Introduction
Understanding how to effectively leverage Ex US data for FDA submissions can truly transform the approval processes for companies. With the FDA increasingly recognizing the value of high-quality foreign research, mastering the nuances of this data not only boosts the chances of successful submissions but also meets the rigorous U.S. standards for safety and efficacy. Yet, navigating the complexities of Ex US data comes with its own challenges.
How can companies ensure compliance while steering clear of common pitfalls that might delay their applications? This guide outlines five essential steps to effectively harness Ex US data, paving the way for a smoother FDA submission experience.
Understand the Role of Ex US Data in FDA Submissions
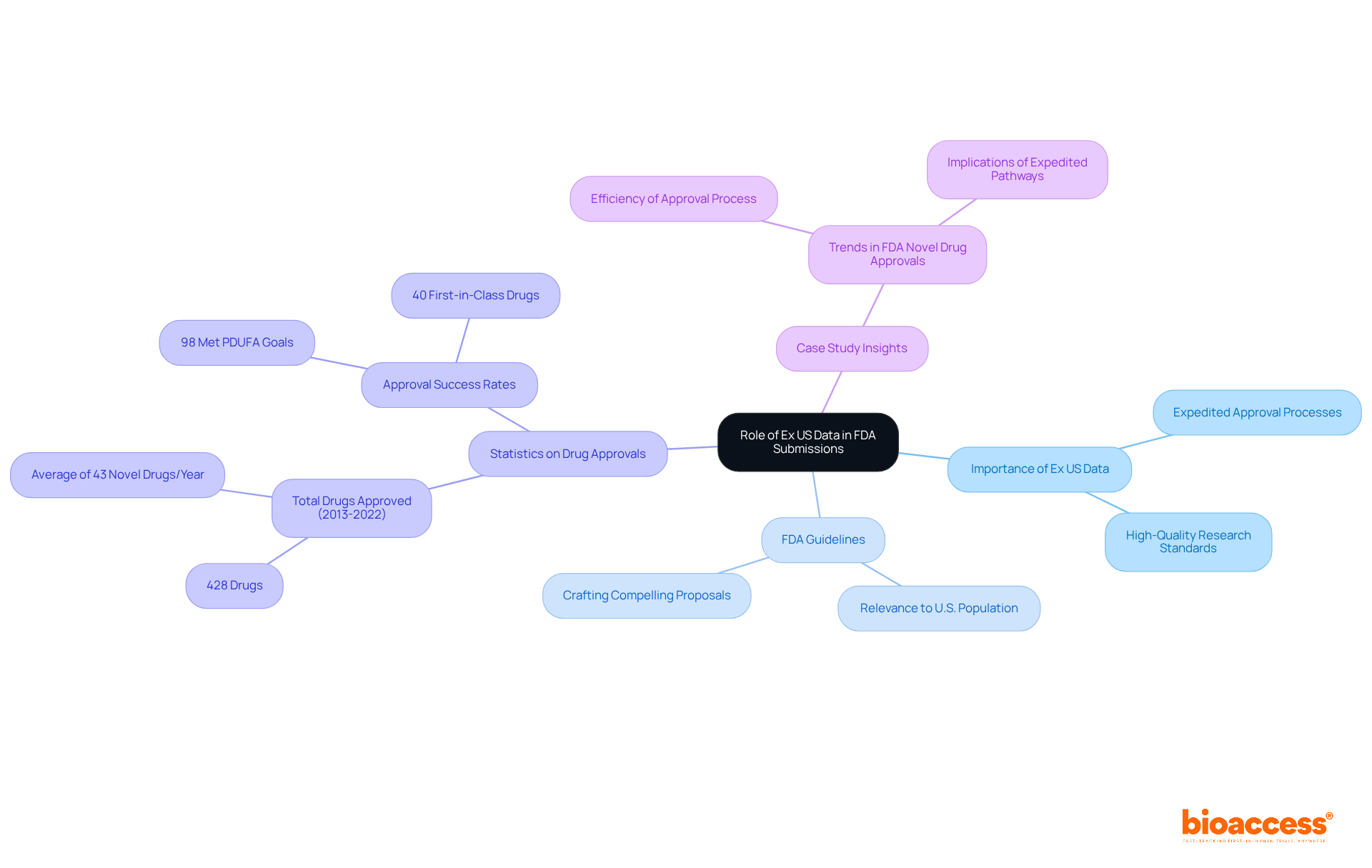
Ex US information refers to medical data collected outside the United States that explains for . Understanding its role is crucial for companies aiming to . The FDA has increasingly recognized the significance of , especially when it meets . Notably, an impressive 98% of novel drug approvals successfully meet the PDUFA review goal dates, showcasing the effectiveness of the approval process. This information can be pivotal in demonstrating a product’s effectiveness, particularly when U.S. are impractical or when faster timelines are necessary.
