Introduction
Informed consent stands as a fundamental pillar in the ethical and legal framework governing research involving human subjects. It is designed to uphold participants’ autonomy by ensuring they are fully informed about the study they are engaging in, including potential risks and benefits. This process of transparency not only fosters trust between researchers and participants but also enhances the overall quality and integrity of the research.
Historical unethical research practices, such as those during World War II and the Tuskegee Syphilis Study, led to the establishment of regulatory safeguards, including Institutional Review Boards (IRBs), which protect the rights and dignity of participants. These bodies serve as an objective third party to ensure ethical research practices are adhered to in compliance with federal regulations.
The primary goal of informed consent documents is to aid potential subjects in deciding whether to participate in a research study by presenting necessary information clearly and comprehensibly. However, the increasing complexity of these documents has posed challenges to clinical trial enrollment, particularly among underserved minority populations. Despite these hurdles, informed consent remains crucial for maintaining public trust and ensuring ethical conduct in research.
serves as a cornerstone in both ethical and legal frameworks for studies involving human subjects. It upholds individual autonomy by ensuring they are fully informed about the study they are involved in, including the potential risks and benefits. This openness not only builds confidence between scholars and subjects but also of the study.
Historical unethical practices, such as those seen during World War II and the Tuskegee Syphilis Study, led to the establishment of . These safeguards, including , are in place to protect the rights and dignity of participants. IRBs act as an impartial third party to guarantee that studies are carried out ethically and in compliance with federal regulations.
The main purpose of is to help prospective participants determine if they wish to engage in a study by clearly providing essential information in an understandable way. However, these documents have become increasingly complex, often presenting obstacles to , particularly among underserved minority populations. Regardless of these obstacles, knowledgeable agreement is crucial for sustaining public confidence and guaranteeing .
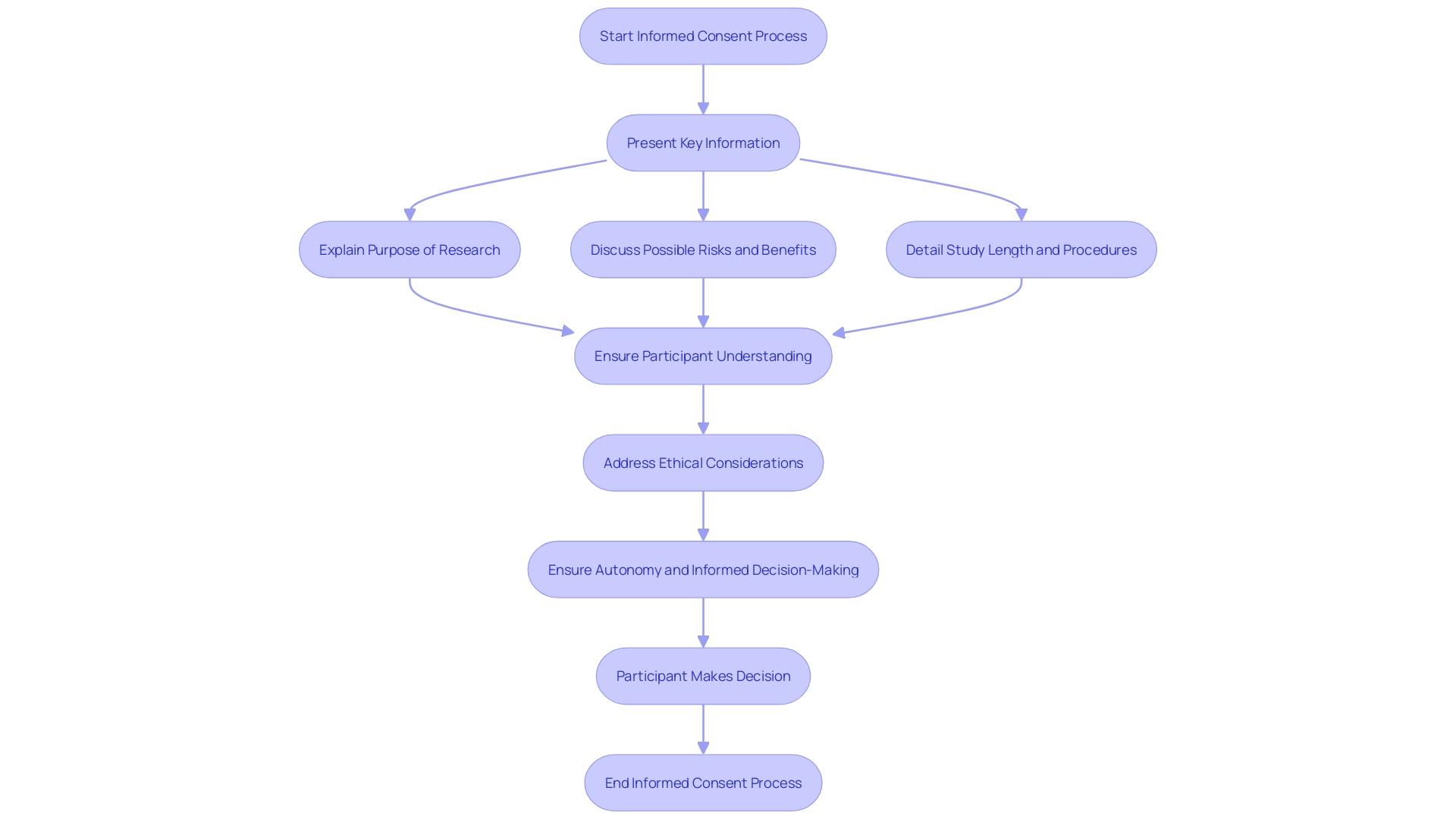
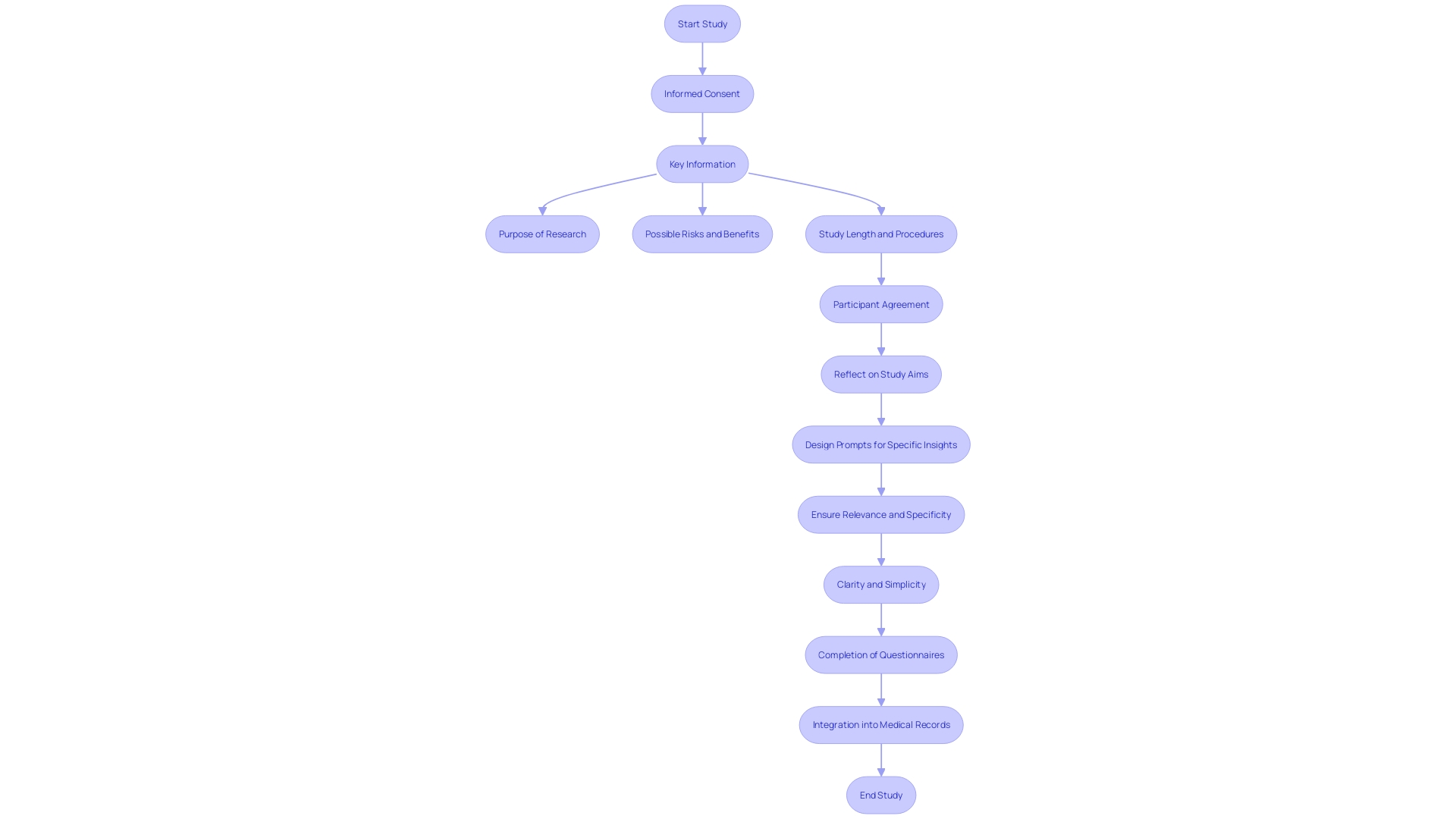
Informed agreement includes several to ensure participants fully understand the study process. These elements include a clear statement indicating that the study involves investigation, an explanation of its purposes, the expected duration of participation, and a description of the procedures involved.
