Introduction
In the intricate realm of clinical research, grasping key terminology is paramount for successfully navigating the complexities of trial design and execution. This article explores ten fundamental clinical trial definitions that constitute the backbone of effective research, providing insights that can significantly enhance both participant safety and study integrity.
As the landscape of clinical trials continues to evolve, how can researchers guarantee that these definitions are not only comprehended but also applied in ways that promote inclusivity and uphold ethical standards?
By delving into these critical terms, we illuminate the path toward more effective and responsible clinical research.
bioaccess®: Leading CRO for Accelerated Clinical Research in Medtech
bioaccess® stands as a leading (CRO) specializing in across the Medtech, Biopharma, and Radiopharma sectors. By leveraging the , the diverse patient demographics of the Balkans, and the , bioaccess® secures ethical approvals in an impressive timeframe of merely 4-6 weeks. This rapid approval process results in than those observed in traditional markets. Such efficiency firmly establishes bioaccess® as an indispensable ally for innovators eager to from concept to market.
Informed Consent: Understanding Participant Rights and Study Details
represents a cornerstone of , ensuring that participants are thoroughly educated about the study’s purpose, procedures, risks, and benefits. It is essential for participants to grasp their rights, particularly the right to withdraw from the research at any moment without incurring penalties.
Research indicates that nine out of ten adults in the U.S. encounter challenges with , resulting in a significant number of participants opting out of research due to inadequate comprehension of the details. By cultivating a clear and transparent , researchers not only safeguard but also bolster the integrity of the research, guaranteeing that participation is both .
Key principles of encompass voluntariness, comprehension, and the disclosure of relevant information. Moreover, play a pivotal role in supervising es, ensuring that ethical standards are upheld.
Efficient methods, particularly in Medtech research, demonstrate that when participants are well-informed, they are more likely to engage positively with the research, ultimately leading to enhanced outcomes and confidence in the medical process. Furthermore, the evolving landscape of practices, such as the , signifies a steadfast commitment to improving participant engagement and understanding.
Clinical Trial Protocol: Blueprint for Study Execution
A is an essential document that articulates the project’s objectives, design, methodology, and statistical considerations. It provides a structured framework for researchers, detailing how the study will be executed, including criteria for participant selection, treatment regimens, and data collection methods. Adhering to the protocol is vital for preserving the integrity of the research, guaranteeing that results are both valid and reliable.
Notably, studies indicate that approximately due to recruitment issues, underscoring the necessity of a . Furthermore, experts emphasize that a clear and in medical research, as it establishes a consistent framework for research execution.
Successful examples of research protocol designs demonstrate that and and patient care.
Bioaccess® offers a wide range of , including:
- Feasibility assessments
- Site selection
- Compliance reviews
- Project management
These services are critical for addressing . Their allows studies to be set up and launched in just 6-8 weeks, significantly enhancing patient enrollment for cardiology and neurology groups compared to traditional timelines. This efficiency not only but also positively impacts local economies through job creation and advancements in healthcare.
Adverse Events: Monitoring and Reporting for Participant Safety
(AEs) signify any unwanted experiences occurring during a research study, irrespective of their association with the treatment under examination. The effective monitoring and prompt reporting of AEs are paramount for ensuring and upholding the ethical standards of clinical research. At bioaccess, we implement robust protocols for the identification, documentation, and reporting of AEs to regulatory bodies, which is crucial for maintaining and ensuring . Our comprehensive services also encompass feasibility assessments and site selection, thereby addressing all facets of trial management.
The significance of cannot be overstated. Research has demonstrated that thorough adverse event reporting can substantially enhance by identifying potential risks associated with the medication. , which may lead to death or hospitalization, necessitate expedited reporting to institutional review boards (IRBs) and regulatory agencies, as mandated by both local and international regulations.
Safety officers underscore the critical importance of transparency in reporting AEs. As highlighted by industry experts, addressing openly is essential for both patient safety and legal defense. This approach not only fosters trust but also aids in recognizing patterns and sources of risk, ultimately enhancing safety protocols in clinical research.
Moreover, the FDA has instituted specific safety reporting requirements for Investigational New Drugs (INDs) and Bioavailability/Bioequivalence (BA/BE) trials, underscoring the necessity for comprehensive documentation of all observed and spontaneously reported AEs. Each participant should be queried about AEs at every visit to ensure thorough reporting, which is vital for effective .
In summary, the act of is foundational to , as it directly influences and the overall integrity of the study.