Companies should familiarize themselves with on for , particularly regarding its relevance to the U.S. population and medical practices. This understanding will aid in crafting a compelling proposal that aligns with FDA expectations. For instance, the case study on ” illustrates how 428 drugs received approval during this period, with 40% classified as first-in-class. This highlights the importance of employing thorough information in submissions.
Identify Required Ex US Data for Submission
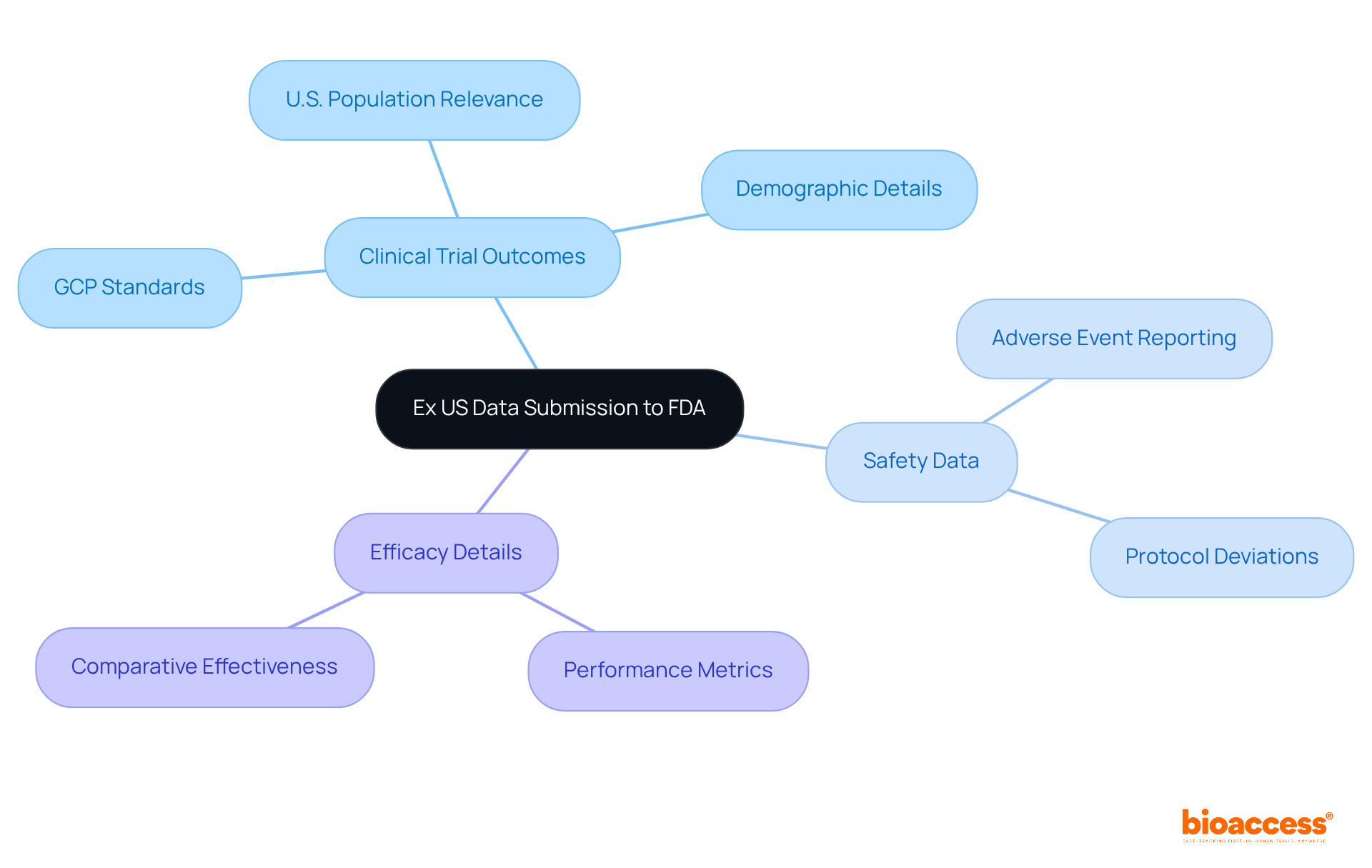
To effectively submit Ex US information to the FDA, it’s essential to understand how to use and identify the . This typically encompasses:
- Safety data
- Efficacy details
These elements collectively illustrate the product’s performance within a relevant patient population. Companies must ensure that their information collection adheres to and accurately reflects the U.S. population. Additionally, any submitted information should include a thorough explanation of its relevance to the U.S. context, which involves providing and clarifying how the study design aligns with U.S. medical practices.
