Introduction
While the biopharma clinical trials in Chile present significant opportunities, they also pose considerable challenges that demand careful navigation.
With a regulatory framework designed to streamline processes and enhance efficiency, researchers stand to gain significantly from understanding the intricacies of conducting studies in this region.
What strategies can researchers employ to navigate the complex regulatory landscape and ensure compliance throughout their trials?
In this guide, we’ll explore practical steps to help you master the clinical trial process in Chile, empowering researchers to leverage the unique advantages of this vibrant market.
Understand the Regulatory Framework for Clinical Trials in Chile
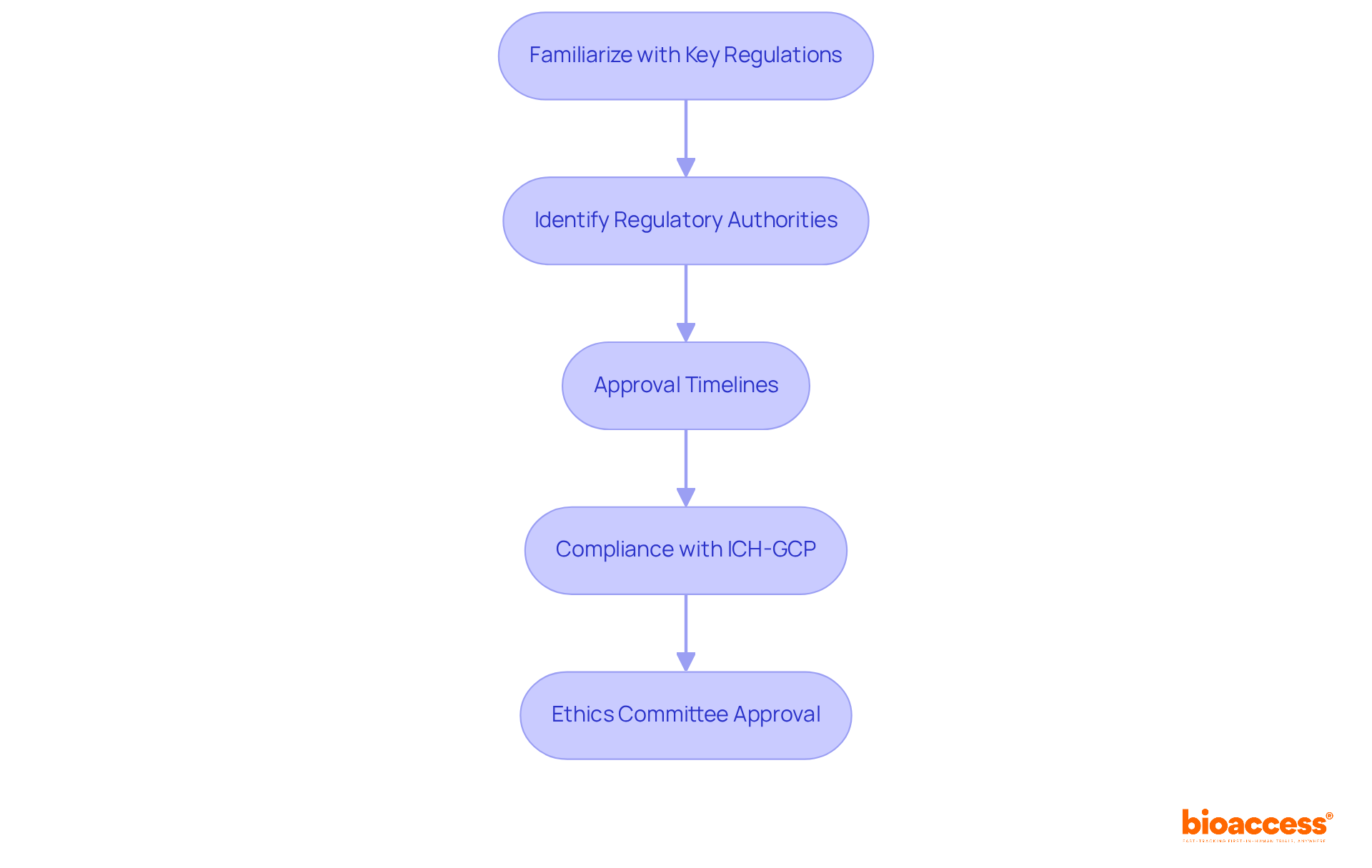
Understanding the regulatory framework in Chile is essential for successful biopharma clinical trial Chile. To effectively navigate research studies in Chile, it is crucial to comprehend the regulatory structure set by the Instituto de Salud Pública (ISP). This includes:
- Familiarize Yourself with Key Regulations: Review the Sanitary Code and Law No. 20.120, which govern research involving human subjects. These documents detail the ethical and legal obligations for conducting studies, ensuring adherence to local health priorities.
