Journal of Pharmacology (https://journals.lww.com/iphr/fulltext/2023/55020/reporting_of_adverse_events_related_to_medical.7.aspx)
- Implementation of adverse event reporting for medical devices, India – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC7047028)
- Health-care Professionals’ Perception toward Medical Device … : Indian Journal of Public Health (https://journals.lww.com/ijph/fulltext/2024/07000/health_care_professionals__perception_toward.14.aspx)
- Study of Adverse Events Associated With the Use of Medical Devices Reported Under the Materiovigilance Programme of India in a Tertiary Care Teaching Hospital – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC12883217)

Leave a Reply