Introduction
Understanding the complex landscape of gene and cell therapy is crucial for stakeholders navigating Montenegro’s rapidly evolving regulatory framework. As advancements in these innovative treatments promise to transform healthcare, grasping the underlying concepts and compliance requirements is vital. Yet, with the intricacies of regulatory processes and ethical considerations at play, how can organizations ensure they meet necessary standards while promoting patient safety and access to groundbreaking therapies?
The Medtech landscape is shifting, and bioaccess plays a pivotal role in addressing the key challenges that arise. By staying informed and adapting to these changes, stakeholders can better navigate the complexities of clinical research. Collaboration among organizations is essential to foster innovation and ensure that patients benefit from these advancements.
In summary, understanding the regulatory landscape is not just about compliance; it’s about paving the way for future breakthroughs in healthcare. Stakeholders must take proactive steps to engage with these developments, ensuring that they are well-equipped to meet the demands of this evolving field.
Clarify Gene and Cell Therapy Concepts
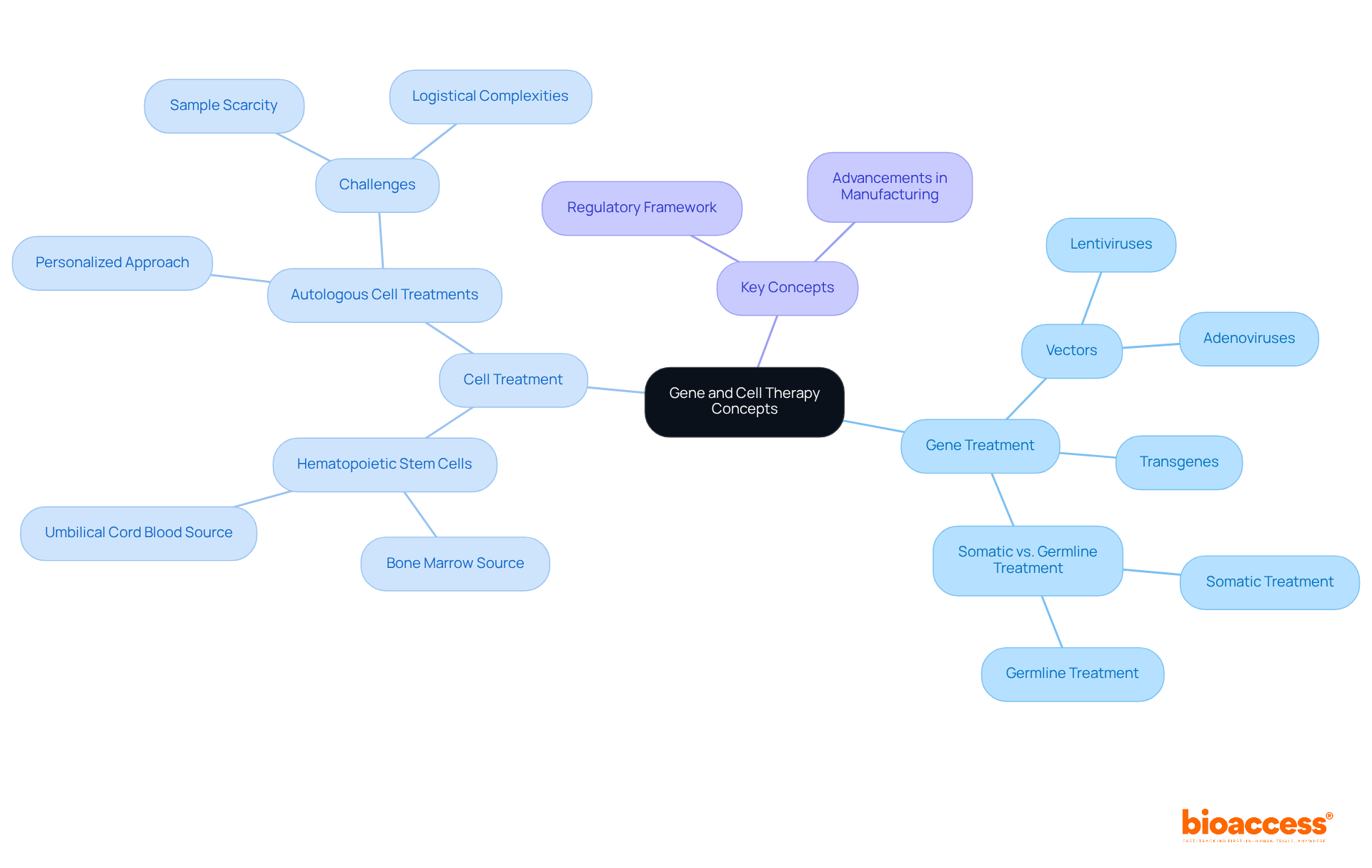
involves introducing genetic material into a patient’s tissues to address or avert diseases. This is achieved through methods such as , which deliver therapeutic genes directly into target tissues. In contrast, employs living organisms to replace or mend damaged tissues and enhance the immune response against illnesses. Understanding these definitions is crucial, as they form the foundation of the in Montenegro that governs these therapies.