Interestingly, 85% to 95% of 510(k) applications do not require clinical information. However, for the remaining applications, . It’s also important to remember that the FDA has a , which can significantly impact your study schedule. By carefully identifying and preparing the essential information, and ensuring that the is kept current with any amendments, companies can greatly enhance their chances of a successful submission by learning how to use ex US data for .
Compile and Format Ex US Data for FDA Submission
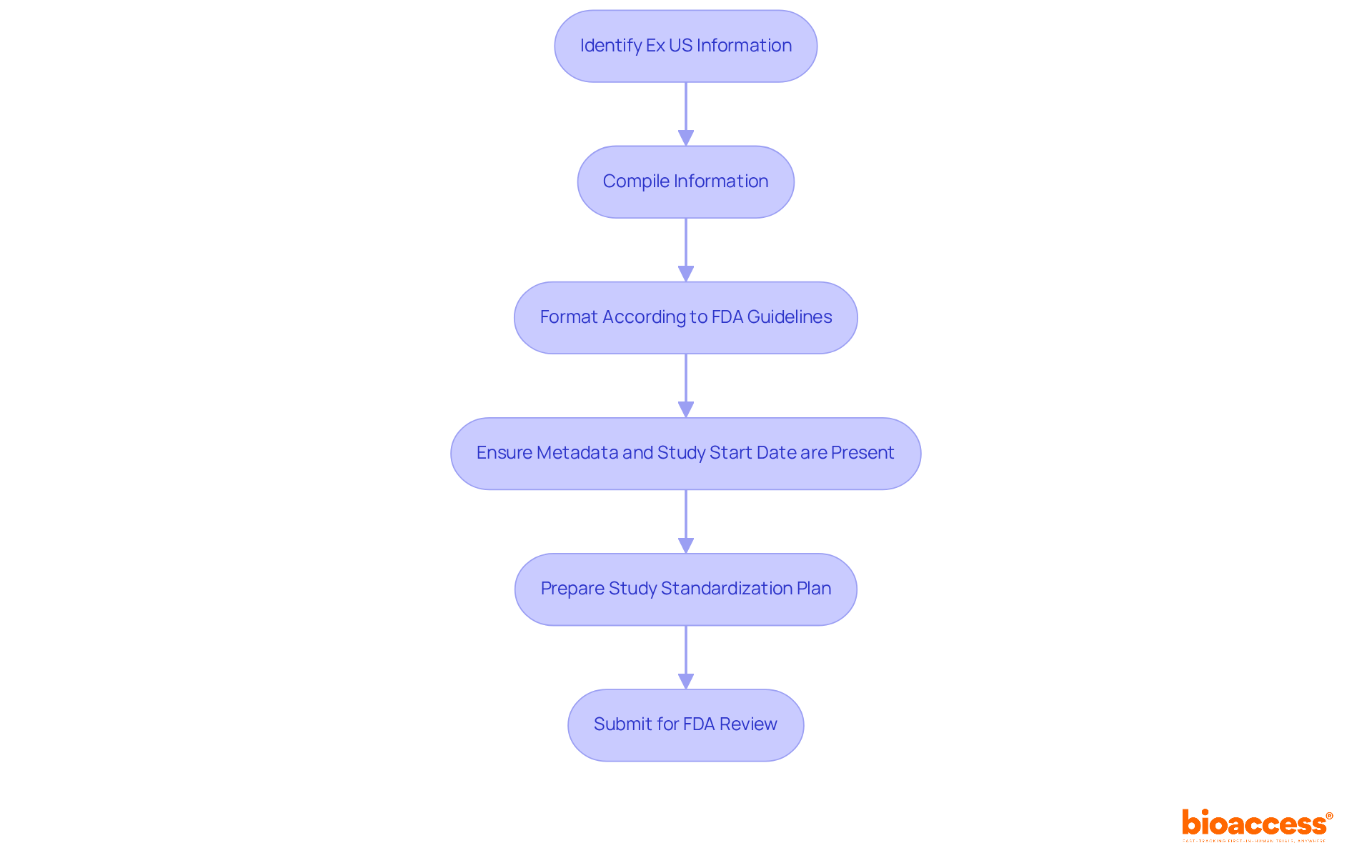
Once the necessary Ex US information has been identified, the next step is to compile and format it according to . This crucial process involves arranging the information into a that is easy for reviewers to navigate. Companies should adopt the for applications, as it is the standard accepted by the FDA. Each dataset must be clearly labeled and include metadata that outlines the contents and context of the information. Furthermore, the Study Start Date (TSPARMCD=”SSTDTC”) must be present in the SDTM TS dataset to ensure compliance with submission requirements.
In addition, preparing a for sent to CBER is essential, as this will aid in maintaining information integrity. Ensuring that all information is accurate, complete, and verifiable is paramount; even minor inconsistencies can lead to significant validation issues, potentially delaying the review process. By adhering to these guidelines and aligning with the , companies can understand how to use ex us data for FDA submission in a manner that facilitates FDA review.
