Introduction
The landscape of clinical research is evolving rapidly, marked by increasing complexities and stringent regulatory demands that are reshaping the role of clinical auditors. With the growing reliance on contract research organizations, there emerges a pressing necessity for auditors to master a diverse set of skills that not only ensure compliance but also enhance the integrity of medical trials.
This article delves into the ten essential skills every clinical auditor must master, exploring how these competencies not only safeguard patient rights but also drive the success of research initiatives.
What challenges do auditors face in this dynamic environment? How can they effectively navigate these challenges to uphold the highest standards of clinical research?
bioaccess®: Expertise in Clinical Research Facilitation
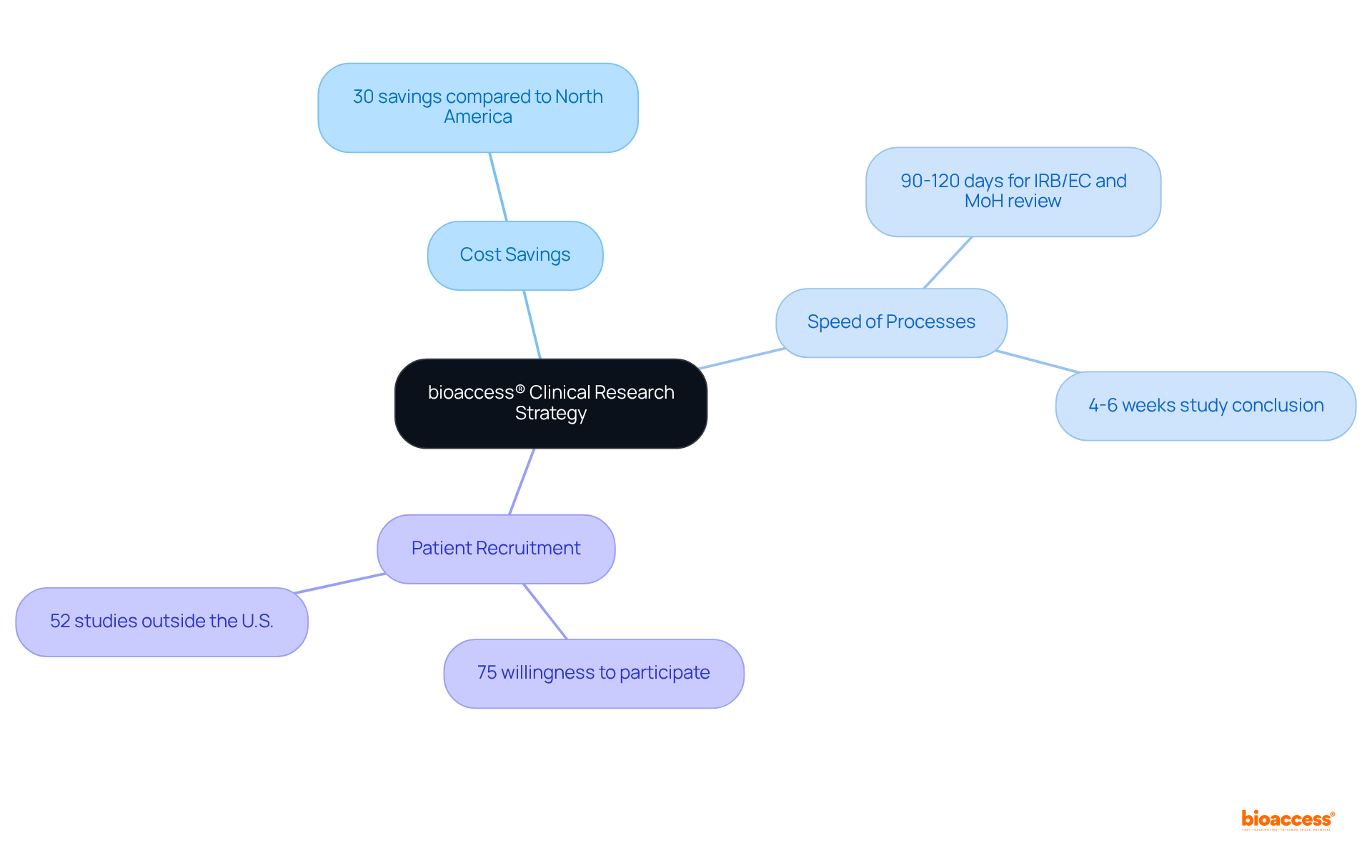
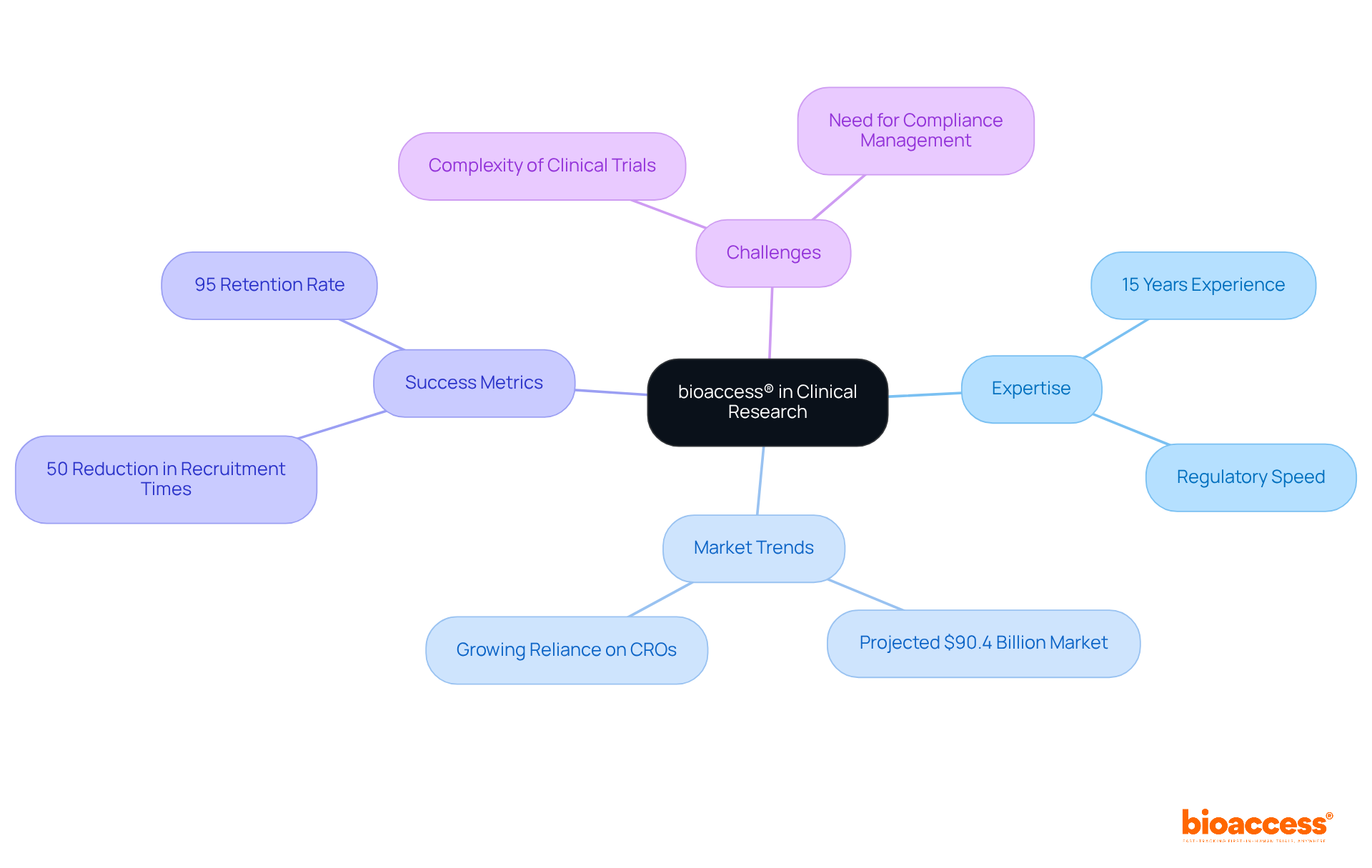
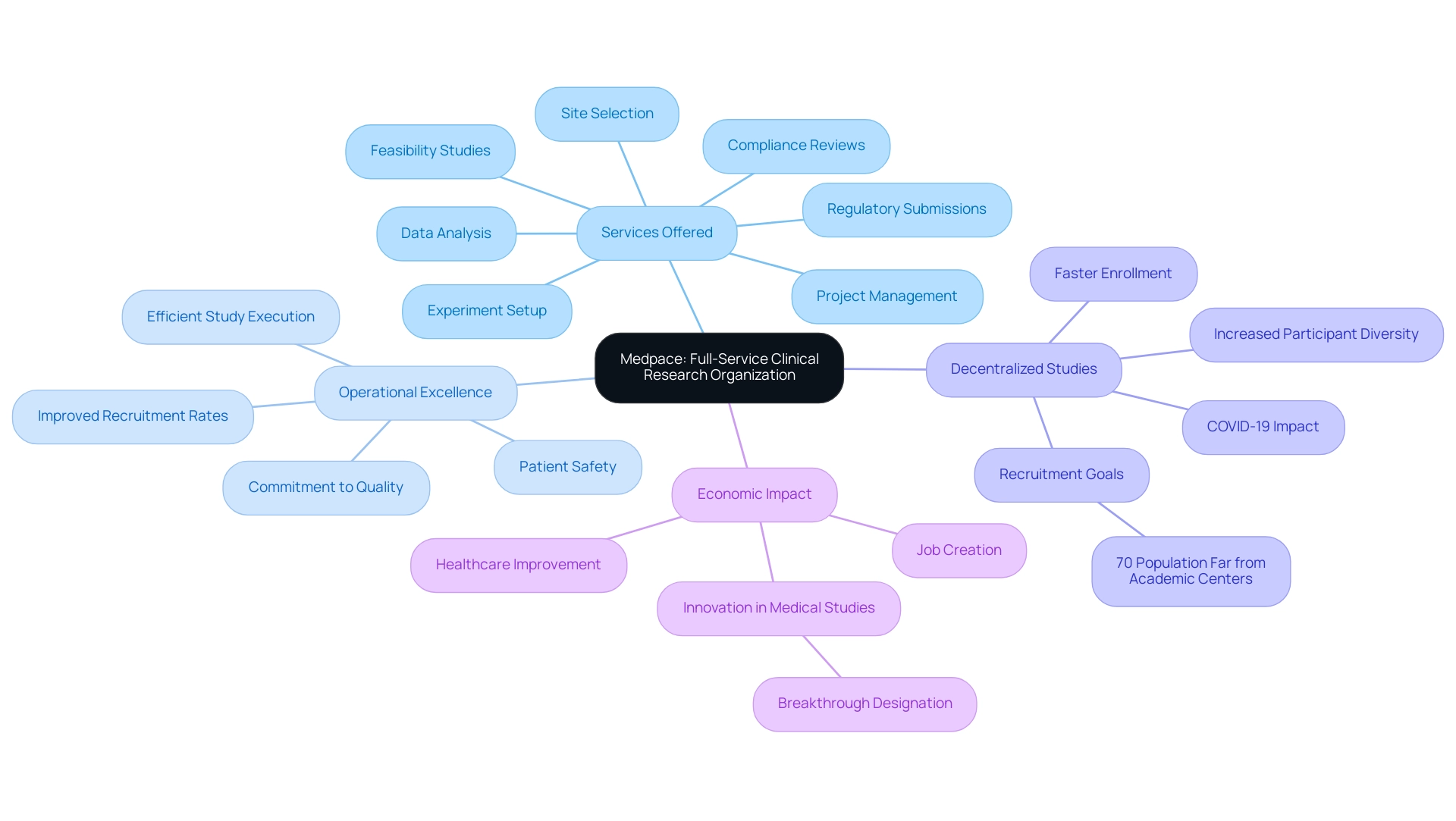
bioaccess® stands as a recognized leader in within the Medtech, Biopharma, and Radiopharma sectors. With over 15 years of experience, the organization leverages regulatory speed and diverse patient populations to expedite research studies, achieving ethical approvals in a mere 4-6 weeks. This rapid turnaround is crucial, especially given that . The likelihood of success varies significantly across therapeutic fields, underscoring the need for effective support in .