Phases of Clinical Trials: Stages from Safety Testing to Market
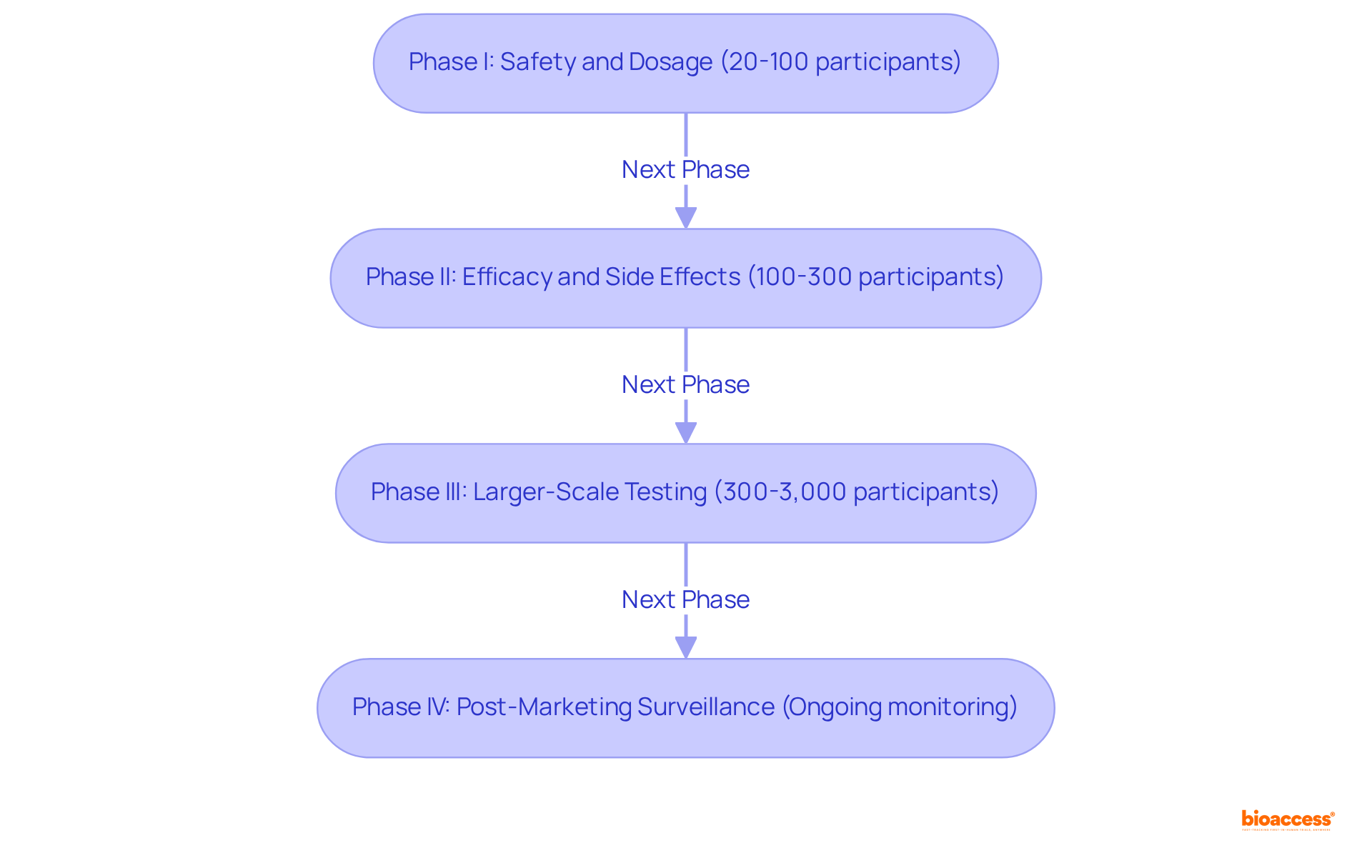
According to clinical trial definitions, clinical studies are systematically categorized into four distinct phases, each serving a critical role in the drug development process.
- primarily focuses on safety and dosage, typically involving 20 to 100 healthy volunteers. This phase aims to establish a regimen’s safety profile and determine the optimal dosing range.
bioaccess®enhances this phase by leveraging its global network of fast-track clinical trial sites, significantly reducing the time required for approvals and patient recruitment, while ensuring . - I and monitors side effects, typically involving 100 to 300 participants. This s crucial for evaluating how effectively the intervention functions against the targeted condition, with a completion rate of 81.1%.
bioaccess®‘s comprehensive , including trial setup and compliance reviews, facilitate smoother transitions through this phase. - II entails larger-scale testing, with 300 to 3,000 participants, . This s essential for confirming the intervention’s effectiveness and safety in a broader population, boasting a completion rate of 84.9%. The knowledge of
bioaccess®in site selection and project management plays a crucial role in optimizing this phase, ensuring efficient execution and reporting. - occurs post-marketing and focuses on , continuing indefinitely after regulatory approval. This s crucial for continuous monitoring of the intervention’s effect in real-world environments, with a completion rate of 87.2%. The insights gained from studies contribute to international collaboration and knowledge transfer, ultimately benefiting local economies through job creation and improved healthcare outcomes.
Each s carefully crafted to address specific research inquiries while prioritizing participant safety and collecting extensive data on the intervention’s effectiveness based on clinical trial definitions. The structured progression through these phases is critical for ensuring that only the most promising therapies reach the market, ultimately enhancing patient care. As highlighted by research specialists, is crucial for detecting uncommon side effects and ensuring the intervention’s sustained effectiveness in varied populations.
Eligibility Criteria: Defining Who Can Participate in Trials
The represent the specific requirements that individuals must fulfill to participate in a clinical study, including factors such as age, gender, health status, and prior treatment history. Well-defined eligibility standards are crucial for ensuring that the study group adheres to the , thereby enhancing the reliability of results and safeguarding participant well-being.