The primary goal of is to assist prospective subjects in making an informed decision about participation by presenting necessary information clearly and facilitating comprehension. Yet, as time has passed, these documents have grown more intricate, extensive, and challenging for all involved parties, including IRBs, physicians, clinical trial sponsors, research subjects, and regulators. The list of mandatory items can run to over 270 words, and documents have expanded from three to four pages to over twenty pages in many cases. They are often written at a reading level too high for many participants and have become more legalistic to comply with legislation, posing an obstacle to , especially among underserved minority populations.
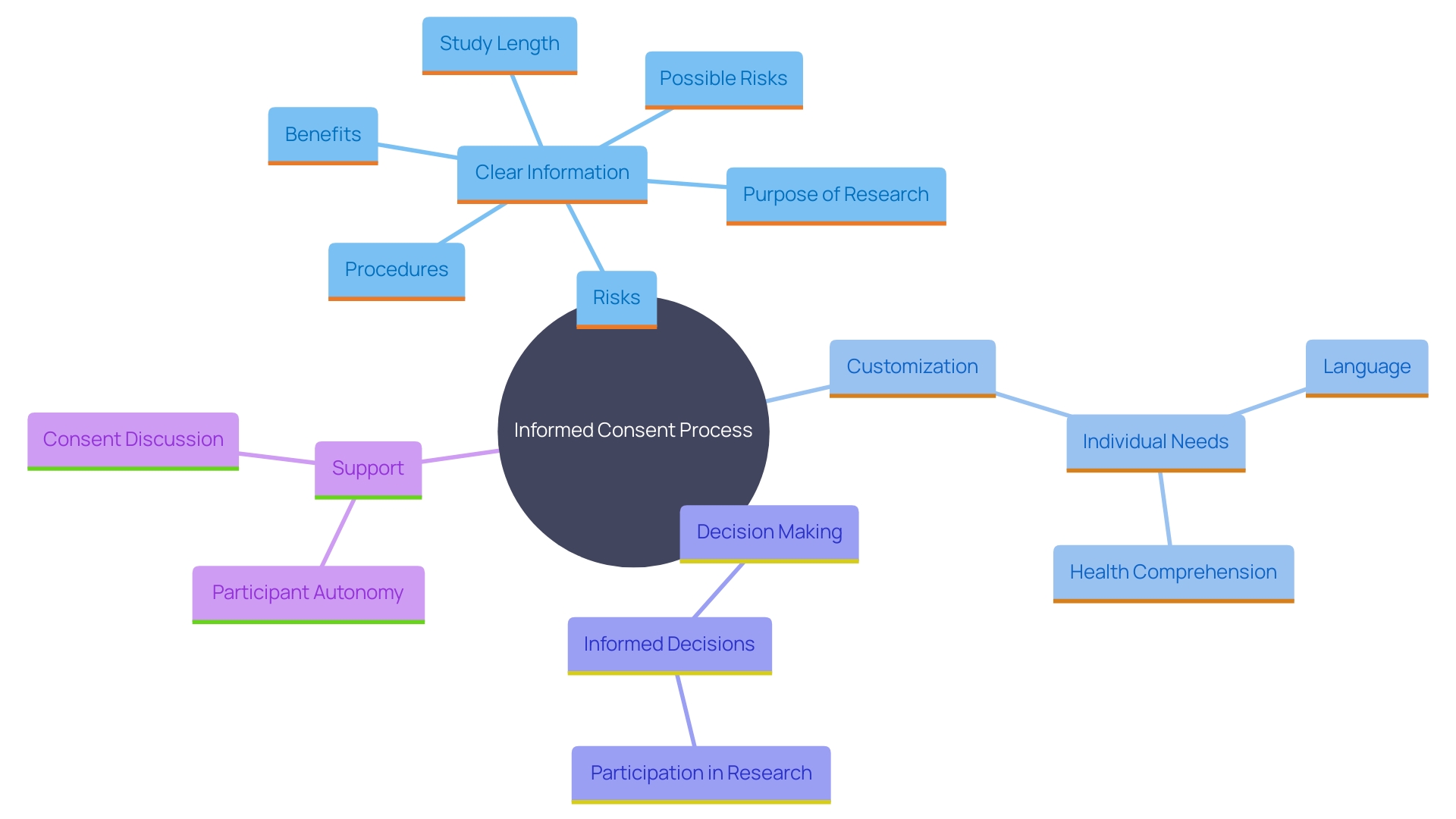
should be presented in a at the beginning of the informed agreement document. This includes the purpose of the research, possible risks and benefits, and the study’s length and procedures. The inclusion of key information can be a valuable resource for current study individuals and support the agreement discussion between the investigator and potential subjects.
The National Organization for Rare Disorders (NORD) has praised draft guidance that permits creative awareness methods, such as videos, to make the process more accessible. It is essential to customize consent to individuals’ unique needs, considering factors like language barriers, hearing or vision impairments, developmental delays, and health literacy competencies. By utilizing simple phrases, plain language principles, and organizational tools like the bubble format, researchers can significantly enhance participants’ comprehension of the information.
Statement that the Study Involves Research
Participants must be clearly informed that their involvement is part of a study. This distinction is crucial for , as it differentiates investigative activities from standard medical treatments. According to the updated , it is vital that individuals understand the purpose of the research, the potential risks and benefits, and the procedures involved. This clarity ensures that participants can make a about their participation.
The significance of knowledgeable agreement is profoundly embedded in ethical structures like the and the , which highlight regard for people and their right to make choices for themselves. These documents have been essential in forming human study ethics, highlighting the importance of understanding agreement at both the beginning and during the study process.
Recent advancements highlight the need for presenting key information in a concise and understandable manner. The draft guidance suggests incorporating subjects like the aim of the research, anticipated length, and possible risks and advantages right at the start of the approval document. This method promotes a clearer comprehension for those involved, assisting them in considering their choice to engage in the study.
Furthermore, creative methods for , including the use of videos and other easily accessible formats, are promoted to address the varied requirements of individuals. This flexibility is crucial for making sure that all potential contributors, regardless of language obstacles or sensory challenges, can fully understand the study they are being invited to join.
By following these principles and guidelines, scholars can maintain , safeguard individuals’ rights, and improve the validity and reliability of their findings.
Explanation of the Purposes of the Research
A comprehensive explanation of the is crucial for ensuring participants understand the significance of the investigation and how their involvement may contribute to broader . Effective communication of these objectives fosters an environment of trust and transparency, which is foundational in . Rooted in frameworks like the Declaration of Helsinki and the Belmont Report, emphasizes respect for individuals and their right to self-determination. This method corresponds with the moral standards that direct , where individuals are not simply subjects but active contributors whose involvement can lead to significant results.
Patient and public involvement (PPI) in studies underscores the importance of collaborative partnerships between researchers and participants. These partnerships are vital in designing, conducting, and disseminating studies that address real-world issues. For instance, the Multi-Regional Center at Brigham and Women’s Hospital and Harvard highlights the evolving nature of these partnerships, ensuring that investigations are both relevant and ethically sound.
Statistics indicate that greatly affect and results, making it essential that those involved are fully informed about the study’s objectives. This openness not only improves the of studies but also guarantees that contributors understand how their involvement can result in progress in medical understanding and patient results. Engaging participants through clear communication and shared decision-making reinforces their role in the research process, ultimately leading to more effective and impactful scientific discoveries.
Expected Duration of Participation
Participants must be thoroughly informed about the , encompassing the length of individual sessions and any follow-up periods. , as highlighted by the and the , necessitates clarity and transparency from the onset. The recently updated Guidance by the Multi-Regional Center emphasizes presenting key information in a manner that enhances comprehension. This encompasses information regarding the study’s length, which is essential for individuals to make about their engagement. ‘Ensuring individuals are aware of the duration not only respects their autonomy but also aligns with ethical standards that underscore the importance of throughout the research process.’.
Description of Procedures to be Followed
A detailed account of the procedures individuals will undergo is crucial. This encompasses any interventions, assessments, or tests required during the study. For instance, individuals might need to complete at various stages, such as baseline urinary and erectile function assessments, which are directly added to their . ‘The clarity in defining these steps ensures that individuals are well-informed about the specific aspects of the study, which is important for maintaining and compliance with guidelines.’. As highlighted in recent updates to the International Council for Harmonization (ICH) E6 guidelines, the reliability of depends on the and the integrity of procedures such as randomization and dosing escalation.
Description of Any Reasonably Foreseeable Risks or Discomforts
It is crucial for researchers to provide individuals with clear, concise information about any or discomforts associated with the study. This openness is essential for , enabling individuals to make choices based on a comprehensive understanding of the possible effects. As stated by the National Organization for Rare Disorders (NORD), the consent process should be available and customized to address the of individuals, taking into account elements like language obstacles and . By doing so, researchers can ensure that individuals are fully aware of the risks and benefits, thereby supporting their autonomy and right to make educated decisions.