Specialist viewpoints highlight that the complexity of medical studies often leads pharmaceutical firms to delegate tasks to like bioaccess®, necessitating the involvement of a . This trend is propelled by the demand for specialized knowledge and resources to navigate the intricate . By 2025, , reflecting a growing reliance on CROs to enhance operational efficiencies and improve trial outcomes.
Successful examples of research facilitation are evident in Latin America and Australia, where bioaccess® has effectively reduced recruitment times by over 50% and achieved an impressive 95% retention rate in studies. These accomplishments illustrate the organization’s commitment to enhancing patient engagement and streamlining processes, which are vital for maintaining compliance and ensuring high-quality research outcomes. As the landscape of medical studies continues to evolve, the role of evaluators becomes increasingly critical in ensuring that these investigations adhere to regulatory standards and yield reliable results.
Regulatory Knowledge: Understanding Compliance Standards
A clinical auditor must possess a comprehensive understanding of , particularly emphasizing and local regulations. The role of a clinical auditor is crucial for evaluating whether and in alignment with established guidelines, thereby and safety throughout the research process.
Notably, underscore the necessity for ongoing training and compliance with evolving regulations, reflecting the dynamic nature of medical research. Alarmingly, statistics indicate that GCP adherence for observational studies averages only 79.5%, significantly lower than the 92.2% for interventional studies, which underscores the urgent need for .
Regulatory specialists, including Ana Criado, Director of Regulatory Affairs and CEO of Mahu Pharma, alongside Katherine Ruiz, a recognized authority in Regulatory Affairs for Medical Devices and In Vitro Diagnostics in Colombia, advocate for a robust framework that not only meets standards but also fosters , ensuring that all participants are treated with respect and transparency.
Furthermore, the can result in severe consequences, such as penalties and revocation of approvals for assessments, making it imperative for clinical auditors to prioritize adherence to regulations. , such as those offered by bioaccess, can enhance visibility and control throughout organizations, further supporting regulatory efforts.
Analytical Skills: Evaluating Data and Identifying Issues
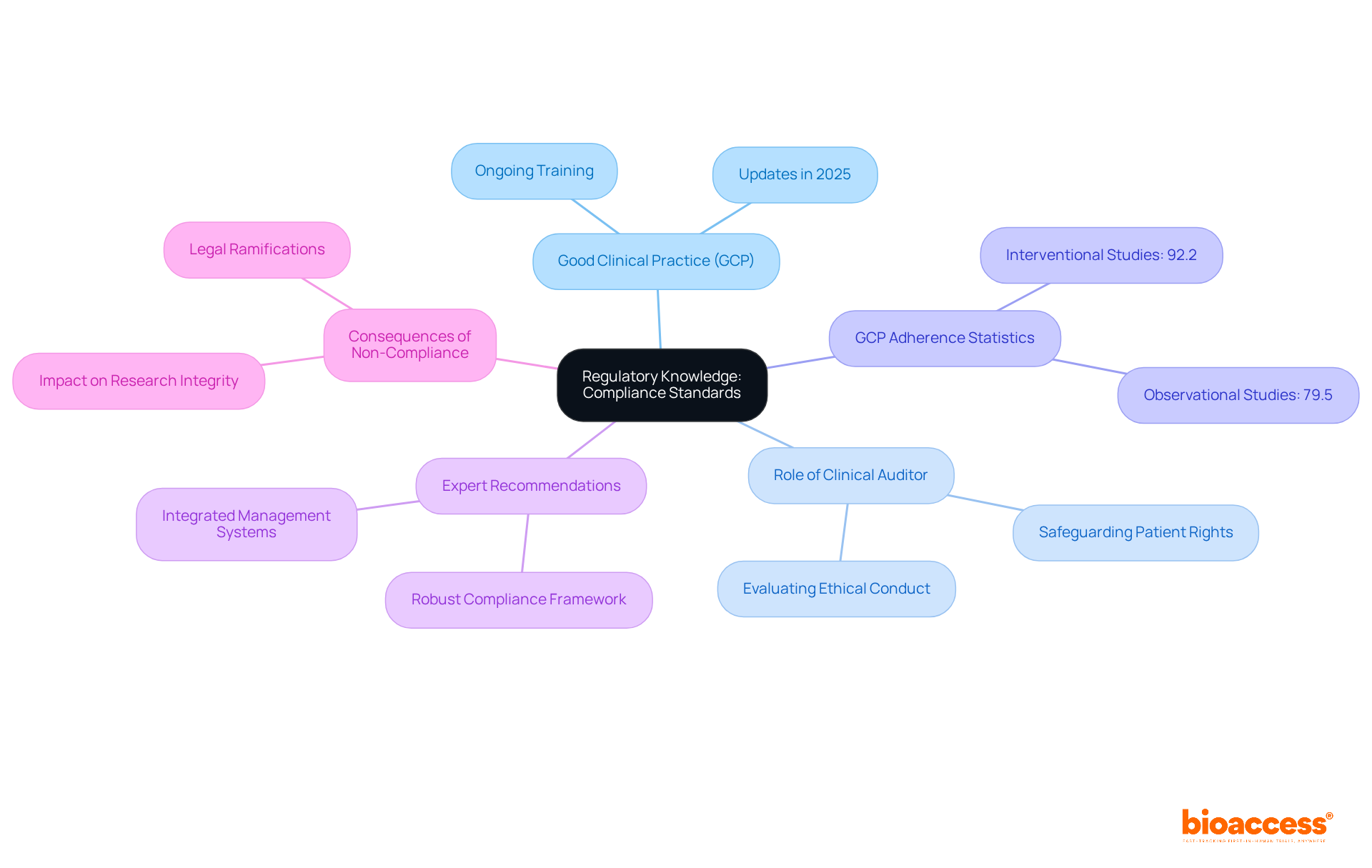
Skilled clinical reviewers excel in examining , identifying patterns, and spotting irregularities. This process necessitates a meticulous review of study protocols, patient records, and regulatory documentation to ensure accurate reporting and prompt resolution of any issues. For instance, examiners often discover common issues such as discrepancies in patient enrollment figures or inconsistencies in data reporting, which can significantly affect .
As highlighted by industry specialists, is crucial; one auditor emphasized that ‘the key duties involve performing and guaranteeing adherence to federal and industry standards.’ Moreover, comprehending is essential for assessing data efficiently.
