Introduction
In the rapidly evolving landscape of clinical research, the quest for efficiency and effectiveness has never been more critical. As organizations strive to accelerate their studies and improve patient outcomes, understanding the nuances of various research strategies becomes paramount. This article delves into ten compelling head-to-head comparisons that illuminate the strengths and weaknesses of distinct clinical research methodologies. How can stakeholders harness these insights to optimize their approaches and ultimately enhance the success of medical trials?
bioaccess®: Accelerate Clinical Research with Global-First Agility
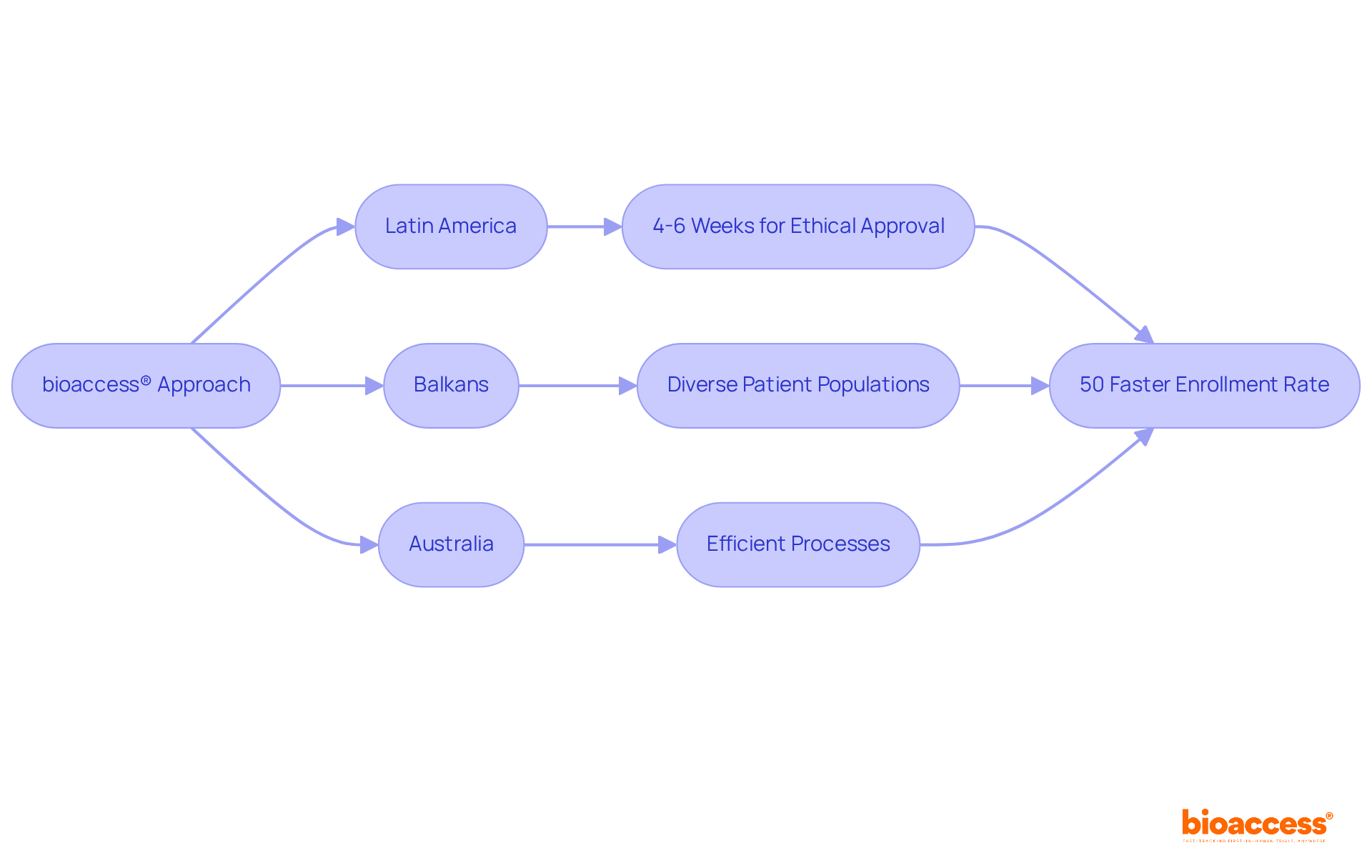
bioaccess® distinguishes itself in the medical investigation landscape by providing unmatched flexibility through its . By harnessing the , where ethical approvals can be secured in just 4-6 weeks, and combining this with the in the Balkans and the , bioaccess® achieves a remarkable compared to traditional markets. This exceptional efficiency positions bioaccess® as the ideal partner for innovators who are eager to expedite their research timelines. The region’s rich diversity not only enhances the across various ethnic groups but also fosters a robust recruitment environment, establishing it as a vital hub for .