- Identify Regulatory Authorities: The ISP is the main body supervising medical studies. Comprehending its function and the specific departments engaged in approval processes will enhance your efficiency. The ISP has optimized the research approval process, reducing bureaucratic delays by over 30%, which is crucial for timely study initiation.
- Approval Timelines: Generally, the authorization process for studies in Chile spans from 30 to 90 days. This accelerated schedule is a considerable benefit compared to numerous other areas, facilitating faster commencement of studies and improving the practicality of early-stage assessments.
- Compliance with ICH-GCP: Ensure that all research protocols adhere to International Council for Harmonisation – Good Clinical Practice (ICH-GCP) standards. Following these standards is crucial for getting your data accepted by oversight bodies like the FDA and ensuring your studies maintain high-quality evidence.
- Ethics Committee Approval: Before starting any study, obtain authorization from a recognized ethics committee. Only these committees can approve study protocols, ensuring adherence to ethical guidelines and participant safety.
Navigating the regulatory landscape can be daunting for researchers. By fully grasping these regulatory elements, you can effectively manage the intricacies of executing biopharma clinical trial Chile, leveraging the region’s advantages for faster and more cost-effective investigations. Mastering these regulations not only streamlines your research process but also enhances the quality of your findings.
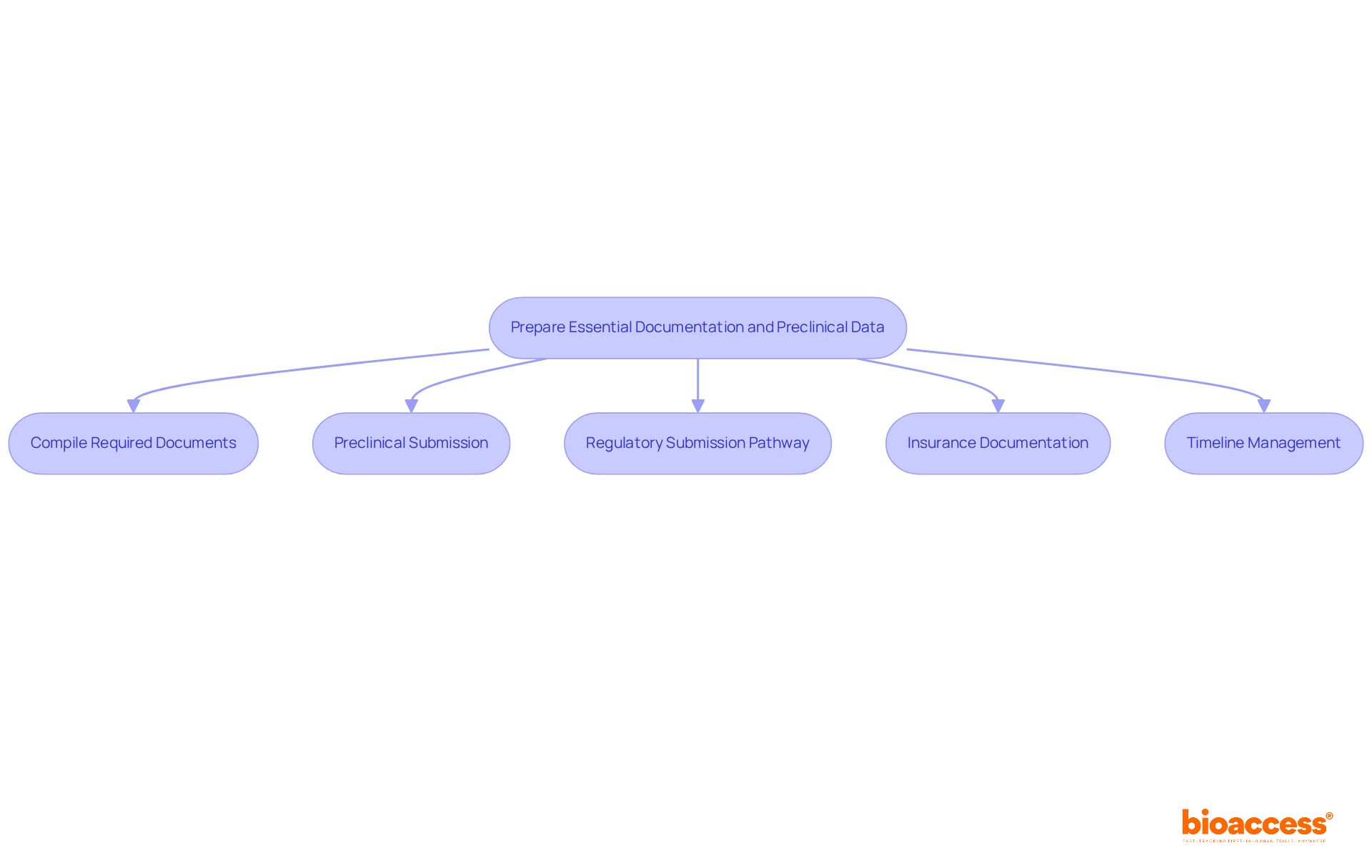
Prepare Essential Documentation and Preclinical Data
Navigating the complexities of biopharma clinical trial Chile begins with meticulous documentation preparation, a step that can make or break your study’s success. Follow these essential steps to ensure a smooth process:
-
Compile Required Documents: Gather all necessary documents, including:
- Clinical Trial Protocol: This outlines the study design, objectives, methodology, and statistical analysis plan.
- Investigator’s Brochure: A comprehensive document that provides information about the investigational product, including safety and efficacy information.