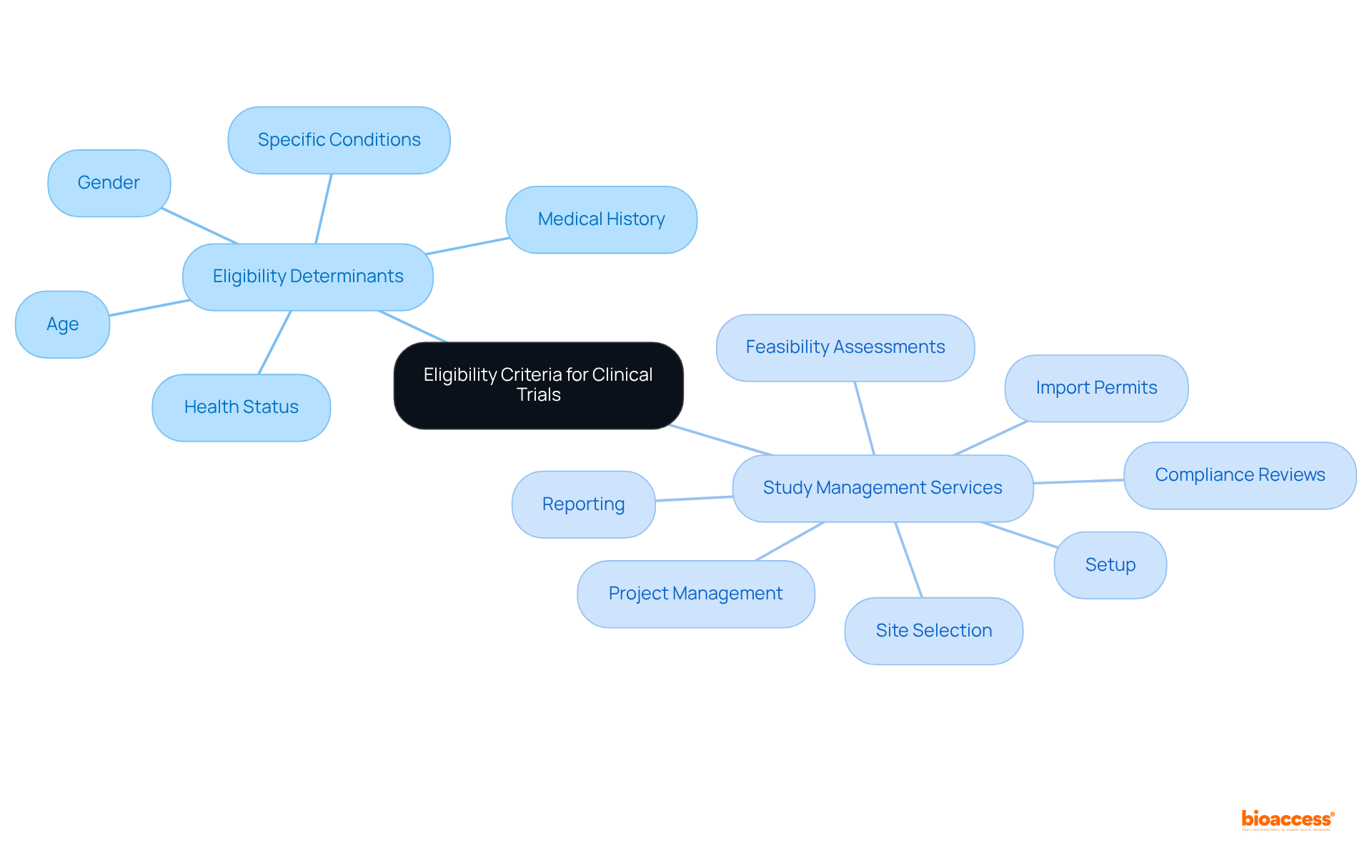
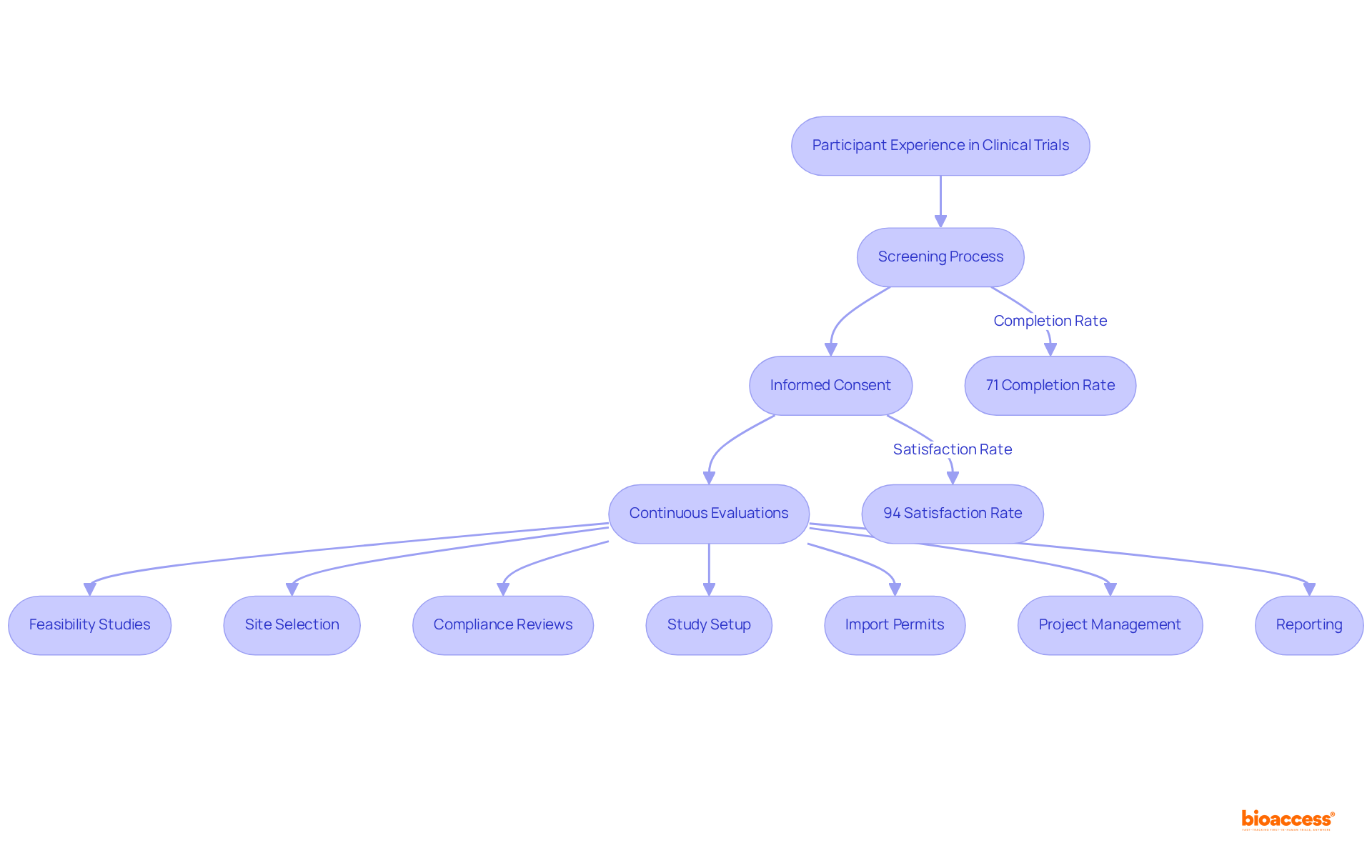
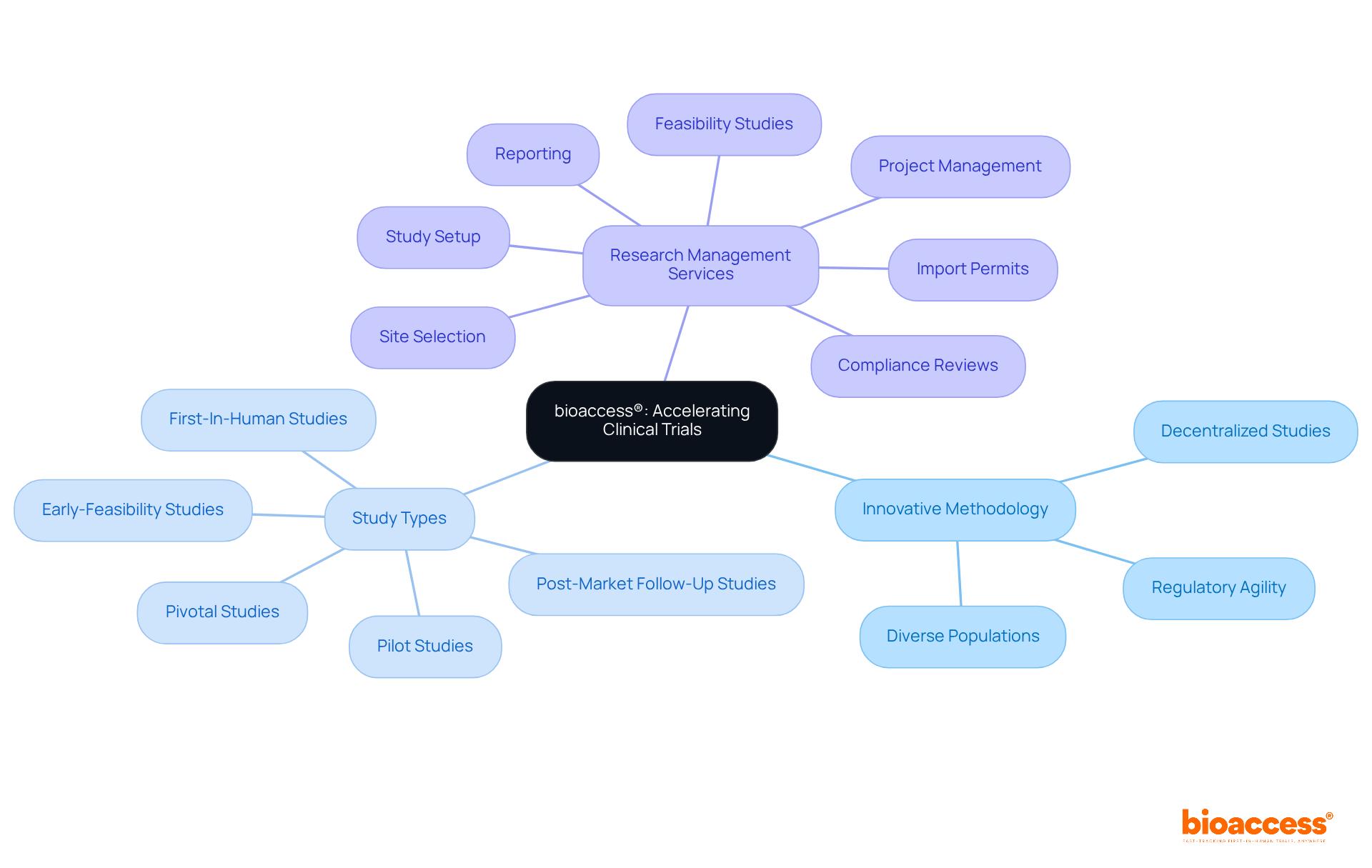
The extensive provided by bioaccess—including feasibility studies, site selection, compliance reviews, setup, and project management—directly contribute to the success of studies by ensuring that all processes align with and best practices. By utilizing strong analytical methods, evaluators can offer valuable insights that improve the integrity of medical trials and aid in the overall success of research initiatives.
Additionally, the —through job creation, economic growth, and healthcare improvement—underscores the importance of effective auditing by a in fostering international collaboration and innovation in the field. To further enhance their effectiveness, professionals should concentrate on and remain informed about industry standards.