Second-Generation Nucleic Acid Amplification Tests: A Comparative Analysis
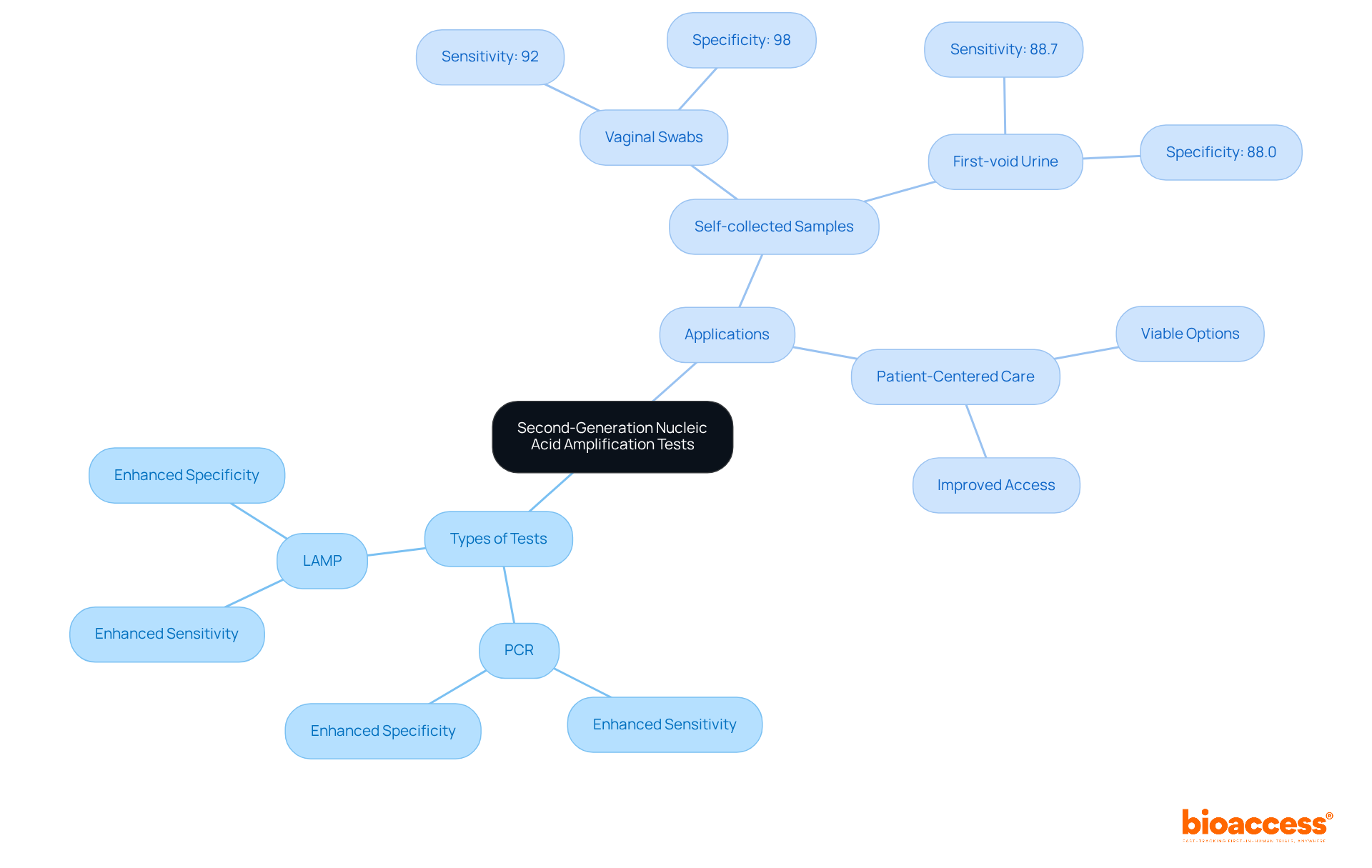
Second-generation (NAATs) have revolutionized the detection of . These tests, including PCR and LAMP, demonstrate compared to traditional methods, establishing their credibility in . Notably, studies indicate that , such as vaginal swabs and first-void urine, yield , positioning them as viable options for . This analysis underscores the critical importance of selecting the based on the medical context and individual requirements, prompting healthcare professionals to consider in their practice.