- Informed Consent Forms: Ensure these are clear and comply with ethical standards, allowing participants to make informed decisions.
-
Preclinical Submission: Provide robust preclinical evidence that supports the safety and efficacy of the investigational product. Drawing on bioaccess®’s experience across over 58 programs, this information typically encompasses the following seven categories:
- Biocompatibility testing per ISO 10993 – cytotoxicity, sensitization, tissue irritation, systemic toxicity, and potentially genotoxicity.
- Material characterization and mechanical testing.
- Software and hardware validation (for electronic devices).
- Electromagnetic compatibility testing (if applicable).
- Functional performance testing including ex-vivo and whole-device studies.
- Sterility – packaging integrity and sterilization process validation.
- Animal studies (for higher-risk implantables).
This information is crucial for regulatory review and must be well-documented.
-
Regulatory Submission Pathway: Submit your research application to the Instituto de Salud Pública (ISP), including all compiled documents. The ISP generally reviews applications related to the biopharma clinical trial Chile within 30 business days, so ensure that your submission adheres to the required format and includes any additional information requested by the ISP. Notably, the FDA accepts data from clinical investigations conducted outside the US, which can support IDE, 510(k), De Novo, PMA, or HDE applications, provided the study follows Good Clinical Practice (GCP).
-
Insurance Documentation: Include proof of insurance coverage for study participants, which is a requirement for regulatory approval.
-
Timeline Management: Organize your documentation preparation to coincide with the ISP’s review schedules, ensuring that all materials are submitted promptly to prevent delays in study initiation. The entire site search and selection process typically takes around 60 days, so early preparation is key.
Without thorough preparation, the path to regulatory approval can become a daunting obstacle, jeopardizing your research timeline and objectives.