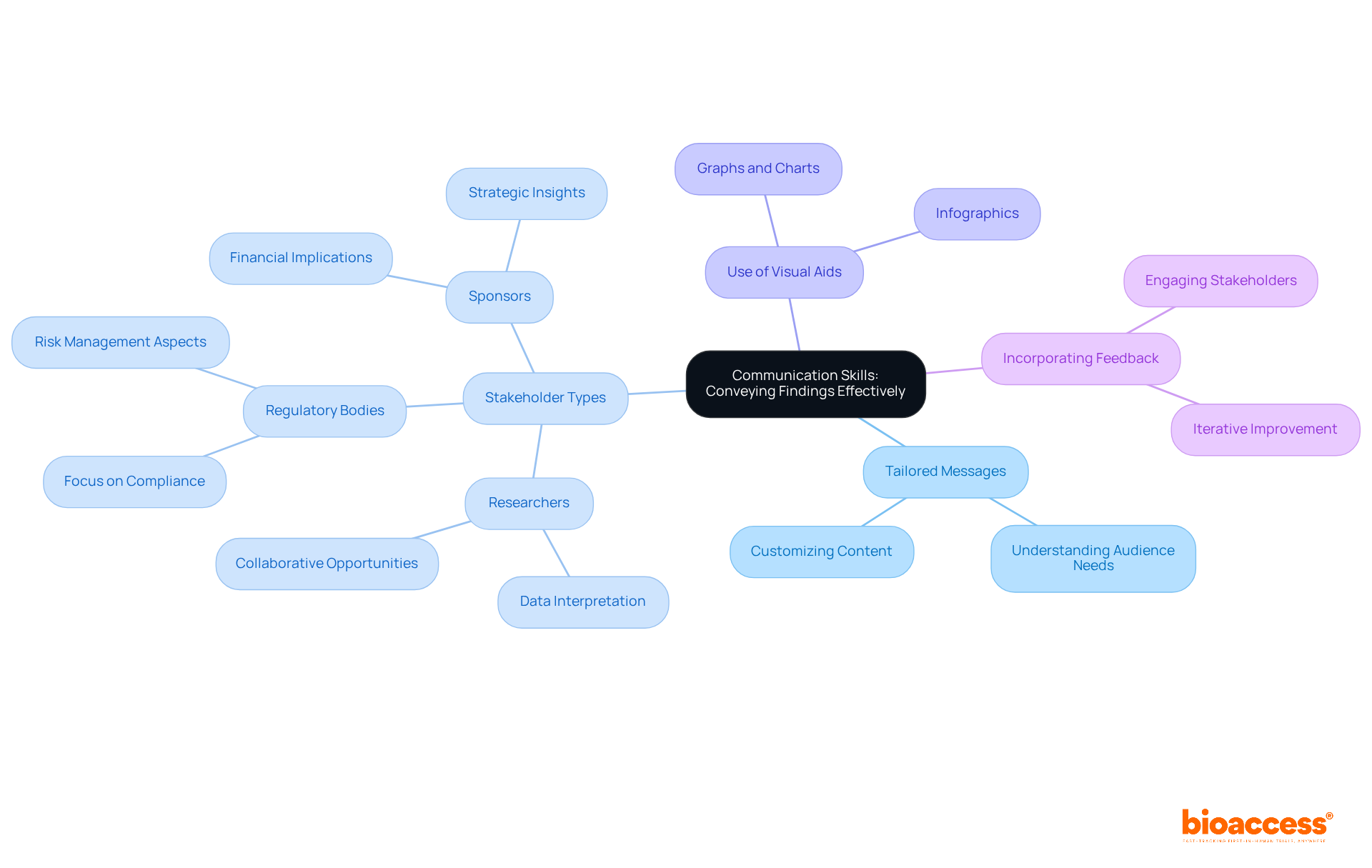
Communication Skills: Conveying Findings Effectively
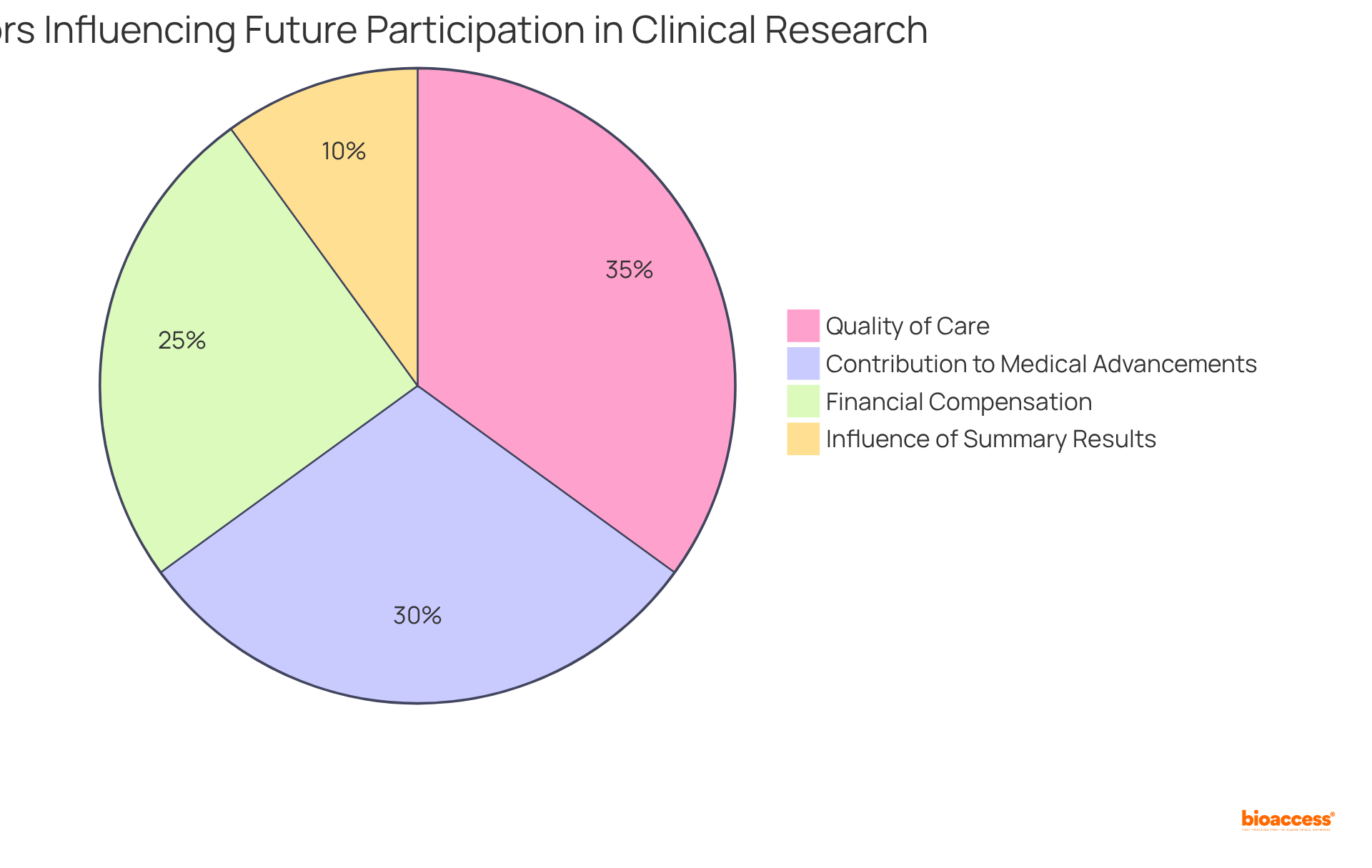
Clinical evaluators play a pivotal role in ensuring that their findings are communicated effectively to a range of stakeholders, including researchers, sponsors, and . Effective communication transcends mere clarity in reporting; it involves to meet the specific needs of diverse audiences. Research indicates that can significantly enhance , with studies showing that 70% of stakeholders feel more informed when messages are aligned with their interests and concerns.
To convey , a must prioritize clarity and relevance. For instance, when presenting to , it is crucial to emphasize compliance and risk management aspects, while discussions with sponsors may require a focus on . Utilizing , such as graphs and charts, can enhance understanding, as they distill .
Examples from industry leaders underscore the importance of this approach. A research leader observed that tailoring presentations for various stakeholders not only but also promotes a collaborative environment, encouraging stakeholders to be more receptive to the findings. Furthermore, incorporating feedback from stakeholders during presentations can lead to more productive discussions and actionable outcomes.
In summary, mastering the art of communication is essential for a . By employing and actively engaging with stakeholders, examiners can ensure that their findings are not only acknowledged but also comprehended and acted upon, ultimately fostering enhancements in research practices.