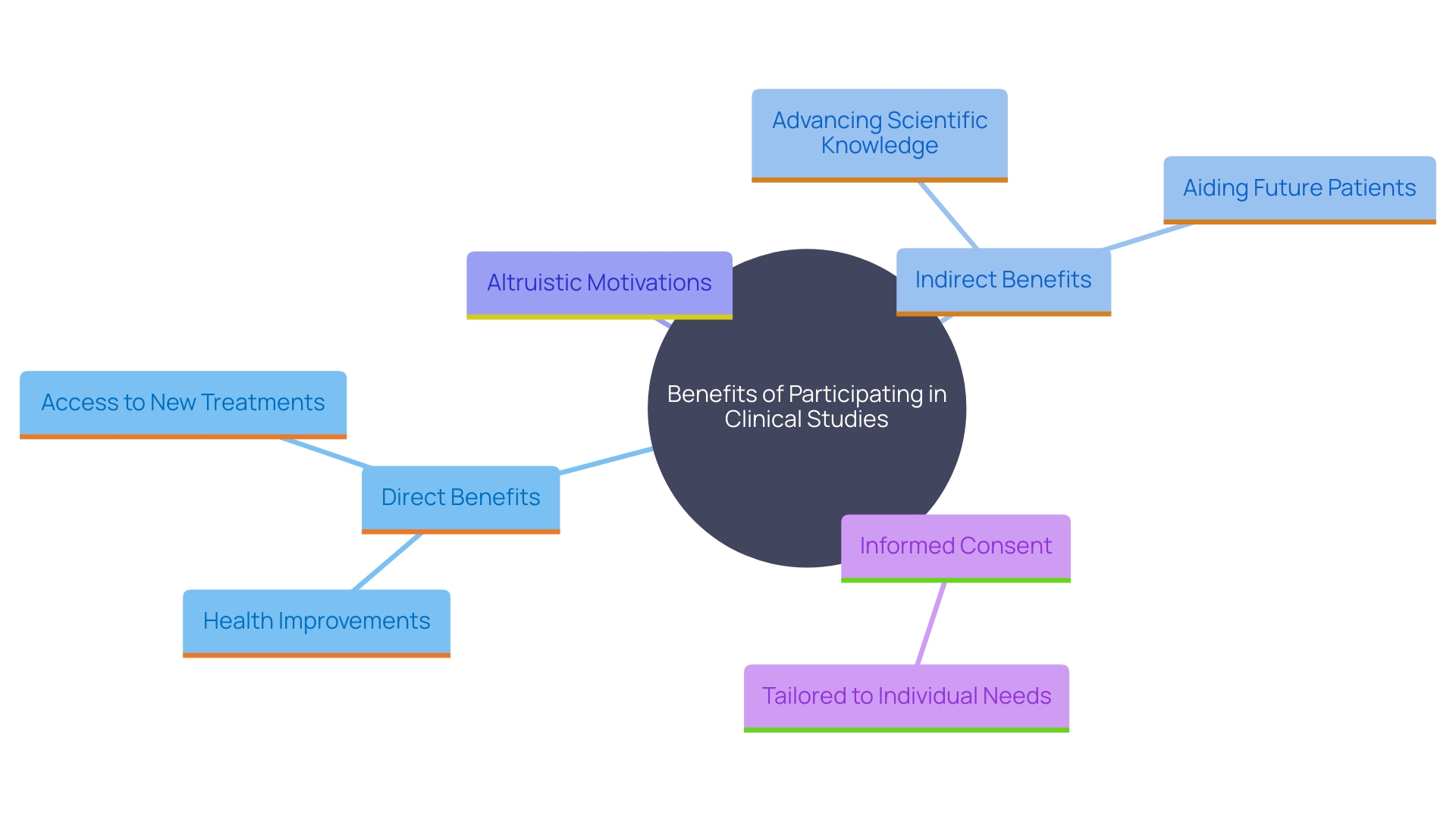
Description of Any Benefits to the Subject or Others
Participants should be completely aware of both direct and indirect advantages they may receive from their participation in . Direct benefits often include or access to new treatments. Indirect benefits encompass the broader impact their participation can have on advancing and potentially aiding future patients. Based on the 2023 Perceptions and Insights Study by the Center for Information and Study on Clinical Research Participation (CISCRP), a considerable number of individuals take part in studies mainly to assist in . This altruistic motivation underscores the importance of clearly communicating the potential of . As highlighted by the National Organization for Rare Disorders (NORD), knowledgeable agreement must be provided in accessible formats customized to individuals’ specific needs, ensuring they completely comprehend the risks and benefits of involvement. This thorough method for not only honors individuals’ independence but also promotes a clear and principled setting for inquiry.
Disclosure of Appropriate Alternative Procedures or Courses of Treatment
Informed agreement must include information about any or procedures available outside the research study. This transparency ensures individuals can make well-informed decisions about their involvement. As highlighted by the National Organization for Rare Disorders (NORD), presenting in accessible manners—such as through videos—can significantly enhance individuals’ understanding of their options. Furthermore, the draft guidance released by the FDA and OHRP highlights the significance of conveying clearly and succinctly, including the aim of the study, , as well as the duration and methods of the investigation. Incorporating this practice not only aligns with ethical standards but also facilitates , empowering individuals to make choices that best suit their needs.
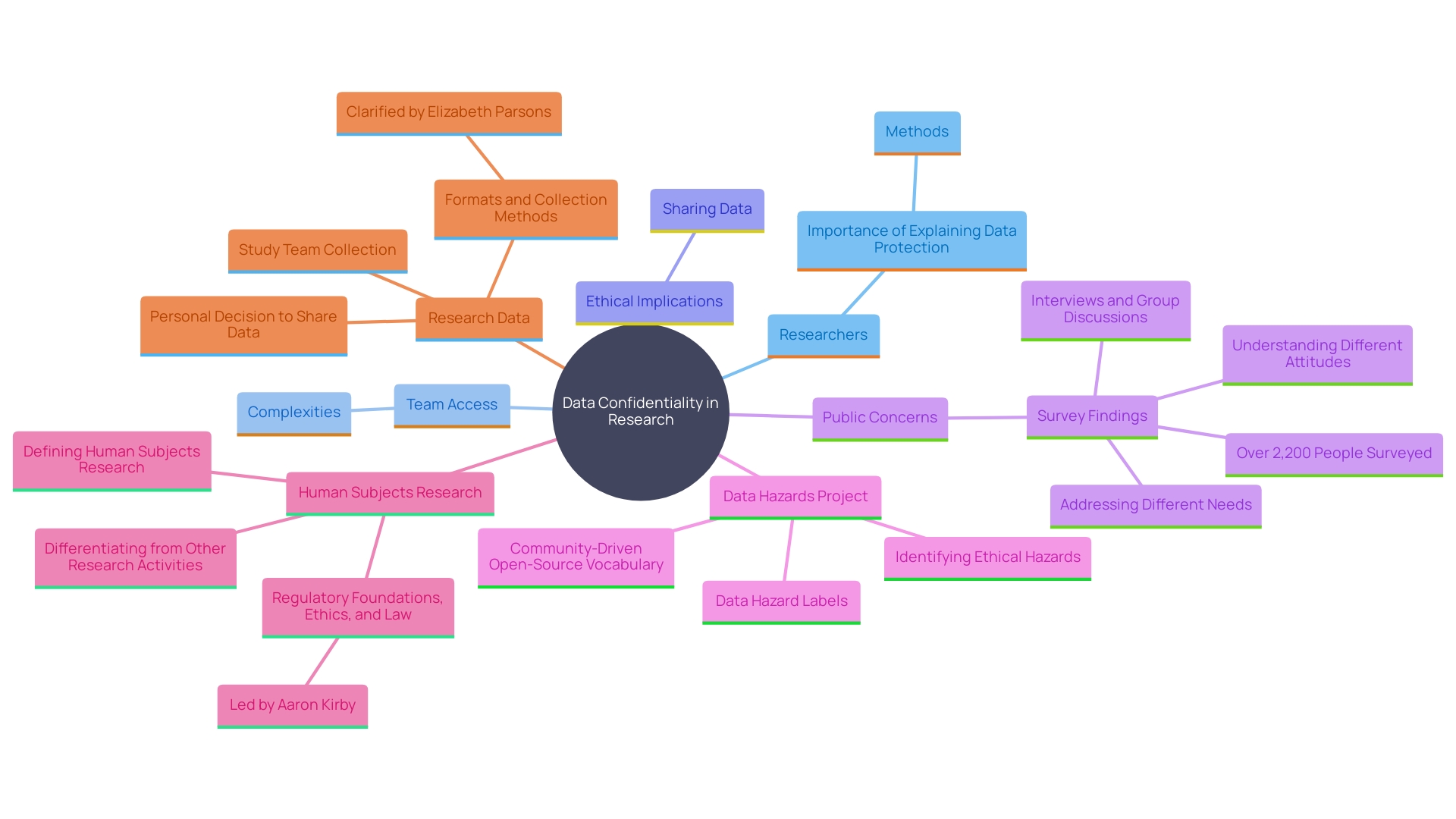
Statement Describing the Extent of Confidentiality of Records
Participants require assurance about the and data collected during the study. Researchers must clearly explain the methods for data storage, access limitations, and protection measures. As stated by Joe Zurba, head of security and regulatory adherence at Harvard Medical School, a study includes various team members, which can complicate it for individuals to grasp who will have access to their information. Therefore, it’s crucial to outline the protocols for . ‘Elizabeth Parsons, IRB administrator for the Harvard University Area IRB, emphasizes that sharing data can enhance public health but deciding to share private information is a personal choice.’. Making sure that individuals are aware of how their information will be protected can help foster trust and encourage . This is particularly important given the , as highlighted by a recent Pew Research Center survey indicating increased unease about how personal data is used by companies and the government.
Beyond the basic elements of , there are additional considerations that may be relevant depending on the study’s context. These include , which acknowledges the time and effort of those taking part. is another critical aspect, ensuring that individuals receive necessary care if they experience any adverse effects. Moreover, the return of to those involved is gaining attention, emphasizing the importance of transparency and respect for the individuals concerned. The and the Belmont Report have long emphasized these , and recent updates by the Multi-Regional Center provide further guidance on these matters. The National Organization for Rare Disorders (NORD) advocates for creative methods to secure consent, such as employing videos to enhance the process’s accessibility, emphasizing the necessity to customize consent documents to the distinct requirements of participants.