AI-Based Coronary Stenosis Algorithms: Evaluating Inter-Reader Agreement
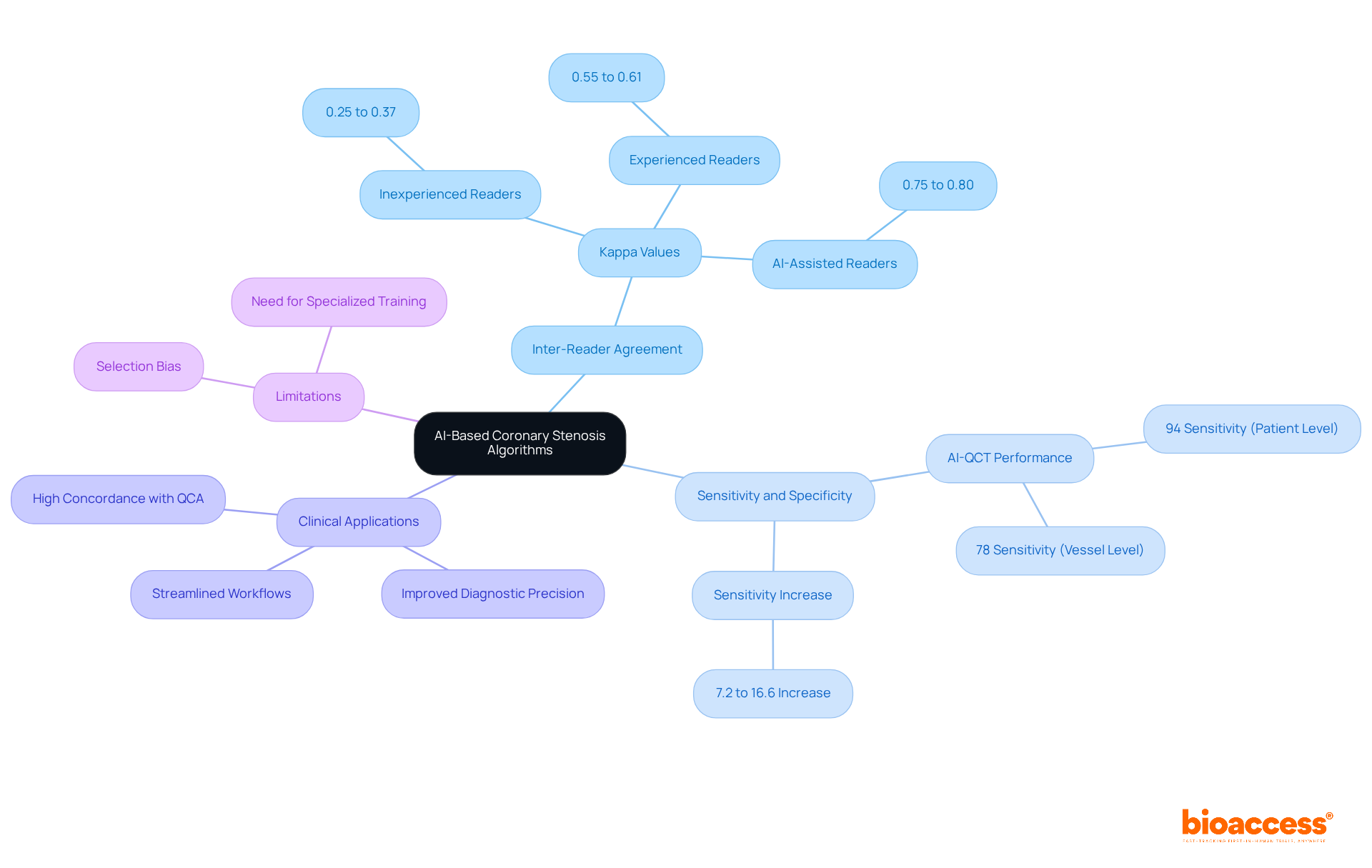
have significantly improved inter-reader agreement among radiologists. Recent studies highlight that . Notably, one study reported a sensitivity increase from 7.2% to 16.6% for AI-assisted readers compared to their inexperienced counterparts. This progress not only enhances diagnostic precision but also in , ultimately leading to .
For instance, AI-QCT has demonstrated a remarkable 94% sensitivity at the individual level and 78% at the vessel level, surpassing conventional methods and showcasing AI’s potential to minimize variability in evaluations. Furthermore, , underscoring its effectiveness in clinical applications.
However, it is essential to acknowledge potential limitations, such as selection bias stemming from the use of invasive coronary angiography (ICA) as the reference standard. Additionally, , particularly for inexperienced readers. The study involved 196 individuals who underwent both coronary computed tomography angiography (CCTA) and invasive coronary angiography (ICA) within a six-month period, providing critical context for these findings. , thereby facilitating more reliable diagnoses and timely interventions.