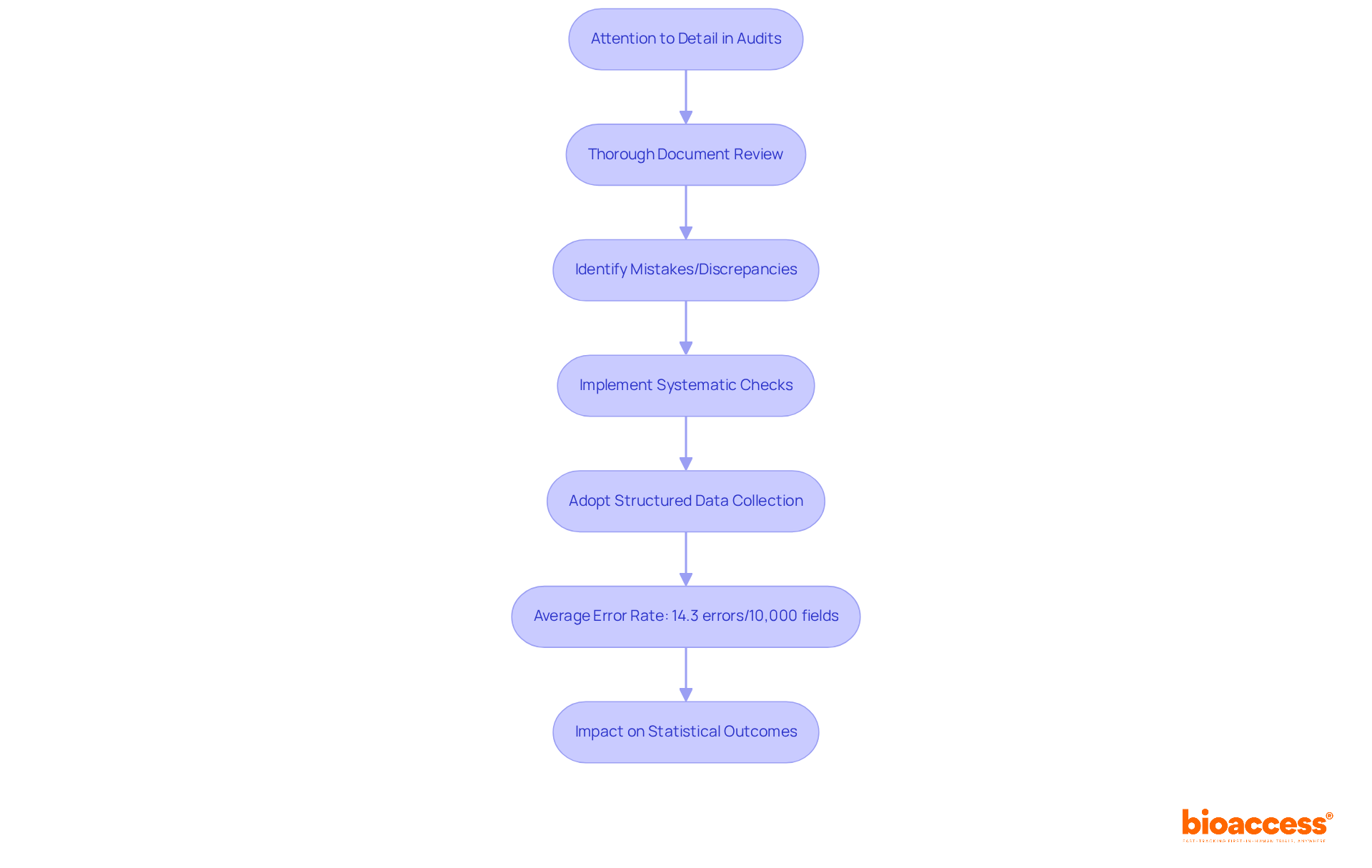
Attention to Detail: Ensuring Accuracy in Audits
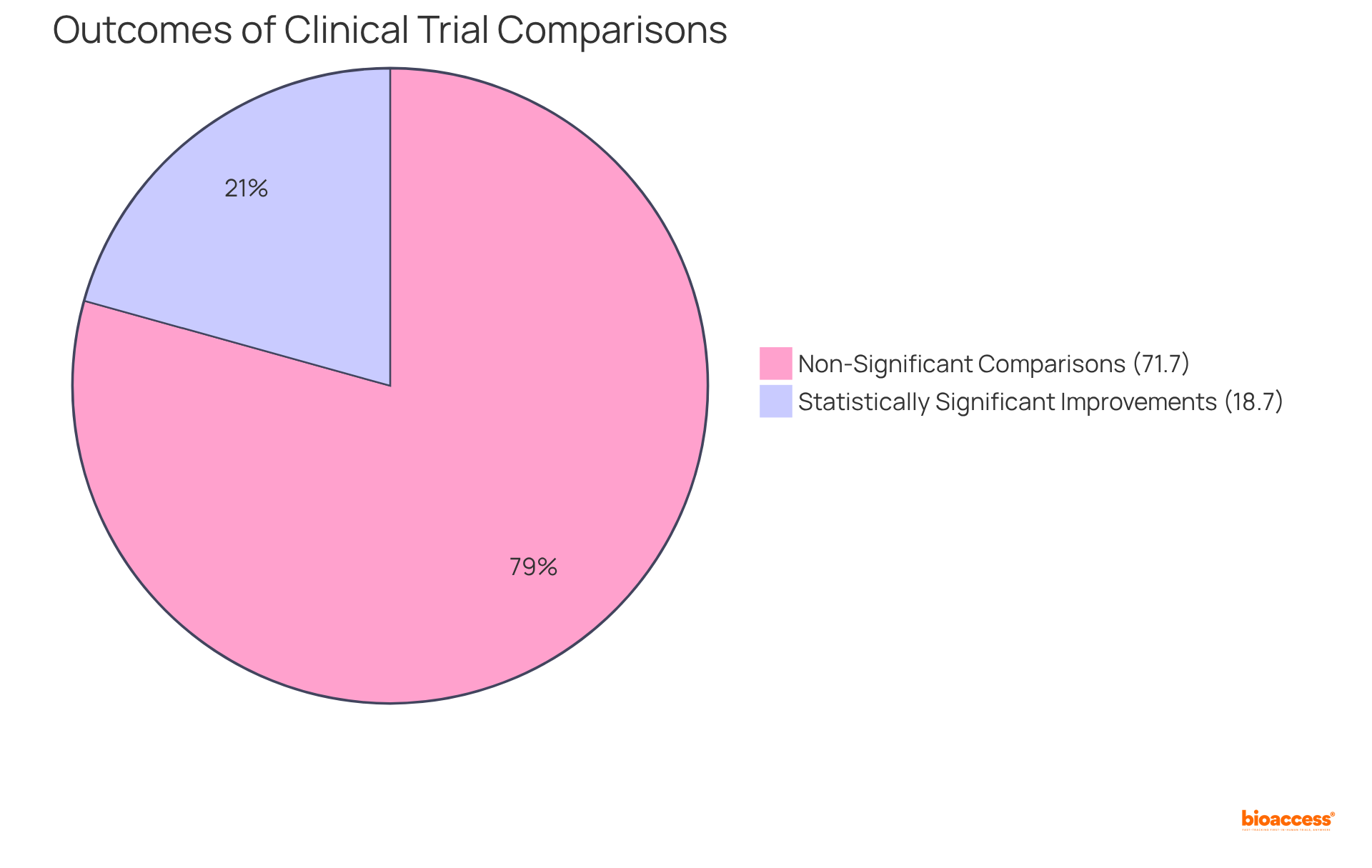
Outstanding is crucial for a successful auditor, as it encompasses a thorough review of documents, data, and processes to ensure precision. This careful method is essential for recognizing possible mistakes or discrepancies that could threaten the validity of and their results. Research indicates that the can reach concerning levels, with some studies reporting as many as 40.5 errors per 10,000 fields. Such discrepancies can significantly alter statistical outcomes, affecting p-values and correlation coefficients, which underscores the necessity for .
Industry leaders emphasize that forms the foundation for reliable study conclusions. Poor not only threatens the validity of findings but also complicates the generalizability of results. To ensure precision in research documentation, reviewers should implement , such as programmed edit checks, which can catch potential mistakes before final data submission. Additionally, adopting a structured approach to data collection and processing can mitigate the risk of errors, as evidenced by the lower error rates associated with electronic data capture systems compared to traditional methods.
Ultimately, promoting a within research teams is essential. By prioritizing , healthcare reviewers can improve , thus aiding the successful implementation of and the progress of medical advancements.
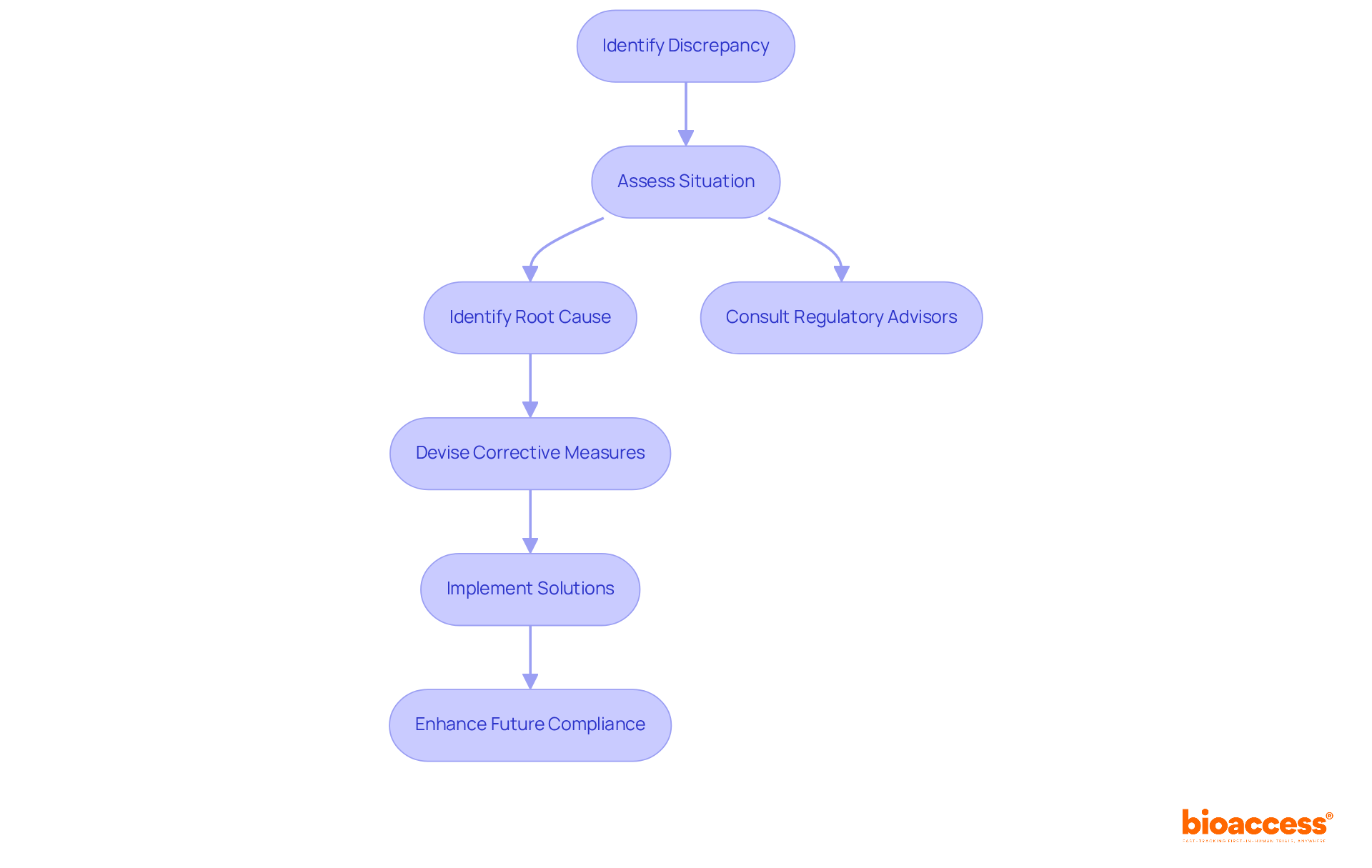
Problem-Solving Skills: Navigating Audit Challenges
Clinical reviewers frequently encounter challenges that demand quick decision-making and adept problem-solving skills. Discrepancies in , such as inconsistencies in reporting methods or analysis discrepancies, . For example, three studies revealed in 46% to 82% of RCTs, underscoring the prevalence of these issues.