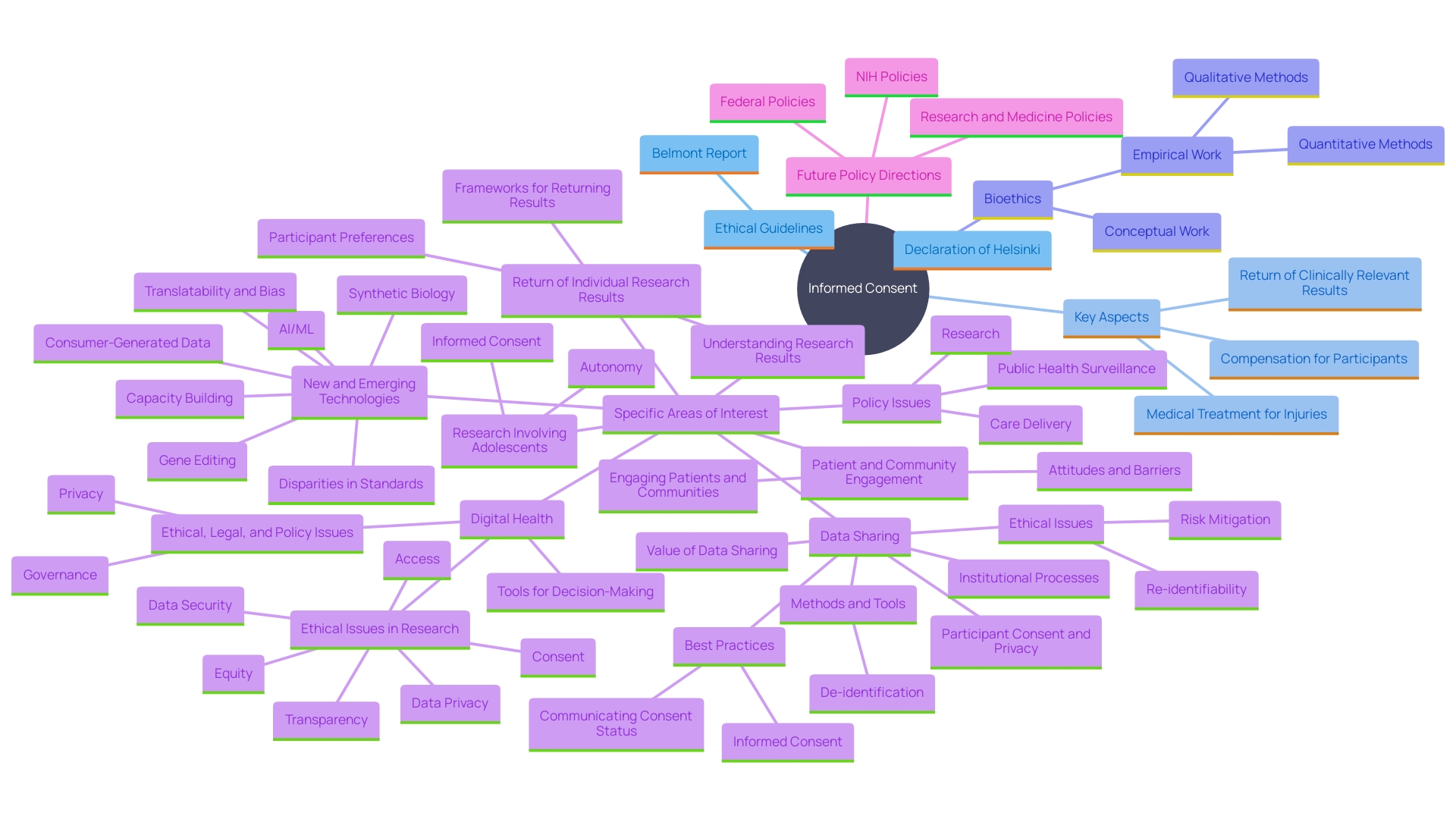
Participants must be fully informed about any compensation they will receive for their participation, as well as the available in case of research-related injuries. This is vital not only for ethical reasons but also to adhere to such as those outlined in the , which has been a cornerstone of medical ethics for decades. Recent surveys indicate that public trust in clinical studies is significantly influenced by transparency regarding . For example, a national survey commissioned by Research!America and ACRO found that 77% of respondents prefer receiving information about from their healthcare providers, emphasizing the need for clear and thorough communication. Furthermore, real-life accounts like that of Barbara, who discovered an undiagnosed heart condition through participation in a clinical trial, highlight the importance of informing participants about available medical follow-ups. These measures are essential for maintaining and fostering trust in the investigation process.

Supplying is a crucial element of consent in healthcare and studies. This transparency ensures that participants can ask questions or voice concerns about the study before and during their participation. According to the Declaration of Helsinki, established by the World Medical Association, respect for the individual and their right to self-determination are paramount. This approach is supported by the ‘s , which are foundational to ethical considerations in .
In practice, including contact information facilitates patient and public involvement and engagement, which are critical for the success and ethical integrity of . It enables collaborative partnerships and shared decision-making, ensuring that the study is pertinent, well-executed, and that results are communicated effectively to those who will use them to make . For example, in the United Kingdom, in research often includes prioritizing research questions, design, delivery, oversight, analysis, and dissemination.
A case study from the Bangladesh Hypertension Control Initiative (BHCI) emphasizes the significance of . Despite the initiative’s aim to control hypertension, it faced significant challenges with follow-up, as 44% of registered patients had not visited the clinic for three months or more by the end of 2021. Offering clear and accessible contact information could assist in resolving such issues, ensuring individuals remain engaged and well-informed throughout the study.
Moreover, the Multi-Regional Center of Brigham and Women’s Hospital and Harvard emphasizes the necessity of returning individual data and results to those involved, further underlining the importance of open lines of communication. This practice not only honors the participants’ contributions but also improves the validity and generalizability of the findings by ensuring that participants stay engaged and informed throughout the study duration.
Voluntary Participation Statement
A clear affirmation that is fundamental in any study. that they have the or withdraw from the study at any point, without suffering any penalties. This principle is deeply rooted in the established by the , which emphasizes respect for individuals and their autonomy in medical studies. It is essential for participants to feel enabled to make choices about their involvement freely and without coercion, ensuring the of the process.
Procedures for Withdrawal from the Study
Participants must be completely educated about the procedures for withdrawing from a study. This includes detailed steps they need to take and assurances that their decision will not impact their . The highlights the importance of knowledgeable agreement throughout the research process, emphasizing respect for the individual’s right to self-determination. The Belmont Report further reinforces this by advocating for the , beneficence, and justice. Ensuring clear communication about withdrawal procedures is not only an but also a legal one, as highlighted by the Multi-Regional Center’s updated guidance. Given the complexity and demanding nature of —where individuals in , for instance, may attend up to 20 visits and undergo numerous procedures—their without any repercussions is paramount. Addressing this explicitly in the awareness agreement process supports the ethical framework and respects the substantial investment of time, energy, and resources made by participants.
Ensuring Understandability and Voluntariness
Making certain that the promotes is essential. This involves presenting in a clear, concise manner, emphasizing the purpose of the research, its , and the procedures involved. Utilizing lay language, visual aids, and allowing ample time for questions are . As stated by the National Organization for Rare Disorders (NORD), knowledgeable agreement must be available and customized to personal requirements, taking into account elements such as language obstacles and health understanding. This method not only promotes improved comprehension but also tackles issues raised by stakeholders concerning the complexity and legalistic character of current , which often exceed twenty pages and obstruct .