Coronary CT Angiography Techniques: A Head-to-Head Comparison
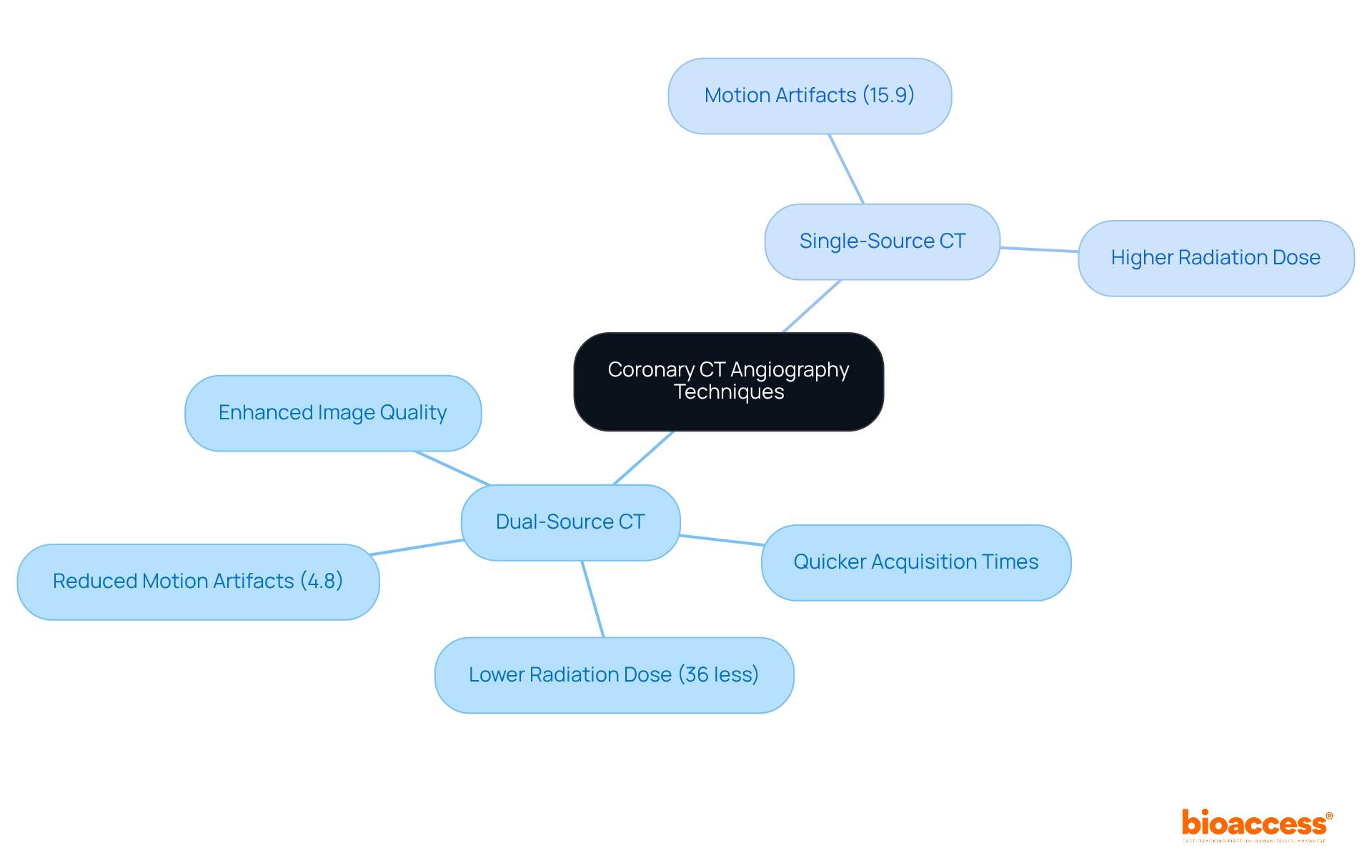
A of coronary CT angiography techniques, including dual-source and single-source CT, reveals significant differences in . Notably, , making it more suitable for individuals with elevated heart rates, high calcium scores, or obesity. Recent advancements in dual-source technology, such as iterative reconstruction algorithms and improved temporal resolution, have significantly reduced motion artifacts. Studies indicate a . Moreover, the , reinforcing its role in optimizing safety while maintaining high diagnostic accuracy. As highlighted in the literature, “,” emphasizing the importance of selecting the appropriate technology based on medical requirements. Clinicians are encouraged to incorporate the into their evaluations to enhance diagnostic strategies effectively.
Customer Value Analysis in Clinical Research: Key Comparisons
underscores the critical importance of , particularly patients, sponsors, and regulatory agencies. Organizations that prioritize consistently report higher satisfaction rates and improved . Research indicates that by addressing their concerns and minimizing barriers to involvement. By , healthcare organizations can enhance their service offerings, ultimately fostering stronger connections with clients and elevating overall study success.