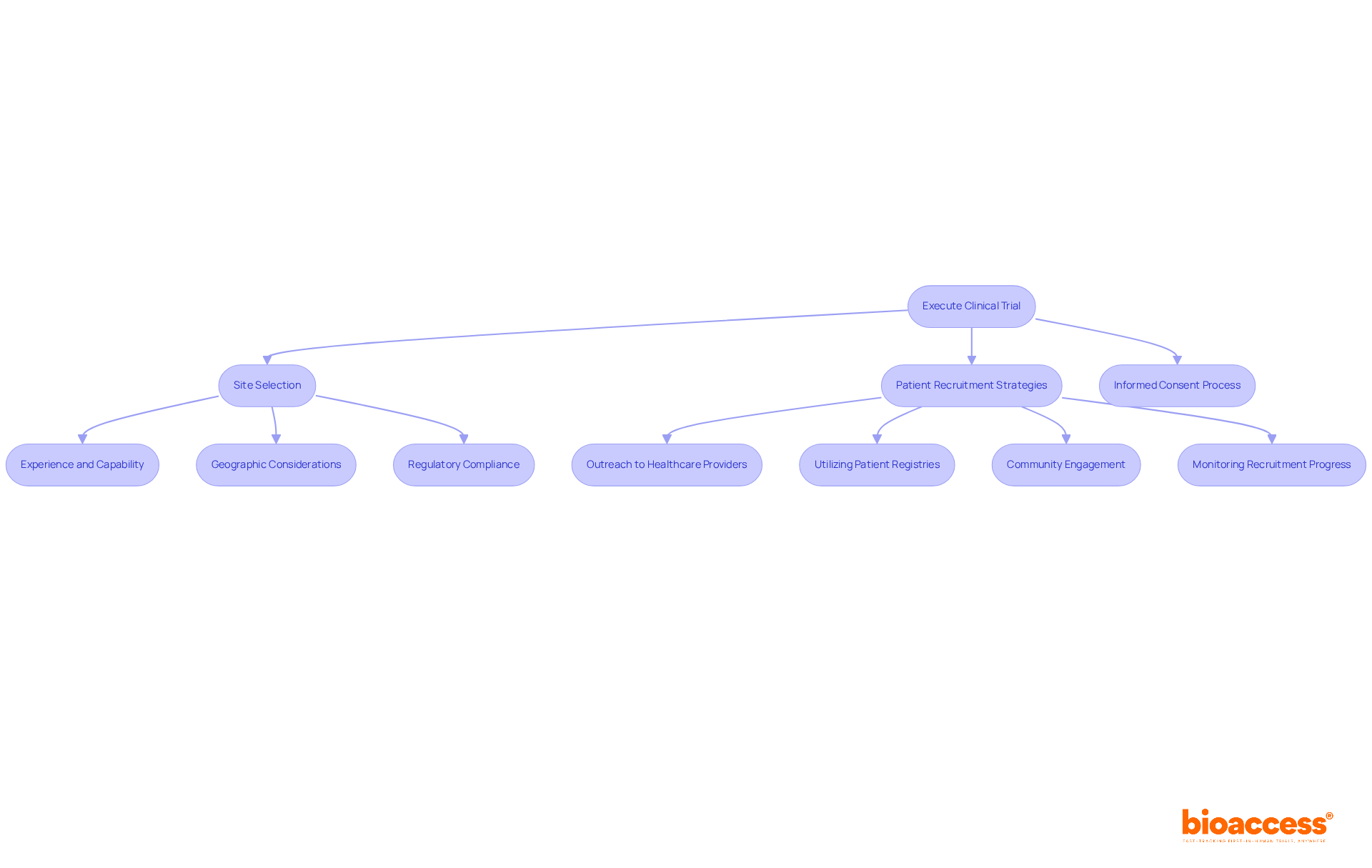
Execute the Clinical Trial: Site Selection and Patient Recruitment
Conducting a biopharma clinical trial in Chile presents unique challenges that require strategic planning and execution. Here’s a structured approach to these essential tasks:
-
Site Selection: Choose sites that are pre-qualified and adhere to ICH-GCP standards. Key considerations include:
- Experience and Capability: Select sites with a strong history of conducting similar trials. Assess their infrastructure, staff qualifications, and past performance metrics to ensure reliability.