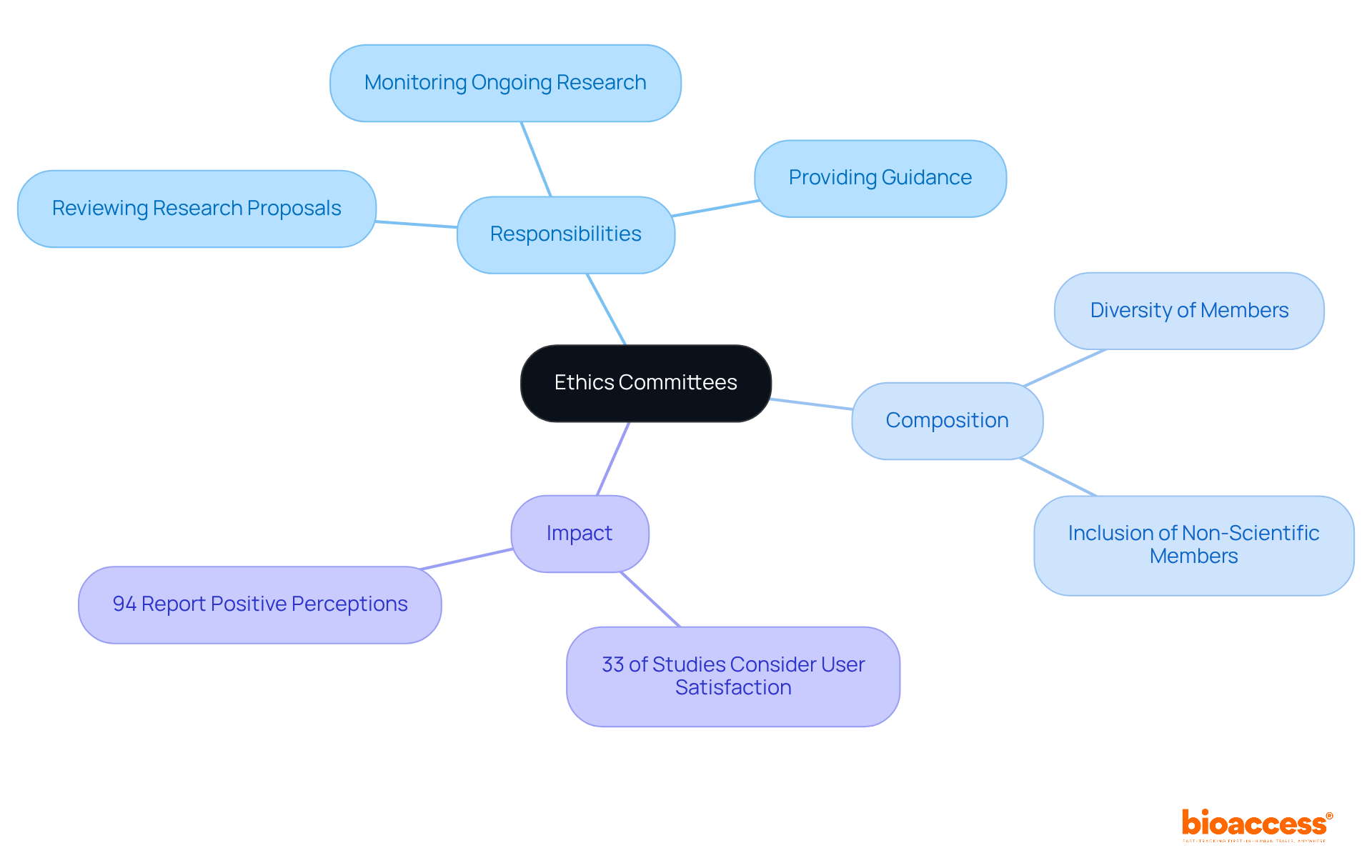
The Role of the Institutional Review Board (IRB)
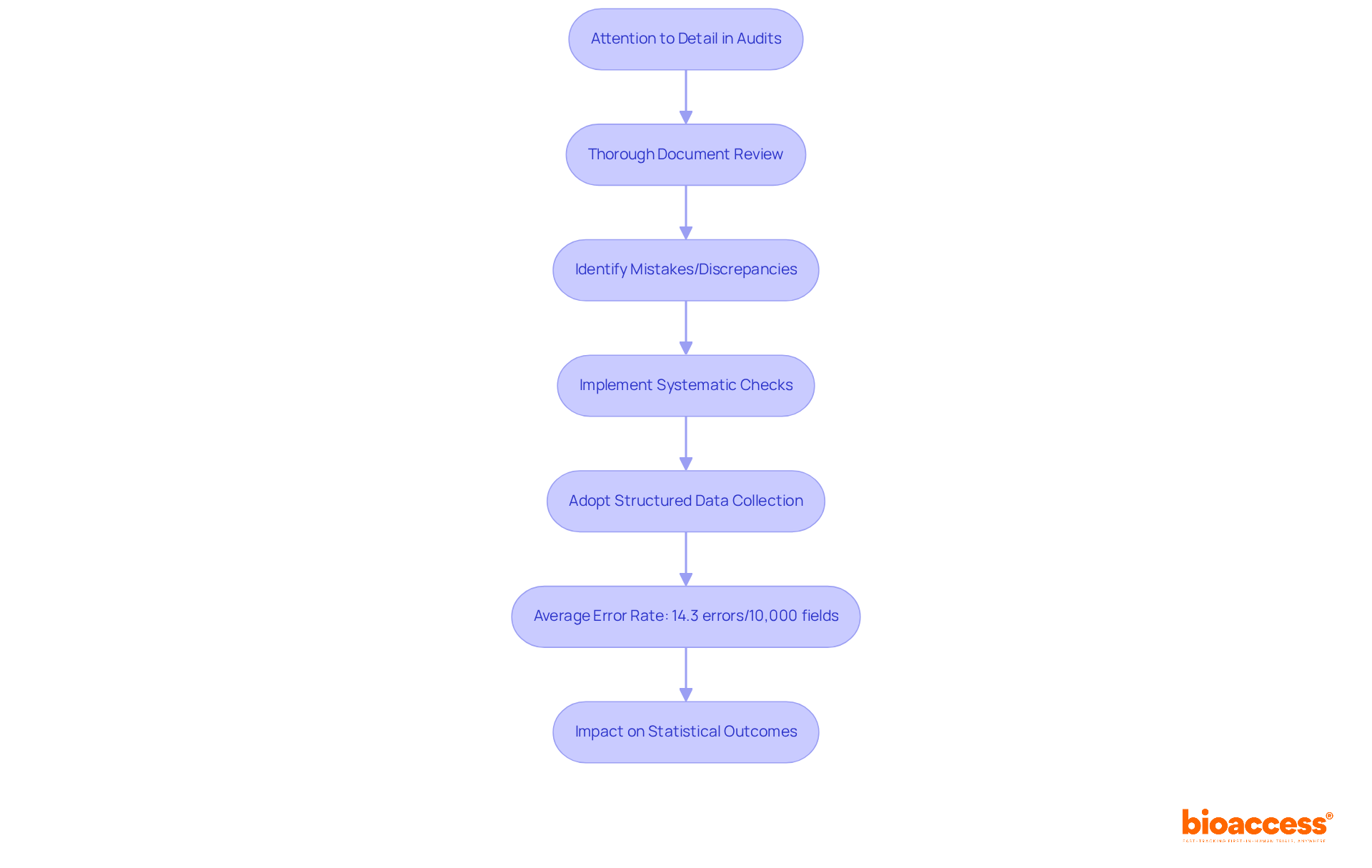
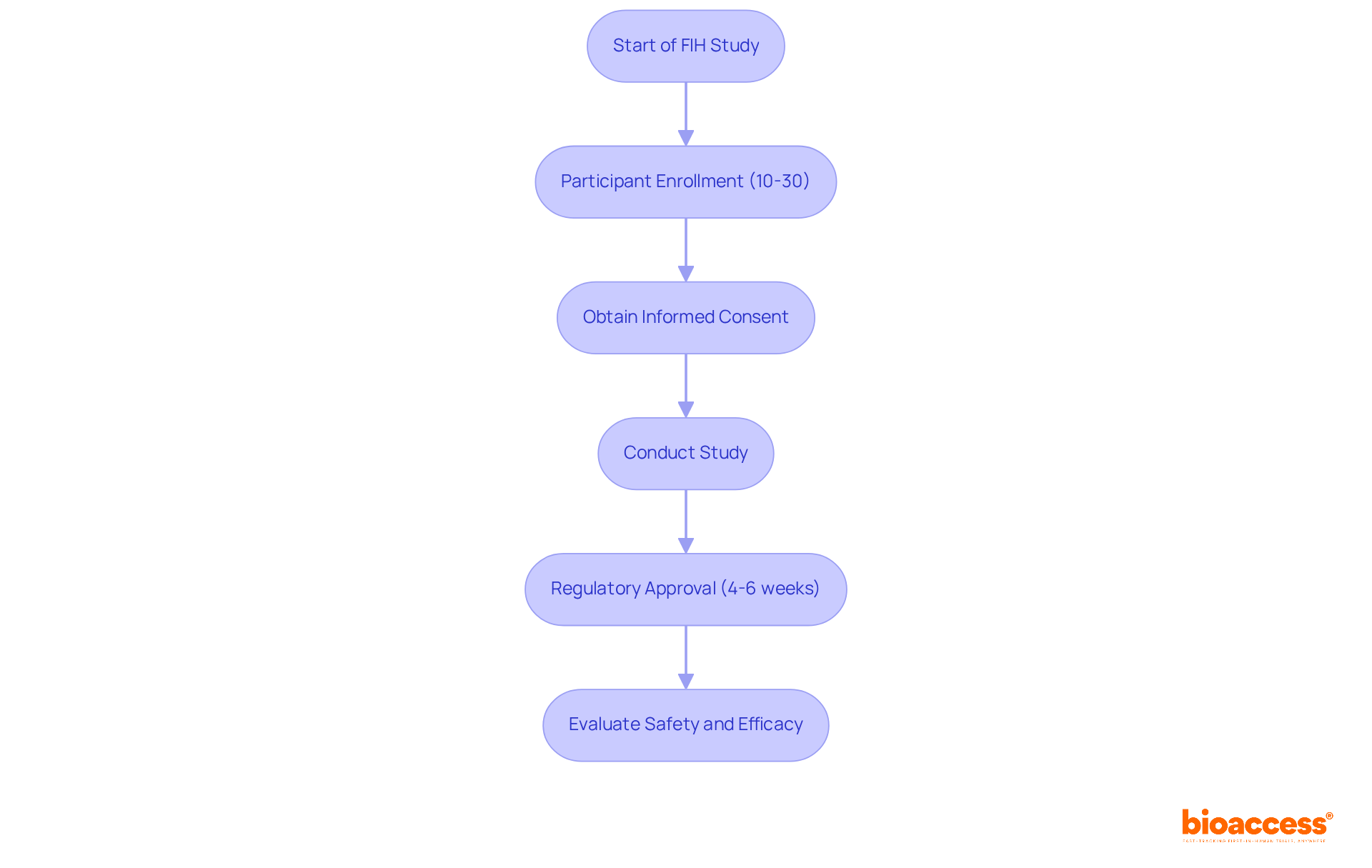
The is crucial in ensuring the involving . To protect participants’ rights and welfare, IRBs carefully examine , which include detailed descriptions of the investigation’s purpose, procedures, risks, benefits, and approval documents. This rigorous process, codified in the National Research Act of 1974, aims to prevent unethical practices in studies that have marred history, such as the Tuskegee Syphilis Study. In this egregious case, Black men were denied treatment for syphilis for 40 years, leading to over 100 deaths.