Moreover, the , which facilitate remote involvement, illustrates how can yield more effective recruitment strategies and enhanced retention rates. This comprehensive approach not only boosts but also aligns medical studies with the evolving landscape of patient expectations. As the environment continues to change, it is imperative for organizations to embrace these strategies to ensure successful outcomes.
Ethical Approvals in Clinical Trials: A Comparative Overview
in research studies exhibit substantial variability across regions, significantly . Notably, distinguishes itself with , typically achieving approvals within 4 to 6 weeks. This efficiency offers a marked advantage compared to North America and Europe, where stringent regulatory frameworks often result in , occasionally exceeding six months.
For example, ‘s regulatory environment is recognized for its swift assessments, with the IRB/EC and INVIMA review processes completed in just 90 to 120 days. Understanding these is crucial for sponsors aiming to enhance , as of the pace of can directly influence the success of medical initiatives.
Enrollment Strategies in Clinical Trials: Regional Comparisons
Enrollment approaches in research studies vary significantly across regions, shaped by cultural, economic, and regulatory factors. In Latin America, community engagement is crucial for . Local partnerships, exemplified by the collaboration between , play a vital role in fostering trust and awareness, which in turn enhances enrollment rates. This collaboration aims to position Barranquilla as a , supported by Colombia’s Minister of Health, who advocates for the expansion of research initiatives in the region. Remarkably, this partnership has achieved and boasts a 95% retention rate, underscoring its effectiveness.
Research indicates that can rival those in North America, where digital marketing and patient registries are more commonly utilized. However, nearly 80% of medical studies globally fail to meet their initial enrollment targets, with delays costing sponsors between $600,000 and $8 million each day. ; for instance, while 16% of the U.S. population is Hispanic, they constitute only 1% of research study participants. Similarly, although African-Americans make up 12% of the U.S. population, they represent only 5% of research participants. This disparity highlights the urgent need for .
Engaging local healthcare providers and employing culturally relevant messaging can enhance participation rates and ensure a more representative sample in medical studies. The experience of leaders like Dushyanth Surakanti, Founder & CEO of Sparta Biomedical, during bioaccess®’s inaugural human trial in Colombia further illustrates the potential for successful outcomes through .
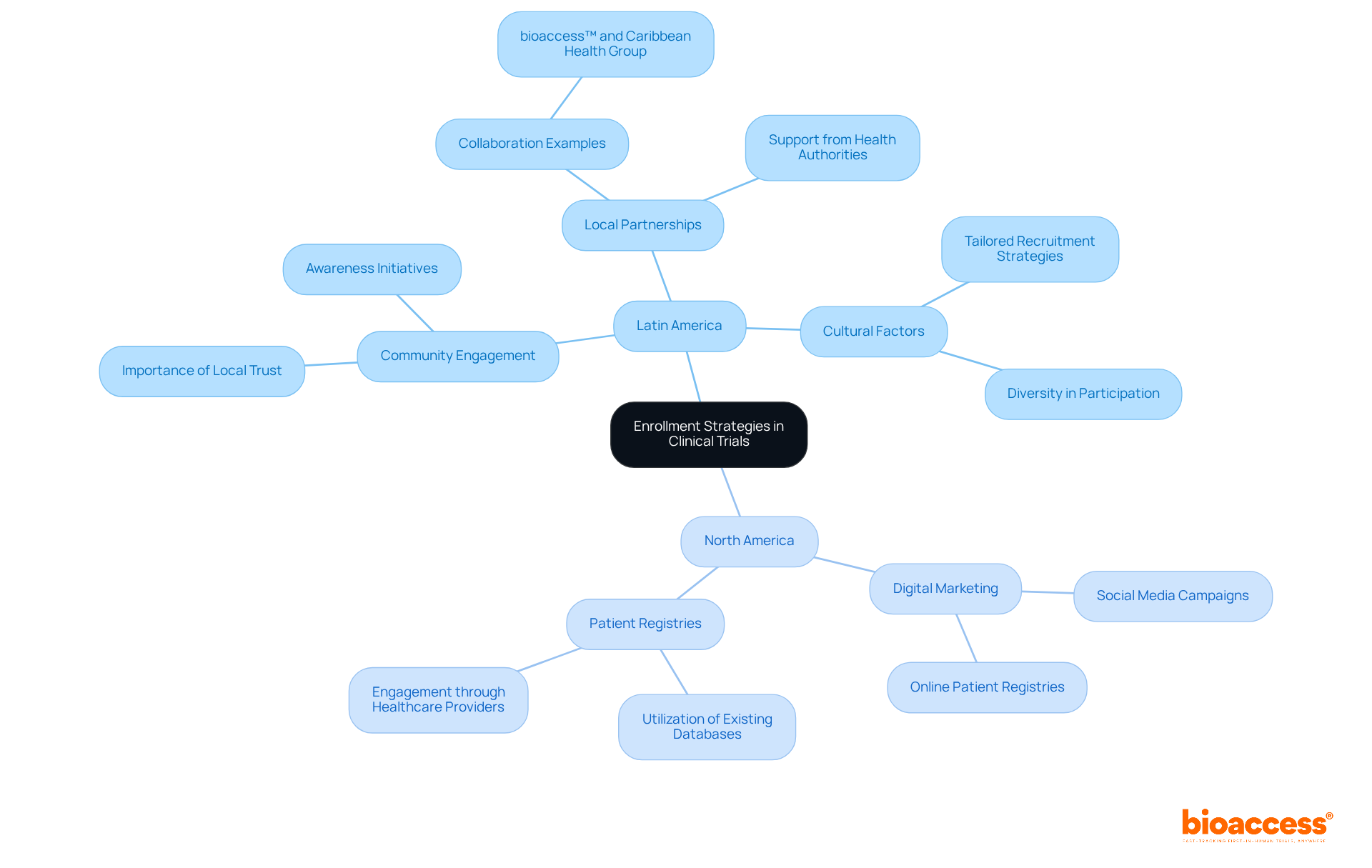
Regulatory Pathways in Clinical Research: A Comparative Study
A comparative examination of shows significant differences in approval procedures and timelines through head to head comparisons. The , enhance access to investigational therapies in the U.S., allowing for approvals in as little as six months. In contrast, other regions often adhere to more stringent frameworks, which can considerably extend the approval timeline. For instance, over the past decade, 51 , underscoring the efficiency of the FDA’s expedited programs. Moreover, in 2018 and 2019, 73% and 60% of novel drugs, respectively, received expedited approval, reflecting the FDA’s commitment to addressing unmet medical needs.