At bioaccess, we understand the urgency for startups. Our trials are and audit-ready, ensuring that your is not only robust but also accelerates your . This support is vital in assisting you to secure the funding you require.
Ensure Regulatory Compliance for Ex US Data
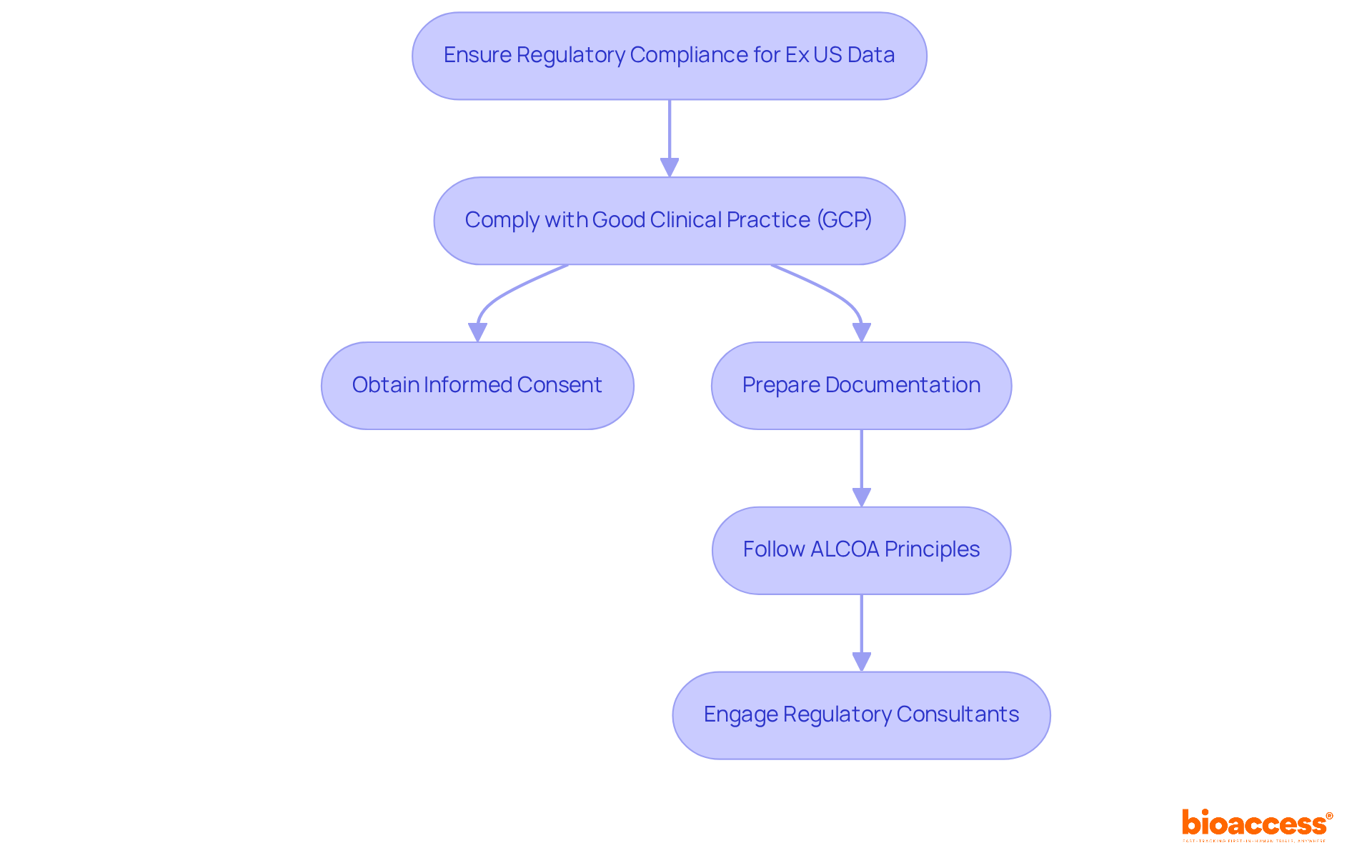
To ensure that Ex US information aligns with FDA regulations, it is crucial to know while adhering to several key principles. First, all information collection must comply with , which mandates obtaining informed consent from participants and ensuring robust ethical oversight. Statistics indicate that account for 6% of critical GCP non-compliances in US trials, underscoring the importance of this step. Notably, in 2025, a California investigator received an FDA warning for enrolling subjects before , highlighting the in clinical trials.
Businesses should also prepare extensive documentation demonstrating where the information was collected, especially regarding how to use ex us data for , as the FDA emphasizes the need for thorough record-keeping and protocol adherence. The principle is essential for ensuring that documentation meets FDA standards. Furthermore, a is essential to understand and confirm that it meets FDA standards for quality and integrity. Engaging regulatory consultants or legal experts specializing in FDA applications can provide valuable insights and guidance.