Navigating these challenges necessitates that professionals are well-versed in and equipped to implement effective solutions that uphold the integrity of the audit. Collaborating with knowledgeable regulatory advisors, as illustrated in the case study ‘,’ can further assist reviewers in , ensuring that all elements of the review adhere to established guidelines.
Moreover, leveraging comprehensive , such as those provided by bioaccess—including feasibility studies, site selection, regulatory reviews, trial setup, import permits, project management, and reporting—can enhance reviewers’ capacity to navigate these complexities.
When discrepancies arise, examiners must promptly assess the situation, identify the root cause, and that not only rectify the current issue but also bolster . This proactive approach is essential for fostering a culture of transparency and accountability within clinical research organizations.
As Sarah Lee aptly stated, ‘ is not merely a technical goal—it is a crucial aspect of safeguarding patient health and advancing medical innovation.’ Furthermore, the potential repercussions of non-compliance, such as financial penalties, legal actions, audit denials, and delays in outcomes, highlight the critical nature of the reviewers’ role in ensuring adherence to .
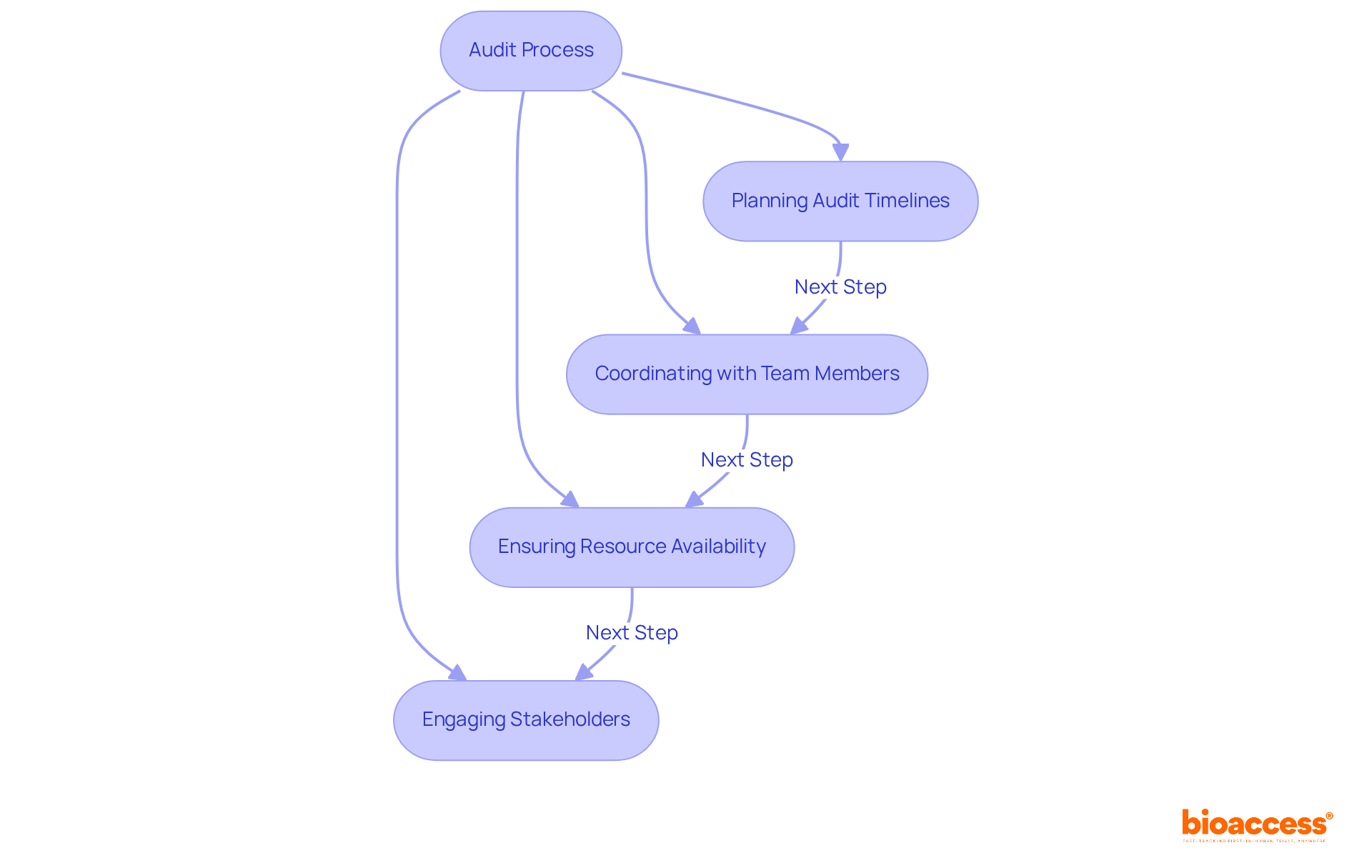
Project Management Skills: Coordinating Audit Activities
To effectively oversee the from inception to completion, s must exhibit robust . This includes meticulous planning of audit timelines, seamless coordination with team members, and ensuring the availability of all necessary resources. Effective project management streamlines the auditing process and enhances the efficiency and effectiveness of all activities involved, particularly for the .
For instance, organizations that adopt structured methodologies attain an impressive , underscoring the importance of organized project management in audits. Furthermore, proficient project managers are associated with enhanced , which are crucial for the success of research studies. As the , cultivating these skills within medical research becomes progressively essential.
Engaging stakeholders and maintaining open communication are vital components that contribute to the overall success of audit activities performed by the , ensuring that all parties are aligned and informed throughout the process. In the context of progressing medical equipment evaluations, clinical reviewers play a vital role in site feasibility and investigator selection, fundamental to overcoming regulatory obstacles and recruitment challenges.
Additionally, they are involved in , obtaining , and reporting on adverse events, all critical for compliance and project success. As Samantha Ferguson, Head of Content at Project.co, states, ‘Yes, project management is in extremely high demand,’ further emphasizing the need for skilled professionals in this field. Moreover, with 80% of studies facing delays due to recruitment issues, cannot be overstated.
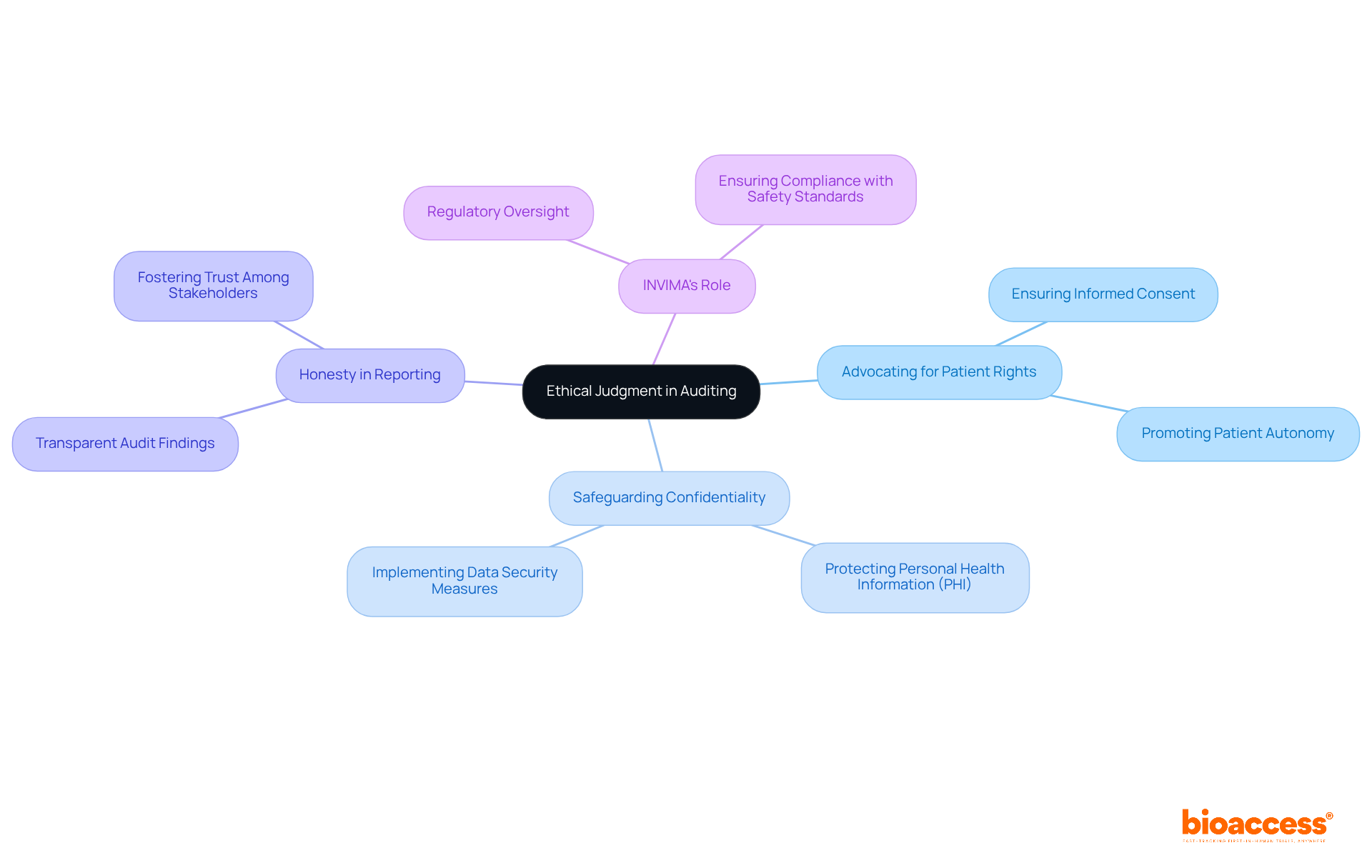
Ethical Judgment: Upholding Integrity in Auditing
Clinical reviewers must demonstrate , ensuring their actions and decisions align with established and regulations. This responsibility includes:
- , thereby fostering trust among stakeholders
In , the (Instituto Nacional de Vigilancia de Medicamentos y Alimentos), which plays a crucial role in the inspection and supervision of health products, including medical devices. Recognized as a Level 4 health authority by the Pan American Health Organization and World Health Organization, INVIMA guarantees that adhere to stringent safety, efficacy, and quality standards. Understanding these is essential for professionals to in their auditing processes.