Key concepts include:
- Vectors: Tools like lentiviruses or adenoviruses that facilitate the delivery of genetic material into cells.
- Transgenes: Specific genes transferred into the patient’s tissues to achieve therapeutic effects.
- : Somatic treatment targets non-reproductive units, while germline treatment affects reproductive units, potentially passing changes to future generations.
Recent advancements in have yielded promising results, particularly in regenerative medicine. For instance, derived from bone marrow or umbilical cord blood have shown effectiveness in treating hematological cancers, underscoring their vital role in tissue regeneration. Furthermore, , which utilize a patient’s own cells, are gaining traction for their personalized approach, despite challenges such as sample scarcity.
As Dr. David DiGiusto observes, “The advancement of manufacturing methods and QC assays is vital for guaranteeing the safety and effectiveness of these treatments.” By grasping these concepts, stakeholders can navigate the intricate gene and cell therapy in Montenegro, overseeing these innovative treatments more effectively, promoting compliance and encouraging progress in care alternatives.
Explore Montenegro’s Regulatory Landscape
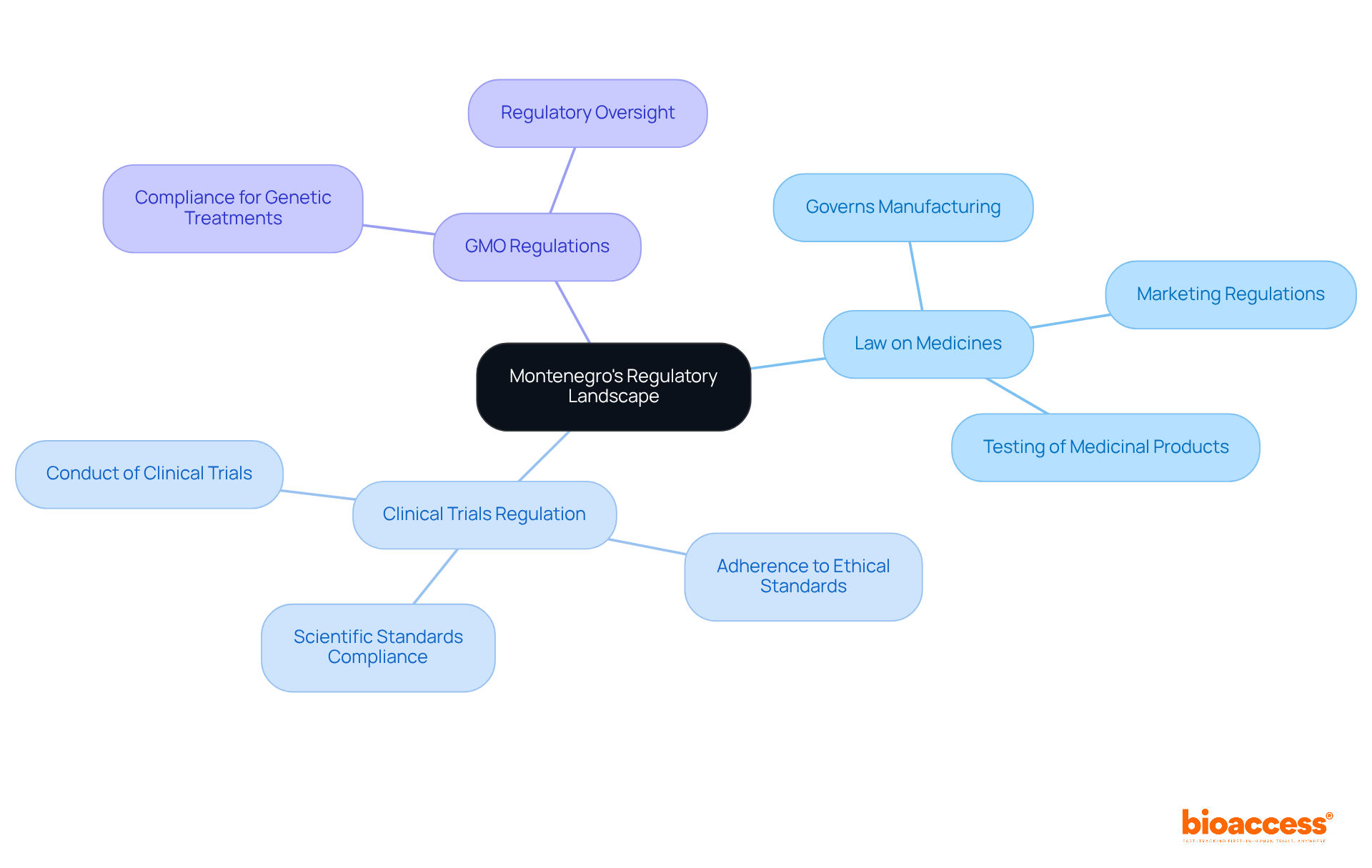
The gene and cell therapy in Montenegro is rapidly advancing, demonstrating significant alignment with EU standards. The primary regulatory body overseeing these treatments is the (CInMED), which plays a crucial role in ensuring compliance with both national and EU regulations.
Key components of this framework include:
- : This legislation governs the manufacturing, marketing, and testing of medicinal products, including .