By prioritizing adherence to GCP principles, companies can significantly reduce risks related to their applications and enhance the likelihood of successful approval.
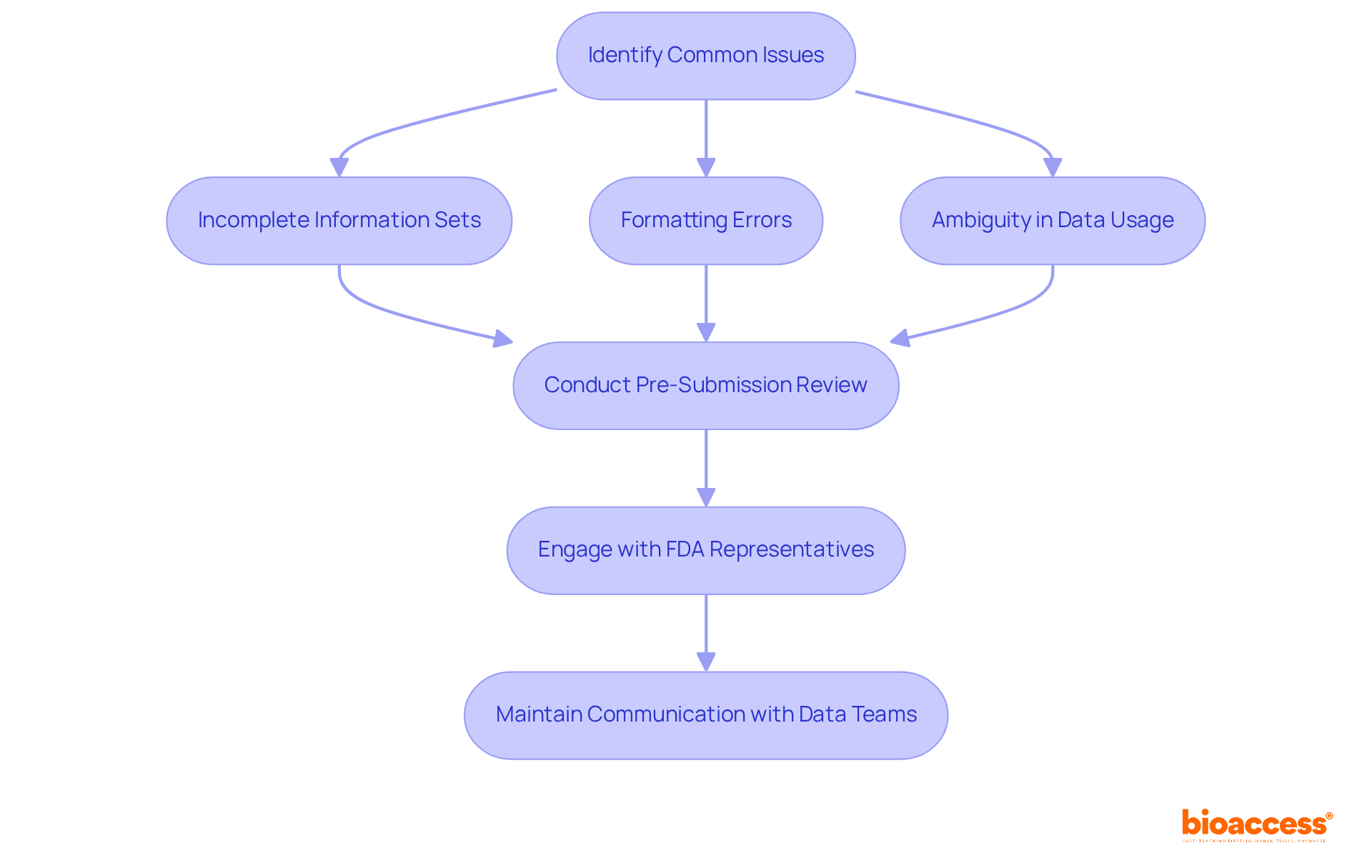
Troubleshoot Common Issues with Ex US Data Submissions
Even with meticulous preparation, challenges can emerge when understanding how to use ex US data for . include:
- Incomplete information sets
- Formatting errors
- Ambiguity regarding as it pertains to the U.S. population
To effectively address these challenges, companies should:
- Conduct a thorough to pinpoint any .
- early in the process to yield valuable insights and clarify expectations.
- Maintain with data management teams to ensure that any discrepancies are resolved swiftly.
By taking a , companies can significantly reduce the risk of delays and enhance the overall quality of their submissions.
Conclusion
Understanding how to effectively use Ex US data for FDA submissions is essential for companies aiming to navigate the complexities of the approval process. By leveraging high-quality foreign research that meets U.S. safety and efficacy standards, businesses can significantly enhance their chances of obtaining timely approvals. This guide outlines the crucial steps to ensure that Ex US data is not only relevant but also presented in a way that aligns with FDA expectations.
Key insights include:
- The necessity of identifying the required types of Ex US data, such as clinical trial outcomes and safety information, while ensuring compliance with Good Clinical Practice standards.
- The importance of compiling and formatting data according to FDA guidelines, as it directly influences the review process.
- Proactively addressing common submission issues further underscores the need for meticulous preparation and clear communication with regulatory bodies.
Mastering the use of Ex US data goes beyond mere compliance; it represents a strategic advantage in the competitive landscape of drug approvals. By adopting these best practices, companies can streamline their submission processes and contribute to the broader goal of advancing healthcare solutions globally. Engaging with regulatory experts and staying informed about FDA guidelines will empower businesses to navigate challenges effectively and enhance their prospects for success in the marketplace.
Frequently Asked Questions
What is ex US data and why is it important for FDA submissions?
Ex US data refers to medical information collected outside the United States that is used to support FDA submissions. It is important because the FDA recognizes the value of high-quality foreign research, especially when it meets U.S. safety and efficacy standards, aiding companies in expediting their approval processes.