This understanding is vital for sponsors aiming to effectively and capitalize on the advantages offered by these . Bioaccess® links pioneering Medtech, Biopharma, and Radiopharma startups with leading research facilities in Latin America, Eastern Europe, and Australia. By utilizing bioaccess’s extensive management services for —including feasibility assessments, site selection, , setup, import permits, project oversight, and reporting—sponsors can accelerate their research processes and ensure adherence to regulatory standards. Ultimately, this collaboration results in .
Patient Recruitment Methods in Clinical Trials: A Comparative Analysis
A comparative analysis of reveals a significant shift from , such as physician referrals and community outreach, to innovative , including social media campaigns and online registries. Research indicates that , which blend traditional and digital methods, are particularly effective. These models yield faster enrollment and .
For example, studies demonstrate that can successfully recruit a younger demographic, with median ages significantly lower than those achieved through traditional methods. Moreover, the integration of digital tools has been linked to improved engagement and retention rates, effectively addressing logistical challenges faced by potential participants.
This examination underscores the importance of tailored to the unique requirements of specific groups, ensuring broader representation and enhancing the overall success of .
Diverse Patient Pools in Clinical Research: Comparative Insights
are essential for guaranteeing that study findings are relevant to a wide population. Trials conducted in regions with , such as , provide more representative data, enhancing the validity of findings. The partnership between bioaccess™ and Caribbean Health Group aims to establish Barranquilla as a premier location for medical studies in , supported by Colombia’s Minister of Health. This initiative not only enhances access to various patient groups but also addresses the , which has led to significant disparities in health outcomes.
For instance, African Americans and Hispanics represent a disproportionately low percentage of research study participants, despite their higher prevalence of specific diseases. Studies in these regions have demonstrated that including underrepresented groups can significantly . The FDA has underscored the necessity for increased , reinforcing the urgency of this issue. The inclusion of varied groups not only enriches the information gathered but also fosters trust in the research process, as communities see their needs represented in medical studies.
Moreover, bioaccess® offers comprehensive , including feasibility assessments, site selection, compliance evaluations, and project oversight, which are crucial for . Highlighting diversity in recruitment is vital for the success of trials, yielding more across various demographics. This approach aligns with the growing recognition that health equity is a fundamental aspect of clinical research, ensuring that all populations benefit from advancements in medical science.
Conclusion
The exploration of head-to-head comparisons in clinical research strategies reveals a critical need for innovation and adaptability in the medical field. By examining various methodologies and approaches, it becomes evident that leveraging regional advantages and technological advancements can significantly enhance the efficiency and effectiveness of clinical trials. This strategic agility is essential for organizations aiming to accelerate research timelines and improve patient outcomes.