- : Montenegro has established regulations that dictate the conduct of , ensuring adherence to ethical and scientific standards.
- : Given that many genetic treatments involve genetically modified organisms, compliance with is essential.
Stakeholders must grasp these regulatory nuances to ensure their treatments meet the necessary legal requirements for approval. This understanding facilitates and enhances patient access.
Navigate the Approval Process for Therapies
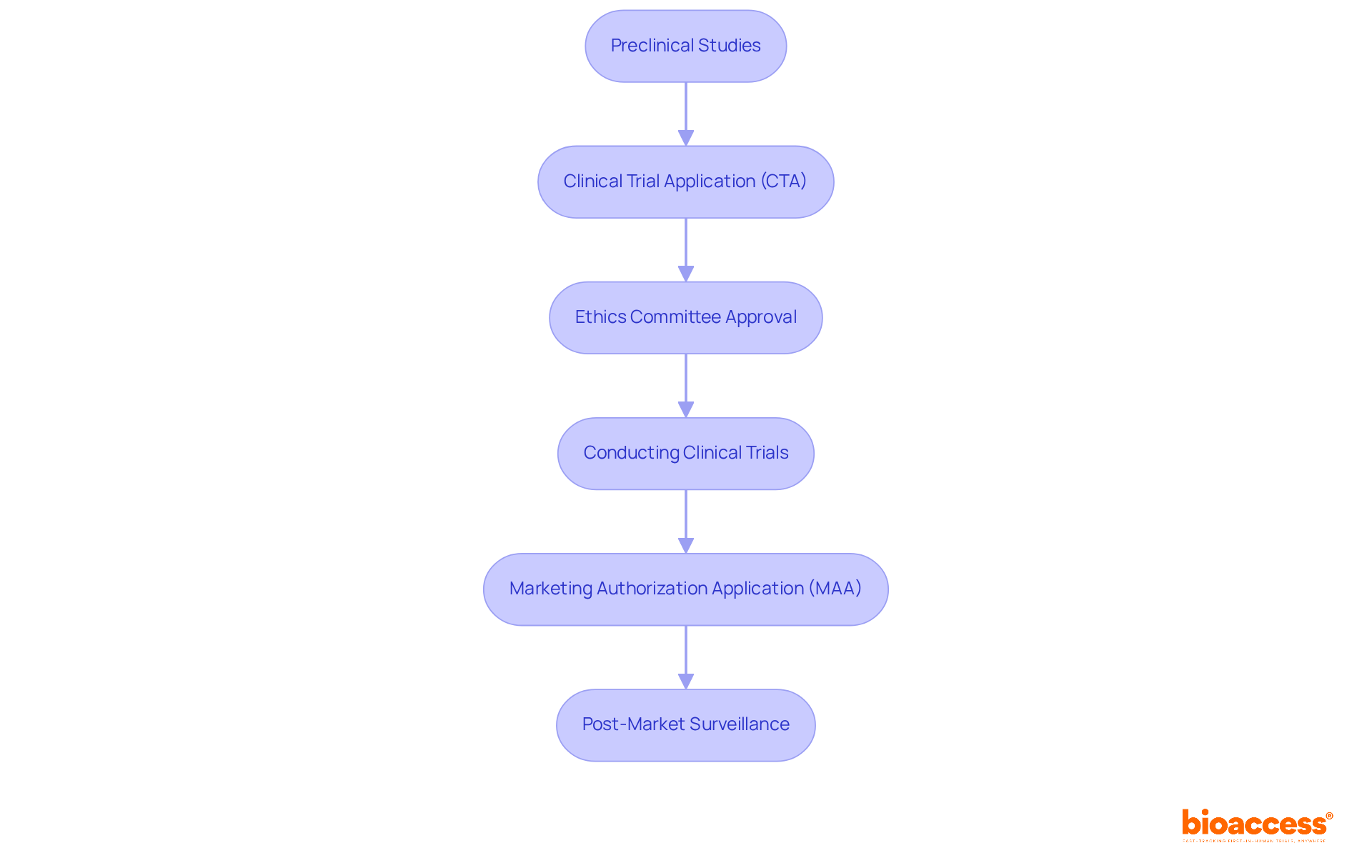
The approval process within the gene and cell therapy regulatory framework in Montenegro is structured around several that are crucial for ensuring safety and efficacy in clinical research.
- : Comprehensive are vital to demonstrate the treatment’s safety and efficacy before submitting an application. These studies form the foundation for , with success rates in preclinical phases significantly influencing later stages.
- (CTA): A must be submitted to the Institute for Medicines and Medical Devices (CInMED), including all necessary documentation such as study protocols, informed consent forms, and investigator brochures. This application is pivotal in initiating the and ensuring compliance with regulatory standards.
- : Gaining approval from an ethics committee is mandatory before commencing clinical trials. This step guarantees that the proposed research adheres to ethical standards and protects participant rights, fostering trust in the research process.
- : During the trial, strict adherence to the approved protocol is essential. This ensures compliance with all regulatory requirements and maintains the integrity of the study, which is critical for valid results.