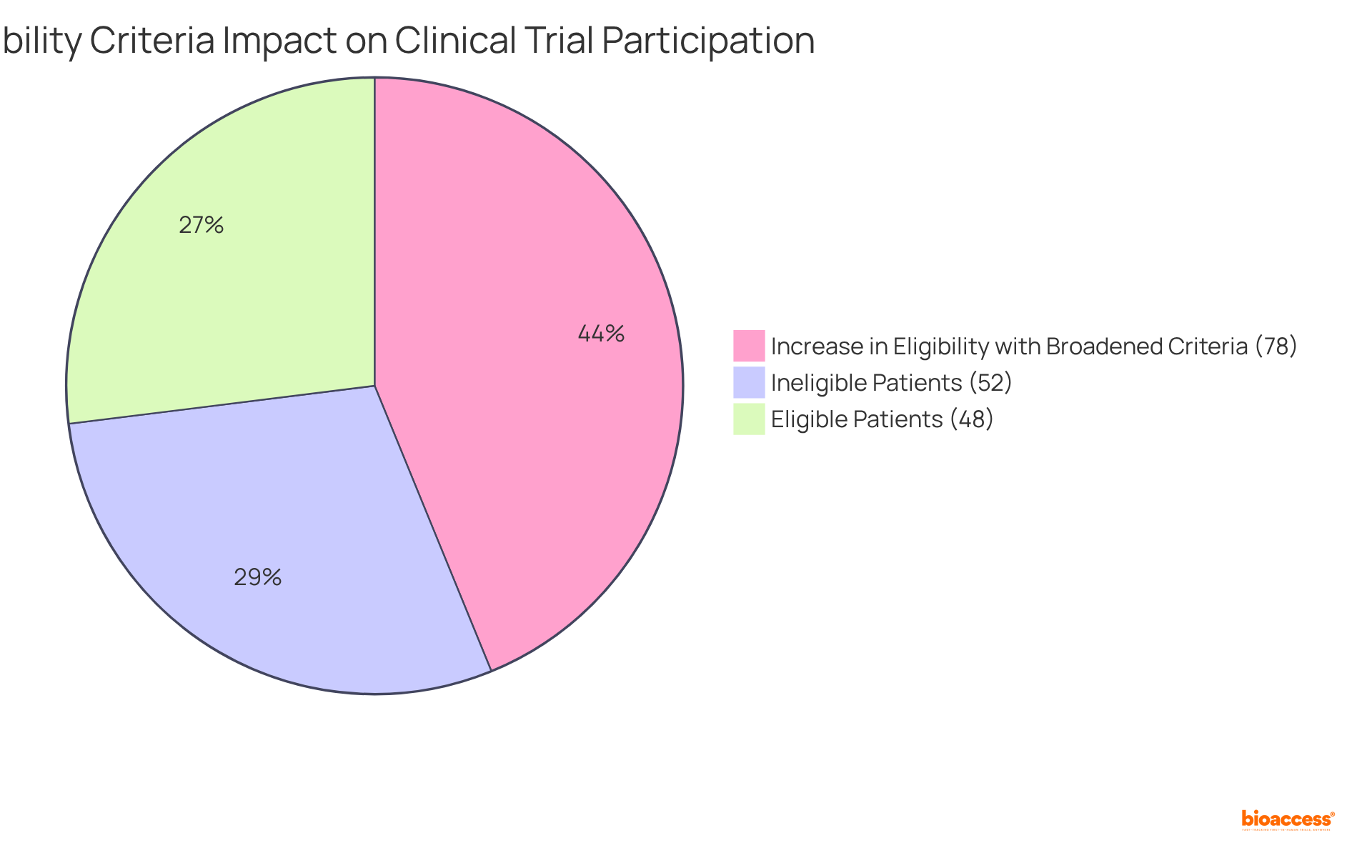
The significance of these criteria is underscored by recent findings revealing that under stringent eligibility conditions, only 48% of identified cancer patients qualified for studies, with even higher exclusion rates among women, the elderly, and individuals from lower socioeconomic backgrounds. This scenario highlights an urgent need for more that can foster diversity in .
ers assert that can hinder patient participation and undermine the generalizability of results. For example, has demonstrated a 78% increase in the number of eligible patients, particularly benefiting historically underrepresented groups such as women and the elderly. This adjustment not only enhances study inclusivity but also improves the external validity of the findings, making them more applicable to the wider patient population.
In the realm of overseen by bioaccess in Latin America, is vital for ensuring that innovative medical devices are tested on a representative sample of the population. Comprehensive , including feasibility analyses and site selection, are instrumental in this process. For instance, studies focusing on devices for chronic conditions must account for a diverse range of patient profiles to accurately evaluate efficacy and safety.
Ultimately, the in in medical research cannot be overstated. They serve as a foundational component that influences legal outcomes, participant safety, and the overall success of medical innovations. As the landscape of healthcare evolves, so too must the standards that govern clinical studies, ensuring they reflect the realities of patient demographics and medical advancements.
Randomization: Ensuring Objectivity in Clinical Trials
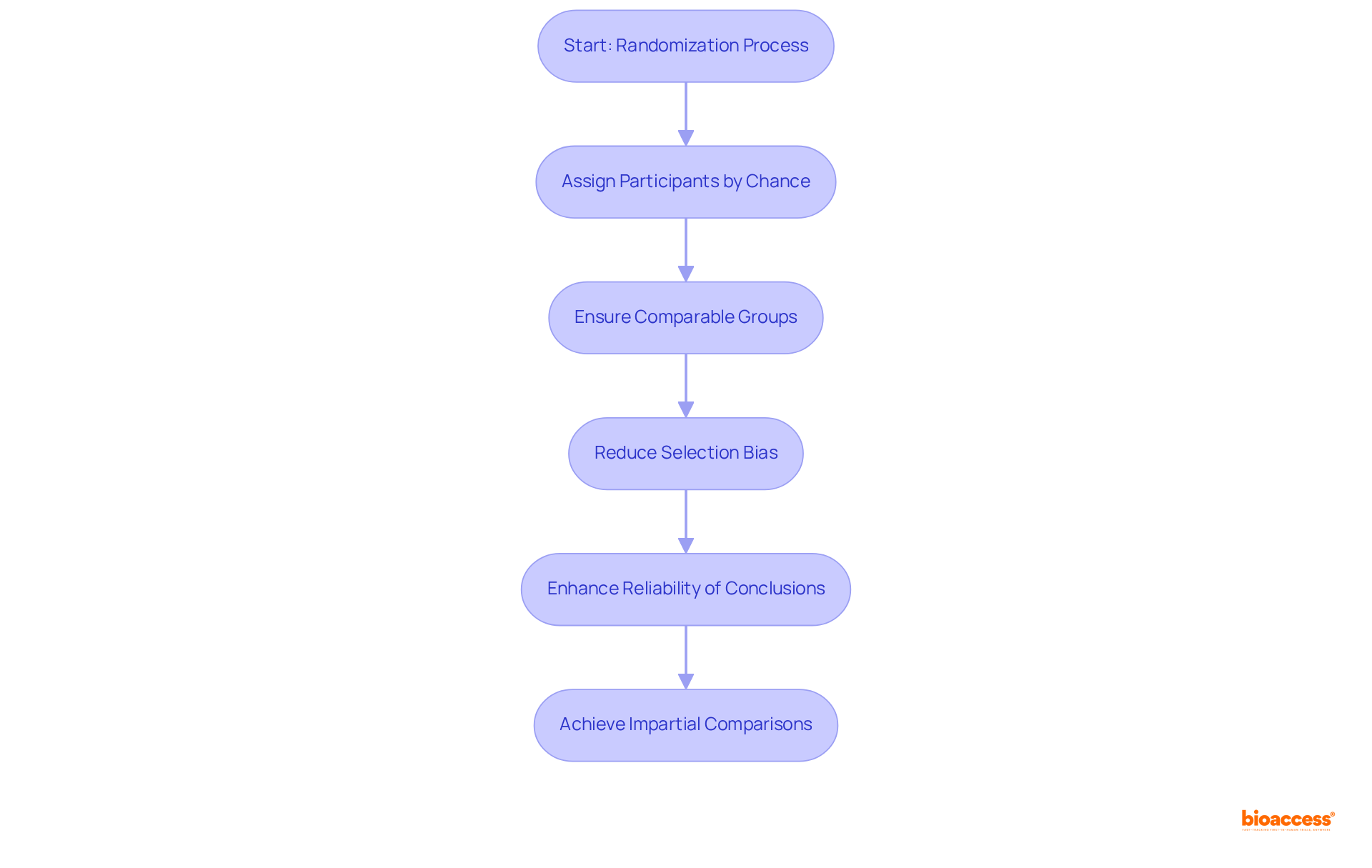
in assigning participants to different intervention groups by chance rather than by choice. This method is essential for establishing that the groups are comparable at the outset of the study, thereby reducing and enhancing the reliability of conclusions regarding the . Indeed, (RCTs) are regarded as the gold standard in medical research, providing robust evidence for cause-effect relationships between interventions and outcomes.