IRB reviews can take weeks or even months, as proposals are often sent back for revisions to meet ethical standards. As Dr. Steven Kritz, a retired physician and current IRB Chair, emphasizes, the inclusion of consent is non-negotiable in any research involving pharmaceutical products. This steadfast commitment ensures that participants are fully informed about the study, including its purpose, potential risks, benefits, and duration. The IRB’s role is not just about ticking boxes but about fostering an environment where are paramount, ultimately contributing to .
 review process for studies involving human subjects, highlighting key components such as protocol examination, revisions, and the importance of informed consent. This flowchart illustrates the steps involved in the Institutional Review Board (IRB) review process for studies involving human subjects, highlighting key components such as protocol examination, revisions, and the importance of informed consent.](https://tely.blob.core.windows.net/telyai/this-flowchart-illustrates-the-steps-involved-in-the-institutional-review-board-irb-review-process-for-studies-involving-human-subjects-highlighting-key-components-such-as-protocol-examination-revisions-and-the-importance-of-informed-consent.jpg)
Conclusion
Informed consent is essential for ethical research involving human subjects, serving as a safeguard for participants’ autonomy and ensuring they are well-informed about the studies they engage in. This process fosters trust between researchers and participants, while also enhancing the integrity of the research. The necessity of informed consent is underscored by historical unethical practices, which have led to the establishment of regulatory bodies like Institutional Review Boards (IRBs) that oversee compliance with ethical standards.
Key components of informed consent include clear communication regarding the study’s purpose, expected duration, procedures, risks, and potential benefits. The growing complexity of consent documents poses challenges, particularly for underserved populations, highlighting the need for accessible and comprehensible materials. Innovations such as multimedia presentations can aid in bridging these gaps, ensuring participants have the information required to make informed decisions about their involvement.
Ultimately, informed consent is not merely a legal formality; it is a fundamental ethical obligation that respects the rights of individuals. By prioritizing transparency and understanding, researchers can uphold ethical standards, maintain public trust, and contribute to the advancement of medical knowledge. Ensuring that participants are fully informed encourages meaningful engagement, which is vital for the success of clinical trials and the broader research landscape.
Ensure your research meets ethical standards and enhances participant trust—contact bioaccess™ today for expert guidance on informed consent processes!
Frequently Asked Questions
What is informed consent and why is it important?
Informed consent is a process that ensures participants in clinical studies are fully informed about the study they are involved in, including potential risks and benefits. It is essential for upholding individual autonomy and fosters trust between researchers and participants while ensuring ethical and legal compliance.
What are the historical contexts that led to the establishment of informed consent regulations?
Historical unethical practices, such as those seen during World War II and the Tuskegee Syphilis Study, prompted the creation of regulatory safeguards like Institutional Review Boards (IRBs) to protect participants’ rights and dignity in research.
What are Institutional Review Boards (IRBs)?
IRBs are impartial entities that review study protocols to ensure ethical conduct, compliance with federal regulations, and the protection of participants’ rights. Their role is crucial in maintaining the integrity of clinical research.
What should be included in informed consent documents?
Informed consent documents should include: a clear statement indicating that the study involves investigation, an explanation of the study’s purpose, the expected duration of participation, a description of the procedures involved, and information about potential risks and benefits.
Why have informed consent documents become complex?
These documents have grown increasingly intricate, often exceeding twenty pages, due to legal requirements and the need to include extensive information. This complexity can hinder understanding, particularly among underserved populations.
How can researchers improve participant comprehension of informed consent?
Researchers can enhance comprehension by presenting essential information in a clear and concise manner at the beginning of the document, using plain language and simple phrases, incorporating creative methods like videos for better engagement, and customizing consent documents to meet individual needs, such as language and health literacy levels.
What is the significance of understanding the study’s objectives?
Clear communication of the study’s objectives is essential for fostering trust and transparency among participants, as it highlights the significance of their involvement in broader scientific advancements.
What types of benefits should participants be informed about?
Participants should be made aware of both direct benefits (e.g., health improvements or access to new treatments) and indirect benefits (e.g., contributions to scientific knowledge and future patient care).
What ethical considerations must researchers address regarding participant data?
Researchers must ensure participants are informed about the confidentiality of their personal information, data storage and access limitations, and protection measures for their data. Transparency in these areas is vital for fostering trust and ethical research practices.
Are participants allowed to withdraw from a study?
Yes, participants have the right to decline or withdraw from the study at any point without facing penalties. This right is fundamental to maintaining ethical standards and respecting individual autonomy.
What should participants know about compensation for their involvement?
Participants should be informed about any compensation they will receive and medical treatments available for research-related injuries. Transparency in these areas can significantly influence public trust in clinical studies.
How can researchers ensure participants can communicate their concerns?
Providing contact information for researchers or ethics committee members is essential for encouraging participants to ask questions or voice concerns about the study, thereby promoting transparency and engagement.
What is the role of ethical frameworks like the Declaration of Helsinki and the Belmont Report in informed consent?
These frameworks emphasize respect for individuals, their autonomy, and the importance of informed decision-making throughout the research process, reinforcing the ethical considerations of informed consent.
Why is it important to return clinically relevant results to participants?
Returning results to participants not only respects their contributions but also enhances the validity of the research findings by keeping participants engaged and informed throughout the study.
List of Sources
- The Importance of Informed Consent in Research
- mailchi.mp (https://mailchi.mp/jhu/scienceethics1-2519855)
- raps.org (https://raps.org/news-and-articles/news-articles/2024/5/stakeholders-welcome-fda-s-informed-consent-‘key-i?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- automatedresearch.org (https://automatedresearch.org/news/news-briefing-1-12-july-2024)
- frontiersin.org (https://frontiersin.org/articles/10.3389/fgene.2023.1261623/full?utm_source=S-TWT&utm_medium=SNET&utm_campaign=ECO_FGENE_XXXXXXXX_auto-dlvrit)
- jamanetwork.com (https://jamanetwork.com/journals/jama/article-abstract/2820280)
- researchmethodscommunity.sagepub.com (https://researchmethodscommunity.sagepub.com/blog/ethics-challenged-world)
- bioethicstoday.org (https://bioethicstoday.org/blog/academic-and-private-partnership-to-improve-informed-consent-forms-using-a-data-driven-approach)
- The rewards of being a research professional (https://ctsi.umn.edu/news/rewards-being-research-professional)
- catalyst.harvard.edu (https://catalyst.harvard.edu/regulatory/video-series-participant-data?utm_source=Twitter&utm_medium=Social%20&utm_campaign=Participant%20Data%20Videos%20)
- umass.edu (https://umass.edu/research/compliance/human-subjects-irb/irb)
- Basic Elements of Informed Consent
- bioethicstoday.org (https://bioethicstoday.org/blog/academic-and-private-partnership-to-improve-informed-consent-forms-using-a-data-driven-approach)
- nam.edu (https://nam.edu/regenerative-medicine-case-study-for-understanding-and-anticipating-emerging-science-and-technology)
- nam.edu (https://nam.edu/telehealth-and-mobile-health-case-study-for-understanding-and-anticipating-emerging-science-and-technology)
- federalregister.gov (https://federalregister.gov/documents/2024/03/01/2024-04377/key-information-and-facilitating-understanding-in-informed-consent-draft-guidance-for-sponsors)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- us.sagepub.com (https://us.sagepub.com/en-us/nam/publication-ethics-and-research-integrity-policy-guidelines-for-authors)
- disabilityhealth.medicine.umich.edu (https://disabilityhealth.medicine.umich.edu/positionality-statements-brief)
- catalyst.harvard.edu (https://catalyst.harvard.edu/regulatory/video-series-participant-data?utm_source=Twitter&utm_medium=Social%20&utm_campaign=Participant%20Data%20Videos%20)
- mailchi.mp (https://mailchi.mp/jhu/scienceethics1-2519855)
- raps.org (https://raps.org/news-and-articles/news-articles/2024/5/stakeholders-welcome-fda-s-informed-consent-‘key-i?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- catalyst.harvard.edu (https://catalyst.harvard.edu/consent-template-instructions-for-simple-minimal-risk-sber-studies?utm_source=Twitter&utm_medium=Social%20&utm_campaign=SBER%20Template)
- urban.org (https://urban.org/research/publication/do-no-harm-guide-crafting-equitable-data-narratives?utm_medium=twitter&utm_source=urban_social)