Key insights from the comparisons underscore the importance of:
- Diverse patient pools
- Expedited ethical approvals
- Modern recruitment strategies
The advantages of utilizing regions with faster regulatory processes, such as Latin America, combined with advanced diagnostic technologies and AI-driven methodologies, demonstrate the potential for optimizing clinical research. Furthermore, a patient-centric approach that prioritizes stakeholder engagement can lead to improved recruitment and retention rates, ultimately enhancing the overall success of studies.
As the landscape of clinical research continues to evolve, it is imperative for organizations to embrace these innovative strategies. By committing to diversity, efficiency, and technological integration, the medical community can ensure that research outcomes are not only relevant but also equitable. This proactive approach will address existing disparities in health outcomes and foster trust within communities, paving the way for groundbreaking advancements in medical science.
Frequently Asked Questions
What is bioaccess® and how does it enhance clinical research?
bioaccess® is a platform that accelerates clinical research by providing unmatched flexibility through a global-first approach. It leverages the regulatory speed of Latin America, where ethical approvals can be obtained in just 4-6 weeks, and combines this with diverse patient populations in the Balkans and efficient processes in Australia, achieving a 50% faster enrollment rate compared to traditional markets.
Why is the diversity of patient populations important for clinical trials?
The rich diversity of patient populations enhances the understanding of treatment effects across various ethnic groups and fosters a robust recruitment environment, making it a vital hub for clinical trials.
What are second-generation nucleic acid amplification tests (NAATs)?
Second-generation NAATs, including PCR and LAMP, are advanced diagnostic tests that have improved the detection of infectious diseases with enhanced sensitivity and specificity compared to traditional methods.
How do self-collected samples compare to traditional collection methods in NAATs?
Studies indicate that self-collected samples, such as vaginal swabs and first-void urine, yield comparable results to traditional collection methods, positioning them as viable options for patient-centered care.
What advancements have AI-based algorithms brought to the assessment of coronary stenosis?
AI-based algorithms have significantly improved inter-reader agreement among radiologists, enhancing sensitivity and specificity in detecting obstructive coronary artery disease. For example, AI-QCT demonstrated a 94% sensitivity at the individual level.
What are some limitations of using AI-based algorithms in clinical settings?
Potential limitations include selection bias from using invasive coronary angiography as the reference standard and the need for specialized training to enhance diagnostic performance, especially for inexperienced readers.
What was the context of the study involving AI-QCT and coronary angiography?
The study involved 196 individuals who underwent both coronary computed tomography angiography (CCTA) and invasive coronary angiography (ICA) within a six-month period, providing critical context for evaluating the effectiveness of AI-assisted evaluations.
List of Sources
- bioaccess®: Accelerate Clinical Research with Global-First Agility
- Quotes on Medicine and Drug Development – Related Articles – Therapy, Diagnosis, Life Sciences, and Medical Research Discoveries and News – Discovery Medicine (https://discoverymedicine.com/related/2/907)
- bioaccessla.com (https://bioaccessla.com/blog/exploring-latin-america-medical-trial-regions-an-essential-guide-for-researchers)
- TOP 10 CLINICAL TRIALS QUOTES | A-Z Quotes (https://azquotes.com/quotes/topics/clinical-trials.html)
- statista.com (https://statista.com/statistics/1013599/latin-america-clinical-trials?srsltid=AfmBOooyzj4-w_GuS91NSUA2mgM_2q0aaWGInBV__I8CA7l-RxT6_ez5)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- Second-Generation Nucleic Acid Amplification Tests: A Comparative Analysis
- ncbi.nlm.nih.gov (https://ncbi.nlm.nih.gov/books/NBK373484)
- nature.com (https://nature.com/articles/s41598-024-61358-y)
- pubs.acs.org (https://pubs.acs.org/doi/10.1021/acs.analchem.3c05225)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC87609)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC4097753)
- AI-Based Coronary Stenosis Algorithms: Evaluating Inter-Reader Agreement
- mdpi.com (https://mdpi.com/2075-4418/15/3/282)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC8851787)
- link.springer.com (https://link.springer.com/article/10.1007/s10554-024-03256-y)
- pace-cme.org (https://pace-cme.org/news/performance-of-ai-versus-humans-in-detecting-coronary-stenosis/2474187)
- openheart.bmj.com (https://openheart.bmj.com/content/12/1/e003115)