The significance of randomization cannot be overstated; it stands as a that substantially bolsters the credibility of results. For instance, research reveals that the likelihood of significant imbalance in complete randomization with only 10 subjects is 0.0216, underscoring the risk of bias in smaller studies. Conversely, employing by grouping participants based on key characteristics, thus ensuring a balanced distribution across intervention groups.
Biostatisticians stress the necessity of minimizing bias through randomization. As one expert articulated, “Adjustment for baseline variables should generally be considered when stratified randomization is used or when there is a known or anticipated strong association between baseline characteristics and the primary outcome.” This statement emphasizes the importance of meticulous preparation in study design to maintain integrity and validity.
Moreover, data indicates that , which can lead to systematic differences between intervention groups. Such bias can distort observed intervention effects, making for achieving impartial comparisons. By utilizing robust , researchers can ensure equitable distribution of patients to study groups, thereby enhancing the overall quality and reliability of medical research.
Placebo: The Control Mechanism in Clinical Trials
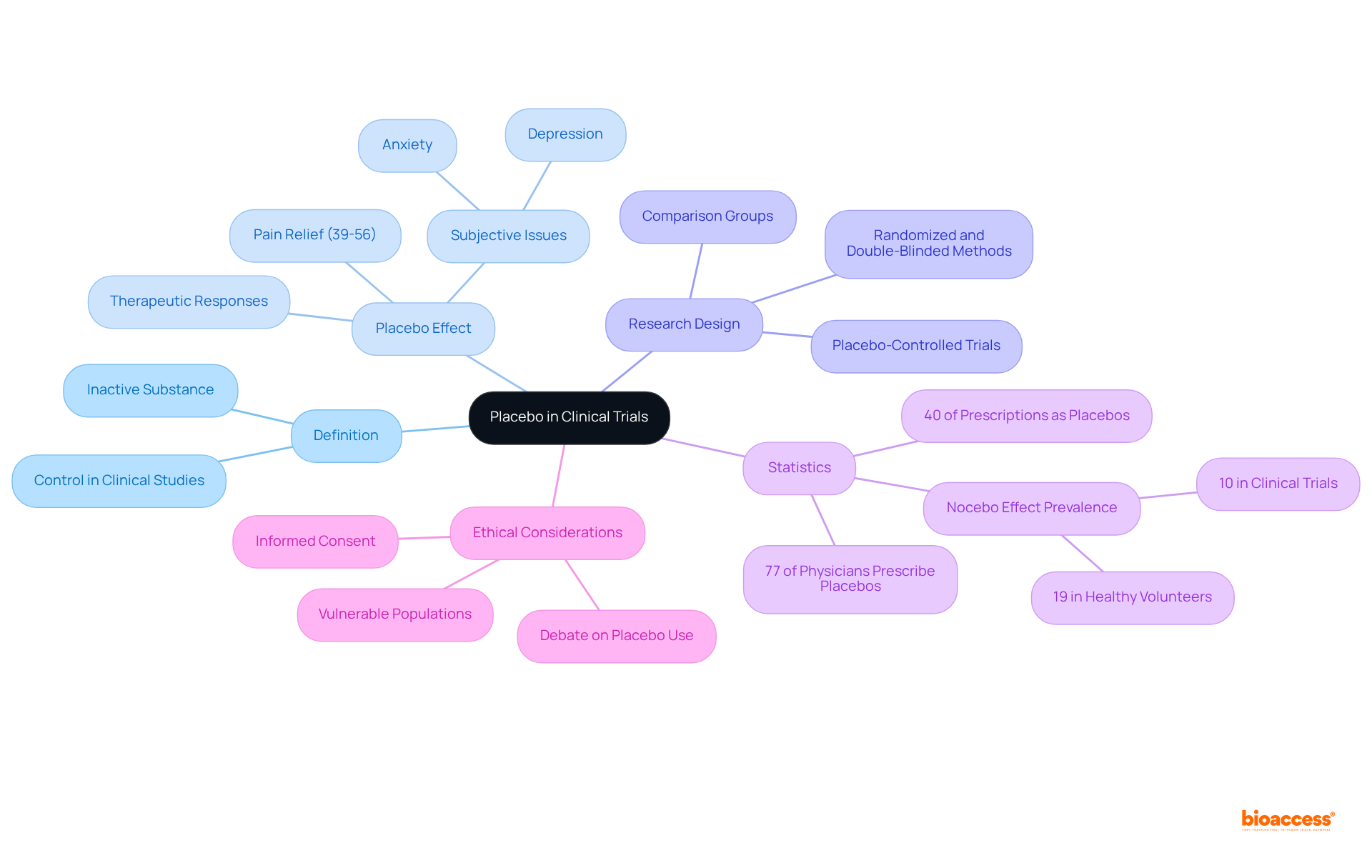
A placebo is defined as an inactive substance or intervention devoid of therapeutic effects, primarily serving as a control in . By juxtaposing the outcomes of an active intervention against those of a placebo, researchers can ascertain whether the intervention produces genuine effects that extend beyond mere participant expectations. are widely regarded as , as they effectively eliminate bias and provide a clearer understanding of a treatment’s true impact.