- Statement that the Study Involves Research
- nam.edu (https://nam.edu/regenerative-medicine-case-study-for-understanding-and-anticipating-emerging-science-and-technology)
- nature.com (https://nature.com/articles/s41746-024-01205-6)
- clinicalleader.com (https://clinicalleader.com/doc/returning-individual-trial-data-to-patients-how-and-why-it-needs-to-happen-0001)
- bmcmedresmethodol.biomedcentral.com (https://bmcmedresmethodol.biomedcentral.com/articles/10.1186/s12874-024-02323-1)
- researchamerica.org (https://researchamerica.org/press-releases-statements/large-majority-of-americans-say-consideration-of-clinical-trial-participation-should-be-a-part-of-regular-health-care)
- clinicaltrialsarena.com (https://clinicaltrialsarena.com/news/oct-europe-data-governence-clarity)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- mailchi.mp (https://mailchi.mp/jhu/bioethics-in-the-academic-literature-2520524)
- raps.org (https://raps.org/news-and-articles/news-articles/2024/5/stakeholders-welcome-fda-s-informed-consent-‘key-i?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- nature.com (https://nature.com/articles/s41591-023-02665-1)
- bioethicstoday.org (https://bioethicstoday.org/blog/academic-and-private-partnership-to-improve-informed-consent-forms-using-a-data-driven-approach)
- catalyst.harvard.edu (https://catalyst.harvard.edu/regulatory/video-series-participant-data?utm_source=Twitter&utm_medium=Social%20&utm_campaign=Participant%20Data%20Videos%20)
- Explanation of the Purposes of the Research
- psychnews.psychiatryonline.org (https://psychnews.psychiatryonline.org/toc/pn/59/07)
- mailchi.mp (https://mailchi.mp/jhu/bioethicsbulletin-2519368-8a3ajiivxd-2520380)
- nature.com (https://nature.com/articles/s41591-023-02588-x|)
- plantae.org (https://plantae.org/science-communication-is-an-important-skill-for-scientists)
- ssrc.org (https://ssrc.org/mercury-project/2024/06/09/seven-tips-from-experts-on-communicating-your-research)
- scientia.global (https://scientia.global/mr-anthony-keyes-understanding-and-improving-clinical-trial-compliance)
- reports.statnews.com (https://reports.statnews.com/collections/30-39-reports-collection/products/stat-guide-interpreting-clinical-trials-update?variant=40308516356199&utm_medium=social&utm_source=twitter&utm_campaign=twitter_organic)
- jamanetwork.com (https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2811814)
- aakinshin.net (https://aakinshin.net/posts/cs-social-survey)
- bmjopen.bmj.com (https://bmjopen.bmj.com/content/14/6/e088737)
- arxiv.org (https://arxiv.org/abs/2310.17506)
- researchinvolvement.biomedcentral.com (https://researchinvolvement.biomedcentral.com/articles/10.1186/s40900-023-00530-6)
- clinicalleader.com (https://clinicalleader.com/doc/returning-individual-trial-data-to-patients-how-and-why-it-needs-to-happen-0001)
- bmcmedresmethodol.biomedcentral.com (https://bmcmedresmethodol.biomedcentral.com/articles/10.1186/s12874-024-02323-1)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- bmj.com (https://bmj.com/content/386/bmj.q1406?utm_campaign=usage&utm_content=tbmj_sprout&utm_id=BMJ005&utm_medium=social&utm_source=twitter)
- Expected Duration of Participation
- clinicalleader.com (https://clinicalleader.com/doc/returning-individual-trial-data-to-patients-how-and-why-it-needs-to-happen-0001)
- nature.com (https://nature.com/articles/s41591-023-02665-1)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- researchinvolvement.biomedcentral.com (https://researchinvolvement.biomedcentral.com/articles/10.1186/s40900-023-00530-6)
- cloudresearch.com (https://cloudresearch.com/resources/seminars)
- niaid.nih.gov (https://niaid.nih.gov/grants-contracts/delayed-start-and-delayed-onset-human-subjects-research)
- web.fibion.com (https://web.fibion.com/articles/effective-esm-prompt-crafting-tips-examples)
- raps.org (https://raps.org/news-and-articles/news-articles/2024/5/stakeholders-welcome-fda-s-informed-consent-‘key-i?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- innotechtoday.com (https://innotechtoday.com/how-tech-could-reshape-the-clinical-trial-process)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- Description of Procedures to be Followed
- hccr.ccrhindia.in (https://hccr.ccrhindia.in/en)
- ncbi.nlm.nih.gov (https://ncbi.nlm.nih.gov/pmc/articles/PMC2656491)
- nejm.org (https://nejm.org/doi/full/10.1056/NEJMoa2309149)
- nexus.od.nih.gov (https://nexus.od.nih.gov/all/2024/08/16/nih-all-about-grants-podcast-considering-language-access-for-participants-in-nih-supported-clinical-research)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- nature.com (https://nature.com/articles/s41591-023-02665-1)
- catalyst.harvard.edu (https://catalyst.harvard.edu/regulatory/video-series-participant-data?utm_source=Twitter&utm_medium=Social%20&utm_campaign=Participant%20Data%20Videos%20)
- scientia.global (https://scientia.global/mr-anthony-keyes-understanding-and-improving-clinical-trial-compliance)
- bmj.com (https://bmj.com/content/387/bmj-2023-077418?utm_campaign=usage&utm_content=tbmj_sprout&utm_id=BMJ005&utm_medium=social&utm_source=twitter)
- pharmafile.com (https://pharmafile.com/appointments/richmond-promotes-new-director-of-communications-and-participant-engagement)
- clinicaltrialsarena.com (https://clinicaltrialsarena.com/news/oct-europe-data-governence-clarity)
- web.fibion.com (https://web.fibion.com/articles/effective-esm-prompt-crafting-tips-examples)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- Description of Any Reasonably Foreseeable Risks or Discomforts
- clinicalleader.com (https://clinicalleader.com/doc/returning-individual-trial-data-to-patients-how-and-why-it-needs-to-happen-0001)
- catalyst.harvard.edu (https://catalyst.harvard.edu/regulatory/video-series-participant-data?utm_source=Twitter&utm_medium=Social%20&utm_campaign=Participant%20Data%20Videos%20)
- nejm.org (https://nejm.org/doi/full/10.1056/NEJMimc2306268)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- raps.org (https://raps.org/news-and-articles/news-articles/2024/5/stakeholders-welcome-fda-s-informed-consent-‘key-i?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- rethinkingclinicaltrials.org (https://rethinkingclinicaltrials.org/news/grand-rounds-february-2-2024-strategies-for-improving-public-understanding-of-fda-and-the-products-it-regulateswhy-should-we-care-and-what-might-we-do-susan-c-winckler-rph-esq)
- researchamerica.org (https://researchamerica.org/press-releases-statements/large-majority-of-americans-say-consideration-of-clinical-trial-participation-should-be-a-part-of-regular-health-care)
- sagebionetworks.pubpub.org (https://sagebionetworks.pubpub.org/pub/fvderdfq/release/1)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- Description of Any Benefits to the Subject or Others
- raps.org (https://raps.org/news-and-articles/news-articles/2024/5/stakeholders-welcome-fda-s-informed-consent-‘key-i?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- The rewards of being a research professional (https://ctsi.umn.edu/news/rewards-being-research-professional)
- nature.com (https://nature.com/articles/d41573-024-00107-2)
- gh.bmj.com (https://gh.bmj.com/content/9/9/e014971.full)
- catalyst.harvard.edu (https://catalyst.harvard.edu/regulatory/video-series-participant-data?utm_source=Twitter&utm_medium=Social%20&utm_campaign=Participant%20Data%20Videos%20)
- nam.edu (https://nam.edu/regenerative-medicine-case-study-for-understanding-and-anticipating-emerging-science-and-technology)
- abpi.org.uk (https://abpi.org.uk/partnerships/working-with-the-nhs/nhs-partnership-case-studies-library-key-aims)
- researchinvolvement.biomedcentral.com (https://researchinvolvement.biomedcentral.com/articles/10.1186/s40900-023-00530-6)
- The rewards of being a research professional (https://ctsi.umn.edu/news/rewards-being-research-professional)
- bmcmedresmethodol.biomedcentral.com (https://bmcmedresmethodol.biomedcentral.com/articles/10.1186/s12874-024-02323-1)
- researchamerica.org (https://researchamerica.org/press-releases-statements/large-majority-of-americans-say-consideration-of-clinical-trial-participation-should-be-a-part-of-regular-health-care)
- Disclosure of Appropriate Alternative Procedures or Courses of Treatment
- transparimed.org (https://transparimed.org/single-post/outcome-switching-research-misconduct)
- federalregister.gov (https://federalregister.gov/documents/2024/03/01/2024-04377/key-information-and-facilitating-understanding-in-informed-consent-draft-guidance-for-sponsors)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- raps.org (https://raps.org/news-and-articles/news-articles/2024/5/stakeholders-welcome-fda-s-informed-consent-‘key-i?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- nam.edu (https://nam.edu/regenerative-medicine-case-study-for-understanding-and-anticipating-emerging-science-and-technology)
- bmj.com (https://bmj.com/content/387/bmj-2023-077418?utm_campaign=usage&utm_content=tbmj_sprout&utm_id=BMJ005&utm_medium=social&utm_source=twitter)
- jamanetwork.com (https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2811814)
- researchamerica.org (https://researchamerica.org/press-releases-statements/large-majority-of-americans-say-consideration-of-clinical-trial-participation-should-be-a-part-of-regular-health-care)
- Statement Describing the Extent of Confidentiality of Records
- How Americans View Data Privacy (https://pewresearch.org/internet/2023/10/18/how-americans-view-data-privacy)
- catalyst.harvard.edu (https://catalyst.harvard.edu/regulatory/video-series-participant-data?utm_source=Twitter&utm_medium=Social%20&utm_campaign=Participant%20Data%20Videos%20)
- tahsn.ca (https://tahsn.ca/documents-resources)
- clinregs.niaid.nih.gov (https://clinregs.niaid.nih.gov/country/india)
- mailchi.mp (https://mailchi.mp/jhu/bioethicsbulletin-2519368-8a3ajiivxd-2520380)
- medcitynews.com (https://medcitynews.com/2024/01/startupdates-new-developments-from-healthcare-startups-88)
- datascience.nih.gov (https://datascience.nih.gov/news/may-data-sharing-and-reuse-seminar-2024)
- datahazards.com (https://datahazards.com)
- catalyst.harvard.edu (https://catalyst.harvard.edu/regulatory/video-series-participant-data?utm_source=Twitter&utm_medium=Social%20&utm_campaign=Participant%20Data%20Videos%20)