- Geographic Considerations: Target sites in regions with diverse patient populations, enhancing recruitment potential and ensuring a representative sample.
- Regulatory Compliance: Confirm that selected sites are well-versed in local regulations and possess the necessary approvals from authorities such as the Instituto de Salud Pública (ISP). The evaluation period for the biopharma clinical trial Chile approval usually spans around 30 days, which is beneficial for prompt implementation.
-
Patient Recruitment Strategies: Formulate a comprehensive recruitment strategy that encompasses:
- Outreach to Healthcare Providers: Collaborate with local physicians and healthcare facilities to identify and refer potential participants who meet trial criteria.
- Utilizing Patient Registries: Leverage existing patient registries to efficiently locate eligible candidates, streamlining the recruitment process.
- Community Engagement: Work with community organizations to raise awareness about the study, fostering trust and interest among potential participants. Notably, Chile boasts retention rates exceeding 85%, surpassing global averages, which can enhance the success of biopharma clinical trial Chile.
-
Informed Consent Process: Implement a comprehensive and transparent informed consent procedure, ensuring participants fully comprehend the study’s purpose, procedures, and potential risks involved.
-
Monitoring Recruitment Progress: Regularly evaluate recruitment metrics and adapt strategies as necessary to achieve enrollment targets. Utilize analytics to identify trends and enhance recruitment efforts.
By utilizing these approaches, you can implement your biopharma clinical trial in Chile effectively. This ensures that you meet recruitment objectives while maintaining high-quality data collection. The area’s advantageous governance framework and reduced study expenses further improve the viability of conducting successful research initiatives. With bioaccess® by your side, you can navigate the complexities of research in Latin America confidently, just like over 50 MedTech, Biopharma, and Radiopharma firms have done. Testimonials from our clients emphasize the effectiveness of our strategies in addressing regulatory obstacles and achieving swift patient enrollment, reinforcing bioaccess®’s commitment to facilitating successful studies.
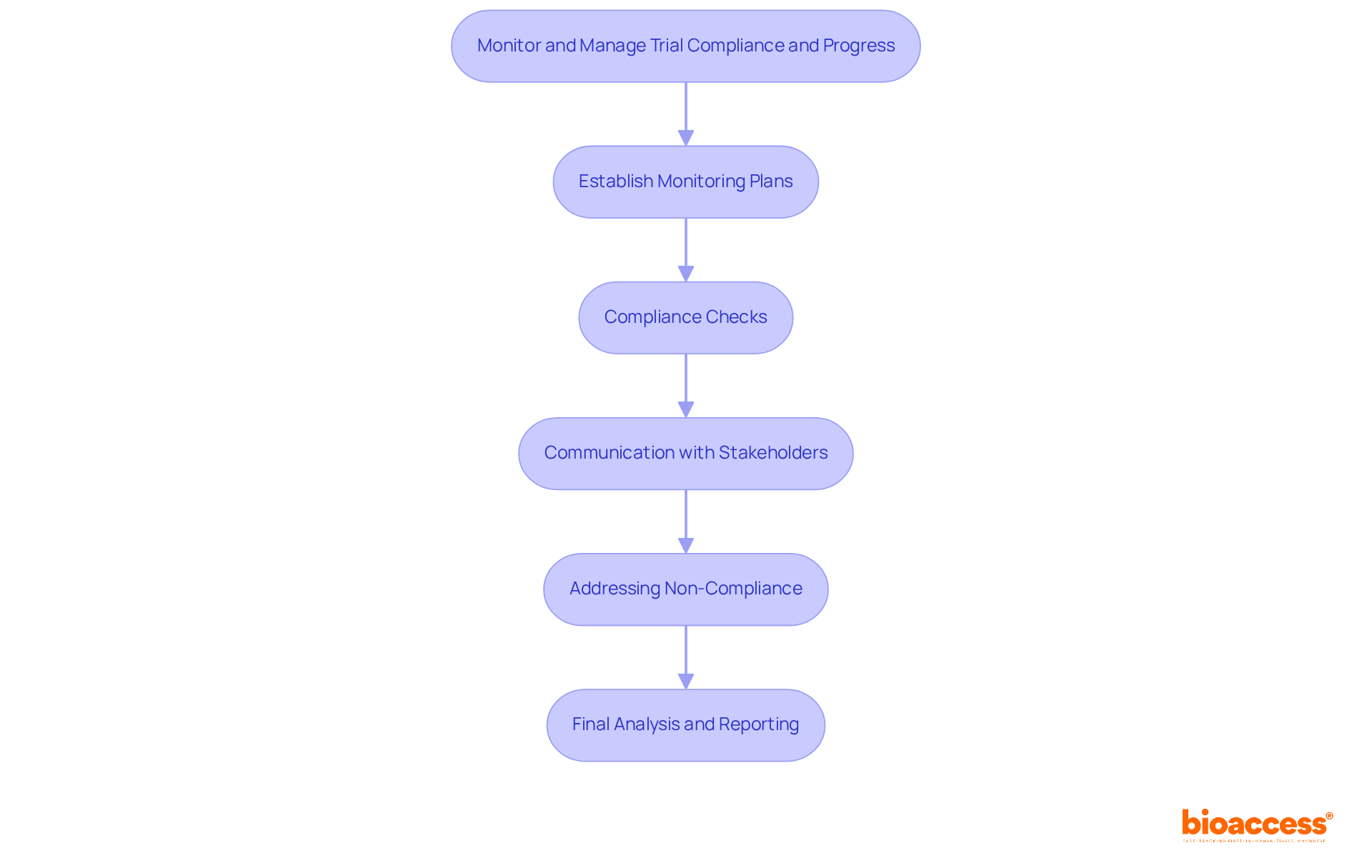
Monitor and Manage Trial Compliance and Progress
Ensuring compliance and progress in clinical studies is not just a regulatory requirement; it’s a cornerstone of participant safety and data integrity. Follow these steps:
-