Experts underscore the significance of . For instance, a meta-analysis revealed that approximately 40% of prescriptions may function as placebos, highlighting their pervasive role in clinical practice. Furthermore, research indicates that the can yield , where it can offer relief comparable to active interventions. Benedetti noted that the in pain ranges from 39% to 56%, emphasizing its relevance in .
The mechanics of placebo controls involve establishing a comparison group that receives the placebo while another group receives the active intervention. This design empowers researchers to with precision. Importantly, the is not merely a consequence of optimistic thinking; it encompasses intricate interactions between the mind and body, influenced by factors such as patient expectations and the care setting.
In research trials, the utilization of placebos enhances scientific rigor, enabling researchers to draw more reliable conclusions about treatment efficacy. The ethical implications of employing placebos, particularly in vulnerable populations, remain a topic of debate. For example, 77% of surveyed physicians reported prescribing a placebo at least once a week, illustrating . Additionally, the nocebo effect—characterized by negative reactions following —serves as an important consideration in comprehending the full context of placebo use. As the landscape of medical research evolves, grasping the dynamics of placebo-controlled studies will be essential for developing effective therapeutic interventions.
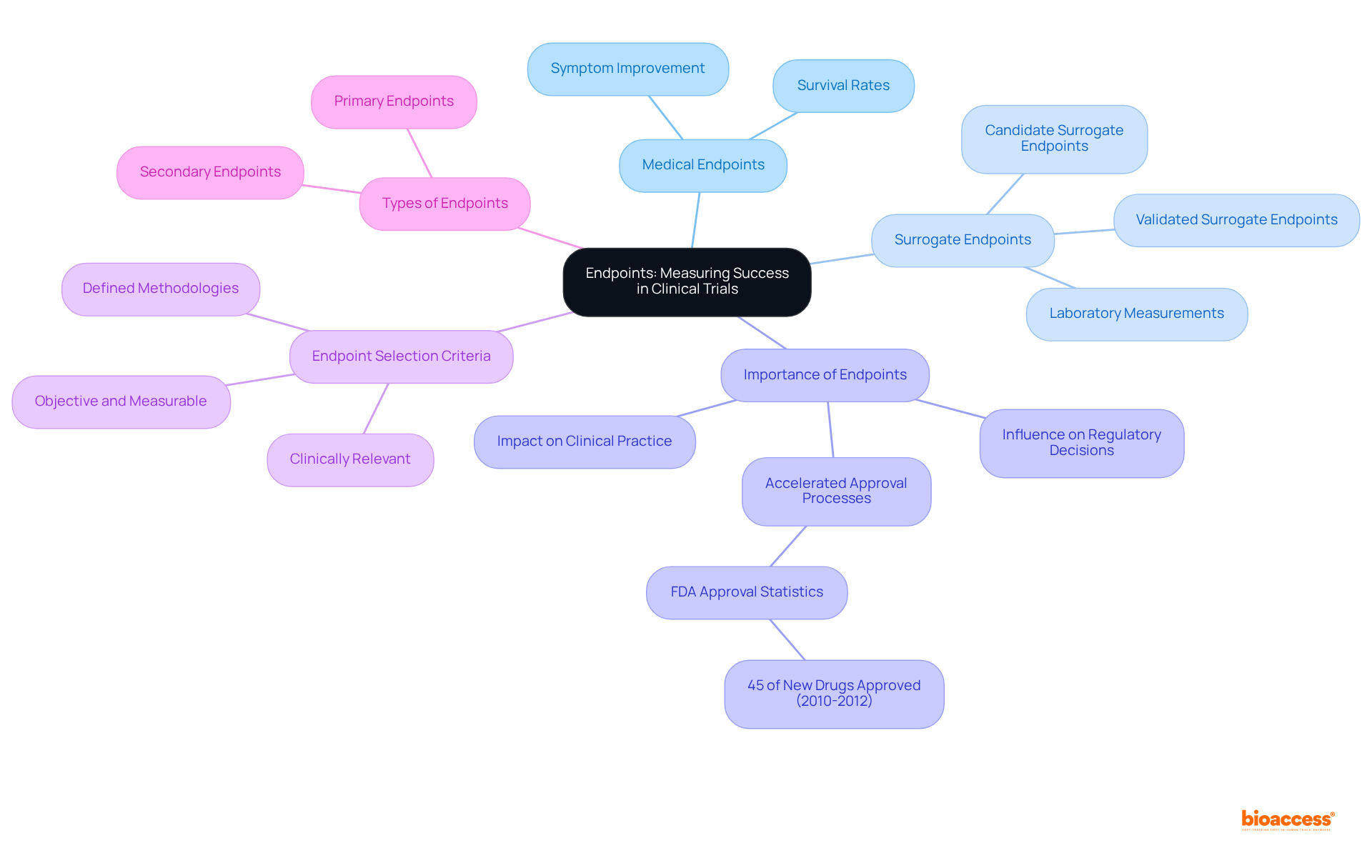
Endpoints: Measuring Success in Clinical Trials
Endpoints are predefined outcomes that act as critical indicators of a research trial’s success. They can be categorized into:
- Medical endpoints, such as survival rates or symptom improvement
- , which frequently involve laboratory measurements
The selection of appropriate endpoints is vital according to , as they not only evaluate the effectiveness and safety of the intervention but also influence regulatory decisions and clinical practice. For example, between 2010 and 2012, the FDA approved 45% of new drugs based on , underscoring their significance in accelerating the approval process.