- digital.nhs.uk (https://digital.nhs.uk/data-and-information/keeping-data-safe-and-benefitting-the-public/public-attitudes-to-data-in-the-nhs-and-social-care)
- Additional Elements of Informed Consent
- clinicalleader.com (https://clinicalleader.com/doc/returning-individual-trial-data-to-patients-how-and-why-it-needs-to-happen-0001)
- jamanetwork.com (https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2811814)
- researchamerica.org (https://researchamerica.org/press-releases-statements/large-majority-of-americans-say-consideration-of-clinical-trial-participation-should-be-a-part-of-regular-health-care)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- raps.org (https://raps.org/news-and-articles/news-articles/2024/5/stakeholders-welcome-fda-s-informed-consent-‘key-i?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- grants.nih.gov (https://grants.nih.gov/news-events/calendar-of-events/66df6d87b39d40f5660714e2)
- statnews.com (https://statnews.com/2023/11/28/clinical-trials-data-readouts-spin?utm_campaign=rss)
- niaid.nih.gov (https://niaid.nih.gov/grants-contracts/bioethical-research-and-capacity-building-efforts)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- grants.nih.gov (https://grants.nih.gov/grants/guide/notice-files/NOT-OD-24-031.html)
- Compensation and Medical Treatment for Research-Related Injury
- nam.edu (https://nam.edu/regenerative-medicine-case-study-for-understanding-and-anticipating-emerging-science-and-technology)
- researchamerica.org (https://researchamerica.org/press-releases-statements/large-majority-of-americans-say-consideration-of-clinical-trial-participation-should-be-a-part-of-regular-health-care)
- The rewards of being a research professional (https://ctsi.umn.edu/news/rewards-being-research-professional)
- jamanetwork.com (https://jamanetwork.com/journals/jama/article-abstract/2820280)
- federalregister.gov (https://federalregister.gov/documents/2024/03/01/2024-04377/key-information-and-facilitating-understanding-in-informed-consent-draft-guidance-for-sponsors)
- nucats.northwestern.edu (https://nucats.northwestern.edu/about/news/2024/tnn-success.html)
- automatedresearch.org (https://automatedresearch.org/news/news-briefing-1-12-july-2024)
- mailchi.mp (https://mailchi.mp/jhu/bioethicsbulletin-2519368-8a3ajiivxd-2520380)
- researchamerica.org (https://researchamerica.org/press-releases-statements/large-majority-of-americans-say-consideration-of-clinical-trial-participation-should-be-a-part-of-regular-health-care)
- researchamerica.org (https://researchamerica.org/blog/americans-decreasingly-willing-to-share-health-information-but-place-high-value-on-clinical-trials)
- usertesting.com (https://usertesting.com/resources/library/industry-reports/us-consumer-perceptions-ai-healthcare)
- Contact Information for Questions or Concerns
- pharmafile.com (https://pharmafile.com/appointments/richmond-promotes-new-director-of-communications-and-participant-engagement)
- bmchealthservres.biomedcentral.com (https://bmchealthservres.biomedcentral.com/articles/10.1186/s12913-024-11166-x)
- nature.com (https://nature.com/articles/s41371-024-00942-1)
- nature.com (https://nature.com/articles/s41591-024-03243-9)
- jamanetwork.com (https://jamanetwork.com/journals/jama/fullarticle/2822037?utm_source=twitter&utm_campaign=content-shareicons&utm_content=article_engagement&utm_medium=social&utm_term=080624)
- researchinvolvement.biomedcentral.com (https://researchinvolvement.biomedcentral.com/articles/10.1186/s40900-023-00530-6)
- clinicalleader.com (https://clinicalleader.com/doc/returning-individual-trial-data-to-patients-how-and-why-it-needs-to-happen-0001)
- Voluntary Participation Statement
- catalyst.harvard.edu (https://catalyst.harvard.edu/regulatory/video-series-participant-data?utm_source=Twitter&utm_medium=Social%20&utm_campaign=Participant%20Data%20Videos%20)
- clinicalleader.com (https://clinicalleader.com/doc/returning-individual-trial-data-to-patients-how-and-why-it-needs-to-happen-0001)
- nam.edu (https://nam.edu/regenerative-medicine-case-study-for-understanding-and-anticipating-emerging-science-and-technology)
- jamanetwork.com (https://jamanetwork.com/journals/jama/article-abstract/2820280)
- hqhumanreproductiveprogramme.cmail20.com (https://hqhumanreproductiveprogramme.cmail20.com/t/d-e-elhuddl-ikelhejk-dh)
- mailchi.mp (https://mailchi.mp/jhu/scienceethics1-2519855)
- gh.bmj.com (https://gh.bmj.com/content/9/9/e014971.full)
- Procedures for Withdrawal from the Study
- ssrc.org (https://ssrc.org/mercury-project/2024/06/09/seven-tips-from-experts-on-communicating-your-research)
- healthing.ca (https://healthing.ca/partners/clinical-trials-ontario/6-clinical-trial-myths-busted?utm_campaign=healthing_promo&utm_medium=postmedia_organic&utm_source=Twitter)
- clinicalleader.com (https://clinicalleader.com/doc/returning-individual-trial-data-to-patients-how-and-why-it-needs-to-happen-0001)
- mailchi.mp (https://mailchi.mp/jhu/bioethics-in-the-academic-literature-2520524)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- rethinkingclinicaltrials.org (https://rethinkingclinicaltrials.org/news/grand-rounds-february-2-2024-strategies-for-improving-public-understanding-of-fda-and-the-products-it-regulateswhy-should-we-care-and-what-might-we-do-susan-c-winckler-rph-esq)
- raps.org (https://raps.org/news-and-articles/news-articles/2024/5/stakeholders-welcome-fda-s-informed-consent-‘key-i?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- greenlight.guru (https://greenlight.guru/blog/how-to-set-up-clinical-studies-to-comply-with-us-fda-regulations)
- walgreens.com (https://walgreens.com/health-services/clinical-trials)
- Ensuring Understandability and Voluntariness
- bioethicstoday.org (https://bioethicstoday.org/blog/academic-and-private-partnership-to-improve-informed-consent-forms-using-a-data-driven-approach)
- catalyst.harvard.edu (https://catalyst.harvard.edu/regulatory/video-series-participant-data?utm_source=Twitter&utm_medium=Social%20&utm_campaign=Participant%20Data%20Videos%20)
- federalregister.gov (https://federalregister.gov/documents/2024/03/01/2024-04377/key-information-and-facilitating-understanding-in-informed-consent-draft-guidance-for-sponsors)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- raps.org (https://raps.org/news-and-articles/news-articles/2024/5/stakeholders-welcome-fda-s-informed-consent-‘key-i?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- mailchi.mp (https://mailchi.mp/jhu/scienceethics1-2519855)
- mailchi.mp (https://mailchi.mp/jhu/bioethics-in-the-academic-literature-2520524)
- jamanetwork.com (https://jamanetwork.com/journals/jama/article-abstract/2812877)
- nejm.org (https://nejm.org/doi/10.1056/NEJMcp2304436?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200pubmed)
- nexus.od.nih.gov (https://nexus.od.nih.gov/all/2024/08/16/nih-all-about-grants-podcast-considering-language-access-for-participants-in-nih-supported-clinical-research)
- federalregister.gov (https://federalregister.gov/documents/2024/03/01/2024-04377/key-information-and-facilitating-understanding-in-informed-consent-draft-guidance-for-sponsors)
- The Role of the Institutional Review Board (IRB)
- mailchi.mp (https://mailchi.mp/jhu/scienceethics1-2519855)
- statnews.com (https://statnews.com/2024/07/18/institutional-review-boards-must-assess-trials-scientific-merit?utm_campaign=rss)
- catalyst.harvard.edu (https://catalyst.harvard.edu/regulatory/video-series-participant-data?utm_source=Twitter&utm_medium=Social%20&utm_campaign=Participant%20Data%20Videos%20)
- federalregister.gov (https://federalregister.gov/documents/2024/03/01/2024-04377/key-information-and-facilitating-understanding-in-informed-consent-draft-guidance-for-sponsors)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)
- mailchi.mp (https://mailchi.mp/jhu/scienceethics1-2519883)
- jonesday.com (https://jonesday.com/en/insights/2024/01/fda-final-rule-irb-waiver-of-informed-consent-for-minimalrisk-investigations)
- undark.org (https://undark.org/2024/02/26/institutional-review-board-slow)
- brownstone.org (https://brownstone.org/articles/the-loosening-of-rules-on-informed-consent)
- niaid.nih.gov (https://niaid.nih.gov/grants-contracts/delayed-start-and-delayed-onset-human-subjects-research)
- FDA Works to Make Informed Consent Easier to Understand (https://fda.gov/news-events/fda-voices/fda-works-make-informed-consent-easier-understand)




. Follow the arrows to see how interim data influences decisions and adjustments throughout the trial process. The flowchart illustrates the key steps in adaptive trial designs. Follow the arrows to see how interim data influences decisions and adjustments throughout the trial process.](https://images.tely.ai/telyai/ngheozaj-the-flowchart-illustrates-the-key-steps-in-adaptive-trial-designs-follow-the-arrows-to-see-how-interim-data-influences-decisions-and-adjustments-throughout-the-trial-process.webp)



 influences [long-term complications](https://bioaccessla.com/blog/10-benefits-of-clinical-trial-remote-monitoring-for-research-directors), helping us see the connections that led to improved diabetes management. The center represents the DCCT trial. The branches show how intensive insulin therapy influences long-term complications, helping us see the connections that led to improved diabetes management.](https://images.tely.ai/telyai/znpozpjk-the-center-represents-the-dcct-trial-the-branches-show-how-intensive-insulin-therapy-influences-long-term-complications-helping-us-see-the-connections-that-led-to-improved-diabetes-management.webp)














 review process for studies involving human subjects, highlighting key components such as protocol examination, revisions, and the importance of informed consent. This flowchart illustrates the steps involved in the Institutional Review Board (IRB) review process for studies involving human subjects, highlighting key components such as protocol examination, revisions, and the importance of informed consent.](https://tely.blob.core.windows.net/telyai/this-flowchart-illustrates-the-steps-involved-in-the-institutional-review-board-irb-review-process-for-studies-involving-human-subjects-highlighting-key-components-such-as-protocol-examination-revisions-and-the-importance-of-informed-consent.jpg)