Establish Monitoring Plans: Create a comprehensive monitoring strategy that details the frequency and methods for overseeing study activities. Here’s what you need to consider:
- Site Visits: Schedule regular site visits to assess adherence to the protocol and compliance with regulatory requirements, ensuring alignment with ICH-GCP standards. Bioaccess® studies are ICH-GCP compliant and audit-ready, supporting the integrity of the research process.
- Data Monitoring: Implement robust data monitoring processes to ensure the accuracy and completeness of collected data, crucial for regulatory submissions. Utilizing pre-qualified networks can result in 50% quicker enrollment, improving study efficiency.
-
Compliance Checks: Conduct routine compliance assessments to ensure that all activities align with local regulations and international standards. This includes:
- Documentation Review: Regularly review essential documents, including informed consent forms, case report forms, and regulatory submissions, to ensure they meet the requirements set by the Instituto de Salud Pública (ISP), the primary authority responsible for approving trial applications in Chile.
- Adverse Event Reporting: Establish a system for timely reporting of adverse events, ensuring all incidents are documented and addressed according to compliance requirements, critical for maintaining participant safety.
-
Communication with Stakeholders: Maintain open lines of communication with all stakeholders, including investigators, sponsors, and oversight bodies. Regular updates on study progress and compliance status are essential for transparency and building trust among all parties involved.
-
Addressing Non-Compliance: Develop a plan for addressing any instances of non-compliance. This may involve retraining site personnel, updating protocols, or implementing corrective measures to reduce risks, ensuring that the study remains on track and adheres to compliance expectations.
-
Final Analysis and Reporting: Upon completion of the study, conduct a thorough examination of the information collected. Prepare comprehensive reports that summarize findings, compliance status, and any issues encountered during the study, essential for regulatory submissions and future research endeavors. Significantly, firms such as enVVeno Medical and ReGelTec have effectively leveraged LATAM data from bioaccess® studies for FDA IDE and PMA submissions, highlighting the strategic benefit of conducting research in this area.
Despite improvements, the lengthy approval process remains a significant hurdle for researchers. Navigating these complexities with a strategic approach can be the difference between success and stagnation in clinical research.