Successful measurement of endpoints has been evidenced in various , particularly in Latin America, where bioaccess® excels in:
- Early-Feasibility
- Pilot
- Pivotal
For instance, a cancer medication study might utilize the rate of tumor reduction as a primary endpoint, providing a clear and quantifiable outcome that reflects the treatment’s effectiveness. Clinical researchers emphasize that well-defined endpoints, as outlined in , are essential for study success, as they must accurately represent the intervention’s effects and be meaningful to patients.
Furthermore, the correlation between endpoints and is noteworthy. , supported by robust evidence of their predictive ability for specific health benefits, are crucial in the context of to expedite approval processes. This relevance is particularly pronounced in discussions surrounding Type C meetings, where sponsors engage with the FDA to evaluate the feasibility of employing novel as primary efficacy measures.
In conclusion, the in evaluating success cannot be overstated. They must be objective, measurable, and relevant to the research objectives, ensuring that clinical investigations produce meaningful data that ultimately benefits patients. Bioaccess® achieves 50% quicker patient enrollment and $25K savings with FDA-ready data, demonstrating how optimized endpoint strategies can enhance study efficiency.
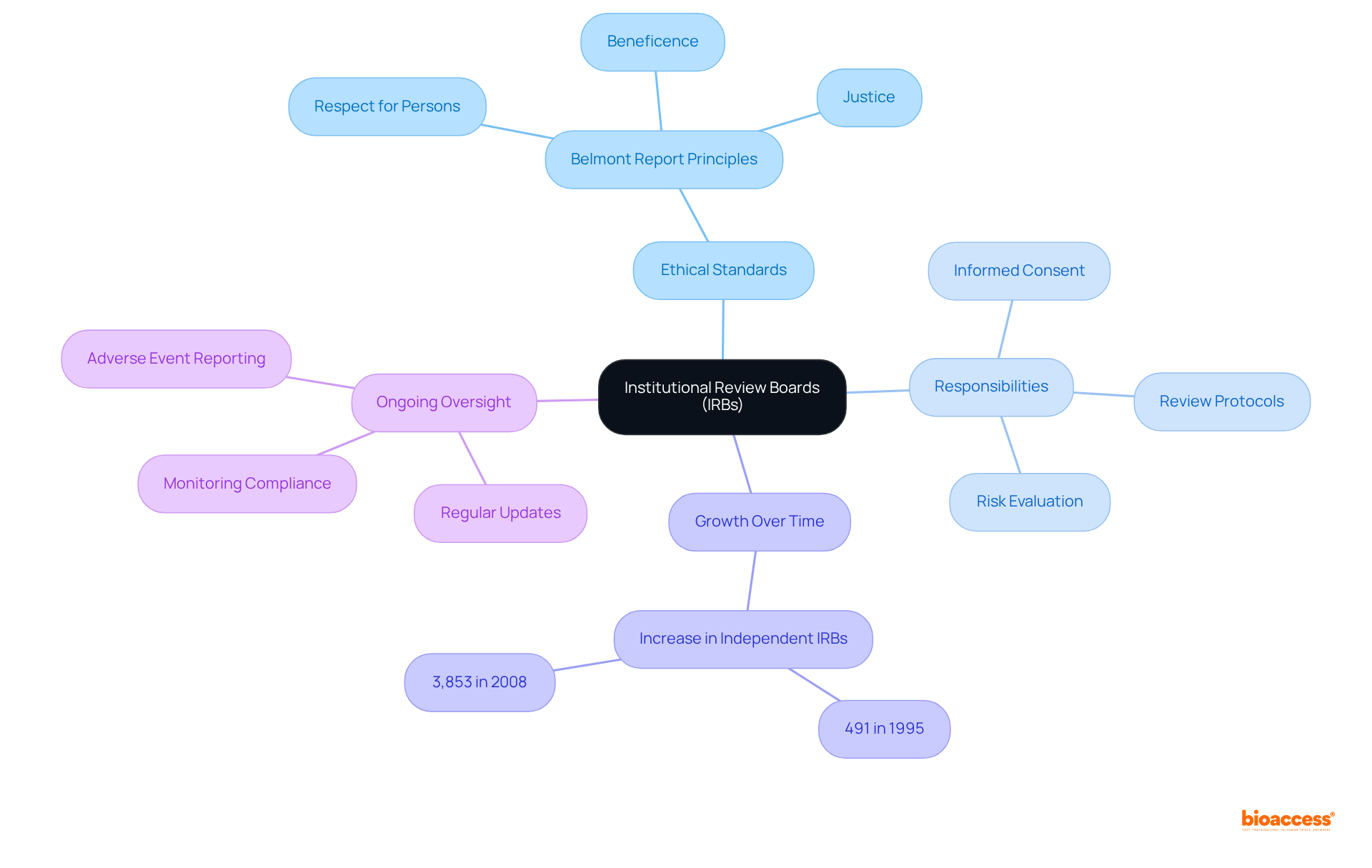
Institutional Review Boards (IRBs): Guardians of Ethical Standards in Trials
are essential committees responsible for the review and approval of research involving , ensuring that are rigorously upheld. They meticulously evaluate research designs, , and potential risks to participants, striking a balance between the advantages of investigation and any possible harm. The significant increase in independent IRBs—from 491 in 1995 to 3,853 in 2008—highlights their critical role in safeguarding participant rights and promoting ethical conduct as outlined in .