- Coronary CT Angiography Techniques: A Head-to-Head Comparison
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC11450059)
- pubmed.ncbi.nlm.nih.gov (https://pubmed.ncbi.nlm.nih.gov/19304713)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC5124932)
- sciencedirect.com (https://sciencedirect.com/science/article/abs/pii/S2173510713000621)
- nature.com (https://nature.com/articles/s41569-025-01191-6)
- Customer Value Analysis in Clinical Research: Key Comparisons
- lindushealth.com (https://lindushealth.com/blog/the-importance-of-patient-centricity-in-improving-clinical-trial-retention)
- Patient Engagement Statistics: Data That Proves Impact (https://nclusiv.co.uk/blog/f/patient-engagement-statistics-data-that-proves-impact)
- tandfonline.com (https://tandfonline.com/doi/full/10.1080/17576180.2025.2458453)
- Patient Experience, Satisfaction and Billing Statistics | CallMiner (https://callminer.com/blog/101-statistics-on-patient-experience-satisfaction-billing-and-more)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC11006977)
- Ethical Approvals in Clinical Trials: A Comparative Overview
- ecancer.org (https://ecancer.org/en/journal/article/1379-timelines-to-initiate-a-phase-iii-trial-across-the-globe-a-sub-analysis-of-the-aphinity-trial)
- Global comparison of research ethical review protocols: insights from an international research collaborative – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11975204)
- bioaccessla.com (https://bioaccessla.com/blog/mastering-clinical-trial-regulation-benefits-in-latin-america-a-comprehensive-guide)
- Experts’ Views on FDA Regulatory Standards for Drug and High-Risk Medical Devices: Implications for Patient Care – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC9708961)
- bmcmedethics.biomedcentral.com (https://bmcmedethics.biomedcentral.com/articles/10.1186/s12910-015-0022-z)
- Enrollment Strategies in Clinical Trials: Regional Comparisons
- veristat.com (https://veristat.com/blog/multi-regional-clinical-trials-key-recommendations-for-a-global-clinical-development-program)
- Enrollment and Retention: A Strategic Imperative for Clinical Trial Success – ConfidenceResearch (https://confidenceresearch.com/enrollment-and-retention-a-strategic-imperative-for-clinical-trial-success)
- researchgate.net (https://researchgate.net/publication/396650978_Regional_variation_in_clinical-trial_risks_a_large-scale_analysis_of_585_clinical_trials)
- appliedclinicaltrialsonline.com (https://appliedclinicaltrialsonline.com/view/enrollment-performance-weighing-facts)
- Enrollment in Clinical Trials: Statistics and Patient Recruitment Strategies | Power (https://withpower.com/guides/enrollment-in-clinical-trials-statistics-and-patient-recruitment-strategies)
- Regulatory Pathways in Clinical Research: A Comparative Study
- jamanetwork.com (https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2798005)
- A Comprehensive Review of US-FDA Novel Drug Approvals from 2013-2022: An Observational Study (https://biomedpharmajournal.org/vol18no3/a-comprehensive-review-of-us-fda-novel-drug-approvals-from-2013-2022-an-observational-study)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC10391045)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC9332089)
- Experts’ Views on FDA Regulatory Standards for Drug and High-Risk Medical Devices: Implications for Patient Care – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC9708961)
- Patient Recruitment Methods in Clinical Trials: A Comparative Analysis
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- From AI to telemedicine: Transforming clinical trial recruitment in 2024 (https://labiotech.eu/expert-advice/clinical-trial-recruitment)
- Representation is power: traditional, hybrid, and digital recruitment results from a non-randomized clinical trial engaging adolescents – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC12504426)
- clinicaltrialsarena.com (https://clinicaltrialsarena.com/sponsored/how-hybrid-models-and-tech-are-shaping-the-future-of-clinical-research)
- Improving Participant Recruitment in Clinical Trials: Comparative Analysis of Innovative Digital Platforms (https://jmir.org/2024/1/e60504)
- Diverse Patient Pools in Clinical Research: Comparative Insights
- Top 5 Reasons Why Diversity in Clinical Trials Is Crucial (https://studypages.com/blog/the-importance-of-diversity-in-clinical-trials)
- obviohealth.com (https://obviohealth.com/resources/diversity-in-clinical-trials)
- antidote.me (https://antidote.me/blog/why-racial-diversity-in-clinical-trials-is-so-important)
- Key Trends in Demographic Diversity in Clinical Trials – Improving Representation in Clinical Trials and Research – NCBI Bookshelf (https://ncbi.nlm.nih.gov/books/NBK584392)
- The Importance of Diversity in Clinical Trials and the Impact of FDA Guidance – Clinical Trial Listing Database & Insights | CenterWatch (https://wcgclinical.com/insights/the-importance-of-diversity-in-clinical-trials-and-the-impact-of-fda-guidance)

Leave a Reply