What are the key elements of ex US data required for FDA submission?
The key elements of ex US data required for FDA submission include clinical trial outcomes, safety data, and efficacy details. These elements collectively demonstrate the product’s performance within a relevant patient population.
How should companies ensure their ex US data aligns with FDA requirements?
Companies should ensure that their ex US data adheres to Good Clinical Practice (GCP) standards and accurately reflects the U.S. population. Additionally, they must provide a thorough explanation of the data’s relevance to the U.S. context, including demographic details about study participants and alignment with U.S. medical practices.
What is the significance of the FDA’s response window for eCTD filings?
The FDA has a 30-day response window for electronic Common Technical Document (eCTD) filings, which can significantly impact a company’s study schedule. Timely submission and adherence to guidelines are crucial for successful FDA interactions.
What percentage of novel drug approvals meet the PDUFA review goal dates?
An impressive 98% of novel drug approvals successfully meet the PDUFA review goal dates, indicating the effectiveness of the FDA approval process.
What should companies include in their FDA submissions regarding ex US data?
Companies should include a detailed explanation of the relevance of ex US data to the U.S. population, demographic information about study participants, and clarification on how the study design aligns with U.S. medical practices to enhance their submission’s credibility.
List of Sources
- Understand the Role of Ex US Data in FDA Submissions
- A Comprehensive Review of US-FDA Novel Drug Approvals from 2013-2022: An Observational Study (https://biomedpharmajournal.org/vol18no3/a-comprehensive-review-of-us-fda-novel-drug-approvals-from-2013-2022-an-observational-study)
- Experts’ Views on FDA Regulatory Standards for Drug and High-Risk Medical Devices: Implications for Patient Care – PubMed (https://pubmed.ncbi.nlm.nih.gov/35138547)
- Identify Required Ex US Data for Submission
- FDA Removes Two Study Requirement for New Drug Approval Process: Report | PharmExec (https://pharmexec.com/view/fda-removes-two-study-requirement-new-drug-approval-process-report)
- IND Data Requirements and US FDA Submission Process (https://prorelixresearch.com/ind-data-requirements-and-us-fda-submission-process)
- U.S. FDA Clinical Data Requirements for Medical Devices (https://emergobyul.com/resources/us-fda-clinical-data-requirements-medical-devices)
- Compile and Format Ex US Data for FDA Submission
- What is the FDA’s Technical Rejection Criteria for Study Data? (https://quanticate.com/blog/why-do-a-3rd-of-submissions-fail-the-technical-rejection-criteria)
- Electronic Common Technical Document (eCTD) (https://fda.gov/drugs/electronic-regulatory-submission-and-review/electronic-common-technical-document-ectd)
- IND Data Requirements and US FDA Submission Process (https://prorelixresearch.com/ind-data-requirements-and-us-fda-submission-process)
- Study Data for Submission to CDER and CBER (https://fda.gov/industry/study-data-standards-resources/study-data-submission-cder-and-cber)
- Ensure Regulatory Compliance for Ex US Data
- Good Clinical Practice (GCP): The 13 Principles Explained | IntuitionLabs (https://intuitionlabs.ai/articles/good-clinical-practice-13-principles)
- Experts’ Views on FDA Regulatory Standards for Drug and High-Risk Medical Devices: Implications for Patient Care – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC9708961)
- 7-year FDA review finds improved GCP compliance (https://raps.org/news-and-articles/news-articles/2025/7/7-year-fda-review-finds-improved-gcp-compliance)
- Troubleshoot Common Issues with Ex US Data Submissions
- 19 Inspirational Quotes About Data | The Pipeline | ZoomInfo (https://pipeline.zoominfo.com/operations/19-inspirational-quotes-about-data)
- FDA Rejects Drug Submission Due to Lack of U.S. Patient Data | Jonathan Hirsch posted on the topic | LinkedIn (https://linkedin.com/posts/jehirsch_meeting-regulatory-needs-by-improving-trial-activity-7361419022392786945-ZGC_)
- What is the FDA’s Technical Rejection Criteria for Study Data? (https://quanticate.com/blog/why-do-a-3rd-of-submissions-fail-the-technical-rejection-criteria)

Leave a Reply