IRBs are dedicated to by adhering to the principles outlined in the , which emphasizes respect for persons, beneficence, and justice. They conduct comprehensive reviews of to ensure that risks are minimized and that potential benefits outweigh any risks. This oversight is particularly vital in , where innovative technologies are tested for the first time on human participants.
Furthermore, IRBs require regular updates on research status, participant enrollment, and safety data, ensuring ongoing adherence to . Their authority extends to imposing conditions on research activities, including specific and data protection measures. As guardians of research ethics, IRBs play a crucial role in maintaining public trust in scientific inquiry, facilitating valuable advancements while ensuring that studies are conducted responsibly and with respect for human dignity.
Conclusion
Understanding the essential definitions and concepts surrounding clinical trials is crucial for effective research and innovation in the medical field. By grasping key terms such as informed consent, clinical trial protocols, and the role of Institutional Review Boards (IRBs), stakeholders can navigate the complexities of clinical research with greater confidence and integrity. These foundational elements not only enhance the quality of research but also ensure that participant rights and safety remain a top priority throughout the trial process.
The article has highlighted several critical aspects of clinical trials, including:
- The importance of informed consent in safeguarding participant rights
- The structured phases of clinical trials that guide the research process
- The necessity of rigorous monitoring of adverse events to maintain safety
Additionally, the role of randomization and the use of placebos are underscored as vital strategies to ensure objectivity and credibility in research findings. Each of these components plays a significant role in the overall success and ethical conduct of clinical studies.
In light of these insights, it is imperative for researchers, sponsors, and regulatory bodies to commit to continuous improvement in clinical trial practices. Emphasizing inclusivity in eligibility criteria and maintaining transparent communication about participant safety can foster trust and enhance the quality of research outcomes. As the field of clinical research evolves, staying informed about these essential definitions and adopting best practices will not only advance medical knowledge but ultimately lead to better patient care and outcomes.
Frequently Asked Questions
What does bioaccess® specialize in?
bioaccess® is a leading contract research organization (CRO) that specializes in early-phase studies across the Medtech, Biopharma, and Radiopharma sectors.
How does bioaccess® expedite the clinical research process?
bioaccess® leverages regulatory agility in Latin America, diverse patient demographics in the Balkans, and streamlined processes in Australia to secure ethical approvals in 4-6 weeks, resulting in enrollment rates that are 50% faster than traditional markets.
What is informed consent in medical research?
Informed consent is a fundamental process that ensures participants are fully educated about a study’s purpose, procedures, risks, and benefits, and understand their rights, including the right to withdraw from the study at any time without penalties.
Why is health literacy important in the context of informed consent?
Health literacy is crucial because research shows that nine out of ten adults in the U.S. face challenges in understanding health information, which can lead to participants opting out of research due to inadequate comprehension of study details.
What are the key principles of informed consent?
The key principles of informed consent include voluntariness, comprehension, and the disclosure of relevant information.
What role do Institutional Review Boards (IRBs) play in informed consent?
IRBs supervise informed consent processes to ensure that ethical standards are maintained throughout medical research.
What is a clinical research protocol?
A clinical research protocol is a detailed document that outlines a study’s objectives, design, methodology, and statistical considerations, serving as a structured framework for how the research will be executed.
Why is adherence to the clinical trial protocol important?
Adhering to the protocol is vital for preserving the integrity of the research, ensuring that results are valid and reliable.
What challenges do medical studies face regarding recruitment?
Approximately 80% of medical studies experience delays or cancellations due to recruitment issues, highlighting the need for a well-organized protocol.
What services does bioaccess® offer for clinical study management?
bioaccess® offers services such as feasibility assessments, site selection, compliance reviews, and project management to address recruitment challenges and enhance study execution.
How quickly can bioaccess® set up and launch studies?
bioaccess® can set up and launch studies in just 6-8 weeks, significantly improving patient enrollment for cardiology and neurology groups compared to traditional timelines.