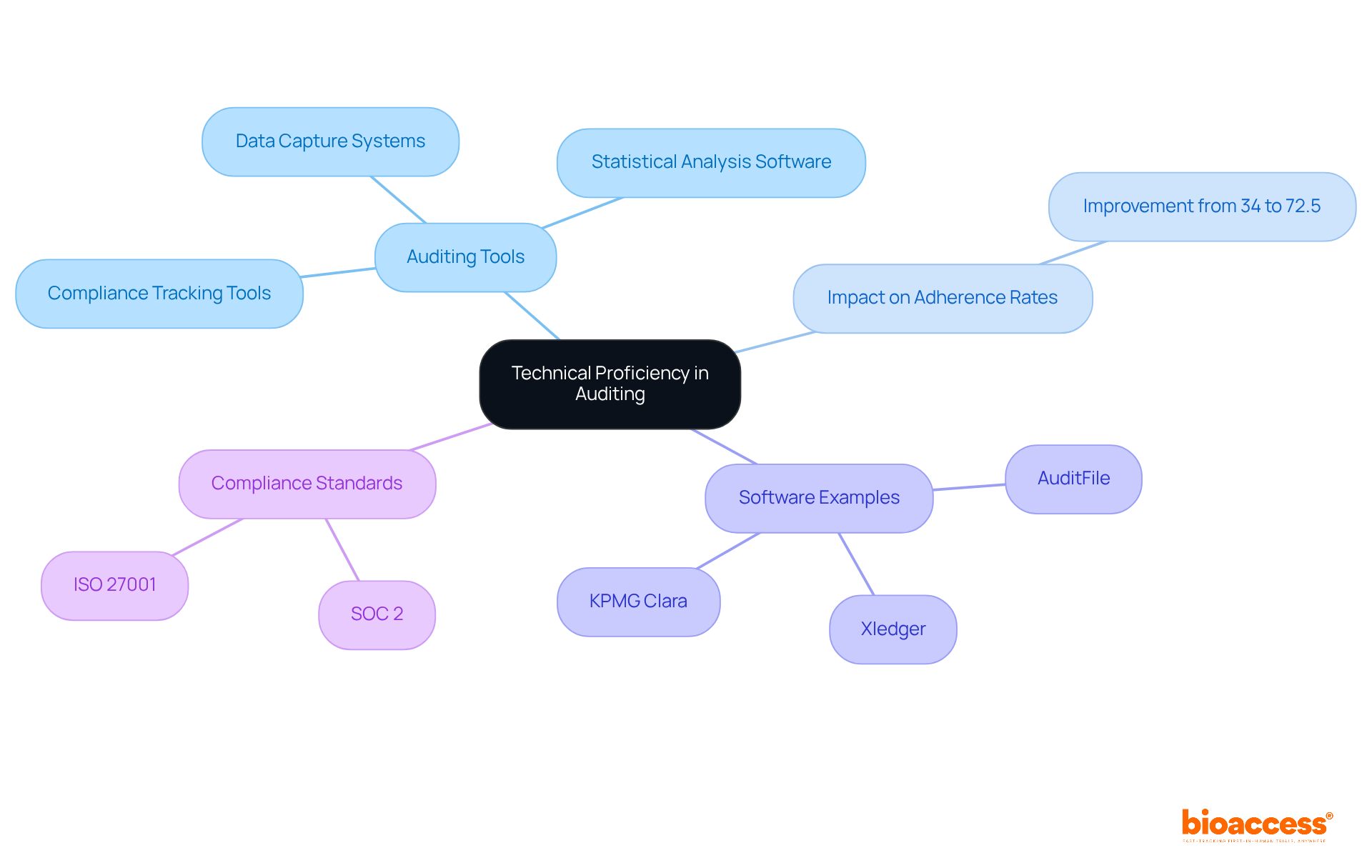
Technical Proficiency: Utilizing Auditing Tools Effectively
A must excel in utilizing a variety of that streamline data collection, analysis, and reporting. Proficiency in is essential for a , as these platforms improve the accuracy and efficiency of audits. Research conducted at Al-Karak Governmental Hospital clearly illustrates this point: organizations employing experienced a remarkable increase in adherence rates, with conformity to guidelines improving from 34% to 72.5% after implementing these audits.
Furthermore, the integration of and empowers the to conduct comprehensive evaluations, leading to more reliable findings. Industry leader Mohammad Abu-Jeyyab emphasizes that the significant improvements in adherence to guidelines after the second loop of audits underscore the effectiveness of these auditing tools.
Additionally, the use of specific auditing software, such as AuditFile and Xledger, can further enhance and real-time reporting capabilities. As technology advances, the field of medical auditing is evolving, making it essential for a to stay informed about the latest tools and methods to ensure effective oversight and quality assurance.
is also crucial in this context, as it underscores the importance of maintaining high standards in auditing practices.
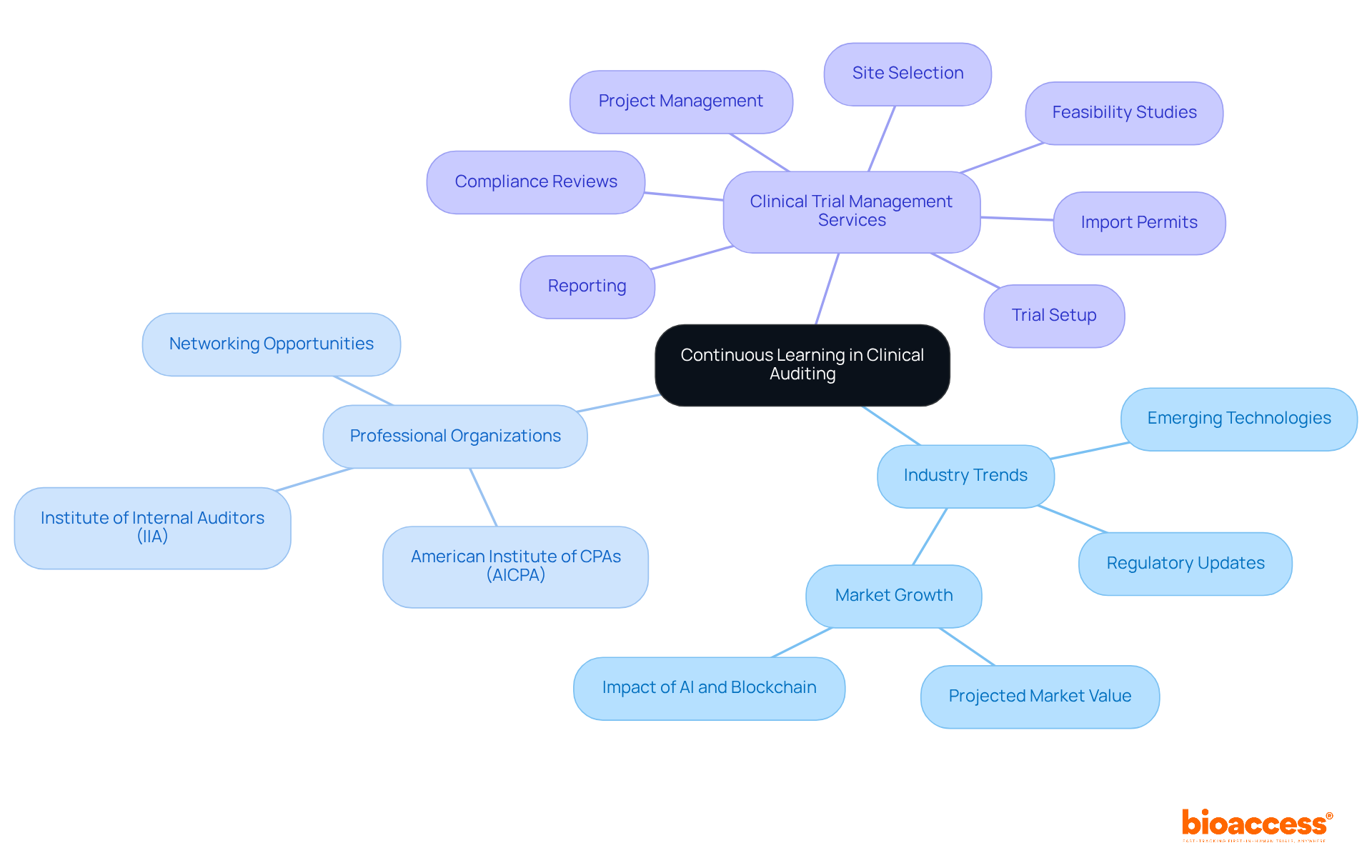
Continuous Learning: Staying Updated with Industry Trends
To maintain their effectiveness, clinical auditors must prioritize , actively engaging with the latest industry trends, regulatory updates, and . This commitment to professional growth not only hones the skills of a clinical auditor but also enables them to provide valuable insights and suggestions that significantly enhance the success of medical trials. With the projected to exceed $80 billion by 2025, it is essential for clinical auditors to remain knowledgeable in order to navigate the complexities of evolving regulations and adherence requirements. Industry leaders emphasize that embracing change and adapting to new methodologies are crucial for professionals to stay relevant in a rapidly transforming landscape.