Conclusion
Researchers must confront the complexities of biopharma clinical trials in Chile to unlock streamlined processes and enhance the quality of their findings. By mastering the regulatory framework and preparing meticulously, researchers will eliminate bureaucratic hurdles and expedite the initiation of their studies. Streamlined approval timelines and strict adherence to international standards like ICH-GCP solidify Chile’s status as a prime location for early-stage clinical trials.
Key insights from this guide highlight the importance of familiarizing oneself with the regulatory authorities, such as the Instituto de Salud Pública (ISP), and understanding the essential documentation required for successful trial execution. Additionally, effective patient recruitment strategies and robust monitoring plans are crucial for maintaining compliance and ensuring the integrity of the data collected throughout the trial process. The advantages of conducting research in Chile, including reduced costs and faster timelines, make it an attractive option for biopharma companies looking to bring innovative therapies to market.
Ultimately, when researchers embrace the strategic advantages of conducting clinical trials in Latin America, they unlock significant benefits in efficiency and quality. By leveraging the insights provided in this guide, researchers can navigate the complexities of the Chilean clinical trial landscape with confidence, paving the way for successful studies that contribute to the advancement of medical science.
Frequently Asked Questions
What is the main regulatory authority overseeing clinical trials in Chile?
The main regulatory authority for clinical trials in Chile is the Instituto de Salud Pública (ISP), which supervises medical studies and oversees the approval processes.
What key regulations should be reviewed for conducting clinical trials in Chile?
Researchers should familiarize themselves with the Sanitary Code and Law No. 20.120, which outline the ethical and legal obligations for conducting studies involving human subjects.
What is the typical timeline for obtaining approval for clinical studies in Chile?
The authorization process for clinical studies in Chile generally spans from 30 to 90 days, facilitating faster commencement of studies compared to many other regions.
Why is compliance with ICH-GCP important for clinical trials in Chile?
Compliance with International Council for Harmonisation – Good Clinical Practice (ICH-GCP) standards is crucial for ensuring that research protocols are accepted by oversight bodies like the FDA and for maintaining high-quality evidence in studies.
Is ethics committee approval required before starting a clinical trial in Chile?
Yes, obtaining authorization from a recognized ethics committee is mandatory before starting any study, ensuring adherence to ethical guidelines and participant safety.
How does understanding the regulatory framework benefit researchers conducting clinical trials in Chile?
By fully grasping the regulatory elements, researchers can effectively manage the complexities of executing biopharma clinical trials, leveraging Chile’s advantages for faster and more cost-effective investigations while enhancing the quality of their findings.
List of Sources
- Understand the Regulatory Framework for Clinical Trials in Chile
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Prepare Essential Documentation and Preclinical Data
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOoruKpv9PjUnUWhR0FcAjX2fBw5-M82j13VlVxm2SZuTDIsUePHx)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Understanding the Regulatory Requirements for Preclinical Studies: A Comprehensive Guide – Infinix Bio (https://infinixbio.com/glossary/understanding-the-regulatory-requirements-for-preclinical-studies-a-comprehensive-guide)
- Safety First: The Importance of Preclinical Research in Contemporary Pharmacology – AIHCP (https://aihcp.net/2025/08/25/safety-first-the-importance-of-preclinical-research-in-contemporary-pharmacology)
- The Importance of Quality Data for Regulatory Submissions | Pharmaceutical Technology (https://pharmtech.com/view/the-importance-of-quality-data-for-regulatory-submissions)
- Execute the Clinical Trial: Site Selection and Patient Recruitment
- Trends in clinical trials performed in Chile (https://scielo.cl/article_plus.php?pid=S0034-98872021000100110&tlng=en&lng=es)
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- Monitor and Manage Trial Compliance and Progress
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Chile Clinical Trial Supply & Logistics Market Size & Outlook, 2030 (https://grandviewresearch.com/horizon/outlook/clinical-trial-supply-logistics-market/chile)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOordeUWhJKjbqKm_JJ5F9rdqN_te1p8aNhZjEbKZWjzZDrg0aM7r)
- Clinical Research Monitoring 101 Quotes by Dan Sfera (https://goodreads.com/work/quotes/54920341)

Leave a Reply