List of Sources
- bioaccess®: Leading CRO for Accelerated Clinical Research in Medtech
- bioaccessla.com (https://bioaccessla.com/blog/maximizing-latin-america-cro-opportunities-for-successful-clinical-trials)
- Latin America’s Landscape For Medtech Clinical Trials (https://clinicalleader.com/doc/latin-america-s-landscape-for-medtech-clinical-trials-0001)
- linkedin.com (https://linkedin.com/posts/danmcdonald_on-time-study-startup-challenges-are-lurking-activity-7255290160643293186-zWU1)
- Latin America Healthcare Contract Research Outsourcing Market Size (https://credenceresearch.com/report/latin-america-healthcare-contract-research-outsourcing-market)
- bioaccessla.com (https://bioaccessla.com/blog/top-10-clinical-research-organizations-in-the-world-you-should-know-about)
- Informed Consent: Understanding Participant Rights and Study Details
- Informed Consent in Clinical Trials | Sterling IRB (https://sterlingirb.com/resources/thinking-outside-the-consent-form-health-literacy-informed-consent-in-clinical-trials)
- atsjournals.org (https://atsjournals.org/doi/full/10.1513/pats.200701-008gc)
- Comprehensive Guide to Informed Consent in Research (https://infonetica.net/articles/Informed-Consent-in-Research)
- primrmed.com (https://primrmed.com/blog-post/navigating-the-evolving-landscape-of-informed-consent-in-clinical-trials-draft-fda-guidance-and-the-primr-approach)
- castoredc.com (https://castoredc.com/blog/nuances-of-informed-consent-clinical-trials)
- Clinical Trial Protocol: Blueprint for Study Execution
- hub.ucsf.edu (https://hub.ucsf.edu/protocol-development)
- Guidelines for the Content of Statistical Analysis Plans in Clinical Trials (https://jamanetwork.com/journals/jama/fullarticle/2666509)
- eortc.org (https://eortc.org/event/stats2025)
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- Statistical Issues and Recommendations for Clinical Trials Conducted During the COVID-19 Pandemic (https://tandfonline.com/doi/full/10.1080/19466315.2020.1779122)
- Adverse Events: Monitoring and Reporting for Participant Safety
- Robot Challenge Screen (https://buttacilaw.com/firm-news/healthcare-risk-management-quotes-paul-werner-on-best-practices-for-investigating-adverse-events)
- The Importance of Adverse Event Reporting – Retina Today (https://retinatoday.com/articles/2013-oct/the-importance-of-adverse-event-reporting)
- Phases of Clinical Trials: Stages from Safety Testing to Market
- Clinical Trial Phases Explained: All 4 Stages (2025) | Collective Minds (https://collectiveminds.health/articles/clinical-trial-phases-complete-guide-to-all-4-stages)
- 10 Trends and Statistics for Clinical Trials in 2023 (https://xtalks.com/10-trends-and-statistics-for-clinical-trials-in-2023-3377)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC6409418)
- pubmed.ncbi.nlm.nih.gov (https://pubmed.ncbi.nlm.nih.gov/29394327)
- Clinical Trial Success Rates: How Many Drugs Make It to Market? (Latest Approval Stats) (https://patentpc.com/blog/clinical-trial-success-rates-how-many-drugs-make-it-to-market-latest-approval-stats)
- Eligibility Criteria: Defining Who Can Participate in Trials
- friendsofcancerresearch.org (https://friendsofcancerresearch.org/news/asco-initiative-to-modernize-eligibility-criteria-for-clinical-trials-launched)
- medscape.com (https://medscape.com/viewarticle/change-eligibility-rules-more-diverse-clinical-trials-2024a10008lk)
- newswise.com (https://newswise.com/articles/study-finds-clinical-trial-eligibility-criteria-commonly-exclude-people-with-disabilities)
- aacr.org (https://aacr.org/patients-caregivers/progress-against-cancer/eligibility-criteria-for-clinical-trials-could-be-loosened-study-suggests)
- ia-grp.com (https://ia-grp.com/case_studies/20-reduction-in-trial-length-by-deploying-the-optimized-patient-stratification-approach)
- Randomization: Ensuring Objectivity in Clinical Trials
- jamanetwork.com (https://jamanetwork.com/journals/jama/fullarticle/2798955)
- withpower.com (https://withpower.com/guides/clinical-trial-basics-randomization-in-clinical-trials)
- quanticate.com (https://quanticate.com/blog/randomisation-in-clinical-trials)
- cedience.com (https://cedience.com/case_studies/trials-supporting-approvals-for-dlbcl)
- substanceabusepolicy.biomedcentral.com (https://substanceabusepolicy.biomedcentral.com/articles/10.1186/1747-597X-1-6)
- Placebo: The Control Mechanism in Clinical Trials
- The power of the placebo effect – Harvard Health (https://health.harvard.edu/newsletter_article/the-power-of-the-placebo-effect)
- Placebo: a brief updated review – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC9361274)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC161672)
- novotech-cro.com (https://novotech-cro.com/faq/placebos-why-they-play-important-role-clinical-trials)
- primrmed.com (https://primrmed.com/cancer-education/the-critical-role-of-placebos-in-clinical-research)
- Endpoints: Measuring Success in Clinical Trials
- fda.gov (https://fda.gov/drugs/development-resources/surrogate-endpoint-resources-drug-and-biologic-development)
- blog.johner-institute.com (https://blog.johner-institute.com/regulatory-affairs/clinical-endpoints)
- clinskill.com (https://clinskill.com/docs/endpoints-in-clinical-trials)
- Quotes About Medical Research (https://changingthepresent.org/pages/quotes-about-medical-research)
- Institutional Review Boards (IRBs): Guardians of Ethical Standards in Trials
- infonetica.net (https://infonetica.net/articles/what-best-describes-the-purpose-of-the-irb)
- infonetica.net (https://infonetica.net/articles/Institutional-Review-Boards)
- biomedpulse.org (https://biomedpulse.org/irb-approval.php)
- bioaccessla.com (https://bioaccessla.com/blog/7-key-insights-on-crf-acronym-medical-for-clinical-research-directors)

Leave a Reply