- (MAA): After successful trials, a must be submitted, encompassing comprehensive data on safety, efficacy, and manufacturing processes. This application is crucial for obtaining market access and bringing innovative treatments to patients.
- : Once approved, establishing a robust strategy is essential. This strategy tracks the treatment’s performance and reports any adverse effects, ensuring and confidence in the therapy.
By meticulously following these steps, organizations can effectively navigate the approval process outlined in the gene and cell therapy regulatory framework in Montenegro, ensuring compliance and promoting timely access to the market for groundbreaking treatments.
Address Ethical and Compliance Considerations
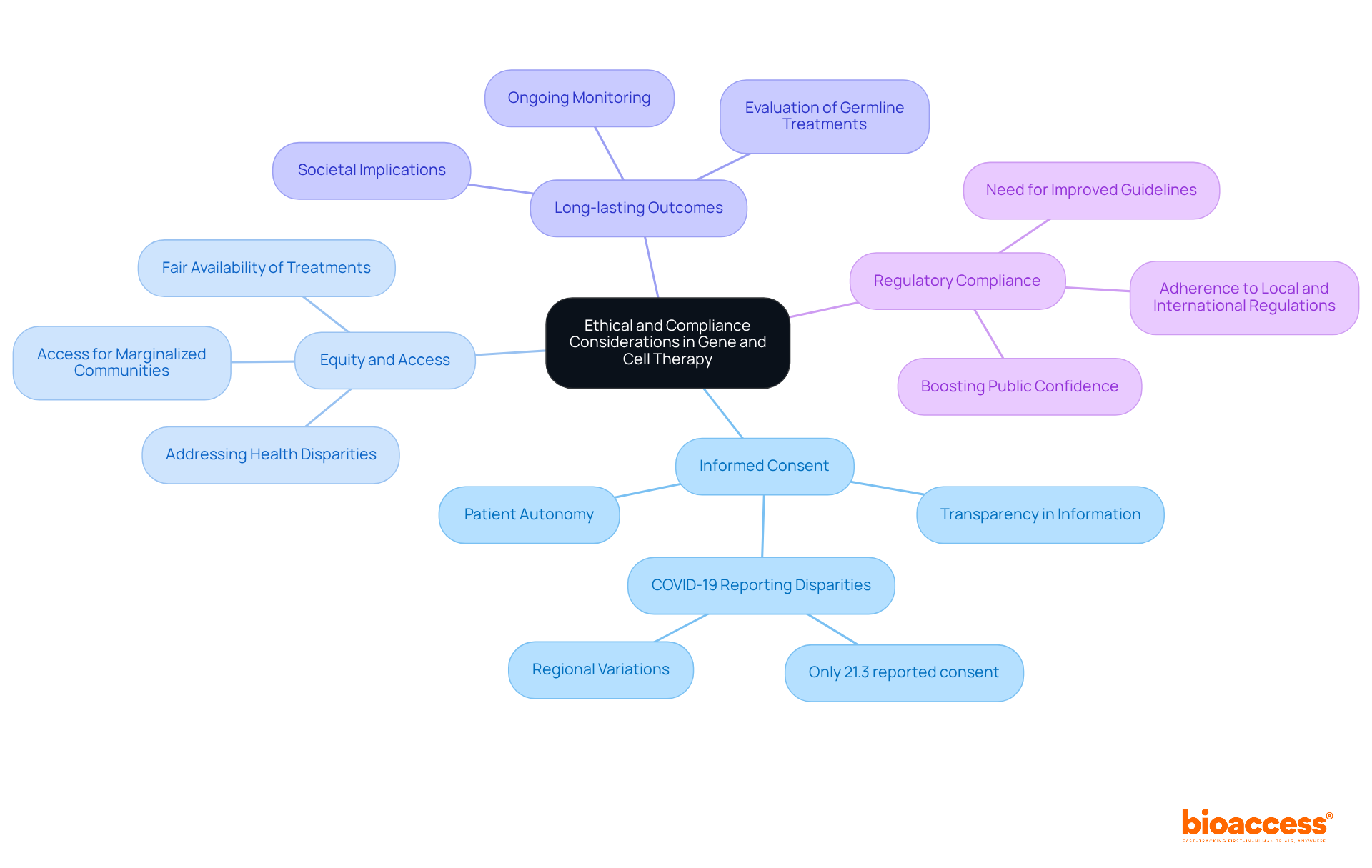
encompass several critical issues that demand attention:
- : Patients must receive comprehensive information about the therapy’s nature, potential risks, and benefits before enrolling in . This transparency is essential for protecting patient autonomy and ensuring ethical research conduct. Notably, a systematic review found that only 21.3% of studies reported during the COVID-19 pandemic, underscoring the urgent need for rigorous adherence to protocols.
- Equity and Access: Ensuring is vital to prevent exacerbating existing health disparities. Efforts focused on enhancing access to genetic treatments, especially in marginalized communities, are essential for promoting . Frameworks that address and ethics regulations during crises can significantly enhance access for these groups.
- : Investigators must thoroughly evaluate the enduring consequences of genetic modification, particularly in germline treatments that may affect subsequent generations. This necessitates ongoing monitoring and evaluation to understand the broader societal and health implications of these interventions.
- : Adhering to both local and international regulations is essential for maintaining research integrity and guaranteeing . Compliance with ethical standards not only safeguards participants but also boosts public confidence in biological and cellular treatments. The under-reporting of ethical aspects in multicenter studies highlights the need for improved guidelines and practices in and data sharing.
By addressing these ethical and compliance considerations, stakeholders can promote a responsible approach to the development and implementation of the gene and cell therapy regulatory framework in Montenegro. This ultimately enhances .
Conclusion
Mastering the gene and cell therapy regulatory framework in Montenegro is not just essential; it’s a critical step for stakeholders eager to navigate this rapidly evolving field. Understanding the intricacies of gene and cell therapy – definitions, methodologies, and the regulatory landscape – is crucial for ensuring compliance and fostering innovative treatments. Montenegro’s alignment with EU standards showcases a robust framework that supports the safe and effective development of these therapies.
In this article, we explored key components such as the roles of regulatory bodies, the approval process, and ethical considerations in detail. The importance of preclinical studies, clinical trial applications, and post-market surveillance emerged as critical steps in ensuring the safety and efficacy of treatments. Moreover, ethical issues like informed consent and equitable access were highlighted as vital considerations for responsible research and practice.