Moreover, participation in professional organizations such as:
- the
- the American Institute of CPAs (AICPA)
along with attending conferences, can provide professionals with networking opportunities and access to the latest advancements in research integrity. Understanding , such as those offered by bioaccess—including:
- site selection
- import permits
- reporting
can further equip clinical auditors to meet the challenges of 2025 and beyond.
Conclusion
Mastering the essential skills of clinical auditing is crucial for professionals navigating the complexities of medical research. These skills not only ensure compliance with regulatory standards but also enhance the integrity and success of clinical trials. A clinical auditor’s expertise in areas such as:
- Regulatory knowledge
- Analytical skills
- Communication
- Attention to detail
- Problem-solving
- Project management
- Ethical judgment
- Technical proficiency
- Continuous learning
collectively contribute to their effectiveness in the field.
Throughout the article, key insights were highlighted, including:
- The importance of understanding Good Clinical Practice (GCP)
- The necessity of robust analytical methods
- The critical nature of effective communication with stakeholders
- The role of attention to detail and problem-solving capabilities, as these traits are vital for identifying discrepancies and ensuring accurate audit outcomes
- The demand for project management skills, given their significance in coordinating audit activities and overcoming recruitment challenges in clinical studies
As the landscape of clinical research continues to evolve, the significance of these skills cannot be overstated. Clinical auditors are at the forefront of safeguarding patient rights and ensuring the ethical conduct of research. By prioritizing continuous learning and staying updated with industry trends, auditors can adapt to the changing environment and maintain their effectiveness. Embracing these essential skills will not only enhance individual careers but also advance the integrity and success of clinical research as a whole.
Frequently Asked Questions
What is bioaccess® and what sectors does it operate in?
bioaccess® is a recognized leader in research facilitation within the Medtech, Biopharma, and Radiopharma sectors, with over 15 years of experience in expediting research studies.
How quickly can bioaccess® achieve ethical approvals for research studies?
bioaccess® can achieve ethical approvals in a mere 4-6 weeks, which is crucial given that only about 25% of drugs in the R&D pipeline successfully reach the market.
Why do pharmaceutical firms delegate tasks to contract research organizations (CROs) like bioaccess®?
Pharmaceutical firms often delegate tasks to CROs due to the complexity of medical studies and the need for specialized knowledge and resources to navigate the intricate regulatory landscape.
What is the projected global market size for contract research outsourcing by 2025?
The global market for contract research outsourcing is projected to reach $90.4 billion by 2025.
What are some successful outcomes bioaccess® has achieved in research facilitation?
In Latin America and Australia, bioaccess® has effectively reduced recruitment times by over 50% and achieved a 95% retention rate in studies.
What is the role of a clinical auditor in medical trials?
A clinical auditor evaluates whether medical trials are conducted ethically and in alignment with regulatory guidelines, safeguarding patient rights and safety.
What are Good Clinical Practice (GCP) standards, and why are they important?
Good Clinical Practice (GCP) standards are regulatory requirements that ensure ethical and scientific quality in clinical trials. Adherence to GCP is crucial for protecting participants and ensuring reliable results.
What are the compliance statistics for observational and interventional studies?
The adherence to GCP for observational studies averages only 79.5%, while interventional studies have a higher adherence rate of 92.2%.
What are the potential consequences of non-compliance with regulatory standards?
Non-compliance can result in severe consequences, including penalties and the revocation of approvals for assessments.
What skills are essential for skilled clinical reviewers?
Skilled clinical reviewers must excel in examining clinical study data, identifying patterns, spotting irregularities, and ensuring adherence to federal and industry standards.
How does bioaccess® support the success of clinical studies?
bioaccess® provides extensive research management services, including feasibility studies, site selection, compliance reviews, setup, and project management, ensuring alignment with regulatory standards.
What impact do Medtech clinical studies have on local economies?
Medtech clinical studies contribute to local economies through job creation, economic growth, and healthcare improvement, highlighting the importance of effective auditing in fostering collaboration and innovation.
List of Sources
- bioaccess®: Expertise in Clinical Research Facilitation
- labiotech.eu (https://labiotech.eu/trends-news/clinical-trials-success-rate)
- 2025 Clinical Data Trend Report | Veeva (https://veeva.com/2025-clinical-data-trend-report)
- statista.com (https://statista.com/topics/6756/clinical-trials)
- 10 Trends and Statistics for Clinical Trials in 2023 (https://xtalks.com/10-trends-and-statistics-for-clinical-trials-in-2023-3377)
- bioaccessla.com (https://bioaccessla.com/blog/10-key-features-of-clinical-trial-software-for-medtech-success)
- Regulatory Knowledge: Understanding Compliance Standards
- The Importance of Regulatory Compliance in Clinical Trials (https://lindushealth.com/blog/the-importance-of-regulatory-compliance-in-clinical-trials)
- Clinical Research Compliance: 5 Critical Factors for 2024 Success (https://infonetica.net/articles/clinical-research-compliance)
- fdli.org (https://fdli.org/2025/02/compliance-challenges-for-clinical-research-sites)
- goodreads.com (https://goodreads.com/work/quotes/14007542)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC4936070)
- Analytical Skills: Evaluating Data and Identifying Issues
- Statistical analysis and significance tests for clinical trial data (https://sciencedirect.com/science/article/pii/S1357303925000787)
- aamc.org (https://aamc.org/news/whats-stake-when-clinical-trials-research-gets-cut)
- Understanding Statistical Analysis in Clinical Trials (https://lindushealth.com/blog/understanding-clinical-trial-statistics-a-comprehensive-guide)
- nature.com (https://nature.com/articles/d41586-023-02299-w)
- medicalbillgurus.com (https://medicalbillgurus.com/professional-medical-claims-auditing)
- Communication Skills: Conveying Findings Effectively
- tealhq.com (https://tealhq.com/skills/auditor)
- ecampusontario.pressbooks.pub (https://ecampusontario.pressbooks.pub/internalauditing/chapter/10-02-communicating-findings-to-stakeholders)
- 50 Quotes About Communication | Chanty (https://chanty.com/blog/communication-quotes)
- Communication Quotes to Inspire Connection, Collaboration & Teamwork | SUCCESS (https://success.com/communication-quotes)
- eoxs.com (https://eoxs.com/new_blog/how-to-communicate-findings-effectively-in-audit-reports)
- Attention to Detail: Ensuring Accuracy in Audits
- Quantifying Data Quality for Clinical Trials Using Electronic Data Capture (https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0003049)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC10775420)
- status.net (https://status.net/articles/attention-detail-best-examples)
- Problem-Solving Skills: Navigating Audit Challenges
- chartrequest.com (https://chartrequest.com/articles/healthcare-auditing-challenges)
- numberanalytics.com (https://numberanalytics.com/blog/avoiding-type-ii-errors-medical-trials)
- fdli.org (https://fdli.org/2025/02/compliance-challenges-for-clinical-research-sites)
- journals.plos.org (https://journals.plos.org/plosmedicine/article/info:doi/10.1371/journal.pmed.1001666)
- link.springer.com (https://link.springer.com/article/10.1007/s43441-022-00388-y)
- Project Management Skills: Coordinating Audit Activities
- What Makes An Effective Clinical Trial Project Manager? – Imperial Clinical Research Services Blog (https://imperialcrs.com/blog/business-insights/what-makes-an-effective-clinical-trial-project-manager)
- project.co (https://project.co/project-management-statistics)
- projectmanagertemplate.com (https://projectmanagertemplate.com/post/project-management-stats-worldwide-insights-and-trends)
- bioaccessla.com (https://bioaccessla.com/blog/understanding-the-clinical-project-manager-role-and-responsibilities)
- Ethical Judgment: Upholding Integrity in Auditing
- msaonline.depaul.edu (https://msaonline.depaul.edu/blog/ethical-standards-in-auditing)
- internationalaccountingbulletin.com (https://internationalaccountingbulletin.com/news/kpmg-fined-for-audit-breaches)
- Healthcare Data Breach Statistics (https://hipaajournal.com/healthcare-data-breach-statistics)
- researchgate.net (https://researchgate.net/publication/346530718_Review_of_Ethics_Issues_related_to_Clinical_Audit_and_Quality_Improvement_Activities_Clinical_audit_tool_to_promote_quality_for_better_health_services)
- Technical Proficiency: Utilizing Auditing Tools Effectively
- vintti.com (https://vintti.com/blog/top-5-audit-software-a-detailed-review-for-accounting-firms)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC8317325)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC10961103)
- Top 10 Quality Control Quotes to Inspire Your Team (https://movley.com/blog/top-10-quality-control-quotes-to-inspire-your-team)
- appliedclinicaltrialsonline.com (https://appliedclinicaltrialsonline.com/view/clinical-study-audits-the-quality-management-approach)
- Continuous Learning: Staying Updated with Industry Trends
- linkedin.com (https://linkedin.com/advice/3/what-best-ways-healthcare-professionals-stay-pnmpf)
- 2025 Clinical Research Workforce Report (https://ccrps.org/clinical-research-blog/2025-clinical-research-workforce-report)
- tealhq.com (https://tealhq.com/skills/auditor)
- Clinical Research Compliance: 5 Critical Factors for 2024 Success (https://infonetica.net/articles/clinical-research-compliance)
- 5 Important Things for Life Sciences Companies to Consider | MasterControl (https://mastercontrol.com/gxp-lifeline/quality-inspiration)



. Follow the arrows to see how interim data influences decisions and adjustments throughout the trial process. The flowchart illustrates the key steps in adaptive trial designs. Follow the arrows to see how interim data influences decisions and adjustments throughout the trial process.](https://images.tely.ai/telyai/ngheozaj-the-flowchart-illustrates-the-key-steps-in-adaptive-trial-designs-follow-the-arrows-to-see-how-interim-data-influences-decisions-and-adjustments-throughout-the-trial-process.webp)