Ultimately, the advancement of gene and cell therapies holds immense promise for transforming healthcare in Montenegro. By adhering to regulatory requirements and ethical standards, stakeholders can build public trust and enhance patient safety. Engaging with the evolving landscape of gene and cell therapy regulations not only paves the way for innovative treatments but also ensures that these advancements are accessible and beneficial to all segments of the population. What steps will you take to be part of this transformative journey?
Frequently Asked Questions
What is gene therapy?
Gene therapy involves introducing genetic material into a patient’s tissues to address or prevent diseases, often using methods such as viral vectors to deliver therapeutic genes directly into target tissues.
How does cell therapy differ from gene therapy?
Cell therapy employs living organisms to replace or repair damaged tissues and enhance the immune response against illnesses, while gene therapy focuses on the introduction of genetic material.
What are vectors in the context of gene therapy?
Vectors are tools, such as lentiviruses or adenoviruses, that facilitate the delivery of genetic material into cells.
What are transgenes?
Transgenes are specific genes that are transferred into the patient’s tissues to achieve therapeutic effects.
What is the difference between somatic and germline treatment?
Somatic treatment targets non-reproductive cells, while germline treatment affects reproductive cells, potentially passing genetic changes to future generations.
What recent advancements have been made in cellular treatment?
Recent advancements in cellular treatment, particularly in regenerative medicine, include the effectiveness of hematopoietic stem cells (HSCs) derived from bone marrow or umbilical cord blood in treating hematological cancers.
What are autologous cell treatments?
Autologous cell treatments utilize a patient’s own cells, offering a personalized approach to therapy, although challenges such as sample scarcity exist.
Why is the advancement of manufacturing methods and quality control (QC) assays important in gene and cell therapy?
The advancement of manufacturing methods and QC assays is vital for ensuring the safety and effectiveness of gene and cell therapies.
How can understanding gene and cell therapy concepts help stakeholders in Montenegro?
Understanding these concepts allows stakeholders to navigate the gene and cell therapy regulatory framework in Montenegro, promoting compliance and encouraging progress in innovative treatment options.
List of Sources
- Clarify Gene and Cell Therapy Concepts
- Cell Therapy Types, Uses, and Future Impacts (https://the-scientist.com/cell-therapy-types-uses-and-future-impacts-74014)
- Case studies in cell & gene therapy process optimization (https://insights.bio/cell-and-gene-therapy-insights/webinars/65/Case-studies-in-cell-gene-therapy-process-optimization)
- Explore Montenegro’s Regulatory Landscape
- FDA removes barriers to cell, gene therapy development (https://pharmaphorum.com/news/fda-removes-barriers-cell-gene-therapy-development)
- Why gene and cell therapies are stalling at the FDA (https://drugdiscoverynews.com/why-gene-and-cell-therapies-are-stalling-at-the-fda-16527)
- Navigate the Approval Process for Therapies
- Navigating Cell and Gene Therapy Regulations | IDT (https://idtdna.com/page/support-and-education/decoded-plus/navigating-the-regulatory-landscape-a-business-perspective-on-cell-and-gene-therapy-approvals)
- Case studies in cell & gene therapy process optimization (https://insights.bio/cell-and-gene-therapy-insights/webinars/65/Case-studies-in-cell-gene-therapy-process-optimization)
- Montenegro Pharmacovigilance Reporting Requirements – Clinical Trials & Post-Marketing | RegIntelHub (https://regintelhub.com/Montenegro-18b49d840dce80d0a1c4fc5367c3b7d9)
- Address Ethical and Compliance Considerations
- Addressing the Barriers to Equitable Access in Cell and Gene Therapies | Targeted Oncology – Immunotherapy, Biomarkers, and Cancer Pathways (https://targetedonc.com/view/addressing-the-barriers-to-equitable-access-in-cell-and-gene-therapies)
- Structural under-reporting of informed consent, data handling and sharing, ethical approval, and application of Open Science principles as proxies for study quality conduct in COVID-19 research: a systematic scoping review – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10254958)
- Structural under-reporting of informed consent, data handling and sharing, ethical approval, and application of Open Science principles as proxies for study quality conduct in COVID-19 research: a systematic scoping review (https://gh.bmj.com/content/8/5/e012007)
- As genome-editing trials become more common, informed consent is changing (https://genome.gov/news/news-release/As-genome-editing-trials-become-more-common-informed-consent-is-changing)
- Creating Accessible Cell & Gene Therapies | Azenta Life Sciences (https://azenta.com/learning-center/blog/creating-accessible-cell-and-gene-therapies-the-world-cant-wait)

Leave a Reply